Back to Journals » Clinical Ophthalmology » Volume 8
A prospective, multicenter, noninterventional study of Optive Plus® in the treatment of patients with dry eye: the prolipid study
Authors Kaercher T , Thelen U, Brief G, Morgan-Warren R, Leaback R
Received 29 November 2013
Accepted for publication 7 January 2014
Published 17 June 2014 Volume 2014:8 Pages 1147—1155
DOI https://doi.org/10.2147/OPTH.S58464
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Thomas Kaercher,1 Ulrich Thelen,2 Gerrett Brief,3 Robert J Morgan-Warren,4 Richard Leaback4
1Augenarztpraxis, Heidelberg, Germany; 2University of Münster, Münster, Germany; 3Ruhr University, Bochum, Germany; 4Allergan Holdings Ltd, Marlow, UK
Objective: The aim was to evaluate the efficacy of Optive Plus®, an artificial tear containing castor oil, in patients with dry eye, in a routine clinical setting.
Methods: This was a prospective, noninterventional study of patients with dry eye who switched from a prior therapy or who were naïve to treatment (n=1,209). Patients were issued Optive Plus® artificial tears. Dry eye severity, tear break-up time (TBUT), Schirmer score, Ocular Surface Disease Index (OSDI) score, and patient assessment of symptoms were recorded at baseline and at the follow-up visit (4 weeks after starting Optive Plus®).
Results: The cause of dry eye was determined to be aqueous deficiency, lipid deficiency, or a mixture of aqueous and lipid deficiency (in 19.5%, 20.1%, and 47.8%, respectively, of the total study population). The severity of dry eye decreased from baseline to the follow-up visit, showing a decrease of the more severe levels (2–4) and a concurrent increase in mild level (1) of the rating scale. Patients reported an improvement in dry eye symptoms over the duration of the study, specifically 74.2% (n=152), 85.4% (n=182), and 82.4% (n=417) of patients in the aqueous-deficient, lipid-deficient, and mixed-deficiency groups, respectively. TBUT was measured in 475 patients. Baseline measurements for mean and standard deviation were 9.0±3.5, 7.1±3.6, and 6.6±3.0 seconds for the aqueous-deficient, lipid-deficient, and mixed-deficiency groups, respectively. These increased to 10.5±3.5, 10.0±3.6, and 9.2±3.1 seconds at the final visit. Overall, 92.5% of all patients were satisfied with the use of Optive Plus®, and 86% said they would purchase Optive Plus®. Ten percent of patients reported adverse events, and 1.8% of all patients experienced treatment-related adverse events.
Conclusion: Optive Plus® was well tolerated and effective in reducing the signs and symptoms of all types of dry eye but is recommended for lipid-deficient dry eye patients.
Keywords: dry eye, lipid-deficient, aqueous-deficient, meibomian, hyperosmolarity, evaporative
A Letter to the Editor has been received and published for this article.
Introduction
Dry eye (also known as keratoconjunctivitis sicca or dysfunctional tear syndrome) is an ocular surface disease affecting millions of people worldwide.1,2 Symptoms of dry eye vary between patients and can include itching, gritty feeling, burning, foreign body sensation, dryness, photosensitivity, pain, blurred vision, and contact lens intolerance.3 Dry eye can have an adverse effect on patient quality of life, affecting, in particular, reading, computer use, watching television, and driving.4 These detrimental effects on quality of life can seriously affect the ability of patients to cope with their disease; feelings of disappointment, frustration, anxiety, and depression have been reported to be common.5 Defined as a “multifactorial disease of the tears and ocular surface,” dry eye and subsequent ocular damage occurs in a vicious circle.2,6 Entry to the vicious circle can be caused by a number of external factors, such as blepharitis, meibomian gland dysfunction (MGD), allergy, ocular surgery, or Sjögren’s syndrome. Tear film instability leads to drying and hyperosmolarity of the ocular surface, which in turn leads to apoptosis of epithelial surface cells.6 A cascade of inflammatory mediators leads to the loss of mucin-producing goblet cells.6 The instability of the tear film is exacerbated, and the circle continues as dry eye symptoms develop and persist.6
Dry eye can take on two forms: aqueous-deficient dry eye (synonymous with tear-deficient dry eye and lacrimal tear deficiency) results from reduced tear secretion and volume, while lipid-deficient dry eye (synonymous with evaporative dry eye) results from a disrupted lipid layer in the tear film and excessive evaporation from the ocular surface.2,7,8 Lipid-deficient dry eye can be classified into several subgroups based upon various causes: MGD, blepharitis, lid disorders, low blink rate, ocular surface disorders, use of contact lens, ocular surface disease, allergic conjunctivitis, or environmental causes (as in “office” dry eye).2,9 MGD is the most common cause of lipid-deficient dry eye.10,11
Lipids secreted from the meibomian glands prevent hyperevaporation of tears and excessive tearing.12 A blockage of some or all of these ducts reduces the availability of lipids to the tear film, leading to lipid-deficient dry eye.13 Since the secretion of lipids is blink-dependent, the tear film is most deprived of lipids upon waking from sleep; consequently, symptoms of lipid-deficient dry eye tend to be more pronounced in the morning, whereas those associated with aqueous-deficient dry eye get worse during the day.12 Dry eye severity is calculated, by the Delphi and International Dry Eye WorkShop (DEWS) panels, on a scale of 1 to 4, with the latter being the most severe.1,2
The current mainstay for treatment of stage 1 dry eye is the use of artificial tears, which are predominantly aqueous-based.11 Given the lipid layer is compromised in certain forms of dry eye, it may be beneficial to add lipids to compensate. However, the majority of artificial tears do not contain a lipid component and probably only provide momentary relief from symptoms and therefore, low patient satisfaction.11 Therapy might be more effective if it could be tailored to the specific type of dry eye. Artificial tears with a lipid component are thought to replenish the lipid layer. They have been shown to have a long residence time in the tear film, reduce the tear evaporation rate, improve the signs and symptoms of dry eye, improve the structure of the lipid layer, and to improve diagnostic test results, particularly the Schirmer score and tear break-up time (TBUT).11,14,15 While more than 80% of dry eye patients have some form of compromised lipid layer (lipid deficiency or a combination of aqueous and lipid deficiencies), relatively few artificial tear products on the market are lipid-based.11,16
Optive Plus® artificial tears (Allergan, Inc., Irvine, CA, USA) contain the baseline components of Optive® (Allergan, Inc.) (carboxymethylcellulose, glycerol, L-carnitine, and erythritol) with the addition of a polysorbate-80 vessel that delivers highly pure castor oil to the tear film. Upon mixing with the salts in the tear film, the polymer micelles disintegrate, releasing lipid particles into the environment, where they provide a protective layer for the tear film.17
The objective of this observational study was to collect data on the effect of Optive Plus®, a new lipid-containing artificial tear, in patients with dry eye, in a routine clinical setting.
Methods
Study design
This was a prospective, multicenter, noninterventional, 4-week study of patients with dry eye receiving Optive Plus® in routine clinical care at 166 centers in Germany. Data were collected between September 2012 and March 2013. In accordance with regulations, advice was received from the German Ethics Committee (Landesärztekammer Baden-Württemberg). Ethics approval was granted (reference number F 2012-064). No consent forms were required because German law does not require informed consent for observational studies where treatment is medically indicated by the physician regardless of study participation, and where treatment is restricted to approved indications.
Patient selection and symptom measurements
Patients selected for inclusion in the study were all previously diagnosed with dry eye. Disease severity ranged from mild to severe, using the DEWS classification (measured on a scale of 0 to 4, indicating “none” to “very severe”).2 At the baseline visit, patients who, in the opinion of the treating physician (ophthalmologist), were candidates for Optive Plus® were included in the study. Patients were categorized into subtypes of dry eye (aqueous-deficient, lipid-deficient, or mixed deficiency) by their physician, based on the DEWS guidelines.2 Dry eye severity and symptom grading, Ocular Surface Disease Index (OSDI), TBUT, and Schirmer score were recorded by a physician at baseline. TBUT and Schirmer tests were performed according to individual clinical practice in each clinic. The OSDI questionnaire includes 12 items categorized into three subscales: ocular symptoms (sensitivity to light, gritty eyes, and painful/sore eyes), vision-related function (blurred vision, poor vision, impairment when reading, difficulties when driving at night, difficulties when working with a computer/using a cash machine, and problems when watching TV), and environmental triggers (windy conditions, low humidity, and air-conditioned areas).18 The 12 items were graded, by the patient, on a scale of 0 to 4, where 0= none of the time, 1= some of the time, 2= half of the time, 3= most of the time, and 4= all of the time.18 The OSDI total score was calculated from the statement responses: OSDI = (sum of all scores × 100)/(total number of questions answered × 4), where a higher score represented greater severity.18
All patients were issued Optive Plus® artificial tears, which were administered according to the recommended dosing information. In those patients who were receiving dry eye therapy, Optive Plus® replaced the previous therapy. Patients were followed up approximately 4 weeks after the initial visit, according to routine clinical practice. At the follow-up visit, dry eye severity and symptoms were graded, and OSDI, TBUT, and Schirmer scores were recorded. Patients were asked about adverse events, and patient satisfaction with treatment was measured in response to statements, on a scale from 1 to 5 (strongly agree, agree, neither agree nor disagree, disagree, strongly disagree).
Data entry and analyses were carried out using the statistical software package SAS version 9.1.3 (SAS Institute Inc., Cary, NC, USA) and proprietary software of Syneed Medidata GmbH (Konstanz, Germany). Data analysis was performed descriptively – this included preparation of data listings and summary statistics (extreme values, interquartile section, mean and median values, and standard deviations) or frequency distribution tables, as appropriate for each item. It should be noted that only patients with complete data in the subgroup of interest at both visits were included for analysis.
Prior dry eye treatments
Prior to entry into the study, a selection of patients were using commonly prescribed artificial tears in Germany. These included aqueous-based tears Hylo-Comod® (URSAPHARM Arneimittel GmbH, Saarbrücken, Germany), Wet-Comod® (URSAPHARM Arneimittel GmbH), Hylo-Vision® (OmniVision GmbH, Puchheim, Germany), Artelac Rebalance® (Bausch and Lomb, Inc., North Bridgewater, NJ, USA), Artelac Splash® (Bausch and Lomb, Inc.), Systane® (Alcon Laboratories Inc., Fort Worth, TX, USA), Systane Ultra® (Alcon Laboratories Inc.), Thealoz® (Spectrum Thea Pharmaceuticals, Ltd, Macclesfield, UK), Hyabak® (Spectrum Thea Pharmaceuticals, Ltd), and Vismed® (TRB Chemedica Int SA, Geneva, Switzerland), or lipid-based tears Artelac Lipids® (Bausch and Lomb, Inc.), Cationorm® (Santen Pharmaceutical Co, Ltd, Osaka, Japan), Tears Again® (OCuSOFT, Inc., Rosenberg, TX, USA), and Systane Balance® (Alcon Laboratories Inc.).
Results
Patient baseline characteristics
In total, there were 1,209 patients, all of whom had received Optive Plus® for 4 weeks. As detailed in Table 1, female patients predominated, comprising 66.6% of the study population, while the mean age of participants was 60±16 years. The most common cause of dry eye (as determined by the practicing physician) was a mixture of aqueous and lipid deficiencies (47.8%), while 20.1% had lipid deficiency, and 19.5% had aqueous deficiency (Table 1). Patients were either not taking dry eye medication (36.4%) or were taking artificial tears to alleviate symptoms (64.4%) (data available for 1,189 patients). The lack of efficacy of the existing treatment was the most commonly reported reason for switching to Optive Plus® (66.9% of patients) (data available for 901 patients), and this was more evident when a lipid-deficient form of dry eye was diagnosed (Table 1).
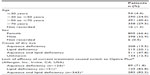  | Table 1 Demographic and background patient data |
Prior dry eye medication
Prior to the start of the monitoring period, 64.4% (n=766) of all study participants with dry eye were treated with artificial tears (data on previous therapies were available for 1,189 patients). The most commonly used artificial tear products were Hylo-Comod® (n=171 [22.3%]), Hylo-Vision® (n=107 [14.0%]), and Systane® (n=92 [12.0%]). Several other drugs were named less frequently (≤10% each). The majority of patients on prior dry eye medication were using an aqueous-based tear (89% of patients on monotherapy). Of all the patients on monotherapy prior to the start of the study, only 10.9% were using lipid-based tears.
Changes in severity
The severity of dry eye, as graded using the DEWS classification, in the total study population decreased during the study period: levels 2–4 (moderate to very severe) decreased, and there was a corresponding increase in the level 1 (mild) cases, from 21.8% to 51.8% (data on severity were available for 1,145 patients) (Figure 1). A similar trend was displayed for the aqueous-deficient, lipid-deficient, and mixed-deficiency patient groups. Figure 1 also demonstrates that baseline severity was considered by physicians to be worse (ie, a greater proportion of patients were assigned to severity levels 2–4) when lipid deficiency was present. The most effective reduction in severity appeared to be in the lipid-deficient patients, for whom severity level 2 decreased by 29%, level 3 by 81%, and level 4 by 86%.
  | Figure 1 Severity level of dry eye in patients at baseline and follow-up visit (data classed as missing has been excluded). |
Patient assessment of symptoms with Optive Plus®
Prior to study commencement and at the final assessment, patients were asked to rate the severity of a number of symptoms, on a scale ranging from 0 to 4, where 0= no symptoms (I do not have this symptom) and 4= very severe symptoms (I always notice this symptom, it does make me uncomfortable, and it does interfere with my activities). The symptoms measured were stinging/burning, itching, sandiness/grittiness, blurred vision, dryness, light sensitivity, pain/soreness, increased lacrimation/ watery eyes, feeling of tension/pressure, and red eyes. After 4 weeks of treatment, the majority of all patients reported improvements in symptoms after using Optive Plus® (data on patient assessment of symptoms were available for 1,194 patients), as illustrated by Figure 2. Specifically, 82.9% (n=990), 74.2% (n=152), 85.4% (n=182), and 82.4% (n=417) of patients reported an improvement in dry eye symptoms, in the total population, aqueous-deficient, lipid-deficient, and mixed-deficiency groups, respectively. The improvement in symptoms appeared to be more prominent in the lipid-deficient patient group.
  | Figure 2 Patient assessment of dry eye symptoms 4 weeks after switching to Optive Plus® (Allergan, Inc., Irvine, CA, USA). |
Patients on prior dry eye medication were asked to rate their symptoms, after using Optive Plus® for 4 weeks, on a scale ranging from much improved to improved, about the same, worse, or much worse (Figure 3). There was some degree of symptom improvement for all prior treatments listed (and for the treatment-naïve group). Previous therapy groups taking Thealoz®, Hyabak®, and Artelac Rebalance®, and the treatment-naïve group had the highest percentage of patients reporting an improvement of symptoms (91.7%, 89.3%, 89.2%, and 88.5% respectively). The Artelac Rebalance®, Artelac Lipids®, Tears Again®, Hylo-Vision®, Hyabak®, and treatment-naïve groups had several patients reporting a worsening of symptoms (14 patients out of a total of the 852 patients with complete data).
Efficacy analyses: TBUT, Schirmer score, and OSDI score
Figure 4A illustrates the TBUT for all the study population groups (data on TBUT were available for 475 patients). Overall, switching to Optive Plus® had a positive effect of increasing the mean TBUT by ≥1.5 seconds. For each group, the TBUT returned to the normal range of ≥10 seconds, with the exception of the mixed-deficiency patient group.2 The greatest change was for the lipid-deficient patient group, with an increase of 2.9 seconds. Figure 4B shows that TBUT improved for lipid-deficient patients who switched from one of the three most widely-used prior treatments (<10% of patients on a prior therapy) – Hylo-Comod®, Hylo-Vision®, or Systane® – to Optive Plus®. Optive Plus® was significantly better at improving TBUT for all patients who had switched from these three treatments (P<0.0001 for Hylo-Comod® and Hylo-Vision®; P=0.0027 for Systane®).
In the total population, Schirmer test scores increased by an average of 21.4% between baseline and the follow-up visit (data available for 307 patients). In the aqueous-deficient, lipid-deficient, and mixed-deficiency groups, Schirmer scores increased on average, between baseline and the follow-up visit, by 15.2%, 12.9%, and 29.7%, respectively (Table 2). Mean OSDI score decreased by 17.5 in the total population (data available for 310 patients), by 18.7 in lipid-deficient patients, 22.0 in the mixed-deficiency group, and 13.9 in aqueous-deficient patients (Table 2).
Patient and physician satisfaction with Optive Plus®
Overall, 92.5% of all patients were satisfied with the use of Optive Plus® for the treatment of their dry eye (data available for 1,191 patients). All patient groups displayed a similar level of satisfaction. In general, patients were at least 65% satisfied with Optive Plus® in relation to certain aspects of their treatment: 83% agreed that their dry eye symptoms had improved (data available for 1,194 patients); 67% agreed that Optive Plus® made their eyes feel more comfortable (data available for 1,182 patients); 65% agreed that Optive Plus® made their eyes feel more comfortable for longer (data available for 1,174 patients); 81% agreed that normal vision returned within a short time after administration of Optive Plus® (data available for 1,187 patients); 93% agreed that Optive Plus® did not cause matted or crusty eyes/eyelashes (data available for 1,189 patients). Consequently, 86% indicated that they would purchase Optive Plus® for dry eye signs and symptoms (data available for 1,150 patients).
With regard to physicians’ overall satisfaction with the effectiveness of Optive Plus®, physicians were “very satisfied” or “satisfied” in 94% of cases, “dissatisfied” in 6%, and “very dissatisfied” in 0.4% (data available for 1,176 patients). In 89% of cases (data available for 465 patients), the physicians recommended continuation of Optive Plus®, whereas continuation of treatment was not recommended in 11% (predominantly due to lack of efficacy/deterioration or patient dissatisfaction/ refusal to continue). This trend appeared to be similar for all patient groups.
Adverse events
Of the 1,209 patients, 123 (10.2%) reported adverse events; however, none of these were classified as serious adverse device effects. Twenty-two (1.8% of all patients) adverse events were identified as being treatment-related. The most frequent adverse events were eye discharge (n=24 [2.0%]), eye pruritus (n=23 [1.9%]), dry eye (n=22 [1.8%]), foreign body sensation in eyes (n=20 [1.7%]), increased lacrimation (n=20 [1.7%]), and blurred vision (n=20 [1.7%]). The aqueous-deficient patient group reported the most number of adverse events (n=29 [13.9%]); however, only three of these were thought to be treatment-related. The lipid-deficient patient group recorded 21 (9.8%) patients as having adverse events.
Discussion
This study met the objective of collecting routine patient data on the effect of Optive Plus® and demonstrated the benefits of using this device on patients suffering from dry eye. Optive Plus® use was associated with increased TBUT and Schirmer score, reduced OSDI score, and improvement of patient symptoms and patient and physician satisfaction. In terms of comfort, patients were satisfied, and moderate, severe, and very severe symptoms were reduced. This study demonstrated that Optive Plus® is particularly effective in symptom control in lipid-deficient patients, is nontoxic, improves parameters associated with dry eye, and is effective in maintaining the tear film over the eye. Indeed, patient statements confirm they would purchase Optive Plus® in the future. Improvements in signs and symptoms were most marked in patients with lipid-deficient dry eye, and these patients benefited more from lipid-containing artificial tears than the aqueous-deficient patient group.
The Optive® family was developed with the aim of providing protection against hyperosmolarity. Hyperosmolarity plays a vital role in the formation of dry eye and appears to provide one way of breaking the vicious circle of the disease.6 Osmoprotectant compatible solutes can be included in artificial tear formulations to provide this protection. To this end, Optive® contains several osmoprotectants (glycerol, L-carnitine, and erythritol) and sodium carboxymethylcellulose (CMC), which binds to human corneal epithelial cells, promoting growth and wound healing as well as lubricating the ocular surface.19 The combination of CMC with osmoprotectant-compatible solutes in Optive® significantly reduces the signs and symptoms of dry eye. Several studies have demonstrated the effectiveness of Optive® at improving the results of clinical tests, as well as improving patient symptoms of dry eye.20–23
The lipid layer of the tear film is important for the prevention of tear evaporation and for facilitating the spread of the tear film across the surface of the cornea.24 The lipids are secreted from the lid margin by the meibomian glands as meibum and form two distinct layers.25–27 The polar phase, comprised of phospholipids (phosphatidylcholine and phosphatidylethanolamine), sphingomyelin, ceramides, cerebrosides, triglycerides, and free fatty acids, is thought to be important for tear film stability.24–27 The larger, nonpolar phase (comprising wax and cholesterol esters, triglycerides, and hydrocarbons) is important for stabilizing the polar phase and, perhaps more importantly, for controlling the transmission rate of water vapor and various ions.24–27 Disruptions in the lipid layer, for example, caused by MGD, can result in dry eye. Consequently, an artificial tear that can replenish the lipid-depleted tear film is thought to be beneficial.
There have been varying degrees of success at producing lipid-containing artificial tears in the past, the first being TearGard™ (Bio Products Ophthalmics Inc., NY, USA) in 1983, which was successful in protecting the integrity of the tear film and provided relief for dry eye patients.28 Since then, several lipid-based therapies have been developed and have demonstrated tolerability and significant improvements in the signs and symptoms of dry eye.11 The process of developing a lipid-containing artificial tear can be hampered by many factors, including the combination of immiscible liquids, instability, irregular spreading, effects on visual acuity and light scattering.29 Historically, lipid-containing artificial tears have not been well tolerated by patients, due to such difficulties in formulation, and they have provided thick, viscous therapies that cause blurring of vision.30
In addition to the ingredients previously described in Optive®, Optive Plus® contains a lipid component, castor oil, which restores the osmolarity of the tear layer, with a concomitant reduction in evaporation.31 Castor oil, obtained from the plant Ricinus communis, is a pale yellow, flexible, stable triglyceride that is environmentally friendly, making it a valuable addition to artificial tears.32 Castor oil is preserved in the tears mixture by means of polysorbate-80, which forms a “vessel” surrounding the lipid, preventing separation in the bottle and ensuring intact delivery to the tear film.17 Optive Plus® is specifically designed for lipid-deficient dry eye, but improvements have been noted with its use in all types of dry eye disease (an important point, considering that different forms of dry eye can change throughout the course of the disease). The preservative used in Optive® and Optive Plus®, PURITE® (Allergan, Inc.), has a demonstrable safety and tolerability record and is gentle to the ocular surface.21,22,33 The addition of the lipid element in Optive Plus® provides an additional level of protection to the tear layer and thus, further protection for the patient. A 1-month prospective bilateral study of 35 patients demonstrated that the castor oil component of Optive Plus® remained within the tear film for up to 2 hours (1 hour P<0.001; 2 hour P=0.013) and revealed a cumulative effect with increasing castor oil concentration, due to repeated use of Optive Plus®.34 In addition, patients reported a significant decrease in OSDI scores (P<0.001) and a significant increase in ocular comfort (P<0.001).34 In a comparison study of lipid-based artificial tears, Optive Plus® proved to be more effective than Systane Balance® and OCuSOFT Retaine® MGD™ (OCuSOFT, Inc.) (marketed as Cationorm® by Santen Pharmaceuticals Co, Ltd outside the US) in increasing noninvasive TBUT in MGD patients.35
The current study had a number of limitations, due to its nature as a prospective, multicenter, observational study of patients treated in a routine clinical setting. The analysis included those patients who fully completed the study and did not take into account discontinuations due to adverse events. The efficacy of Optive Plus® was compared with baseline measurements, not a control group, and data were only collected at one time point (follow-up visit). Further research should include large randomized head-to-head trials of different artificial tears, preferably with stratification for lipid-deficient and aqueous-deficient etiologies.
To summarize, Optive Plus® was well tolerated and effective during this clinical study. The formulation of Optive Plus® has proven to increase (compared with baseline) TBUT and Schirmer score, reduce OSDI score and patient symptoms, and perform well with regards to patient satisfaction.
Conclusion
This observational study of Optive Plus® in the routine clinical setting has demonstrated this device to be effective across a broad spectrum of patients with dry eye. A number of the quantitative and qualitative measures recorded indicated that Optive Plus® is particularly beneficial for patients with lipid-deficient dry eye.
Acknowledgments
This study was funded by Allergan Ltd. The authors thank Rachel Rankin of Darwin Healthcare Communications for writing and editing support, funded by Allergan Ltd.
Disclosure
Robert Morgan-Warren and Richard Leaback are employed by Allergan Holdings Ltd. The authors report no other conflicts of interest in this work.
References
Behrens A, Doyle JJ, Stern L, et al; Dysfunctional Tear Syndrome Study Group. Dysfunctional tear syndrome: a Delphi approach to treatment recommendations. Cornea. 2006;25(8):900–907. | |
International Dry Eye WorkShop. Report of the International Dry Eye WorkShop (DEWS). Ocul Surf. 2007;5(2):61–204. | |
Nichols KK. Patient-reported symptoms in dry eye disease. Ocul Surf. 2006;4:137–145. | |
Miljanović B, Dana R, Sullivan DA, Schaumberg DA. Impact of dry eye syndrome on vision-related quality of life. Am J Ophthalmol. 2007;143(3):409–415. | |
Li M, Gong L, Sun X, Chapin WJ. Anxiety and depression in patients with dry eye syndrome. Curr Eye Res. 2011;36(1):1–7. | |
Baudouin C, Aragona P, Messmer EM, et al. Role of hyperosmolarity in the pathogenesis and management of dry eye disease: proceedings of the OCEAN group meeting. Ocul Surf. 2013;11(4):246–258. | |
Goto E, Tseng SC. Differentiation of lipid tear deficiency dry eye by kinetic analysis of tear interference images. Arch Ophthalmol. 2003;121(2):173–180. | |
Murube J, Németh J, Höh H, et al. The triple classification of dry eye for practical clinical use. Eur J Ophthalmol. 2005;15(6):660–667. | |
Berta A. The differential diagnosis of the red eye. Int Ophthalmol. 2008;28(Suppl 1):S7–S17. | |
Knop E, Knop N, Millar T, Obata H, Sullivan DA. The international workshop on meibomian gland dysfunction: report of the subcommittee on anatomy, physiology, and pathophysiology of the meibomian gland. Invest Ophthalmol Vis Sci. 2011;52(4):1938–1978. | |
Lee SY, Tong L. Lipid-containing lubricants for dry eye: a systematic review. Optom Vis Sci. 2012;89(11):1654–1661. | |
Petricek I. Dry eye. Int Ophthalmol. 2008;28 (suppl.):18–31. | |
Nichols KK, Foulks GN, Bron AJ, et al. The international workshop on meibomian gland dysfunction: executive summary. Invest Ophthalmol Vis Sci. 2011;52(4):1922–1929. | |
Di Pascuale MA, Goto E, Tseng SC. Sequential changes of lipid tear film after the instillation of a single drop of a new emulsion eye drop in dry eye patients. Ophthalmology. 2004;111(4):783–791. | |
Khanal S, Tomlinson A, Pearce EI, Simmons PA. Effect of an oil-in-water emulsion on the tear physiology of patients with mild to moderate dry eye. Cornea. 2007;26(2):175–181. | |
Lemp MA, Crews LA, Bron AJ, Foulks GN, Sullivan BD. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort: a retrospective study. Cornea. 2012;31(5):472–478. | |
Beard BJ, Simmons PJ, Vehige JG. Mechanism of action of an advanced artificial tear formulation. Presented at the Annual Meeting of the American Academy of Optometry; October 12–15, 2011; Boston, MA. | |
Schiffman RM, Christianson MD, Jacobsen G, Hirsch JD, Reis BL. Reliability and validity of the Ocular Surface Disease Index. Arch Ophthalmol. 2000;118(5):615–621. | |
Garrett Q, Simmons PA, Xu S, et al. Carboxymethylcellulose binds to human corneal epithelial cells and is a modulator of corneal epithelial wound healing. Invest Ophthalmol Vis Sci. 2007;48(4):1559–1567. | |
Rajpal RK, Logan LA, Kislan TP. Evaluation of Optive™ in patients previously using Systane® for the treatment of dry eye signs and symptoms. Poster presented at: the 5th International Conference on the Tear Film and Ocular Surface: Basic Science and Clinical relevance; September 5–8, 2007; Taormina, Italy. | |
Kaercher T, Buchholz P, Kimmich F. Treatment of patients with keratoconjunctivitis sicca with Optive: results of a multicenter, open-label observational study in Germany. Clin Ophthalmol. 2009;3:33–39. | |
Guillon M, Maissa C, Ho S. Evaluation of the effects on conjunctival tissues of Optive eyedrops over one month usage. Cont Lens Anterior Eye. 2010;33(2):93–99. | |
Baudouin C, Cochener B, Pisella PJ, et al. Randomized, phase III study comparing osmoprotective carboxymethylcellulose with sodium hyaluronate in dry eye disease. Eur J Ophthalmol. 2012;22(5):751–761. | |
Stern ME. The normal tear film and ocular surface. In: Pflugfelder SC, Beuerman RVV, Stern ME, editors. Dry Eye and Ocular Surface Disorders. New York, NY: Marcel-Dekker; 2004:41–62. | |
Nicolaides N, Kaitaranta JK, Rawdah TN, Macy JI, Boswell FM, Smith RE. Meibomian gland studies: comparison of steer and human lipids. Invest Ophthalmol Vis Sci. 1981;20(4):522–536. | |
McCulley JP, Shine W. A compositional based model for the tear film lipid layer. Trans Am Ophthalmol Soc. 1997;95:79–88; discussion 88–93. | |
Tiffany JM. The normal tear film. Dev Ophthalmol. 2008;41:1–20. | |
Silverman JJ. The first three-layered tear substitute. Contacto. 1983;27:19–22. | |
Rieger G. Lipid-containing eye drops: a step closer to natural tears. Ophthalmologica. 1990;201(4):206–212. | |
Geerling G, Tauber J, Baudouin C, et al. The international workshop on meibomian gland dysfunction: report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2011;52(4):2050–2064. | |
Goto E, Shimazaki J, Monden Y, et al. Low-concentration homogenized castor oil eye drops for noninflamed obstructive meibomian gland dysfunction. Ophthalmology. 2002;109(11):2030–2035. | |
Ogunniyi DS. Castor oil: a vital industrial raw material. Bioresour Technol. 2006;97(9):1086–1091. | |
Noecker R. Effects of common ophthalmic preservatives on ocular health. Adv Ther. 2001;18(5):205–215. | |
Maissa CA, Guillon M, Bossard B. The effect of a new lipid-based eye drop and its interaction with the lipid layer. Presented at: ARVO 2012; May 6–10, 2012; Fort Lauderdale, FL. | |
Connor CG, Ottenbreit R, Schroeder LK, Rabin JC, Narayanan S. Comparison of three commercially available tear substitutes designed for evaporative dry eye treatment. Presented at: ARVO 2013; May 5–9, 2013; Seattle, WA. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



