Back to Journals » Drug Design, Development and Therapy » Volume 9
The efficacy and safety of panitumumab in the treatment of patients with metastatic colorectal cancer: a meta-analysis from five randomized controlled trials
Received 21 March 2015
Accepted for publication 2 June 2015
Published 7 August 2015 Volume 2015:9 Pages 4471—4478
DOI https://doi.org/10.2147/DDDT.S85178
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Wei Duan
Ruo-feng Liang,1 Lei-lei Zheng2
1General Department, University Hospital, Affiliated to Zhejiang Science-Technology University, Hangzhou, People’s Republic of China; 2Department of Psychiatry, Second Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou, People’s Republic of China
Background: The efficacy of adding panitumumab to chemotherapy remains controversial in the treatment of metastatic colorectal cancer (mCRC). Thus, we conducted this meta-analysis to assess the efficacy and safety of this combination regimen in patients with mCRC.
Methods: The PubMed, Embase, and Web of Science databases were comprehensively searched. Eligible studies included randomized controlled trials (RCTs) that estimated the efficacy of panitumumab with or without chemotherapy in the treatment of patients with mCRC. Hazard ratio (HR), risk ratio (RR), and 95% confidence intervals (CIs) were calculated, and heterogeneity was tested using I2 statistics.
Results: Four studies involving a total of 3,066 patients were included in this meta-analysis. The addition of panitumumab to chemotherapy significantly improved progression-free survival (PFS) (HR =0.84, 95% CI =0.78–0.91, P=0.000) and the objective response rate (ORR) (RR =2.18, 95% CI =1.13–4.22, P=0.021) compared to chemotherapy alone, but no effect was noted on overall survival (OS) (HR =0.97, 95% CI =0.89–1.05, P=0.402). Subgroup analysis based on KRAS gene status revealed that the combined therapy significantly improved PFS (HR =0.71, 95% CI =0.57–0.88, P=0.002) and ORR (RR =2.43, 95% CI =1.21–4.90, P=0.013) in patients with wild-type KRAS tumors. Irinotecan-based chemotherapy plus panitumumab significantly prolonged PFS in patients with mCRC (HR =0.84, 95% CI =0.76–0.94, P=0.002). The combined treatment also increased the incidence of grade 3/4 adverse events.
Conclusion: This meta-analysis indicates that the combination of panitumumab and chemotherapy effectively improved PFS and ORR, but it did not prolong OS. However, as the number of studies in the meta-analysis was limited, more large-scale, better-designed RCTs are needed to assess the combination of panitumumab and chemotherapy.
Keywords: panitumumab, chemotherapy, metastatic colorectal cancer, meta-analysis
Introduction
Colorectal cancer ranks as the third most common cancer worldwide, as approximately 1 million new cases are diagnosed annually.1 The median overall survival (OS) of patients with metastatic colorectal cancer (mCRC) has been improved considerately in recent decades,2,3 mainly because of the introduction of new chemotherapeutic agents and targeted therapies.4,5
Chemotherapeutic agents are widely used for patients with advanced disease to prolong survival, control symptoms, and improve the quality of life.6–9 As the foundation of chemotherapy in advanced mCRC, oxaliplatin and irinotecan have exhibited clinical effects in the treatment of mCRC. Human epidermal growth factor receptor (EGFR) is a transmembrane cell surface glycoprotein10 that plays an important role in the proliferation and metastasis of colorectal cancer by activating various signaling pathways.11 Thus, EGFR has become the primary therapeutic target for this cancer. Panitumumab is a fully human monoclonal antibody directed against EGFR, and its activity in terms of a survival benefit has been proven.12 However, panitumumab appears to produce different clinical outcomes in the treatment of mCRC when combined with different chemotherapeutics. Beneficial effects were observed when panitumumab was combined with irinotecan-based chemotherapy, and negative effects were noted when the drug was combined with oxaliplatin-based chemotherapy. Moreover, the interaction between oxaliplatin and panitumumab remains unknown.
The KRAS gene status is considered a predictive marker of anti-EGFR monoclonal antibodies. Previous research illustrated that patients with mutant KRAS did not experience a clinical benefit of anti-EGFR therapy.13 The aim of this meta-analysis was to explore the efficacy and toxicities of adding panitumumab to chemotherapy in patients with mCRC according to KRAS status and assess the efficacy of panitumumab in combination with different chemotherapeutic partners.
Methods
Literature search and inclusion criteria
The PubMed, Embase, and Web of Science databases were searched to identify studies (published before December 24, 2014) on the addition of panitumumab to chemotherapy in the treatment of mCRC. The following search terms were used: (“panitumumab” [Supplementary Concept] OR “panitumumab” [All Fields]) AND (“secondary” [Subheading] OR “secondary” [All Fields] OR “metastatic” [All Fields]) AND (“colorectal neoplasms” [MeSH Terms] OR (“colorectal” [All Fields] AND “neoplasms” [All Fields]) OR “colorectal neoplasms” [All Fields] OR (“colorectal” [All Fields] AND “cancer” [All Fields]) OR “colorectal cancer” [All Fields]). The search was limited to human studies and randomized controlled trials (RCTs). No language restriction was imposed. We also manually searched the reference lists of the included studies until no potentially eligible articles could be identified. Studies that met the following inclusion criteria were included: 1) study design, RCT; 2) study population, ≥18 years old with a diagnosis of adenocarcinoma of the colon or rectum; 3) intervention, chemotherapy with or without panitumumab; and 4) outcome measure, OS, progression-free survival (PFS), and overall response rate (ORR). If duplicate data were presented in several studies, only the most informative or complete articles were included.
Data extraction and outcome measures
Two authors independently extracted the following data from each study: first author, treatment regimen, the number of patients (intervention/control), age range or mean age of the patients, type of blinding, type of controls, hazard ratios (HRs) with 95% confidence intervals (CIs) for OS, PFS, ORR, and the incidence of adverse events. A standardized Excel file was used for data extraction. Disagreements between the investigators were resolved by discussion and consensus. The primary outcome was PFS. Secondary outcomes included OS, ORR, and the incidence of adverse events.
Quality assessment
The methodological quality of the studies was independently scored by Ruo-feng Liang and Lei-lei Zheng using a validated Jadad five-point scale.14 The scale consists of three items describing randomization (0–2 points), masking (0–2 points), and dropouts and withdrawals (0–1 point) in the report of an RCT.14 A score of 1 point is given for each of the points described. An additional point is given when the method of randomization and/or blinding is given and appropriate, whereas a point is deducted when the method is inappropriate. The quality scale ranges from 0 to 5 points. A higher score indicates better quality. Articles with ≥3 points were considered to have high quality.15
Statistical analyses
We assessed the overall efficacy of adding panitumumab to chemotherapy in the treatment of patients with mCRC based on the data from the included studies. PFS and OS were treated as time-to-event variables, and thus were expressed as HRs with 95% CIs for each study. The ORR and incidence of adverse events were treated as dichotomous variables and were expressed as risk ratios (RRs) with 95% CIs for each study. Heterogeneity across the studies was tested using I2 statistics, which quantitatively measured the degree of inconsistency across studies. I2 values of <25%, approximately 50%, approximately 75%, and approximately 100% indicated no, low, moderate, and high heterogeneity, respectively.16 A fixed-effects model (Mantel–Haenszel method)17 was used, whereas a random-effects model (DerSimonian–Laird method)18 was used when significant heterogeneity existed (I2>50%). A subgroup analysis was conducted on the basis of KRAS status and chemotherapeutic partners. The presence of publication bias was assessed using the Begg’s test.19 A P-value of less than 0.05 was considered statistically significant. All statistical analyses were conducted using STATA software version 12.0 (StataCorp LP, College Station, TX, USA).
Results
Identification of eligible studies
The initial search of the PubMed, Embase, and Web of Science databases yielded 566 relevant publications. Of these, 39 were excluded for duplicate records, and 458 were excluded after reviewing the title or abstract, leaving 69 articles for full-text review. In the review, eleven articles were excluded for the following reasons: one study used panitumumab in both arms,20 one study used bevacizumab and panitumumab in the intervention arm,21 three studies presented preliminary outcomes,12,13,22 and six articles had single-arm study designs.23–28 Finally, four studies29–32 that met the inclusion criteria were included in this meta-analysis. The flowchart of the search strategy is shown in Figure 1.
  | Figure 1 Eligibility of studies for inclusion in the meta-analysis. |
Study characteristics and quality assessment
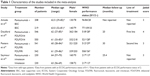
The main characteristics of the four RCTs included in the meta-analysis are presented in Table 1. These studies were published between 2008 and 2014. The sample sizes of the studies ranged from 427 to 1,096 patients (total, 3,066 patients). The clinical characteristics were generally balanced between the intervention and control arms of each study. The four studies tested the KRAS mutation status of the tumors using allele-specific polymerase chain reaction. Adverse events were graded using the common Terminology Criteria for Adverse Events (version 3.0) with modifications for specific skin- and nail-related toxicities.33 The median Jadad score of the included studies was 3 (range: 3–4).
Primary outcome: PFS
All the four RCTs reported PFS in the study patients. The aggregated results of these studies suggested that the addition of panitumumab to chemotherapy significantly prolonged PFS in the treatment of patients with mCRC (HR =0.84, 95% CI =0.78–0.91, P=0.000) (Figure 2). Begg’s test and the funnel plot revealed no evidence of obvious publication bias (Z=0.707, P=0.946). Meanwhile, significant heterogeneity was identified (I2=83.6%, P=0.00). Subsequently, we performed subgroup analyses to explore potential sources of heterogeneity.
  | Figure 2 Meta-analysis exploring the effect of adding panitumumab to chemotherapy on progression-free survival. |
Subgroup analyses were conducted on the basis of KRAS status (wild-type or mutant KRAS). The results demonstrated a 29% reduction in the risk of progression with the addition of panitumumab to chemotherapy (HR =0.71, 95% CI =0.57–0.88, P=0.002; heterogeneity test: I2=79.9%, P=0.002) in patients with wild-type KRAS tumors (Figure 3). However, no beneficial effect was found in patients with KRAS-mutant tumors (HR =1.06, 95% CI =0.87–1.31, P=0.420; heterogeneity test: I2=58.9%, P=0.088).
  | Figure 3 Subgroup analyses of progression-free survival based on KRAS mutation status. |
Subgroup analyses were performed on the basis of the use of oxaliplatin-based chemotherapy and irinotecan-based chemotherapy. Pooled results illustrated that irinotecan-based chemotherapy significantly improved PFS (HR =0.84, 95% CI =0.76–0.94, P=0.002), whereas oxaliplatin-based chemotherapy had no effect (HR =1.05, 95% CI =0.74–1.48, P=0.781).
Secondary outcomes: OS and ORR
Overall survival
All the four RCTs reported OS data. Pooled results revealed that adding panitumumab to chemotherapy did not significantly improve OS (HR =0.97, 95% CI =0.89–1.05, P=0.402) (Figure 4). Begg’s test and the funnel plot revealed no evidence of obvious publication bias (Z=0.06, P=0.07).
  | Figure 4 Meta-analysis exploring the effect of adding panitumumab to chemotherapy on overall survival. |
Moreover, subgroup analyses identified no statistically significant improvement in OS in the subgroup of patients with wild-type KRAS tumors (HR =0.94, 95% CI =0.85–1.04, P=0.216). Nonsignificant results were observed for both irinotecan-based chemotherapy (HR =0.95, 95% CI =0.85–1.06, P=0.340) and oxaliplatin-based chemotherapy (HR =1.01, 95% CI =0.76–1.34, P=0.944).
Overall response rate
All the four RCTs reported ORR data. The pooled analysis using a random-effects model indicated that adding panitumumab to chemotherapy significantly increased ORR (RR =2.18, 95% CI =1.13–4.22, P=0.021) (Figure 5). In the subgroup analysis, a similar outcome was found in patients with wild-type KRAS tumors (RR =2.43, 95% CI =1.21–4.90, P=0.013), but not in patients with mutant KRAS tumors (RR =0.99, 95% CI =0.81–1.20, P=0.894). The assessment of publication bias revealed no potential publication bias among the included studies (Z=0.734, P=0.132).
Adverse events
All the four RCTs reported the occurrence of adverse events. The pooled estimate calculated using a random-effects model demonstrated that the combination of panitumumab and chemotherapy was associated with significantly increased incidence rates of grade 3/4 skin toxicity (RR =19.79, 95% CI =12.65–30.95, P=0.00), hypokalemia (RR =3.31, 95% CI =1.31–8.35, P=0.011), mucositis (RR =3.52, 95% CI =1.84–6.77, P=0.00), hypomagnesemia (RR =18.79, 95% CI =5.90–59.84, P=0.00), diarrhea (RR =1.71, 95% CI =1.19–2.47, P=0.004), and fatigue (RR =2.02, 95% CI =1.30–3.15, P=0.002).
Discussion
The current meta-analysis suggested that, compared with chemotherapy alone, the combination of panitumumab and chemotherapy significantly improved PFS (HR =0.84, 95% CI =0.78–0.91, P=0.000) and ORR (RR =2.18, 95% CI =1.13–4.22, P=0.021), but it did not prolong OS (HR =0.97, 95% CI =0.89–1.05, P=0.402). Additionally, in the subgroup analysis, a PFS benefit was observed in the wild-type KRAS population (HR =0.71, 95% CI =0.57–0.88, P=0.002), but not in the mutant KRAS population (HR =1.06, 95% CI =0.87–1.31, P=0.420). Similarly, the addition of panitumumab to irinotecan-based chemotherapy significantly prolonged PFS compared to chemotherapy alone (HR =0.84, 95% CI =0.76–0.94, P=0.002). By contrast, the addition of panitumumab to oxaliplatin-based chemotherapy conferred no significant benefit concerning PFS (HR =1.05, 95% CI =0.74–1.48, P=0.781).
According to this meta-analysis, patients receiving the combination of panitumumab and chemotherapy experienced significantly prolonged PFS compared to patients treated with chemotherapy alone. However, this significant benefit was not achieved when panitumumab was added to bevacizumab-based chemotherapy. In the RCT conducted by Hecht et al21 patients were randomly assigned to receive bevacizumab-based chemotherapy with or without panitumumab, and panitumumab-treated patients displayed decreased PFS (HR =1.27, 95% CI =1.06–1.52). Although the exact explanation for this result is not clear, the authors postulated several plausible hypotheses: 1) the existence of pharmacokinetic interactions between antibodies or between antibodies and chemotherapy; 2) toxicity was exacerbated by dual-pathway inhibition in the combination arm; and 3) the toxicity induced by the anti-EGFR antibodies and chemotherapy resulted in dose delays and reductions and increased the risk of death in the panitumumab arm.21
In this meta-analysis, we found that irinotecan-based regimens significantly prolonged PFS in the treatment of mCRC. However, this beneficial effect was not found for oxaliplatin-based regimens. The reason why panitumumab is better paired with irinotecan than with other drugs remains unknown. In a recently published meta-analysis conducted by Zhou et al,34 cetuximab and panitumumab were found to have different effects when combined with different fluoropyrimidine regimens. The authors hypothesized that the addition of cetuximab may have reduced the dose intensity and thus affected the efficacy of capecitabine-based therapy.
Skin toxicity is a class effect of anti-EGFR treatment for both monoclonal antibodies and tyrosine kinase inhibitors.35,36 The development of skin toxicity is considered a predictive factor of clinical efficacy. Among panitumumab-treated patients, prolonged PFS and OS and a higher ORR are usually observed in those who develop grade 2–4 skin toxicity compared with those without skin toxicity. However, although the appearance of skin toxicity indicated the on-target activity associated with clinical benefits, it was also found in patients who did not benefit from treatment.
Some potential limitations in this meta-analysis should be considered. First, the data we abstracted were obtained from publications rather than individual patient data, making them less powerful for confirming the findings.37 However, according to the results of a correlation analysis,38 meta-analyses based on abstract data are strongly correlated with those based on individual patient data, indicating that abstract data are acceptable and that they could be used in place of individual patient data. Second, there was considerable heterogeneity among the included studies. The characteristics of the targeted populations, study designs, treatment schedules, and follow-up intervals varied greatly. All of these factors may lead to heterogeneity and potentially affect our results. Third, although we included all large trials assessing the efficacy of panitumumab plus chemotherapy in the treatment of patients with mCRC, caution should be taken when interpreting the results, as the analysis was based on a limited number of RCTs.
In conclusion, this meta-analysis confirmed the effects, as well as the acceptable safety profile, of adding panitumumab to chemotherapy on PFS and ORR in the treatment of patients with wild-type KRAS mCRC. However, considering the limited number of RCTs and the heterogeneity among the included studies, larger, better-designed RCTs are needed to substantiate the findings and investigate the predictive role of KRAS concerning the combination of panitumumab and chemotherapy for patients with mCRC.
Disclosure
The authors report no conflicts of interest in this work.
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. | ||
Wolpin BM, Mayer RJ. Systemic treatment of colorectal cancer. Gastroenterology. 2008;134:1296–1310. | ||
O’Neil BH, Goldberg RM. Innovations in chemotherapy for metastatic colorectal cancer: an update of recent clinical trials. Oncologist. 2008; 13:1074–1083. | ||
Kopetz S, Chang GJ, Overman MJ, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol. 2009;27:3677–3683. | ||
Chau I, Cunningham D. Treatment in advanced colorectal cancer: what, when and how? Br J Cancer. 2009;100:1704–1719. | ||
Simmonds PC. Palliative chemotherapy for advanced colorectal cancer: systematic review and meta-analysis. Colorectal Cancer Collaborative Group. BMJ. 2000;321:531–535. | ||
Labianca RF, Beretta GD, Pessi MA. Disease management considerations: disease management considerations. Drugs. 2001;61:1751–1764. | ||
Labianca R, Pessi MA, Zamparelli G. Treatment of colorectal cancer. Current guidelines and future prospects for drug therapy. Drugs. 1997; 53:593–607. | ||
Midgley R, Kerr D. Colorectal cancer. Lancet. 1999;353:391–399. | ||
Ortega J, Vigil CE, Chodkiewicz C. Current progress in targeted therapy for colorectal cancer. Cancer Control. 2010;17:7–15. | ||
Banck MS, Grothey A. Biomarkers of resistance to epidermal growth factor receptor monoclonal antibodies in patients with metastatic colorectal cancer. Clin Cancer Res. 2009;15:7492–7501. | ||
Douillard JY, Siena S, Cassidy J, et al. Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J Clin Oncol. 2010;28:4697–4705. | ||
Peeters M, Price TJ, Cervantes A, et al. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28:4706–4713. | ||
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17:1–12. | ||
Kjaergard LL, Villumsen J, Gluud C. Reported methodologic quality and discrepancies between large and small randomized trials in meta-analyses. Ann Inter Med. 2001;135:982–989. | ||
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. | ||
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. | ||
Vangel MG, Rukhin AL. Maximum likelihood analysis for heteroscedastic one-way random effects ANOVA in interlaboratory studies. Biometrics. 1999;55:129–136. | ||
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–1101. | ||
Mitchell EP, Piperdi B, Lacouture ME, et al. The efficacy and safety of panitumumab administered concomitantly with FOLFIRI or Irinotecan in second-line therapy for metastatic colorectal cancer: the secondary analysis from STEPP (Skin Toxicity Evaluation Protocol With Panitumumab) by KRAS status. Clin Colorectal Cancer. 2011;10:333–339. | ||
Hecht JR, Mitchell E, Chidiac T, et al. A randomized phase IIIB trial of chemotherapy, bevacizumab, and panitumumab compared with chemotherapy and bevacizumab alone for metastatic colorectal cancer. J Clin Oncol. 2009;27:672–680. | ||
Van Cutsem E, Peeters M, Siena S, et al. Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol. 2007;25:1658–1664. | ||
Berlin J, Posey J, Tchekmedyian S, et al. Panitumumab with irinotecan/leucovorin/5-fluorouracil for first-line treatment of metastatic colorectal cancer. Clin Colorectal Cancer. 2007;6:427–432. | ||
Hecht JR, Patnaik A, Berlin J, et al. Panitumumab monotherapy in patients with previously treated metastatic colorectal cancer. Cancer. 2007;110:980–988. | ||
Kohne CH, Hofheinz R, Mineur L, et al. First-line panitumumab plus irinotecan/5-fluorouracil/leucovorin treatment in patients with metastatic colorectal cancer. J Cancer Res Clin Oncol. 2012;138:65–72. | ||
Cohn AL, Shumaker GC, Khandelwal P, et al. An open-label, single-arm, phase 2 trial of panitumumab plus FOLFIRI as second-line therapy in patients with metastatic colorectal cancer. Clin Colorectal Cancer. 2011;10:171–177. | ||
van Hellemond IE, Creemers GJ, van Warmerdam LJ, de Jong FA, Koornstra RH. Panitumumab monotherapy as a second-line treatment in metastasised colorectal cancer: a single centre experience. Clin Oncol (R Coll Radiol). 2014;26:135–141. | ||
Van Cutsem E, Siena S, Humblet Y, et al. An open-label, single-arm study assessing safety and efficacy of panitumumab in patients with metastatic colorectal cancer refractory to standard chemotherapy. Ann Oncol. 2008;19:92–98. | ||
Amado RG, Wolf M, Peeters M, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26:1626–1634. | ||
Douillard JY, Siena S, Cassidy J, et al. Final results from PRIME: randomized phase III study of panitumumab with FOLFOX4 for first-line treatment of metastatic colorectal cancer. Ann Oncol. 2014;25:1346–1355. | ||
Peeters M, Price TJ, Cervantes A, et al. Final results from a randomized phase 3 study of FOLFIRI {+/−} panitumumab for second-line treatment of metastatic colorectal cancer. Ann Oncol. 2014;25:107–116. | ||
Seymour MT, Brown SR, Middleton G, et al. Panitumumab and irinotecan versus irinotecan alone for patients with KRAS wild-type, fluorouracil-resistant advanced colorectal cancer (PICCOLO): a prospectively stratified randomised trial. Lancet Oncol. 2013;14:749–759. | ||
Cancer Therapy Evaluation Program. Common Terminology Criteria for Adverse Events, Version 3.0. Bethesda, MD: Division of Cancer Treatment and Diagnosis, National Cancer Institute, National Institutes of Health, Department of Health and Human Services; 2006. | ||
Zhou SW, Huang YY, Wei Y, et al. No survival benefit from adding cetuximab or panitumumab to oxaliplatin-based chemotherapy in the first-line treatment of metastatic colorectal cancer in KRAS wild type patients: a meta-analysis. PloS One. 2012;7:e50925. | ||
Orditura M, De Vita F, Galizia G, et al. Correlation between efficacy and skin rash occurrence following treatment with the epidermal growth factor receptor inhibitor cetuximab: a single institution retrospective analysis. Oncol Rep. 2009;21:1023–1028. | ||
Racca P, Fanchini L, Caliendo V, et al. Efficacy and skin toxicity management with cetuximab in metastatic colorectal cancer: outcomes from an oncologic/dermatologic cooperation. Clin Colorectal Cancer. 2008;7:48–54. | ||
Stewart LA, Parmar MK. Meta-analysis of the literature or of individual patient data: is there a difference? Lancet. 1993;341:418–422. | ||
Bria E, Gralla RJ, Raftopoulos H, Giannarelli D. Comparing two methods of meta-analysis in clinical research-individual patient data-based (IPD) and literature-based abstracted data (AD) methods: analyzing five oncology issues involving more than 10,000 patients in randomized clinical trials (RCTs). J Clin Oncol. 2007;25: abstr 6512. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.