Back to Journals » Clinical and Experimental Gastroenterology » Volume 12
Cholestasis In Infants With Down Syndrome Is Not Due To Extrahepatic Biliary Atresia: A Ten-Year Single Egyptian Centre Experience
Authors Kotb MA, Draz I, Basanti CWS , El Sorogy STM, Abd Elkader HM , Esmat H , Abd El Baky H, Mosallam DS
Received 17 May 2019
Accepted for publication 3 September 2019
Published 22 October 2019 Volume 2019:12 Pages 401—408
DOI https://doi.org/10.2147/CEG.S216189
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Everson L.A. Artifon
Magd A Kotb,1 Iman Draz,1 Christine WS Basanti,1 Sally TM El Sorogy,2 Hesham M Abd Elkader,3 Haytham Esmat,4 Hend Abd El Baky,1 Dalia Sayed Mosallam1
1Pediatrics Department, Faculty of Medicine, Kasr Al Ainy, Cairo University, Cairo, Egypt; 2Pediatrics Department, Public Mounira Hospital, Cairo, Egypt; 3Department of Pediatric Surgery, Faculty of Medicine, Ain Shams University, Cairo, Egypt; 4Department of Pediatric Surgery, Cairo University, Cairo, Egypt
Correspondence: Dalia Sayed Mosallam
Pediatrics Department, Faculty of Medicine, Cairo University, Al Manial, Cairo 11562, Egypt
Tel +20 2 33386592
Email [email protected]
Purpose: We aimed to define the clinical presentations, course and outcome of cholestasis in infants with Down syndrome (trisomy 21) who presented to the Pediatric Hepatology Clinic, New Children Hospital, Cairo University, Egypt.
Methods: Retrospective analysis of data of cohort of infants with Down syndrome and cholestasis who followed up during 2005–2015.
Results: Among 779 infants with cholestasis who presented during 2005–2015, 61 (7.8%) had Down syndrome. Six dropped out. Among the 55 who followed-up for a mean duration +SD = 12.1 ± 16.7 months, none had extrahepatic biliary atresia (EHBA), 37 (63.3%) had neonatal hepatitis and 18 (32.7%) had non-syndromic paucity of intrahepatic biliary radicals. Fourteen (25.4%) had associated congenital heart disease. Only 35 (63.3%) cleared the jaundice. Twenty-nine (52.7%) received ursodeoxycholic acid (UDCA); of them, 13 cleared the jaundice, one improved, 14 progressed and one died, compared to 22 who cleared the jaundice of the 26 who did not receive UDCA. Only three of those who did not receive UDCA progressed and none died. UDCA carried a 3.4-fold risk of poor prognosis (p= 0.001). UDCA use was associated with more complications (p= 0.016) in those with Down syndrome and cholestasis.
Conclusion: We did not come across EHBA among neonates and infants with Down syndrome in 10 years. Non-syndromic paucity is associated with favorable outcome in infants with Down syndrome. UDCA use in cholestasis with Down syndrome is associated with poor outcome.
Keywords: cholestasis, extrahepatic biliary atresia, EHBA, neonatal hepatitis, Down syndrome, trisomy 21, ursodeoxycholic acid, UDCA
Introduction
Down syndrome (trisomy 21) is associated with congenital anomalies in 64% of cases. The cardiac anomalies are commonest, followed by digestive system, musculoskeletal system, urinary system, respiratory and other system anomalies.1 Estimated worldwide incidence of Down syndrome is 1:1,000–1:1,100, 0.827:1000 in USA2 and 1.8:1000–1.6:1000 in Egypt.3 Cholestasis was reported to affect 3.9% of neonates and infants with Down syndrome in a population-based study.4 The cholestasis in Down syndrome was reported to be due to the probability of a smaller circulating bile acid pool size, a lower rate of synthesis, reduced recirculation of bile acids and immature function of the canalicular bile acid transporting system.5 The increased susceptibility of cholestasis in Down syndrome was not mapped to chromosome 21, i.e. major genes controlling the uptake, synthesis, or secretion of bile acids in human hepatocytes, synthesis, or ileal enterocytes.6 The aim of this study was to define the spectrum of clinical presentations, course and outcome of cholestasis in infants with Down syndrome.
Subjects And Methods
Subjects
This is an observational study that included a retrospective analysis of data of a cohort of infants with Down syndrome and cholestasis who followed up during 2005–2015 at the Pediatric Hepatology Clinic, New Children Hospital, Cairo University, Egypt. The study was approved by the Pediatric Department Committee for Post-Graduate Studies and Research, and by the Post-Graduate Studies and Research Administration, Faculty of Medicine, Cairo University, Egypt. Parental approval was not applicable to this retrospective, observational, non-interventional cohort study.7 The study complies with the Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects.8
Methods
We revised all files of neonates and infants who presented with cholestasis and clinical features of Down syndrome (trisomy 21) during 2005–2015. We analyzed all data of recruited children, including the history of age at onset of symptoms, age at presentation, presenting symptom, complications and/or associations of liver disease, neurologic disease, age of the patient at the time of the study, weight and height percentiles, and outcome. Anthropometric measures were plotted against Egyptian percentiles for children with Down syndrome weight and height3 and recorded as percentiles for age.
Etiology of cholestasis was studied according to clinical judgment, i.e., virology, bile acids, metabolic screen, imaging and liver biopsy.
The outcome was graded into resolved, improved, stationary, progressive and death. The resolved outcome was when the cholestasis resolved without sequelae; improved, with an improvement of cholestasis but did not resolve completely; stationary was coined to those who did not improve or deteriorate; while progressive was coined to those where cholestasis increased.
Statistical Analysis
All the statistical analyses in this study were conducted using the Statistical Package for Social Sciences version 19 (SPSS, Chicago, IL). Simple frequency, cross-tabulation, descriptive analysis, tests of significance (t-test for parametric data and χ2 tests for non-parametric numbers N5), and correlations were employed.
Results
During 2005–2015 only 61 infants with Down syndrome presented to the Pediatric Hepatology Clinic, Cairo University. Six (9.2%) dropped out and did not show up for a second visit; they were all females (Figure 1). The other 55 were followed up for 12.1 ± 16.7 months. Of them, 28 (51%) were females and 27 (49%) males. Mean ± SD age at onset of cholestasis was 1.23 ± 11.78 months, and at presentation to our medical attention was 2.1 ± 9.2 months. Seventeen (30.1%) were the product of a consanguineous marriage. Fifteen (27.3%) had a history of another family member affected by cholestasis. The symptoms, signs of the studied cohort, their serum bilirubin and liver enzymes are shown in Tables 1 and 2, respectively. Liver biopsy was performed in 32 (58.2%) subjects and the findings are shown in Table 2.
 |
Table 1 Clinical Findings In Down Syndrome Cohort With Cholestasis |
 |
Table 2 Laboratory And Liver Biopsy Findings In Down Syndrome Cohort With Cholestasis |
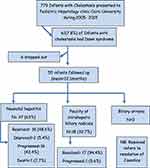 |
Figure 1 Flowchart of studied cohort of neonates and infants with Down syndrome and cholestasis. |
Associated congenital cardiac anomalies were encountered in 14 (25.4%) children (Table 1). No other anomalies were encountered in our studied cohort. None had any bone marrow-associated disease.
Etiology and outcome of cholestasis in the studied cohort of neonates and infants with down syndrome are shown in Figures 2 and 3, respectively.
None of the affected infants had EHBA, 37 (67.3%) had neonatal hepatitis and 18 (32.7%) suffered from non-syndromic paucity of intrahepatic biliary radicals. Cholestasis resolved in 35 (63.6%). None tested positive for viral screening known to cause neonatal hepatitis or metabolic workup and none had progressive familial intrahepatic cholestasis (Figure 2). One child had massive fibrosis, with unidentified underlying pathology, suggesting congenital hepatic fibrosis. Figure 1 depicts the diagnosis and outcome of the studied cohort. They all received fat-soluble vitamin supplements and 29 (52.72%) received UDCA also. Of them, 21 (72.4%) suffered from complications. Both those who received UDCA and those who did not were matched as regards severity of cholestasis (p= 0.17). UDCA use was associated with poorer outcome (p= 0.000) and complications (p= 0.016) (Table 3). UDCA in neonates with Down syndrome and cholestasis carried a 3.4-fold risk of poor prognosis (p= 0.001) (95% confidence interval) (Figure 3).
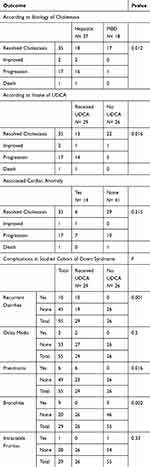 |
Table 3 Outcome And Associated Complications Of The Cohort With Down Syndrome And Cholestasis According To Etiology, UDCA Intake And Association Of Congenital Heart Disease |
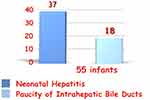 |
Figure 2 Etiology of Cholestasis in Down syndrome. None had biliary atresia. |
 |
Figure 3 The outcome of Cholestasis in Down syndrome. UDCA use was associated with poor outcome (p= 0.000). |
Discussion
During 2005–2015, 7.8% of the neonates and infants who presented with cholestasis to the Pediatric Hepatology Clinic, Faculty of Medicine, Cairo University had the Down syndrome (trisomy 21) phenotype. Clinically, cholestasis was mostly without organomegaly, where only 36.5% and 40% had hepatomegaly and splenomegaly, respectively. The outcome was generally favorable unless UDCA was given.
Congenital Heart Disease Was Encountered In Only 25% Of Cases And None Had Cyanotic Heart Disease
Down syndrome is associated with congenital heart disease (CHD) in 40–60%,9–11 with a dramatic increase from about 20% in the early 1970s to more than 50% in the late 1980s (p = 0.0001) in certain areas.12 In Egypt, studies of the prevalence of CHD in Down syndrome are limited, but the reported range was almost 40%.13,14 It is not clear why our cohort had less CHD compared to other populations of Down syndrome. More studies are required to establish or refute a protective effect of placental metabolism of environmental factors that are responsible for the development of CHD in a developing fetus with Down syndrome and/or cholestasis. The sample size is small, however, to draw sound conclusions, yet it remains an observation that CHD is less prevalent among Down syndrome with cholestasis, and the CHD spectrum did not include cyanotic heart disease.
We Did Not Come Across A Single Case Of EHBA In Down Syndrome During The 10 Years 2005–2015
This comes in congruence with previous literature, as we failed to find any previous reports of Down syndrome associated with EHBA. Kotb recently defined EHBA as aflatoxin-induced cholangiopathy in neonates with GST M1 null deficiency, while damage to bile ducts was mediated through neutrophil elastase. Damage control of the aflatoxin-induced cholangiopathy through neutrophil elastase ends in fibrosis and obliteration of extrahepatic bile ducts.15 Factors involved in the etiology of EHBA were not sought in this cohort, i.e., aflatoxins, glutathione S transferase M1, p53 and neutrophil functions. It seems that Down syndrome protects against the development of biliary atresia. This protective role might be due to compromised neutrophil function in Down syndrome,16,17 which might arrest the inflammatory process of EHBA.
Paucity Of Intrahepatic Biliary Radicals Has Excellent Prognosis In Down Syndrome
Generally, paucity of intrahepatic biliary radicals has a 69% chance of clearance of cholestasis in absence of UDCA intake,18 yet 94.4% of our cohort of neonates and infants with Down syndrome with paucity of intrahepatic biliary radicals cleared the cholestasis. Again, it seems that Down syndrome enhances clearance of cholestasis. This effect could be attributed to the compromised immunity in Down syndrome; this compromise will not mount massive destructive effect in the cholestasis inflammatory process.19
We Could Not Identify The Etiology Of Neonatal Hepatitis In Down Syndrome
The etiology of cholestasis of all of our studied neonates and infants with Down syndrome was idiopathic hepatitis, despite undergoing the battery of investigations to identify etiology (metabolic, congenital and infectious) when appropriate according to clinical situation.18 The etiology of neonatal cholestasis in our studied cohort remained idiopathic, with no overlap in etiology. We did not come across any cases of cystic fibrosis, infections, or galactosemia or any other etiology in our cohort with Down syndrome.
UDCA Is Not Effective And Is Not Safe In Cholestasis In Down Syndrome
UDCA was found to be ineffective in clearing cholestasis in neonates and infants with Down syndrome, and its use was associated with significantly worse outcome. UDCA compromised the outcome of those with Down and cholestasis – only 44.8% of those who received UDCA resolved the cholestasis, compared to 84.6% of those who did not receive UDCA. UDCA generally impedes resolution of cholestasis in neonatal hepatitis compared to no UDCA (44.8% compared to 70.2%, respectively).18 The discouraging effect of UDCA is exaggerated in our studied cohort. It is not clear why UDCA is more toxic in neonates with Down syndrome and cholestasis. The UDCA toxicity in cholestasis in Down syndrome might be attributed to their compromised detoxification of medications, e.g., methotrexate, glucocorticoids, anthracyclines, etc.20 It might be related to trisomy 21 type karyotyping or other genetic makeup that needs further investigation.
Conclusion
Cholestasis complicates Down syndrome. We did not come across EHBA among our studied cohort in 10 years. Down syndrome seems to protect against the development of EHBA. Use of UDCA in cholestasis associated with Down syndrome compromises resolution of cholestasis and its use is associated with poor prognosis. UDCA use in cholestasis associated with Down syndrome should be contraindicated.
Abbreviations
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CHD, congenital heart disease; D.Bil, direct bilirubin; EHBA, extrahepatic biliary atresia; SD, standard deviation; T.Bil, total bilirubin; UDCA, ursodeoxycholic acid.
Compliance With Ethical Statements
This article does not contain any studies with human participants or animals performed by any of the authors. It is a retrospective study including all files of neonates and infants who presented with cholestasis and clinical features of Down syndrome (trisomy 21) during 2005–2015. The study was approved by The Pediatric Department Committee for Post-Graduate Studies and Research, and by Post-Graduate Studies and Research Administration, Faculty of Medicine, Cairo University, Egypt.
What Is Known?
- Exclusion of surgical causes of cholestasis is invasive, yet it is part of workup in every neonate with cholestasis as surgical portoenterostomy should not be delayed beyond 3 months of age to halt the march of biliary cirrhosis.
- Cholestasis associated with trisomy 21 (Down syndrome) has been reported previously.
What Is New?
- Cholestasis in Down syndrome was never found to be due to biliary atresia; we did not come across a single obstructive cholestasis in Down syndrome in 10 years of practice.
- Generally, the outcome of cholestasis is favorable in Down syndrome, especially if the etiology of cholestasis is non-syndromic paucity, unless they receive ursodeoxycholic acid, as its' use is associated with poor outcome, complications and fatality.
Acknowledgments
We acknowledge all Pediatric Hepatology Team Members and archiving secretaries of the Pediatric Hepatology Clinic, New Children Hospital, Cairo University Hospital, Cairo University. There was no funding source for this study. The trial was approved by the Pediatric Department Committee for Post-Graduate Studies and Research, and by Post-Graduate Studies and Research Administration, Faculty of Medicine, Cairo University, Cairo, Egypt. The corresponding author had full access to all of the data and takes full responsibility for the veracity of the data and statistical analysis.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Stoll C, Dott B, Alembik Y, Roth MP. Associated congenital anomalies among cases with Down syndrome. Eur J Med Genet. 2015;58:674–680. doi:10.1016/j.ejmg.2015.11.003
2. Presson AP, Partyka G, Jensen KM, et al. Current estimate of Down Syndrome population prevalence in the United States. J Pediatr. 2013;163(4):1163–1168. doi:10.1016/j.jpeds.2013.06.013
3. Afifi HH, Aglan MS, Zaki ME, Thomas MM, Tosson AM. Growth charts of Down syndrome in Egypt: a study of 434 children 0–36 months of age. Am J Med Genet A. 2012;158A(11):2647–2655. doi:10.1002/ajmg.a.35468
4. Arnell H, Fischler B. Population-based study of incidence and clinical outcome of neonatal cholestasis in patients with Down syndrome. J Pediatr. 2012;161:899–902. doi:10.1016/j.jpeds.2012.04.037
5. Suchy FJ, Balistreri WF, Heubi JE, Searcy JE, Levin RS. Physiologic cholestasis: elevation of the primary serum bile acid concentrations in normal infants. Gastroenterology. 1981;80:1037–1041. doi:10.1016/0016-5085(81)90078-0
6. Halilbasic E, Claudel T, Trauner M. Bile acid transporters and regulatory nuclear receptors in the liver and beyond. J Hepatol. 2013;58:155–168. doi:10.1016/j.jhep.2012.08.002
7. WHO International Standards for Clinical Trial Registries. ISBN 978 92 4 150429 4 (NLM classification: QV 771.4); 2012. Available from: https://apps.who.int/iris/bitstream/handle/10665/76705/9789241504294_eng.pdf;jsessionid=2DA15341BDC67DFDEDE8FC1E1F8B93D5?sequence=1.
8. WMA DECLARATION OF HELSINKI. WMA declaration of Helsinki - ethical principles for medical research involving human subjects, 64th. WMA general assembly; 2013. Available from https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/.
9. Elmagrpy Z, Rayani A, Shah A, Habas E, Aburawi EH. Down syndrome and congenital heart disease: why the regional difference as observed in the Libyan experience? Cardiovasc J Afr. 2011;22:306–309. doi:10.5830/CVJA-2010-072
10. Kim MA, Lee YS, Yee NH, Choi JS, Choi JY, Seo K. Prevalence of congenital heart defects associated with down syndrome in Korea. J Korean Med Sci. 2014;29:1544–1549. doi:10.3346/jkms.2014.29.11.1544
11. Morsy MM, Algrigri OO, Salem SS, Abosedera MM, Abutaleb AR, Al-Harbi KM. The spectrum of congenital heart diseases in down syndrome: a retrospective study from Northwest Saudi Arabia. Saudi Med J. 2016;37:767–772. doi:10.15537/smj.2016.7.14536
12. Khoury MJ, Erickson JD. Improved ascertainment of cardiovascular malformations in infants with Down’s syndrome, Atlanta, 1968 through 1989. Implications for the interpretation of increasing rates of cardiovascular malformations in surveillance systems. Am J Epidemiol. 1992;136:1457–1464. doi:10.1093/oxfordjournals.aje.a116466
13. Afifi HH, Abdel Azeem AA, El-Bassyouni HT, Gheith ME, Rizk A, Bateman JB. Distinct ocular expression in infants and children with down syndrome in Cairo. EgyptMyopia Heart Dis JAMA Ophthalmol. 2013;131:1057–1066. doi:10.1001/jamaophthalmol.2013.644
14. Mokhtar MM, Abdel-Fattah M. Major birth defects among infants with Down’s syndrome in Alexandria, Egypt (1995–2000). J Trop Pediatr. 2002;48:247–249. doi:10.1093/tropej/48.4.247
15. Kotb MA, Kotb A. Extrahepatic biliary atresia is an aflatoxin induced cholangiopathy in infants with null GSTM1 genotype with disrupted P53 and GSTPi to mothers heterozygous for GSTM1 polymorphism: damage control is mediated through neutrophil elastase and CD14+ activated monocytes: kotb disease. Med J Cairo Univ. 2015;83:137–145.
16. Rascón Trincado MV, Lorente Toledano F, Villalobos VS. A study of the functions of polymorphonuclear neutrophil in patients with Down’s syndrome. Allergol Immunopathol (Madr). 1988;16:339–345.
17. Novo E, García MI, Lavergne J. Nonspecific immunity in Down syndrome: a study of chemotaxis, phagocytosis, oxidative metabolism, and cell surface marker expression of polymorphonuclear cells. Am J Med Genet. 1993;46:384–391. doi:10.1002/ajmg.1320460408
18. Kotb MA. Ursodeoxycholic acid in neonatal hepatitis and infantile paucity of intrahepatic bile ducts: review of a historical cohort. Dig Dis Sci. 2009;54:2231–2241. doi:10.1007/s10620-008-0600-8
19. Ram G, Chinen J. Infections and immunodeficiency in Down syndrome. Clin Exp Immunol. 2011;164:9–16. doi:10.1111/j.1365-2249.2011.04335.x
20. Alaama JY, Ahmad MS, Ahmad S, Damanhouri ZA. Altered Metabolism in down Syndrome, Health Problems in down Syndrome. Subrata Dey, IntechOpen; 2015. Available from https://www.intechopen.com/books/health-problems-in-down-syndrome/altered-metabolism-in-down-syndrome. doi:10.5772/60638
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
