Back to Journals » Clinical Ophthalmology » Volume 9
Portable single port 23-gauge vitrectomy in postoperative endophthalmitis
Authors Höhn F, Kretz F, Sheth S, Natarajan S, Singh P, Koch F, Koss M
Received 20 January 2015
Accepted for publication 24 March 2015
Published 13 August 2015 Volume 2015:9 Pages 1457—1461
DOI https://doi.org/10.2147/OPTH.S81262
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Fabian Höhn,1,* Florian TA Kretz,2,* Saumil Sheth,3 S Natarajan,3 Pankaj Singh,4 Frank H Koch,4 Michael J Koss2,5
1Helios Klinikum Pforzheim, Pforzheim, Germany; 2Department of Ophthalmology, Hospital of the Ruprecht-Karls University, Heidelberg, Germany; 3Aditya Jyot Eye Hospital Pvt. Ltd, Mumbai, India; 4Department of Ophthalmology, Hospital of the Goethe University, Frankfurt am Main, Germany; 5Department of Ophthalmology, University of Southern California, Los Angeles, CA, USA
*These authors contributed equally to this work
Aim: To evaluate the safety and effectiveness of the Intrector® for treating postoperative endophthalmitis.
Materials and methods: In a retrospective multicenter study, patients who received a single port 23-gauge core pars plana vitrectomy and isovolumetric injection of vancomycin, ceftazidime, and dexamethasone/amphotericin B using the Intrector® for postoperative endophthalmitis of intermediate severity (grade II or III vitreous inflammation and best-corrected visual acuity between hand movements and 0.3 logMAR [logarithm of the minimum angle of resolution]) were evaluated. Improvement in visual acuity, resolution of intraocular inflammation, the need for additional surgical procedures, and the development of complications were evaluated at a 1-month follow-up examination.
Results: Fifteen patients (mean age 55.6±7.2 years) underwent treatment with the Intrector®. The mean vitreous volume aspirated was 0.78±0.22 mL. The vitreous samples indicated positive microorganism culture results in six of the 15 cases, but the samples were positive when analyzed by real-time polymerase chain reaction in all cases (15/15). The mean best-corrected visual acuity improved significantly (P=0.01) from 0.88±0.29 (logMAR) to 0.32±0.28. Each patient demonstrated at least three lines of visual improvement. No additional medical or surgical interventions were required, and the complete resolution of intraocular inflammation was noted in all patients at the 1-month follow-up examination. No procedure-related complications were observed.
Conclusion: The Intrector® may be a safe and effective treatment option for infectious postoperative endophthalmitis. Larger studies comparing the outcomes of the Intrector® to the traditional treatments for postoperative endophthalmitis need to be conducted before its role in the treatment of postoperative endophthalmitis can be properly defined.
Keywords: Intrector®, postoperative endophthalmitis, 23-gauge vitrectomy
Introduction
The Endophthalmitis Vitrectomy Study (EVS) recommends either a vitreous biopsy with a concomitant injection of intravitreal antibiotics or a three-port pars plana vitrectomy (PPV) with intravitreal antibiotics for the treatment of acute postoperative infectious endophthalmitis following cataract surgery.1 PPV offers better outcomes in more severe cases (visual acuity worse than hand movements at 1 meter) compared with the tap and inject procedure. However, eyes with a pre-existing severely compromised view of the retina, adherent vitreous and fragile retina may also be more prone to other sight-threatening complications inherent to a PPV, such as developing retinal breaks and detachments. For less severe cases with visual acuity of hand movements at 1 meter or better, the tap and inject procedure often completely resolves the intraocular inflammation without the need for further intervention. Additionally, this procedure is less extensive and may be less complication-prone. Occasionally, patients receive either another tap and inject procedure or a PPV if they fail to respond to the initial treatment. The patients who fail to respond to the initial tap and inject procedure typically have very poor visual outcomes.2 One could attempt to identify and target the sub-group of patients that has intermediate severity endophthalmitis with a procedure that is more effective than tap and inject but less elaborate and complication-prone, such as PPV.
The Intrector® (Insight Instruments Inc., Stuart, FL, USA) is a 23-gauge single port, portable vitrector (Figure 1).3 The Intrector® cuts and aspirates vitreous in a quantity equal to that of the drug being infused, thereby enabling large quantities (>0.2 mL) of intravitreal therapeutic agents to be safely delivered without causing sight-threatening rises in intraocular pressure (IOP). This feature may be useful when multiple synergistic therapeutic agents must be delivered simultaneously without compromising their preferred dosages as in the treatment of infectious endophthalmitis, severe occult neovascular membranes,4 refractory vein occlusions, and diffuse diabetic macular edemas. Other reported uses for the Intrector® include pneumatic retinopexy, removal of uncomplicated vitreous media opacities, and vitreous aspiration for diagnostic purposes.3,5 This pilot study evaluates the safety and efficacy of vitreous biopsy, core vitrectomy, and intravitreal antibiotic injection using the Intrector® to determine whether the Intrector® could have a potential role in the treatment of intermediate severity postoperative endophthalmitis, possibly in an in-office setting.
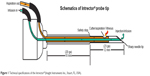  | Figure 1 Technical specifications of the Intrector® (Insight Instruments Inc., Stuart, FL, USA). |
Materials and methods
The leading institutional review board (University of Heidelberg) approved this retrospective, non-randomized study analysis of patients presenting in three different centers with intermediate severity postoperative endophthalmitis (defined in the inclusion criteria). Informed consent was obtained from each subject in accordance with the tenets of the Declaration of Helsinki.
Inclusion criteria
- Patients were diagnosed with infectious, postoperative endophthalmitis within less than 12 weeks following cataract surgery.
- The best-corrected visual acuity (BCVA) ranged between hand movements at 1 meter and 20/80 on the Snellen’s or 0.6 on the logMAR (logarithm of the minimum angle of resolution) chart.
- The vitreous inflammation was grade II or grade III according to the Standardization of Uveitis Nomenclature Working Group.6
Exclusion criteria
- Exudative retinal detachment as observed on B-scan ultrasonography.
- Age less than 18 years old.
- BCVA and vitreous inflammation that was unable to be properly graded because of an intense anterior segment inflammatory response.
- Presence of a relative afferent papillary defect.
- The patient had undergone previous vitreous biopsy or vitrectomy for any indication.
- The patient had a history of an ocular disease that could interfere with the BCVA or inflammatory grading such as uveitis or macular disease.
Presentation
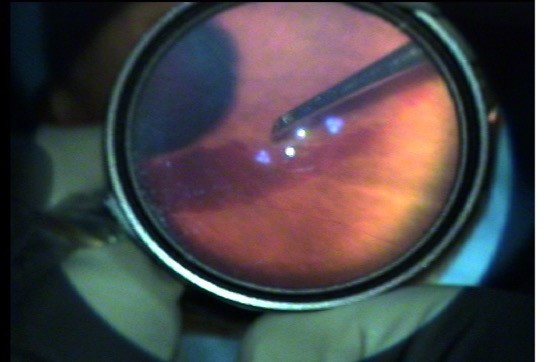
Study subjects underwent a comprehensive ophthalmic examination on initial presentation. The Snellen BCVA was converted to the logMAR values developed by Ferris et al.7 The logMAR values of BCVA of hand movements close to the face was assigned +3.0 logMAR, and the BCVA of hand movements less than 1 meter or counting fingers close to face was assigned +2.0 logMAR, according to the methods published by Holladay.8 IOP was measured by Goldmann applanation tonometry. Grading of the posterior segment inflammation was performed using the slit lamp and indirect ophthalmoscopy. B-scan ultrasonography was performed on all study eyes regardless of media clarity. At the completion of the examination, study patients were taken to the procedure room for treatment with the Intrector®.
Technique
Before surgery, a conjunctival smear with a cotton swab was performed. After applying 5% povidone-iodine to the periocular skin and the conjunctiva for 3 minutes, the patient was draped and a lid speculum was applied. An oblique bi-planar 23-gauge transconjunctival sclerotomy was made 3.5 mm from the superotemporal limbus. A core vitrectomy was performed at a constant rate of 360 cuts per minute, and a vitreous sample was aspirated and collected without dilution of the sample with balanced salt solution (BSS) or drugs. There were two independent cannulas within the probe for aspiration and infusion, which were controlled by an assistant next to the surgeon. The surgeon used one hand to hold the Intrector® and the other to visualize the tip in the vitreous cavity using the indirect ophthalmoscope with a hand-held +20 diopter lens (Figure 2). For all patients, the procedure consisted of three different sequences: 1) undiluted core vitreous sampling without BSS infusion. 2) A thorough vitrectomy in the core vitreous with isovolumic BSS infusion. 3) The infusion with antibiotics alone. Patients presenting with endophthalmitis within 6 weeks of cataract surgery had vancomycin (1 mg/0.1 mL), ceftazidime (2.25 mg/0.1 mL), and dexamethasone (0.4 mg/0.1 mL) infused intravitreally through the Intrector®. Patients presenting with endophthalmitis beyond 6 weeks had vancomycin (1 mg/0.1 mL), ceftazidime (2.25 mg/0.1 mL) and amphotericin B (5–10 μg/0.1 mL) infused intravitreally through the Intrector®. The Intrector® was then withdrawn, and gentle pressure was placed over the sclerotomy site with a cotton tip applicator. Vicryl suture 8-0 was available for any persistent sclerotomy wound leaks.
Post-Intrector® treatment follow-up
Patients were given topical antibiotic/steroid/cycloplegic medications and systemic oral ciprofloxacin (500 mg twice daily for 7 days) following treatment with the Intrector®. The vitreous sample was analyzed by smear, culture, and real-time polymerase chain reaction (PCR) for bacterial and fungal pathogens.9,10 PCR was performed in the local microbiology department using a real-time set-up to determine the infectious organism.
Patients were examined each day following treatment with the Intrector® until clinical improvement was readily apparent. The need for further intervention was based on the clinical appearance 48 hours following treatment with the Intrector®. BCVA, IOP, and intraocular inflammation were assessed during each follow-up examination.
Spectral domain optical coherence tomography (RTVue, Optovue Inc., CA, USA or Spectralis, Heidelberg Engineering, Germany) was performed on all subjects at the 1-month follow-up appointment.
Statistical analysis was performed applying the Wilcoxon matched pairs test for non-parametric before and after comparisons within one group (BiAS® Version 8.2 for Windows®; Epsilon-Verlag, Darmstadt, Deutschland).
Results
Fifteen study eyes underwent Intrector® treatment for postoperative (initial uneventful cataract surgery) endophthalmitis (eight females and seven males, 55.6±7.2 years). There were nine patients (six females, three males) who presented within 6 weeks of cataract surgery (median 12.4 days, range from 5 to 33 days), whereas six patients qualified for beyond 6 weeks after cataract surgery (median 43.2 days, range from 36 to 49 days). A total of 60% (9/15) of these patients had grade II vitritis, and 40% (6/15) had grade III vitritis.
All samples generated positive PCR results (100%; 15/15), which confirmed the earlier positive culture growth in six cases (40%; 6/15; Table 1).
  | Table 1 Microorganism isolation profile of endophthalmitis cases |
The mean volume of the collected vitreous samples was 0.78±0.22 (mean ± standard deviation [SD]) mL (range: 0.5–1 mL) before the assistant had to infuse the eye with BSS and antibiotics. The mean IOP was 14.4±5.4 (mean ± SD) mmHg (range: 8–21 mmHg) on the first post-procedure day. At 1 month, the BCVA improved significantly (P=0.01) from logMAR 0.88±0.29 (mean ± SD) to 0.32±0.28. All cases demonstrated improvement by at least three lines on the logMAR chart.
No cases of hypotony requiring sclerotomy suturing were noted in the post-Intrector® treatment period, and no significant procedure-related complications were observed at 1-month follow-up.
Discussion
The theoretical advantages offered by the Intrector® in the treatment of infectious endophthalmitis include increased microorganism yields for culture by higher volume collection of vitreous sample and improved visual outcomes with faster visual rehabilitation due to the more effective clearing of infective/inflammatory material compared with the vitreous tap and inject method, as observed in the vitreous tap group of the EVS.
The EVS demonstrated positive cultures in 69.3% of cases, whereas only 40% of the cases in this study had positive cultures.11
It is important to state, however, that our study included cases with intermediate severity endophthalmitis only, which has a lower culture positive rate by itself. Positive cultures ranged from 38% to 53.9% in other postoperative endophthalmitis studies.11,12 However, our study did show a 100% microorganism yield by PCR. A study by Varghese et al in 70 eyes with postoperative endophthalmitis gave a much better yield by PCR (49 eyes for bacteria and eleven eyes for fungi) compared with culture (35 eyes for bacteria and three eyes for fungi) with its analysis of samples from three-port PPVs.14 The volume of vitreous aspirate, obtained using our monoport aspiration technique ranged from 0.5 to 1 mL and takes approximately 3 to 10 minutes. This volume was sufficient for culture and PCR testing without the need for additional specimen dilution in all cases. Eighty percent of cases in our study had a BCVA of 20/40 or better at 1-month follow-up with a single procedure. The EVS reported a BCVA of 20/40 or better in 53% of cases at the 9-month follow-up. The number of cases included in our study and our follow-up rate limit the outcomes of this study. It is thus impossible to confirm or dismiss these theoretical advantages of the Intrector®, and our results can hardly be compared with the EVS findings.
Due to all of the surgical advantages of monoport vitrectomy, such as immediate care, short duration of the procedure and the mobility of therapy, the Intrector® does introduce the potential for intra-procedural (eg, loss of cataract parts during complicated cataract surgery) and posttreatment complications (like intermediate endophthalmitis in this study).3,15
Similar to retinal photocoagulation in retinopathy of prematurity (ROP) treatment, the different dexterity is challenging and presents a learning curve. In severe endophthalmitis treatment, retinal breaks may form during the core vitrectomy if vitreous is pulled along or into the instrument during its insertion or in the posttreatment period if vitreous adheres to the sclerotomy site. Iatrogenic retinal breaks caused during the core vitrectomy can be difficult to detect and need to be fixed using the Intrector® by combining this treatment with cryocoagulation. The concept of a monoport vitrectomy is not intended to replace a three-port PPV approach, which is doubtlessly the better option for treating severe immediate postoperative endophthalmitis associated with retinal detachments because it can provide better visualization and surgical control. Treatment with the Intrector® has the advantage of offering a portable, office-based core vitrectomy method for various indications. It is thus suitable for the clinical situation in which a three-port PPV is just too extensive. It is unknown whether the single sclerotomy site of the Intrector® reduces the risk of vitreous adherence to the wound or wound leakage compared with a three-port PPV in endophthalmitis treatment. None of these complications were observed in our study. Moreover, the single 23-gauge port with a bi-planar entry does not necessitate opening the conjunctiva or a suture for its closure in most cases. The intrectomy causes minor conjunctival or scleral trauma and leaves the tissues virgin enough for any further ocular intervention.
Conclusion
In conclusion, the Intrector® was found to be safe and effective in our small case series, and it may offer a viable alternative to the tap and inject procedure for post-cataract surgery endophthalmitis of intermediate severity in terms of microorganism yield, resolution of intraocular inflammation, and BCVA improvement with a single procedure at 1-month follow-up. A larger comparative study is needed to more specifically define the role and feasibility of the Intrector® in the treatment of postoperative endophthalmitis.
Acknowledgment
We acknowledge financial support by Deutsche Forschungsgemeinschaft and Ruprecht-Karls-Universität Heidelberg within the funding program Open Access Publishing.
Disclosure
MJ Koss and FH Koch receive reimbursement for travel expenses from Insight Instruments Inc., Stuart, FL, USA. The remaining authors have no conflicts of interest in this work.
References
No authors listed. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Endophthalmitis Vitrectomy Study Group. Arch Ophthalmol. 1995;113(12):1479–1496. | ||
Shaarawy A, Grand MG, Meredith TA, Ibanez HE. Persistent endophthalmitis after intravitreal antimicrobial therapy. Ophthalmology. 1995;102(3):382–387. | ||
Koch FH, Koss MJ. Microincision vitrectomy procedure using Intrector technology. Arch Ophthalmol. 2011;129(12):1599–1604. | ||
Koss MJ, Scholtz S, Haeusler-Sinangin Y, Singh P, Koch FH. Combined Intravitreal Pharmacosurgery in Patients with Occult Choroidal Neovascularization Secondary to Wet Age-Related Macular Degeneration. Ophthalmologica. 2009;224(2):72–78. | ||
Shukla S, Cohen A, Josephberg R. Nonfamilial Vitreous Amyloidosis diagnosed by portable sutureless vitrectomy. Retin Cases Brief Rep. 2008;2(4):264–265. | ||
Jabs DA, Nussenblatt RB, Rosenbaum JT; Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol. 2005;140(3):509–516. | ||
Ferris FL 3rd, Kassoff A, Bresnick GH, Bailey I. New visual acuity charts for clinical research. Am J Ophthalmol. 1982;94(1):91–96. | ||
Holladay JT. Proper method for calculating average visual acuity. J Refract Surg. 1997;13(4):388–391. | ||
Fenollar F, Lévy PY, Raoult D. Usefulness of broad-range PCR for diagnosis of osteoarticular infections. Curr Opin Rheumtol. 2008; 20(4):463–470. | ||
Choe H, Inaba Y, Kobayashi N, Aoki C, et al. Use of real-time polymerase chain reaction for the diagnosis of infection and differentiation between gram-positive and gram-negative septic arthritis in children. J Pediatr Orthop. 2013;33(3):e28–e33. | ||
Han DP, Wisniewski SR, Wilson LA, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol. 1996;122(1):1–17. | ||
Kratz A, Levy J, Belfair N, Weinstein O, Klemperer I, Lifshitz T. Broth Culture Yield vs Traditional Approach in the Work-up of Endophthalmitis. Am J Ophthalmol. 2006;141(6):1022–1026. | ||
Gupta A, Gupta V, Gupta A, et al. Spectrum and clinical profile of post cataract surgery endophthalmitis in North India. Indian J Ophthalmol. 2003;51(2):139–145. | ||
Varghese B, Rodrigues C, Deshmukh M, et al. Broad-range bacterial and fungal DNA amplification on vitreous humor from suspected endophthalmitis patients. Mol Diagn Ther. 2006;10(5):319–326. | ||
Hilton GF, Josephberg RG, Halperin LS, et al. Office-based sutureless transconjunctival pars plana vitrectomy. Retina. 2002;22(6):725–732. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.