Back to Journals » Clinical Ophthalmology » Volume 12
Comparative study of 0.1% hyaluronic acid versus 0.5% carboxymethylcellulose in patients with dry eye associated with moderate keratitis or keratoconjunctivitis
Authors Groß D, Childs M, Piaton JM
Received 4 January 2018
Accepted for publication 17 April 2018
Published 11 June 2018 Volume 2018:12 Pages 1081—1088
DOI https://doi.org/10.2147/OPTH.S161578
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Dorothea Groß,1 Marc Childs,2 Jean-Marie Piaton3
1Ursapharm Arzneimittel GmbH, Saarbrücken, Germany; 2Laboratoires Delbert, Paris, 3Ophthalmologic Practice, Domont, France
Background: Eye drops containing 0.1% hyaluronic acid (HA) and 0.5% carboxymethylcellulose (CMC) applied one drop three times a day per affected eye were compared in patients with moderate keratitis or keratoconjunctivitis related to dry eye disease (DED).
Patients and methods: This was a prospective, randomized, multicenter, Phase IIIB noninferiority study, with a single-masked phase in parallel mode with two groups over 84 days. The primary efficacy outcome was change in ocular surface (OS) staining between day 0 (D0) and day 35 (D35). The conjunctiva and cornea were stained with lissamine green and fluorescein. Secondary efficacy measures at day 84 (D84) were OS-staining score (SS), ocular comfort index, tear-film breakup time and how patients and investigators rated treatment efficacy and safety.
Results: At D35, 0.1% HA achieved a 46.6% reduction in OS-SS (-2.03±1.35 points, n=39 patients) and 0.5% CMC treatment, followed by a 34.9% reduction (-1.61±1.69 points, n=38 patients) compared to D0. At D84, the SS difference to D0 improved by -2.58±1.45 points (-59.2%) for 0.1% HA and -2.59±2.27 points (-54.4%) for 0.5% CMC. Ocular comfort-index scores improved, with significantly lower (better) values for stinging and itching on D84 for 0.1% HA. Patients assessed treatment with 0.1% HA as significantly better than 0.5% CMC (Likert scale, 4.82 vs 3.97; P=0.018). Four adverse events (AEs) occurred in four of 41 patients (9.8%) treated with 0.1% HA, and three AEs in two of 39 patients (5.1%) treated with 0.5% CMC. No serious AEs were noted.
Conclusion: DED signs and symptoms of DED significantly improved with both eye drops. OS staining improved >54% at D84. Treatment was well tolerated, with only minor AEs <10%. 0.1% HA and 0.5% CMC were equally safe and effective. Significant and nonsignificant results were constantly in favor of 0.1% HA.
Keywords: hyaluronate, carboxymethylcellulose, eye drops, fluorescein, lissamine green
Introduction
Disorders of the ocular surface (OS) are characteristic of dry eye disease (DED) or keratoconjunctivitis sicca. OS disorders affect the conjunctiva and the cornea, as well as the lid apparatus and the lacrimal drainage system.1 Additionally, a qualitative disturbance of the composition of tears, leading to instability in tear film, and a lack of tear volume with an increase in osmolarity of the tears can be observed.2
Currently, it is assumed that during the pathogenesis of dry eye (DE), instability in tear film and increase in osmolarity result in inflammation of the OS.3 Corneal and conjunctival structures can be damaged. This may disturb the integrity of the OS. The International Dry Eye Workshop (2017) formulated the definition of DE as a
multifactorial disease of the OS characterized by a loss of homeostasis of the tear film, accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, OS inflammation and damage, and neurosensory abnormalities play etiological roles.1
The goal of ophthalmological therapy with lubricating eye drops is to slow or stop the progression of DED. Successful DE therapy decreases signs and symptoms in DEs and prevents or delays further damage to the OS.4
Moisturizing therapy attempts to break through the vicious circle described to achieve qualitative and quantitative improvement in tear film. For ocular moisturizing therapy, several types of solutions are used. Eye drops usually contain chemically inactive molecules of high molecular weight that are bound at the OS and retain water molecules.4 A commonly used polymer in lubricating eye drops is sodium hyaluronate or hyaluronic acid (HA).5 HA is a glycosaminoglycan or mucopolysaccharide and has a molecular weight of 50,000–8,000,000 Da. It is a component of the vitreous body, and is also found in physiological tear fluid. For DE therapy, HA concentrations in eye drops range from 0.1% to 0.4%.6–11 Carboxymethylcellulose (CMC) is another frequently used macromolecule for topical DED therapy.12–14 CMC is a polymer composed of glucopyranose subunits. The molecule has an anionic charge and (similar to HA) water-retention and moistening properties.
Untreated DED may cause damage to the corneal and conjunctival epithelium.1,2 Cell defects of the cornea and conjunctiva can be detected and measured by staining, eg, with lissamine green or fluorescein.15 Fluorescein permeates into intercellular spaces and stains corneal lesions. Damaged or devitalized conjunctival cells are stained by lissamine green. Both dyes were used in this clinical study.
Based on previous findings comparing CMC and HA in DE treatment,16,17 the choice of 0.5% CMC as comparator for a 0.1% HA formulation seemed to be of scientific relevance. Staining scores (SSs) were used for the quantification of the clinical benefit of 0.1% HA (HYLO CONFORT; Ursapharm, Saarbrücken, Germany) and 0.5% CMC (OPTIVE; Allergan). The primary goal of this study was to test the noninferiority of efficacy of 0.1% HA in comparison to 0.5% CMC. Combined values of cornea and conjunctiva were used as the primary outcome parameter. Patients with moderate keratitis or keratoconjunctivitis were included in this study. Additionally, treatment-related changes in ocular comfort index (OCI) scores were assessed. Patients and investigators rated their treatment satisfaction. We used similar methods and the same design described in a recent study by Groß et al on HA 0.2% and 0.18%.18
Patients and methods
Participants
The intent-to-treat (ITT) population consisted of 80 patients with moderate DED and uninfectious, nonviral keratoconjunctivitis or keratitis. There were 39 patients in the 0.5% CMC group and 41 in the 0.1% HA group. Inclusion and exclusion criteria have been described in detail in Groß et al.18 The authors compared efficacy, safety, and tolerability of HA 0.2% and 0.18% eye drops in a study with a design similar to the present study. In brief, only patients >18 years of age with moderate DED, despite treatment with lubricating eye drops, were included. Patients’ overall SS for the OS was between ≥3–≤7 on the Oxford SS (OSS), ranging from 0 to 15 points. Patients’ Schirmer values without anesthesia had to be between ≥3 mm and ≤10 mm within 5 minutes and their mean tear-breakup time (TBUT) ≤10 seconds. Exclusion criteria were blepharitis, best-corrected visual acuity <1/10, and DE not related to keratitis or keratoconjunctivitis, as well as medical treatment forms with a potential influence on patients’ DED and hypersensitivity or intolerance to the eye drops tested.
Demographic data
The mean age of patients was 55.8±16.4 years. Of the 80 patients involved, 56 (70.7%) patients were female and 24 (29.3%) male. Both treatment groups were comparable concerning demographic data. Baseline demographic data for the ITT population are shown in Table 1.
  | Table 1 Demographic data at baseline (per protocol population) |
DE in patient history
Patients receiving the 0.1% HA study medication had a diagnosed history of DED for 4.6±4.1 years. Patients receiving 0.5% CMC had a history of DED for 5.4±4.3 years. Groups were not significantly different (P>0.32, Mann–Whitney).
Triggering factors
Triggering factors included cigarette smoking, screen work, air pollution, wind, contact lenses, and chronic medical treatment for anxiety, menopausal symptoms, insomnia, or Parkinson’s disease. Most patients reported one or two factors triggering their DE symptoms, which was comparable between the treatment groups.
Treatment before this study
Patients needed to have received treatment for DE in the previous months. Before this study, the majority of patients had had one treatment of DED with lubricating eye drops or ointments. Data are shown in Table 2.
  | Table 2 Number of lubricating eye drops or ointments before entry in this study |
Study design
This phase IIIB investigation was a single-blind multicenter study randomized with two patient groups run in parallel mode (French ANSM RCB-2015-A00729-40). The protocol followed recommendations of the International Conference on Harmonisation guidelines for Good Clinical Practice and the World Medical Association. All participants gave written informed consent. The study protocol and all other relevant documents were approved by the Comité de Protection des Personnes Ile-de-France VIII, Hôpital Ambroise Paré, Paris.
This study was performed in ten centers in France (average eight patients per center [one to 16]) and comprised two phases. Phase one went from day 0 (D0) to day 35 (D35) and compared the efficacy of the investigational eye drops on the main efficacy criteria. Phase two was a follow-up (D35–day 84 [D84]), and evaluated efficacy and safety of the two products. Treatment was masked throughout the study.
Prior and concomitant treatment
No other topical ophthalmic medications were allowed. Systemic medications were forbidden if they were hormonal treatments. Furthermore, isotretinoin and cyclosporine were not allowed, and neither were pimecrolimus, sirolimus, and tacrolimus.
Study medication
Investigational eye drops comprised a sterile, preservative-free solution for topical ophthalmic use (HYLO CONFORT; Ursapharm), containing 0.1% (1 mg/mL) HA, citrate buffer, sorbitol, and water in a 10 mL bottle. Comparator eye drops comprised a sterile solution for topical ophthalmic use (OPTIVE, Allergan), containing 0.5% (0.5 mg/mL) CMC, glycerol 0.9%, L-carnitine, erythritol, PURITE 0.1 mg (preservative), and water in a multidose bottle of 10 mL. Both products had to be applied three times daily in the affected eye(s).
Clinical investigations
Tests conducted in patients diagnosed with DED were OS on the 15-point OSS, OCI score, Schirmer test, and TBUT. When criteria for inclusion and exclusion were met, patients were recruited at visit 1 (baseline at D0). Follow-up consultation was at visit 2 on D35 (±5 days), where efficacy, safety, and any treatment modifications were evaluated. Patients filled in the symptom questionnaire (OCI), and ophthalmological tests were carried out. This was followed by a clinical interview checking safety issues and patient satisfaction with the treatment. Visit 3 on D84 (±10 days) reassessed symptom frequency and intensity (OCI), as well as ocular staining and the same ophthalmological tests as in visits 1 and 2. Investigators counted returned bottles to evaluate patient compliance at this final visit.
Investigator and patient evaluation
The 15-point scale for the OSS15 is a method of grading the amount of corneal damage and lesions at nasal or temporal conjunctival zones. Dyes were fluorescein for corneal staining and lissamine green for conjunctival staining. The amount of OS-lesion staining was determined. Subscores of the OSS were corneal, nasal, or temporal conjunctival zones on D35 and D84, each subscore ranging 0–5 points. For eyes with the highest scores at D0, we calculated the difference between D35 and D0.
Evolution at D0, D35, and D84 of DE symptoms (keratitis- and keratoconjunctivitis-related) was evaluated by the OCI questionnaire, taking into account the frequency and intensity of symptoms within the previous 7 days of stinging, eye dryness, fatigue, grittiness, pain in the eyes, and itching, with 0 = no symptom and 6 = maximum symptom intensity. TBUT was recorded as the number of seconds that elapsed between the last blink and the appearance of the first dry spot in the tear film. Patients’ satisfaction and efficacy of the topical DED treatment were evaluated by the investigator at D35 and D84 with the qualitative 7-point Likert scale.19
Statistical testing
The data from the most affected eye of each patient underwent statistical analysis. For significance testing, the probability threshold was set at 5% for two-sided tests and two-sided CIs. Statistical analyses were carried out with SAS version 9.4. The primary analysis used the per protocol population. This included all patients from the ITT population that had no major protocol violation. Generally, analyses were performed by treatment group and overall. For adverse events (AEs), only distribution by treatment group is presented. The primary efficacy outcome was change in OSS after 35 treatment days (D0–D35). A difference on the overall OSS of <2 points led to a conclusion of noninferiority of 0.1% HA versus 0.5% CMC. Secondary efficacy outcomes were OSS on D84, TBUT, OCI index with six items, each in frequency and intensity, and evaluation of treatment medications by patients and investigators.
Results
Primary efficacy outcome
Data represent the most affected eye of each individual patient. Staining of OS lesions at D0 (baseline) was 4.76 points for 0.5% CMC (n=38 patients) and 4.36 points for 0.1% HA (n=39 patients) on the OSS (15 points maximum). Figure 1 shows staining values on D0, D35, and D84. On D35, there was a 46.6% reduction in SS (−2±1.35 points, n=39 patients) for individuals treated with 0.1% HA eye drops and a 34.9% reduction (−1.6±1.7 points, n=38 patients) for individuals treated with 0.5% CMC eye drops.
  | Figure 1 Combined staining scores of cornea and conjunctiva (Oxford scale, mean ± SEM) at days 0, 35, and 84. |
The OSS difference between the two treatments on D35 was −0.4204 in favor of 0.1% HA (95% CI −1.1121 to 0.2714). As the upper limit of the 95% CI was below 2, it can be concluded that with respect to this primary efficacy end point, 0.1% HA was not inferior to 0.5% CMC (P<0.0001). At D84, both treatment groups showed improvement OSS. The difference between D84 and baseline (D0) was −2.58 points for 0.1% HA (59.2% decrease) and −2.59 points for 0.5% CMC (54.4% decrease). The difference in efficacy between the two medications observed in favor of 0.1% HA treatment at D35 (Figure 1) was less pronounced at D84.
Secondary efficacy outcomes
All symptoms of the OCI decreased (improved) for both treatments on D35 and D84. The improvement tended to be constantly higher for 0.1% HA treatment compared to 0.5% CMC treatment for each of the six OCI items. Figure 2 shows treatment effects for dryness, grittiness, and stinging. Fatigue, pain, and itching are shown in Figure 3.
  | Figure 3 OCI scores (mean ± SEM) for fatigue, pain, and itching. |
On D35, there was no statistically significant difference in OCI scores between treatments. On D84, OCI items had reduced with 0.1% HA between 44.7% and 51.2% and with 0.5% CMC between 10.2% and 36.4% compared to D0. There was no statistically significant difference between treatments, except for stinging on D84 (P=0.0106) in favor of 0.1% HA and itching on D84 (P=0.0178), also in favor of 0.1% HA.
TBUT values for both treatment groups are listed in Table 3. At D0, patients in the 0.1% HA group had a mean TBUT of 6.8±2.4 seconds, while in the 0.5% CMC group mean TBUT was 7.2±2.4 seconds (not a significant difference). Both treatments slightly improved TBUT values. At D84, the increase in TBUT was 1.5±1.9 seconds in the 0.1% HA group and 1.4±2.1 seconds in the 0.5% CMC group compared to D0. A secondary analysis was carried out using OCI primary end-point data of the ITT population at D35, and confirmed the noninferiority of 0.1% HA vs 0.5% CMC. No other secondary end points showed any statistically significant differences between the two groups.
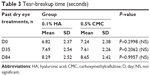  | Table 3 Tear-breakup time (seconds) |
AEs
A total of seven AEs were reported in six patients during the study: four AEs in four of 41 patients (9.7%) in the 0.1% HA group (chronic arthrosis, episodic crackling feeling under the lid, episodic chalazion, chronic eye stinging) and three AEs in two of 39 patients (5.1%) in the 0.5% CMC group (episodic lid edema and episodic eye in one patient stinging, chronic lid eczema in one patient). All AEs resolved without sequelae, except one mentioned as not resolved in the 0.1% HA group (chronic arthrosis).
Two patients discontinued due to AEs before D35 in the 0.1% HA group (chronic arthrosis, chronic eye stinging), and one patient discontinued due to an AE before D84 in the 0.5% CMC group (chronic lid eczema). No serious ocular AE was reported in this study. The pattern of observed AEs was consistent with the known safety profile of both investigational products. There was no statistical significant difference between the groups.
Evaluation of patients and investigators
Evaluation of patients and investigators was assessed on D35 and D84. More patients expressed their satisfaction with 0.1% HA than with 0.5% CMC at D35. Satisfaction levels were 4.82±1.45 for 0.1% HA and 3.97±1.57 for 0.5% CMC. The difference was significant – P<0.018. At D84, there was still a tendency of more favorable evaluation of 0.1% HA, which was not significant (P>0.05). Investigators’ scores for efficacy of the study medications were not significantly different between D35 and D84. See Table 4 for details.
Summary of results
The primary outcome in this study was staining of the cornea and conjunctiva. On D35, staining was reduced by 47.7% in the 0.1% HA treatment group and by 41.2% in patients treated with 0.5% CMC. These values continued to decrease between D35 and D84. By the end of our study, on D84, the 0.1% HA group showed a decrease in staining by 64.5% and the 0.5% CMC group showed a reduction in staining by 56.4% in comparison to baseline values. There was a tendency for less staining and better improvement with 0.1% HA eye drops, with no statistical difference between 0.1% HA and 0.5% CMC. Statistical analysis showed noninferiority of 0.1% HA in comparison to 0.5% CMC. More patients expressed their satisfaction with 0.1% HA than with 0.5% CMC at D35. At D84, there was still a tendency of more favorable patient evaluation of 0.1% HA. Investigators’ scores for treatment efficacy were high for both drugs and not significantly different throughout the study.
Discussion
This study compared the efficacy and safety of 0.1% HA and 0.5% CMC eye drops on clinical signs and symptoms in patients with moderate DED. OSSs for staining of cornea and conjunctiva were significantly improved after treatment in both groups, as well as the six-item OCI and TBUT. The increase in TBUT corresponds well with the data reported in Doughty’s meta-analysis on tear substitutes, where the treatment medications analyzed increased TBUT by 1.4 seconds.20
Decreased extent and severity of lesions of the OS may be viewed as important clinical markers for success in the treatment of DE symptoms.21 Both CMC and HA are used as ingredients in eye drops. HA has excellent moisturizing and mucus-layer adhesive properties and is found in the physiological tear fluid. CMC is an anionic cellulose derivate, provides excellent bioadhesive characteristics, and increases tear-retention time. HA and CMC are available in several viscosity grades, reflecting various concentrations and molecular weights. In this study, 0.1% HA was unpreserved, while 0.5% CMC was preserved with very low toxic PURITE 0.01 mg/mL in a multidose bottle.
Our data show that OS improved with both treatments. This finding is in agreement with other clinical studies on topical DE treatment. To our knowledge, Shimmura et al10 were the first investigators to use two dyes for OS staining. They used rose bengal and fluorescein as markers for treatment effects of HA eye drops. In their study, patients received unpreserved 0.1% HA six times daily for 4 weeks. It was found that staining of the cornea with fluorescein decreased; staining with rose bengal, however, did not change during the study.10 Brjesky et al described clinical outcomes of 0.15% HA treatment. The investigators noted positive results of HA 0.15% treatment on TBUT and reported a decrease in staining values. However, information on the dye used was lacking.22 Clinical research investigating the efficacy of CMC in the treatment of DE has also demonstrated significant beneficial effects: 0.5% CMC improved clinical parameters in mild and moderate forms of DED compared to balanced salt solution.12
Several studies have directly compared CMC and HA in DED treatment. Brignole et al investigated HA and CMC eye drops in the treatment of moderate DE syndrome with superficial keratitis solution for 2 months.23 Both treatments improved OS symptoms and condition. Comfort was significantly higher (P<0.05) in the HA group throughout the study when DED patients were treated either with 0.18% HA or 1% CMC. In the present intervention, HA and CMC concentrations were approximately 50% lower than in Brignole et al. In further clinical investigations performed in similar populations, 0.5% CMC was shown to be noninferior to 0.18% HA and 0.1% HA, in relieving symptoms and staining of DEs in 8216 and 65 patients, respectively.17
In a recent review, Song et al carried out a meta-analysis of five original studies comparing CMC and HA eye drops.13 The authors concluded that CMC was more efficacious than HA in treating DED. These conclusions, however, are obviously limited, since they were based on nonsignificant results and only TBUT test results were analyzed. In our study, no significant differences in TBUT were noted between 0.5% CMC and 0.1% HA treatment. HA molecular weight ranges between 50,000 and 8,000,000 Da. As such, evaluation of clinical results with HA based solely on different concentrations is difficult if no information on molecular weight is available.24 For 0.1% HA as tested in this clinical trial, Simmons et al reported a molecular weight of 2.026 million Da.25 Moreover, CMC is also pharmaceutically used with considerably varying molecular weights.26 This could explain why in contrast to the aforementioned review of Song et al,13 we found a trend for consistently higher improvements for 0.1% HA in comparison to 0.5% CMC in the present study.
Brignole et al concluded that 0.18% HA treatment tended to reduce DE symptoms and alleviate keratitis more quickly than 1% CMC.23 Our data show tendencies in the same direction. Our safety data confirm the safety profile of the two tested eyedrop types. AEs were judged to be related to the underlying DED. The proportion of AEs, including treatment discontinuation, associated with AEs was 9.8% for 0.1% HA and 5.1% for 0.5% CMC. There were no reports on serious AEs throughout the study. We found that the two tested eyedrop types led to clinically and statistically relevant improvements in SSs and OCI values. For several parameters, there was a constantly favorable trend for 0.1% HA in comparison to 0.5% CMC. Our data suggest that 0.1% HA and 0.5% CMC are efficacious in the treatment of patients presenting moderate DE syndrome with keratitis or keratoconjunctivitis. Two of six OCI items (stinging and itching) showed significantly more favorable results for 0.1% HA on D84.
Conclusion
Treatment of DED with 0.1% HA or 0.5% CMC eye drops is efficacious and safe in patients with moderate DED, with a more favorable trend for 0.1% HA eye drops.
Acknowledgments
Ursapharm funded this clinical study. The authors wish to thank Klaus Rudolph of Rudolph Consulting (Freiburg, Germany) for preparing the manuscript and for editorial support, funded by Ursapharm. The authors thank the French study centers involved in this study.
Disclosure
DG is an employee of Ursapharm. MC is the CEO of Laboratoires Delbert. He was appointed as CRO for this clinical study by Ursapharm. JMP reports no conflicts of interest in this work.
References
Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II definition and classification report. Ocul Surf. 2017;15(3):276–283. | ||
[No authors listed]. Management and therapy of dry eye disease: report of the Management and Therapy Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):163–178. | ||
Steven P, Cursiefen C. Antientzündliche Therapie beim Trockenen Auge. [Anti-inflammatory treatment in dry eye disease]. Klin Monbl Augenheilkd. 2012;229(5):500–505. German. | ||
Nebbioso M, Fameli V, Gharbiya M, Sacchetti M, Zicari AM, Lambiase A. Investigational drugs in dry eye disease. Expert Opin Investig Drugs. 2016;25(12):1437–1446. | ||
Klingenberg HO [webpage on the Internet]. Excipient update: a real eye-opener: advances in hyaluronic acid for ophthalmology. 2013. Available from: http://www.drug-dev.com/Main/Back-Issues/EXCIPIENT-UPDATE-A-Real-EyeOpener-Advances-in-Hyal-545.aspx. Accessed April 22, 2018. | ||
Liu XM, Harmon PS, Maziarz EP, Rah MJ, Merchea MM. Comparative studies of hyaluronan in marketed ophthalmic products. Optom Vis Sci. 2014;91(1):32–38. | ||
Lopez-de la Rosa A, Pinto-Fraga J, Arauzo FB, Rodriguez RU, Gonzalez-Garcia MJ. Safety and efficacy of an artificial tear containing 0.3% hyaluronic acid in the management of moderate-to-severe dry eye disease. Eye Contact Lens. 2017;43(6):383–388. | ||
Orsoni JG, Chiari M, Guazzi A, de Carli M, Guidolin D. [Efficacy of hyaluronic acid eyedrops in the treatment of dry eye: cytologic study using an optical microscope and computerized microscope]. Ophtalmologie. 1988;2(4):355–357. | ||
Pinto-Fraga J, Lopez-de la Rosa A, Arauzo FB, Rodriguez RU, Gonzalez-Garcia MJ. Efficacy and safety of 0.2% hyaluronic acid in the management of dry eye disease. Eye Contact Lens. 2017;43(1):57–63. | ||
Shimmura S, Ono M, Shinozaki K, et al. Sodium hyaluronate eyedrops in the treatment of dry eyes. Br J Ophthalmol. 1995;79(11):1007–1011. | ||
Troiano P, Monaco G. Effect of hypotonic 0.4% hyaluronic acid drops in dry eye patients: a cross-over study. Cornea. 2008;27(10):1126–1130. | ||
Bruix A, Adan A, Casaroli-Marano RP. [Efficacy of sodium carboxymethylcellulose in the treatment of dry eye syndrome]. Arch Soc Esp Oftalmol. 2006;81(2):85–92. | ||
Song JK, Lee K, Park HY, et al. Efficacy of carboxymethylcellulose and hyaluronate in dry eye disease: a systematic review and meta-analysis. Korean J Fam Med. 2017;38(1):2–7. | ||
Yao K, Bao Y, Ye J, et al. Efficacy of 1% carboxymethylcellulose sodium for treating dry eye after phacoemulsification: results from a multicenter, open-label, randomized, controlled study. BMC Ophthalmol. 2015;15:28. | ||
Bron AJ, Evans VE, Smith JA. Grading of corneal and conjunctival staining in the context of other dry eye tests. Cornea. 2003;22(7):640–650. | ||
Baudouin C, Cochener B, Pisella PJ, et al. Randomized, phase III study comparing osmoprotective carboxymethylcellulose with sodium hyaluronate in dry eye disease. Eur J Ophthalmol. 2012;22(5):751–761. | ||
Lee JH, Ahn HS, Kim EK, Kim TI. Efficacy of sodium hyaluronate and carboxymethylcellulose in treating mild to moderate dry eye disease. Cornea. 2011;30(2):175–179. | ||
Groß D, Childs M, Piaton JM. Comparison of 0.2% and 0.18% hyaluronate eye drops in patients with moderate to severe dry eye with keratitis or keratoconjunctivitis. Clin Ophthalmol. 2017;11:631–638. | ||
McLeod S [webpage on the Internet]. Likert scale. 2008. Available from: www.simplypsychology.org/likert-scale.html. Accessed September 29, 2016. | ||
Doughty MJ. Fluorescein-tear breakup time as an assessment of efficacy of tear replacement therapy in dry eye patients: a systematic review and meta-analysis. Ocul Surf. 2014;12(2):100–111. | ||
Bron AJ, Tomlinson A, Foulks GN, et al. Rethinking dry eye disease: a perspective on clinical implications. Ocul Surf. 2014;12(2 Suppl):S1–S31. | ||
Brjesky VV, Maychuk YF, Petrayevsky AV, Nagorsky PG. Use of preservative-free hyaluronic acid (Hylabak) for a range of patients with dry eye syndrome: experience in Russia. Clin Ophthalmol. 2014;8:1169–1177. | ||
Brignole F, Pisella PJ, Dupas B, Baeyens V, Baudouin C. Efficacy and safety of 0.18% sodium hyaluronate in patients with moderate dry eye syndrome and superficial keratitis. Graefes Arch Clin Exp Ophthalmol. 2005;243(6):531–538. | ||
Teping C. Hyaluronsäure. Thieme Drug Rep. 2010;2(3):1–12. | ||
Simmons PA, Aragona P, Wang HP, Wang T. Polymer size and other physical properties vary widely among hyaluronic acid-based lubricant eye drops. Poster presented at: Association for Research in Vision and Ophthalmology 2017 Annual Meeting; May 7–11, 2017; Baltimore, MD. | ||
Huan Y, Zhang S, Vardhanabhuti B. Influence of the molecular weight of carboxymethylcellulose on properties and stability of whey protein-stabilized oil-in-water emulsions. J Dairy Sci. 2016;99(5):3305–3315. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


