Back to Journals » Drug Design, Development and Therapy » Volume 9
Synthesis and characterization of a novel chemically designed (Globo)3–DTPA–KLH antigen
Authors Hajmohammadi M, Siadat SD, Ghorbani M , Shafiee Ardestani M , Teimourian S, Asgari V, Ahangari Cohan R, Hajmohammadi M, Hajmohammadi A, Behzadi R, Nezhad SR, Namvar Asl N
Received 10 August 2014
Accepted for publication 2 October 2014
Published 22 December 2014 Volume 2015:9 Pages 217—239
DOI https://doi.org/10.2147/DDDT.S72530
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Shu-Feng Zhou
Mehdi Hajmohammadi,1 Seyed Davar Siadat,2 Masoud Ghorbani,3,4,* Mehdi Shafiee Ardestani,5,* Shahram Teimourian,6 Vahid Asgari,3 Reza Ahangari Cohan,3 Mostafa Hajmohammadi,5 Akram Hajmohammadi,7 Ramezan Behzadi,8 Saied Rajabnezhad,9 Nabiollah Namvar Asl10
1Department of Research and Biotechnology, 2Department of Microbiology, 3Department of Virology, Pasteur Institute of Iran, Tehran Iran; 4Department of Virology, University of Ottawa, Ottawa, ON, Canada; 5Department of Radiopharmacy, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran; 6Department of Medical Genetics, Iran University of Medical Sciences, Tehran, 7Faculty of Pharmacy, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, 8Laboratory Animal Management of North Research Center, Pasteur Institute of Iran, 9Department of Research and Development, Alhavi Pharmaceutical, Tehran, 10Pasteur Institute of Iran, Department of Animal Sciences, Karaj, Iran
*These authors contributed equally to this work
Abstract: In recent years, many experiments have been conducted for the production and evaluation of anticancer glycoconjugated vaccines in developed countries and many achievements have been accomplished with Globo H derivatives. In the current experiment, a new chemically designed triplicate version of (Globo H)3–diethylenetriamine pentaacetic acid (DTPA)–KLH antigen was synthesized and characterized. Immunization with (Globo H)3-DTPA-KLH, a hexasaccharide that is a member of a family of antigenic carbohydrates that are highly expressed in various types of cancers conjugated with DTPA and KLH protein, induced a high level of antibody titer along with an elevated level of IL-4 in mice. Treatment of tumors with the collected sera from immunized mice decreased the tumor size in nude mice as well. None of the immunized mice illustrated any sign of tumor growth after injection of MCF-7 cells compared to the control animals. These findings, based on the newly presented structure of the Globo H antigen, lend exciting and promising evidence for clinical advancement in the development of a therapeutic vaccine in the future.
Keywords: (Globo H)3-DTPA-KLH, glycoconjugate vaccines, breast cancer
Introduction
Cancer is the second leading cause of death after heart disease worldwide, and is considered a dire threat to human life, with a probability of death in more than 50% of cases. Since most cancers show no clinical symptoms at their early stages of development, diagnosis is very difficult or impossible until they become metastatic, when cure becomes virtually impossible. Therefore, developing a vaccine against cancers to produce strong antibodies against small micrometastatic cells could decrease the chance of development of the cancer.1,2
The design of therapeutic vaccines for the treatment of cancers is underway in many laboratories. These vaccines are intended to shrink tumor cells and prevent their regeneration, as well as delay or stop cancer cell growth.3
Normally, the immune system sees the cancer cells as normal cells within the body, and does not consider them as dangerous or foreign. Therefore, it does not conduct a strong attack against them.4 There are many factors that help cancer cells to escape from the immune system. Cancer cells carry the normal self-antigens that help them to hide, as well as releasing some chemical messages that may suppress the activated anticancer immune responses, which allows them to escape even after being recognized by the immune system.5 Recent studies have indicated that carbohydrates have an obvious and undeniable role in causing tumor malignancy. Carbohydrates on the surface of tumor cells are usually present as glycoprotein, mussing, and glycosphingolipid.1 Researchers have recently discovered the presence of detectable antibodies in the serum of cancer patients. Their findings indicated a probable immunity and immune response against the cancerous factors and eventually cancer.6 In recent years, many experiments have been conducted for the production and evaluation of anticancer glycoconjugate vaccines, and many achievements have been accomplished.7 It appears that the production of an effective treatment vaccine is even more difficult than developing a cancer-preventive vaccine.8
It has been suggested that the ideal time for the immune system to respond against cancer cells could be the time immediately after the surgery and chemotherapy period, when the anticancer vaccine can train the immune system in order to identify and kill the remains of cancerous cells.6 If sufficient antibody titer is produced against antigens to omit tumor cells from the blood and lymphatic system and to kill micrometastatic cells, a noticeable change will appear in the curing methods of cancer patients.
Several preclinical studies have been performed to investigate the production of antibodies against carbohydrate antigens on the surface of tumor cells in order to reduce tumor severity.10,11 A number of clinical experiments have shown that prescribing monoclonal antibodies is clinically efficient.12,13 Naturally released antibodies against antigens on cell surfaces, especially carbohydrate antigens, have had a close relationship with the development of new cancer therapies at different clinical stages.14–16 So far, various studies have demonstrated that immunization against polysaccharide glycoconjugates is effective, while immunizing against polysaccharides alone is not.17–19
These findings indicate that polysaccharides could induce immunity, but need to be conjugated in order to be recognized by the immune system and provide immunity. Immunization with polyribosylribitol phosphate (PRP) induces immunity in children above 2 years old, whereas in children under 2 years old who are the target population for Haemophilus influenzae type B, PRP needs to be conjugated with another protein, such as tetanus toxin, for the production of PRP-T vaccine.20 Therefore, an effective vaccination against cancers probably requires tumor-specific glycoconjugates to provide strong immunity against tumor cells and their establishment in the body.9
One of the suggested proteins for conjugating polysaccharides to produce anticancer vaccines is the keyhole limpet hemocyanin (KLH) protein.21 KLH is a large, multisubunit, oxygen-carrying metalloprotein, and is found in the hemolymph of the giant keyhole limpet – Megathura crenulata.
One of the most recently suggested anticancer vaccines is a vaccine against breast cancer, which is the most common cancer in women. “Globo” is one of the known polysaccharides on the surface of cancerous breast cells (MCF-7). Globo can be conjugated with KLH protein to provide a complex that could have an anticancer feature in the humoral immune system against breast cancer.22
Zhu et al23 have been able to stimulate the immune system to elevate IgG and IgM levels against Globo-KLH along with lysis of MCF-7 cells. So far, several derivatives of Globo-KLH vaccine have been derived and evaluated. In all cases, only a single polysaccharide chain of Globo has been used, while the appearance of Globo on the cancer cell surface is a triple chain.
The aim of this project was to produce a newer form of Globo-KLH containing three sugar residues, similar to what appears in nature as a triple chain (Figure 1).
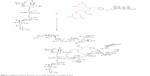  | Figure 1 Suggested chemical structure of a stronger derivation than Globo H KLH. |
Materials and methods
All animal protocols were reviewed and approved by the Pasteur Institute of Iran Animal Care Protocol Review Committee. All chemicals were purchased from Sigma-Aldrich, Canada, unless otherwise stated.
Forty female CB6F1 mice were purchased from the Pasteur Institute of Iran Animal Care facility. CB6F1 hybrid mice are the F1 progeny result of a cross between female BALB/c (H-2d) and C57BL/6 (B6; H-2b) mice. Mice were divided into five groups of eight, and injected with different vaccine regimens.
Synthesis of (Globo)3–diethylenetriamine pentaacetic acid–KLH
All substances were used without purification unless noted. Solvents were distilled under positive-pressure nitrogen stream: tetrahydrofuran (THF) with sodium benzophenone ketyl; ether with LiAlH4, CH2Cl2, and toluene; and benzene with CaH2. All reactions were under N2 pressure.
Column chromatography was performed using either silica gel 60 (40–63 μm) (Merck, Ottawa, Canada) or H-type silica gel (10–40 μm) for the normal phase. For the inverse phase, we used LiChroprep RP-18 (12–25 μm) (catalog number 113901; EMD Millipore, USA).
Synthesis of 3-6-di-O-benzyl-D-glucal
Both D-glucal (8 g, 54.7 mmol) and (Bu3Sn)2O (30.7 mL, 10 M equivalent) were refluxed for 20 hours in dry C6H6 (150 mL) with a Dean–Stark trap apparatus. The mixture was cooled to below boiling point and treated with benzyl bromide (BnBr) (21 mL) and tetra-n butylammonium bromide (TBABr) (35.3 g). The mixture was refluxed for 17 hours and cooled down and dried by evaporation. The residue was then diluted by ethyl acetate (EtOAc; 2×20 mL), and washed using distilled water (3×300 mL). It was then filtered, condensed, and dried by Na2So4. The column chromatography of raw materials was performed using 15%–20% EtOAc in hexanes, which resulted in the desired product (11.66 g, with 65% purity) in the form of a colorless oily residue.
Synthesis of 1,5-anhydro-6-O-(TIPS)-2-deoxy-D-lyxo-hex-1-enopyranose
Under a stream of nitrogen (N2), 6.1 g triisopropylsilyl chloride (TIPSCl; 31.8 mmol), 4.2 g d-glucal (28.8 mmol), and 4.3 g imidazole (63.2 mmol) was added to 20 mL dimethylformamide (DMF) at 0°C and mixed appropriately. The mixture was then kept at room temperature and stirred for 24 hours, followed by the addition of 250 mL EtOAc and 250 mL distilled water. After the separation of layers, the blue (aqueous) layer was extracted by the addition of more EtOAc (2×250 mL). The organic layer was dried later using Na2SO4, filtered, and concentrated under vacuum evaporation. The residue was then purified using column chromatography (eluent, 15%–30% EtOAc:hexane) to obtain the desired compound in the form of a light-yellow viscous solution.
Synthesis of 1,5-anhydro-6-O-(TIPS)-3,4-O-carbonyl-2-deoxy-D-lyxo-hex-1-enopyranose
The aforementioned solution (2.4 g, 14.8 mmol) was then mixed with 2.4 g carbonyldiimidazole (14.8 mmol) in 40 mL dry THF under N2 pressure. Three imidazole crystals were then added and the mixture stirred for 30 minutes, followed by the addition of 1.2 g carbonyldiimidazole (7.4 mmol). The mixture was stirred for 90 more minutes. After evaporation of the solvent under vacuum, the residue was mixed with 200 mL chloride and 100 mL distilled water. After separation of the organic layer, it was washed with 100 mL water, filtered, dried, and condensed using Na2SO4 under vacuum. The residue was then purified by column chromatography using a 15% EtOAc:hexane mixture to obtain the desired compound in the form of a clear syrup.
Synthesis of lactal carbonate 1
The syrup (22 mg) was mixed with 5 mL methylene chloride, followed by the addition of 30 mL dimethyldioxirane (prepared on the same day, as explained by Robert et al)24 slowly in drops at 0°C. The mixture was then stirred for 1 hour at 0°C and analyzed by thin-layer chromatography to verify the complete expansion of the compound. Then, the mixture was condensed under vacuum and azeotroped using 5 mL benzene. Then, 11.6 g of 3-6-di-O-benzyl-D-glucal solution was added and the mixture azeotroped again with 2×5 mL benzene, concentrated under vacuum, and dried using 1.5 mL THF under N2 pressure. A zinc chloride solution (820 μL of a 1 M solution in THF, 0.82 mmol) was added slowly in drops and the mixture returned to room temperature and kept for 24 hours. The mixture was then added to 10 mL NaHCO3 (1 M) solution and extracted by EtOAc (3×20 mL). The extracted organic compound was then filtered, condensed, dried by Na2SO4, and purified by column chromatography (gradient elution, 10%–25% EtOAc:hexanes) to obtain the desired compound.
Synthesis of lactal carbonate
One milliliter of a 60% solution of NaOH in water was added to 30 mL DMF containing 2.7 g of lactal carbonate 1 and 574 μL BnBr at 0°C, followed by stirring for 5 minutes. The mixture was then kept at room temperature and stirred for 2 hours. The total mixture content was later transferred to 70 mL cold distilled water, diluted by EtOAc, washed twice with 70 mL distilled water and once with 70 mL salt water, filtered and dried by Na2SO4, followed by column chromatography of raw materials using 10%–12% EtOAc in hexanes to obtain the desired compound (2.479 g) with purity of 81% in the form of a colorless oil (Figure 2).
  | Figure 2 Synthesis of lactal carbonate. |
Synthesis of lactal
A lactal carbonate solution of TIPS (5.62 mmol, 4.28 g) was treated with 6.75 mL tetra-n-butylammonium fluoride (TBAF; 1.0 M equivalent, 1.0 M) solution mixed with 25 mL THF and 5 mL MeOH. After 6 hours of incubation at room temperature, 4 mL more TBAF was added and the mixture stirred for an additional 3 hours. The mixture was then condensed and subjected to direct chromatography using 4:1 EtOAc-hexane solution to obtain 2.20 g triol. The remains of the solution contained cyclic carbonate and a mixed carbonate mixture that was hydrolyzed in methanol using MeONa (1.0 mL, 25 wt% in MeOH) and purified by chromatography. The total productivity was 3.02 g (93%). The final product was then used directly in the dibenzylation stage.
Synthesis of disaccharide
A mixture containing 2.95 g thioglycol (5.1 mmol), 1.33 g Bu2SnO (1.05 equivalent), and 1.69 mL (Bu3Sn)2O (0.65 equivalent) was refluxed for 5 hours in dry C6H6 (50 mL) under N2 pressure until all the water was evaporated. The mixture was then cooled to under boiling point, treated with 2.43 mL BnBr (4.0 molar equivalent) and 3.29 g TBABr (2.0 equivalent), and refluxed with 10 mL C6H6 for 16 hours. The mixture was loaded directly on a silica bar and eluted by 15%–20% EtOAc-hexanes to obtain the desired compound (3.48 g) with 91% purity in the form of clear oil.
Synthesis of 3-O-(4-methoxybenzyl)-D-glucal
A suspension of 3.70 g D-glucal (25.3 mmol) and 6.30 g Bu2SnO (1.0 equivalent) was refluxed for 5 hours in dry C6H6 (50 mL) under N2 pressure until the water was evaporated completely. The mixture was cooled down to room temperature and treated with 9.1 g p-methoxybenzyl chloride (1.1 equivalent) and 9.1 g TBABr (1.1 equivalent, 9.10 g) and refluxed for 4 hours. The mixture was then filtered through a silica bar and eluted with EtOAc:hexanes (4:1). Fractions containing product were condensed and the residues crystallized in hexane to obtain a white solid crystal as product (4.5 g, approximately 70%).
4,6-Di-O-benzyl-3-O-(4-methoxybenzyl)-D-glucal synthesis
A mixture of 3-O-(4-methoxybenzyl)-D-glucal (8.56 mmol, 2.28 g) and BnBr (3.68 molar equivalent, 3.75 mL) solution was prepared and passed through basic alumina treated by NaH 60% (4.0 molar equivalent, 1.37 g) in dry DMF (30 mL) under N2 pressure at 0°C. The mixture was stirred for 30 minutes at 0°C and for 1 hour at room temperature. The mixture was then poured on ice pieces, diluted by 100 mL of distilled water, and extracted by EtOAc-hexanes (1:1, 3×100 mL). Organic extracts was washed by water (2×100 mL) and dried on Na2SO4. Column chromatography of raw materials was performed using 15% EtOAc-hexanes, and resulted in obtaining the desired product in the form of a clear liquid.
Synthesis of the compound containing fluoride
A mixture of 4,6-di-O-benzyl-3-O-(4-methoxybenzyl)-D-glucal (7.7 mmol, 3.20 g) solution in dry CH2Cl2 (10 mL) was treated with dimethyldioxirane (0.09 M, 80 mL) under N2 pressure at 0°C. The mixture was stirred until the whole glucal was expanded using thin-layer chromatography containing 30% EtOAc in hexanes. The solution was deazeotroped under N2 pressure at 0°C. The residue was then dissolved in dry THF (30 mL) under N2 pressure at 0°C, followed by treatment with 36 mL TBAF (stored over molecular sieves) and stirred for 20 hours at room temperature. The dark-brown solution was filtered through a silica pad (∼4 cm depth) and washed with 200 mL EtOAc, followed by washing twice with 200 mL distilled water, and dried with MgSO4. The residue was then dissolved in 30% EtOAc in hexanes (50 mL) and filtered through a silica bar (10 cm diameter, 4 cm length), and washed again with 1 L of the same solvent. The filtrate was condensed to obtain fluorohydrine in high purity. The residue was then dissolved in dry DMF (30 mL) under N2 pressure at 0°C, treated with BnBr (1.5 equivalent, 958 μL), filtered through basic alumina, and finally was stirred for 30 minutes at 0°C followed by 30 minutes’ stirring at room temperature with NaOH (1.5 equivalent, 60% dispersion, 322 mg). The reaction was stopped after the mixture was poured on 100 g of ice, and the mixture was extracted by EtOAc-hexanes 1:1 (2×150 mL). Column chromatography was performed using 10% EtOAc in hexanes, which resulted in obtaining 2 g (49%) of desired compound in the form of a light-yellow liquid (Figure 3).
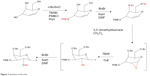  | Figure 3 Synthesis of fluoride. |
Synthesis of the first trisaccharide containing para-methoxybenzyl
The synthesized lactal (1.0 equivalent, 0.791 mmol, 600 mg) and fluorosugar (1.5 equivalent, 679 mg) were synthesized in ether, condensed, dried in vacuum for 2 hours, treated by di-tert-butylpyridine (1.0 equivalent, 177 mL), and dissolved in dry ether (8.5 mL) under N2 pressure. In a separate 25 mL flask, 2 g of 4 Å molecular sieves was added and dried by heat under vacuum and cooled to room temperature. Anhydrous silver perchlorate (1.0 equivalent, 163 mg) and SnCl2 (1.0 equivalent, 150 mg) was added to molecular sieve. The salt mixture was placed in a water bath, and the sugar solution was added to the salt using a double-tipped needle and stirred for 48 hours at room temperature. The mixture was diluted by ether and filtered through a pad of silica, followed by washing with ether. The filtrate (70 mL) was diluted twice with 50 mL sodium bicarbonate solution and dried under Na2SO4. Column chromatography was performed twice using 20% EtOAc in hexanes to obtain 561 mg trisaccharides with a purity of 54%.
Synthesis of the first trisaccharide without para-methoxybenzyl
A para-methoxybenzyl trisaccharide solution containing 0.428 mg trisaccharide (561 mmol) was treated with 830 μL of distilled water and 126 mg 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (1.3 equivalent) at 0°C and stirred for 1 hour at 0°C. The mixture was transferred to 50 mL saturated sodium bicarbonate solution, extracted by EtOAc (2×50 mL), washed with water and saturated sodium bicarbonate solution (2×50 mL), dried with Na2SO4, condensed under vacuum, and purified using column chromatography as previously described to obtain 437 mg of the desired trisaccharide in the form of a colorless oil with 86% purity (Figure 4).
  | Figure 4 Synthesis of trisaccharide. |
Glycal disaccharide synthesis
TIPS galactal carbonate (3.14 mmol, 4.32 g) was dissolved in 20 mL of CH2Cl2 and cooled to 0°C. The mixture was then mixed with 219 mL dimethyldioxirane (∼3.14 mmol) and incubated at 0°C for 20 minutes, followed by condensation using a stream of nitrogen. The residue was then treated with 20 mL C6H6 and dried by high vacuum power for 30 minutes at 0°C. It was then dissolved in 60 mL THF and cooled to −78°C, followed by the addition of 3.32 g (10.95 mmol) dry TIPS galactal carbonate and 26.3 mL ZnCl2 (1.0 M in ether). The mixture was kept at room temperature and stirred for 24 hours. It was then treated with 40 mL saturated NaHCO3 and extracted with 500 mL ether. The combined organic phase was washed with 300 mL salt water, dried by MgSO4, and the raw product was purified by column chromatography using 20% EtOAc in hexanes. The resulting product was 6.20 g white foam.
Glycal trisaccharide synthesis
A mixture of disaccharide (4.08 mmol, 2.64 g) and fucosyl fluoride (3.77 mmol, 1.64 g) was deazeotroped three times by 10 mL C6H6 and dried by high vacuum power for 1 hour. The residue was then dissolved in 20 mL THF and treated with 2,6-di-tert-butylpyridine (11.31 mmol, 2.16 g). The aforementioned mixture was later added to a flask containing AgClO4 (7.54 mmol, 1.56 g), SnCl2 (7.54 mmol, 1.42 g), and 4 Å molecular sieves (4.0 g), and stirred for 30 minutes at 40°C for 34 hours. After the mixture was treated with 40 mL saturated solution of NaHCO3 at 4°C, the mixture was extracted twice with 300 mL EtOAc. The contents of the organic layer were then washed with 200 mL salt water and dried by MgSO4. The residue was then purified using column chromatography as stated earlier to obtain 1.93 g of 47% purified glycal trisaccharide (Figure 5).
  | Figure 5 Synthesis of iodosulfonamide. |
Synthesis of iodosulfonamide
A mixture of glycal trisaccharide (0.11 mmol, 118 mg) and benzenesulfonamide (0.55 mmol, 87 mg) was deazeotroped once by C6H6, dried by high vacuum for 1 hour, and dissolved in 5 mL THF, and then 870 mg freshly activated 4 Å molecular sieves (provided from 4 hours heating in oven at 250°C) was added.
I2 (0.22 mmol, 57.5 mg) was sterilized by 100 mg Ag(sym-coll)2 ClO4 (0.22 mmol) in 1 mL THF at room temperature until the brown color of iodine had disappeared and I(sym-coll)2 ClO4 was obtained. It was then added to the flask containing glycal trisaccharide and benzenesulfonamide at 5°C. The mixture was stirred for 1 hour at 5°C and cooled using 10 mL Na2S2O3 solution. After filtration and extraction by EtOAc (80 mL), the organic layer was washed with 20 mL deuterium-depleted water, saturated with 20 mL Cu2SO4, dried by MgSO4, and purified by column chromatography using silica gel (20% EtOAc in hexanes) to obtain 74 mg iodosulfonamide as the result.
Ethyl-thio-sulfonamide synthesis
Lithium bis(trimethylsylil)amide (1.0 M in THF, 0.81 mL, 0.81 mmol) was added to 10 mL DMF containing etiolate Ethan solution (1.7 mmol, 102 mg) at −42°C. After 5 minutes’ stirring, it was added to a flask containing iodosulfonamide (0.33 mmol, 448 mg) in 10 mL DMF at −42°C. The mixture was then transferred to room temperature and stirred for another 3 hours. It was then diluted with 200 mL diethyl ether, washed with 10 mL salt water and saturated with 1 N NaHCO3 2×10 mL and dried by MgSO4. The desired product was obtained in the form of a white solid (Figure 6).
  | Figure 6 Synthesis of ethyl-thio-sulfonamide. |
Hexasaccharide synthesis
An acceptor mixture containing trisaccharides (1 equivalent, 0.077 mmol, 92 mg), thioglycoside (2 equivalent, 198 mg), and activated molecular sieve (560 mg) suspended in Et2O-CH2Cl2 (2:1) under N2 flow was stirred at room temperature for 10 minutes. The mixture was then cooled to 0°C and treated with 52.4 μL methyl triflate (6.0 equivalent). The mixture was then stirred for 4 hours at 0°C and for 1.5 hours at about 15°C (mild temperature). The reaction was later stopped using 10 mL triethylamine. The mixture was then washed and filtered through a silica pad using diethyl ether. The filtrate (70 mL) was washed again with 2×50 mL saturated NaHCO3 solution, dried with Na2SO4, and purified by high-performance liquid chromatography (HPLC) using a MICROSORB Semi-prep Si 80-120-C5 column (Bio-Rad, California, USA), 17% EtOAc in hexanes, at a flow rate of 15 mL/minute and optical density of 260 nm to obtain 158 mg (85% purity) of the desired product.
Synthesis of azide
A mixture of hexasaccharide glycal and freshly activated 4 Å molecular sieve were treated with 1mL dimethyldioxirane (0.07 M) in 1 mL CH2Cl2 at 0°C under nitrogen stream, and the residue was dried for 20 minutes under high vacuum power. Then, an azidohydrin solution was added to epoxide through a container in THF (0.8 mL) and cooled to −40°C. Then, ZnCl2 in Et2O (1 M, 25 μL) was added, the mixture allowed to cool to room temperature, and then stirred for the next 12 hours. Then, the mixture was diluted by EtOAc (50 mL), was washed with NaHCO3 saturated solution (2×20 mL) and salt water (10 mL), dried by Na2SO4, and the raw material purified by column chromatography (10%–22% EtOAc in hexanes) to provide 31.3 mg of coupled product in the form of a clear oil. To the solution of this product (0.013 mmol, 31.3 mg), 4-dimethylaminopyridine (0.013 mmol, 1.6 mg) and triethylamine (0.0219 mmol, 3.0 μL) was added in CH2Cl2 (1 mL) and Ac2O (0.013 mmol, 1.2 μL) at 0°C. The mixture was allowed to cool to room temperature and stirred for 1 hour. After the solvent had been evaporated, the residue was diluted by EtOAc (30 mL) and was washed with NaHCO3 diluted solution (285 mL) and salt water and dried by Na2SO4. Column chromatography (20% EtOAc in hexanes) of raw materials provided 30 mg (95%) of the desired product (Figure 7).
  | Figure 7 Synthesis of azide. |
Synthesis of amide
A mixture containing 66 mg azide (0.023 mmol), 66 mg Lindlar catalyst, and 23 mg palmitic anhydrate (0.46 mmol) was stirred for 24 hours at room temperature under H2. The reaction mixture was filtered through a silica pad, washed with 20 mL EtOAc, and concentrated under vacuum. The residue was then purified by HPLC using 20% EtOAc in hexanes at a flow rate of 15 mL/minute and detected at 260 nm wavelength using an ultraviolet detector. The final product was 64 mg colorless oil with 90% purity (Figure 8).
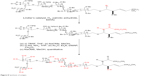  | Figure 8 Synthesis of antigen. |
Synthesis of the final product of (Globo)3-DTPA-KLH
Approximately 1 mmol of KLH (3 g) was mixed with 2 mmol of S-2-(4-Isothiocyanatobenzyl)-diethylenetriamine pentaacetic acid (p-SCN-Bn-DTPA) in phosphate-buffered saline (PBS) and stirred for 24 hours. The mixture was then transferred to a dialysis bag for separation of SCN-DTPA-KLH. The remainder was then added to a mixture containing 10 mL of dissolved DMF, acetic acid anhydride (C4H6O3), and 10 mg pyridine while stirring for 3 hours at 40°C, until the formation of anhydrate. Ten milligrams of 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide plus 2 mL triethylamine were then added to the mixture, the contents placed on ice for 2 hours, and kept at room temperature afterward. The synthesized Globo was later dissolved in 10 mL DMF and the pH adjusted to 10.5 using 1 normal NaOH. After the addition of 125 μL of cyanogen bromide solution (0.2 g in 1 mL H2O) to acetonitrile, the pH was adjusted to 10.5. After 2.5 minutes’ incubation at room temperature, 5 mL of 0.5 molar adipic dihydrazide as well as 5 mL bicarbonate (0.5 molar) were added to the mixture and the pH adjusted to 8.5 using 1 N HCl and kept at 4°C for 24 hours. The activated Globo was then added to the anhydrate and stirred for 2 hours. The conformation of the final product was later determined using 1H nuclear magnetic resonance (NMR), Fourier-transform infrared, and HPLC techniques.
Animal and biological assays
Immunization of mice with vaccine regimens
Mice were divided into five groups of eight, and received different vaccine regimens in 200 μL via intraperitoneal injections. Each mouse received the vaccine four times at 2-week intervals. Group 1 received (Globo)3-DTPA-KLH, group 2 (Globo)3-DTPA-KLH + adjuvant, group 3 was injected with DTPA-KLH as negative control, group 4 received DTPA-KLH + adjuvant, and group 5 was injected with PBS alone.
All animals were monitored for 4 months. All mice were bled prior to the beginning of the experiment and after each immunization. The blood was collected from the saphenous vein in an Eppendorf tube and centrifuged at 1,500 rpm for 15 minutes. The sera were collected and kept at −20°C until use.
Antibody titration
The level of antibodies in the mice sera was detected using enzyme-linked immunosorbent assay (ELISA). The wells of a 96-well plate were coated with either 0.25 μg/well of bovine serum albumin or Globo-DTPA-albumin conjugate separately and incubated overnight. All wells were blocked with 1% skim milk for 1 hour at 37°C. A serial dilution of polled sera (1:50, 1:100, 1:300, 1:900, 1:2,700, and 1:8,100) from each group of mice was prepared, added to the wells, and incubated at 37°C for 1 hour. The plate was then washed three times with PBS containing 0.1% Tween 20. The antibodies were detected using a rabbit antimouse antibody conjugated with horseradish peroxidase at a concentration of 1:1,000 and incubated for 1 hour at 37°C. The plate was then washed three times and 100 μL TMB added to each well and incubated for 15 minutes in the dark. The reaction was stopped by the addition of 100 μL of 0.5 M sulfuric acid to each well, and light intensity was read at 450 nm wavelength using a Bio-Rad ELISA reader.
Cytokine assay
Both IL-4 and IFNγ were detected using ab100710–IL-4 (IL-4) and ab46081-IFNγ mouse ELISA kits (Abcam, Cambridge, MA, USA), respectively, for the detection of cytokines in collected sera from immunized mice.
In vivo tumor-preventive effects
To investigate the antitumor effect of the antibodies in the sera of immunized mice, tumors were developed in two groups of four nude mice (purchased from the Pasteur Institute of Iran), and 20×106 MCF-7 cells were injected into the right limb of each mouse. After the tumor size reached approximately 1 cm, 200 μL of the pooled sera from either the vaccinated mice or the control group was injected around the tumor site in each nude mouse subcutaneously. The tumor size and growth were then monitored during the next 4 weeks.
Statistical analysis
Results are presented as means ± standard error of mean. We used InStat software for analyses of variance, followed by the Student–Newman–Keuls post hoc test. Significant differences are based on P<0.05.
Results
Confirmation of the synthesis of Globo H and Globo H-protected precursor
1H-NMR (solvent, CDCl3)
δ7.67 (2H, d, J =7.2 Hz), 7.35–7.13 (63H, m), 5.65 (1H, d, J =9.0 Hz), 5.63–5.57 (1H, m), 5.43 (1H, br s), 5.33–5.27 (1H, m), 5.11 (2H, t-l1ke, J =4.0 Hz), 4.94–4.90 (2H, m), 4.88–4.79 (4H, m), 4.76–4.73 (2H, m), 4.71–4.64 (5H, m), 4.62–4.58 (3H, m), 4.56–4.49 (5H, m), 4.44 (1H, dd, J =8.6 and 2.2 Hz), 4.40 (1H, d, J =7.0 Hz), 4.36–4.32 (2H, m), 4.30–4.11 (9H, m), 4.02–3.88 (7H, m), 3.81–3.65 (8H, m), 3.60–3.50 (4H, m), 3.45–3.36 (4H, m), 3.34–3.23 (5H, m), 3.09 (1H, br s), 2.10–1.98 (4H, m), 2.04 (3H, s), 1.83 (3H, s), 1.51 (2H, m), 1.30–1.23 (49H, m), 1.10–0.97 (42H, m), 0.88 (6H, t, J =6.9 Hz).
13C-NMR (solvent, CDCl3)
δ172.4, 169.6, 168.5, 153.4, 140.5, 139.3, 139.2, 138.7, 138.6, 138.5, 138.4, 138.3, 138.20, 138.1, 138.0, 136.8, 132.3, 128.9, 128.5, 128.3, 128.3, 128.2, 128.1, 127.9, 127.8, 127.7, 127.6, 127.5, 127.4, 127.3, 127.2, 127.1,127.0, 103.2, 101.1, 99.0, 98.4, 98.1, 79.4, 79.3, 78.8, 77.4, 76.5, 76.3, 76.0, 75.8, 75.2, 74.8, 74.8, 74.3, 74.2, 74.1, 73.5, 73.1, 73.0, 72.9, 72.7, 72.4, 72.3, 72.2, 70.3, 70.2, 69.5, 68.7, 68.3, 68.1, 68.0, 67.9, 67.7, 61.2, 60.9, 53.6, 53.3, 51.5, 36.8, 32.2, 31.9, 29.6, 29.5, 29.4, 29.3, 29.2, 25.6, 22.6, 20.9, 20.7, 18.0, 17.9, 17.9, 17.9, 16.6, 14.0, 11.8 (Figure 9).
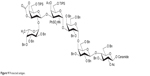  | Figure 9 Protected antigen. |
Globo H (ceramide)
Fourier transform infrared spectroscopy (FTIR) data (KBr)
3,356.16, 2,920.73, 2,851.92, 1,734.56, 1,652.90, 1,547.93, 1,466.19, 1,372.94, 1,260.95, 1,075.45, 1,042.58 cm−1.
1H NMR (solvent, DMSO-4/D2O, 10:1)
δ5.54–5.48 (dt, J =6.7, 15.3 Hz, 1H), 5.37–5.31 (dd, J =7.0, 15.2 Hz, 1H), 4.95 (bs, 1H), 4.81 (d, J =3.7 Hz, 1H), 4.48 (d, J =8.4 Hz, 1H), 4.44 (d, J =8.1 Hz, 1H), 4.25 (d, J =7.6 Hz, 1H), 4.16 (d, J =7.8 Hz, 1H), 4.10–4.04 (m, 2H), 3.98–3.85 (m, 5H), 3.81–3.72 (m, 6H), 3.68–3.35 (m, 22H), 3.32–3.25 (2H, m), 3.04–3.02 (t, J =7.9 Hz, 1H), 2.02 (m, 2H), 1.93 (m, 2H), 1.82 (s, 3H), 1.44 (m, 2H), 1.23 (m, 46H), 1.08 (d, J =6.4, 3H), 0.84–0.83 (t, J =6.6, 6 H).
13C-NMR (solvent, CD3OD)
δ176.0, 174.5, 135.2, 131.4, 105.5, 105.4, 104.4, 103.9, 102.8, 101.0, 81.1, 80.4, 80.0, 79.1, 77.9, 76.8, 76.5, 76.4, 76.2, 75.5, 74.9, 74.7, 73.5, 73.0, 72.6, 72.4, 71.5, 70.6, 70.6, 70.3, 69.9, 69.7, 69.7, 68.1, 62.6, 62.6, 61.7, 61.6, 54.8, 54.8, 53.1, 37.4, 33.5, 33.1, 30.9, 30.9, 30.8, 30.8, 30.8, 30.7, 30.7, 30.6, 30.5, 30.5, 30.4, 27.2, 23.8, 23.7, 16.7, 14.4.
Mass-spectrum data
[M+]=1,532.6, [M+Na]=1,555.8, [M+2H]2+=778.95, [M+3H]3+=519.6 (Figure 10; original data in Supplementary materials).
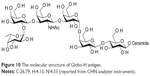  | Figure 10 The molecular structure of Globo-H antigen. |
(Globo H)n=1,3-p-Bn-SCN-DTPA-KLH synthesis confirmation
HPLC procedure
To confirm the attachment of Globo3 to the complex of DTPA-KLH, we used HPLC (Table 1).
CHN analysis
Carbon, hydrogen, and nitrogen analysis showed the total mass of each element residue in the complex, as indicated in Table 2.
  | Table 2 Atomic mass for carbon, hydrogen, and nitrogen elements |
C% calculation = (12.0107× carbon numbers/molecular weight [MW] ×100
H% calculation = (1.0079× hydrogen numbers/MW) ×100
N% calculation = (14.0087× nitrogen numbers/MW) ×100
Calculation of C, H and N numbers from KLH-CHN analysis obtained the data:
C:26.73, H:4.12, N:4.35 (reported from CHN analyzer instrument).
KLH MW = ∼3,000 KDa
C-numbers =26.73×3,000,000/1,201.07=∼66,765
H-numbers =4.12×3,000,000/100.79=∼122,631
N-numbers =4.33×3,000,000/1,400.87=∼9,268
Calculation of C, H, and N numbers from (Globo)3-KLH-CHN analysis obtained the data:
C:26.79, H:4.13, N:4.33 (reported from CHN analyzer instrument).
We knew that in a conjugate, there is an increase in C, H, and N numbers as follows, respectively. In other words, we expected an increase in CHN numbers as: C =258, H =484, and N =22 (new MW = KLH MW +∼5.7 kDa).
C-numbers =26.79×3,005,700/1,201.07=∼67,042
H-numbers =4.13×3,005,700/100.79=∼123,162
N-numbers =4.33×3,005,700/1,400.87=∼9,292
%C-numbers = (67,042–66,765/258) ×100–100=7.3%
%H-numbers = (123,162–122,631/484) ×100–100=10.0%
%N-numbers = (9,292–9268/24) ×100=9.0%
The data statistically confirmed the synthesis of our conjugates.
1H NMR study
As the original results depict in the Supplementary materials, there was a significant sharp peak regarding the anomeric hydrogens of Globo H polysaccharide (4.5–5.5 ppm) in (Globo H)3-DTPA-KLH and Globo H-KLH with an integral ratio of 3:1, which confirmed the presence of 3 M Globo H in the conjugate of (Globo H)3-DTPA-KLH as well.
Immunization results
A significant total of IgM and IgG antibodies was observed only against (Globo H)3-DTPA-KLH and Globo H-KLH compared to other groups (Figure 11). There was also a significant difference (P<0.05) between (Globo H)3-DTPA-KLH and Globo H-KLH. In general (Globo H)3-DTPA-KLH was found to be more potent than Globo H-KLH.
  | Figure 11 Antibody titration. |
Cytokine assay
IL-4 and IFNγ levels were determined in the collected sera after each injection for all groups. Surprisingly, the cytokines were only detected in the mouse group that received (Globo H)3-DTPA-KLH after the third and fourth injections, as listed in Table 3 (Figure 12).
  | Table 3 Amount of IL-4 and IFNγ in the vaccine-administered group after third and fourth injections |
  | Figure 12 Concentration of IL-4 and IFNγ in immunized mice. |
Tumor growth
None of the immunized mice showed any sign of cancer after the injection of MCF-7 cells compared to the control animals (see Supplementary materials).
Discussion
For successful treatment of a tumor or cancer cells, it is ideal to find molecular targets that are not present on normal cells. Meezan et al25 were among the first to demonstrate the differences of tumor glycans from normal cells. It appears that the glycosylation of sugar residue on cancer cells is structurally atypical to that of healthy cells, due to overexpression of specific structures and omission of some others.26 Sugar-residue association with tumors has also been identified recently by monoclonal antibodies and mass spectrometry.27 Association of glycolipids or glycoproteins has also been reported on the surface of certain types of cancer cells.28 The role of surface carbohydrates and its correlation with tumor malignancy is not known yet. However, it appears that these antigens could activate immune responses. One of the carbohydrates associated with tumors was first isolated from breast cancer MCF-7 cells and called Globo H by Hakomori7 and Bremer et al.29 It is now known that Globo H is present on many cancer cells, including prostate, ovary, lung, pancreatic, gastric, and colon cancers,24 and high levels of anti-Globo H antibody can be detected in serum samples of breast cancer patients.30,31 These findings indicate that Globo H could be used as a potential target in cancer therapy and cancer-vaccine development.
In this study, for the first time, a new chemically designated three-branch structure of Globo H antigen conjugated with KLH was synthesized and characterized. The novelty of this structure was regarding the triplicate structure of Globo H, which is highly similar to the nature of this antigen at its binding sites on cancer cells.17 The next advantage of the proposed structure is indicated by its polyamine linker DTPA, which facilitates its presentation to Globo H-binding sites at the cancerous cell surface as well.18,19 Kudryashov et al among others, previously reported the preparation of Globo H-KLH vaccine.22,24,32 They also investigated the immunogenicity of the Globo H-KLH antigen after ozonolysis of the Globo H aglycone that was followed by reductive amination with the KLH carrier protein to generate about 150 carbohydrate units per protein.24
Globo H is a cancer antigen overexpressed in various epithelial cancers. It has been suggested that this antigen can serve as a target in cancer immunotherapy. While vaccines have been developed to elicit antibody responses against Globo H, their anticancer efficacies are unsatisfactory, due to the low antigenicity of Globo H. There is a need for a new vaccine capable of eliciting high levels of immune responses targeting Globo H.
In this study, we successfully synthesized a novel three branch Globo H antigen conjugated with a DTPA-KLH molecule. The triplicate Globo H structure allows this antigen to perform with the same antigenic effects as the sugar residue on tumor cells, and therefore to induce a better immune response against cancer cells.
Administration of (Globo H)3-DTPA-KLH in mice subcutaneously, elevated the level of both IgM and IgG antibody titers significantly (Figure 11). In the meantime, IL-4 and IFNγ levels were also elevated significantly in the sera of vaccinated mice with (Globo H)3-DTPA-KLH compared with the control mice (Figure 12). This phenomenon was confirmed after immunotherapy of nude mice with the sera collected from immunized animals, which prevented the growth of tumor cells or reduced the size of tumors that were already developed (Figure 13).
  | Figure 13 Nude mice injected with pooled sera. |
Altogether, we concluded that the new synthesized (Globo H)3-DTPA-KLH could be a promising vaccine for treatment of cancers. More studies are required to find the best adjuvant to accompany this vaccine to elicit the best immune response against cancer cells. For mass spectrometry results and further information related to the structures of the Globo-H compound, please refer to Supplementary Figures S1–S10 at the end of the article.
Disclosure
The authors report no conflicts of interest in this work.
References
Wilson RM, Warren JD, Querfelli Q, Danishefsky SJ. Synthetic carbohydrate-based antitumor vaccines. In: Roy R, editor. Carbohydrate-Based Vaccines. Washington: American Chemical Society; 2008:258–292. | ||
Brossart P, Keilholz U, Willhauck M, Scheibenbogen C, Möhler T, Hunstein W. Hematogenous spread of malignant melanoma cells in different stages of disease. J Invest Dermatol. 1993;101(6):887–889. | ||
Ghossein RA, Scher HI, Gerald WL, et al. Detection of circulating tumor cells in patients with localized and metastatic prostatic carcinoma: clinical implications. J Clin Oncol. 1995;13(5):1195–1200. | ||
Hoon DS, Wang Y, Dale PS, et al. Detection of occult melanoma cells in blood with a multiple-marker polymerase chain reaction assay. J Clin Oncol. 1995;13(8):2109–2116. | ||
Lloyd KO. Humoral immune responses to tumor-associated carbohydrate antigens. Semin Cancer Biol. 1991;2(6):421–431. | ||
Feizi T. Demonstration by monoclonal antibodies that carbohydrate structures of glycoproteins and glycolipids are onco-developmental antigens. Nature. 1995;314(6006):53–57. | ||
Hakomori S. Tumor-associated carbohydrate antigens. Annu Rev Immunol. 1984;2:103–126. | ||
Pozsgay V, Kubler-Kielb J. Conjugation methods toward synthetic vaccines. In: Roy R, editor. Carbohydrate-Based Vaccines. Washington: American Chemical Society; 2008:36–70. | ||
Baurain JF, Van der Bruggen P, Van den Eynde BJ, Coulie PG, Van Baren N. General principles and first clinical trials of therapeutic vaccines against cancer. Bull Cancer. 2008;95(3):327–335. | ||
Livingston PO. The case for melanoma vaccines that induce antibodies. In: Kirkwood JM, editor. Molecular Diagnosis, Prevention, and Treatment of Melanoma. New York: Marcel Dekker; 1998:39–157. | ||
Zhang H, Zhang S, Cheung NK, Ragupathi G, Livingston PO. Antibodies against GD2 ganglioside can eradicate syngeneic cancer micrometastases. Cancer Res. 1998;58(13):2844–2849. | ||
Fung PY, Madej M, Koganty RR, Longenecker BM. Active specific immunotherapy of a murine mammary adenocarcinoma using a synthetic tumor-associated glycoconjugate. Cancer Res. 1990;50(14):4308–4314. | ||
Jones PC, Sze LL, Liu PY, Morton DL, Irie RF. Prolonged survival for melanoma patients with elevated IgM antibody to oncofetal antigen. J Natl Cancer Inst. 1981;66(2):249–254. | ||
Livingston PO, Ritter G, Srivastava P, et al. Characterization of IgG and IgM antibodies induced in melanoma patients by immunization with purified GM2 ganglioside. Cancer Res. 1989;49(24 Pt 1):7045–7050. | ||
Livingston PO, Wong GY, Adluri S, et al. Improved survival in stage III melanoma patients with GM2 antibodies: a randomized trial of adjuvant vaccination with GM2 ganglioside. J Clin Oncol. 1994;12(5):1036–1044. | ||
MacLean GD, Reddish MA, Koganty RR, Longenecker BM. Antibodies against mucin-associated sialyl-Tn epitopes correlate with survival of metastatic adenocarcinoma patients undergoing active specific immunotherapy with synthetic STn vaccine. J Immunother Emphasis Tumor Immunol. 1996;19(1):59–68. | ||
Springer GF. Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J Mol Med. 1997;75(8):594–602. | ||
Dalmau J, Gultekin HS, Posner JB. Paraneoplastic neurologic syndromes: pathogenesis and physiopathology. Brain Pathol. 1999;9(2):275–284. | ||
Porter CW, Miller J, Bergeron RJ. Aliphatic chain length specificity of the polyamine transport system in ascites L1210 leukemia cells. Cancer Res. 1984;44(1):126–128. | ||
Seiler N. Thirty years of polyamine-related approaches to cancer therapy. Retrospect and prospect. Part 2. Structural analogues and derivatives. Curr Drug Targets. 2003;4(7):565–585. | ||
Pohjanpelto P. Putrescine transport is greatly increased in human fibroblasts initiated to proliferate. J Cell Biol. 1976;68(3):512–520. | ||
Kudryashov V, Ragupathi G, Kim L, et al. Characterization of a mouse monoclonal IgG3 antibody to the tumor-associated Globo H structure produced by immunization with a synthetic glycoconjugate. Glycoconj J. 1998;15(3):243–249. | ||
Zhu J, Warren JD, Danishefsky SJ. Synthetic carbohydrate-based anticancer vaccines: the Memorial Sloan-Kettering experience. Expert Rev Vaccines. 2009;8(10):1399–1413. | ||
Robert C, Guilpin C, Limoge A. Review of neural network applications in sleep research. J Neurosci Methods. 1998;79(2):187–193. | ||
Meezan E, Wu HC, Black PH, Robbins PW. Comparative studies on the carbohydrate-containing membrane components of normal and virus-transformed mouse fibroblasts II. Separation of glycoproteins and glycopeptides by Sephadex chromatography. Biochemistry. 1969;8(6):2518–2524. | ||
Gabius HJ. Biological information transfer beyond the genetic code: the sugar code. Naturwissenschaften. 2000;87(3):108–121. | ||
Shriver Z, Raguram S, Sasisekharan R. Glycomics: a pathway to a class of new and improved therapeutics. Nat Rev Drug Discov. 2004;3(10):863–873. | ||
Dube DH, Bertozzi CR. Glycans in cancer and inflammation – potential for therapeutics and diagnostics. Nat Rev Drug Discov. 2005;4(6):477–488. | ||
Bremer EG, Levery SB, Sonnino S, et al. Characterization of a glycosphingolipid antigen defined by the monoclonal antibody MBr1 expressed in normal and neoplastic epithelial cells of human mammary gland. J Biol Chem. 1984;259(23):14773–14777. | ||
Kim SK, Wu X, Ragupathi G, et al. Impact of minimal tumor burden on antibody response to vaccination. Cancer Immunol Immunother. 2011;60(5):621–627. | ||
Wang CC, Huang YL, Ren CT, et al. Glycan microarray of Globo H and related structures for quantitative analysis of breast cancer. Proc Natl Acad Sci U S A. 2008;105(33):11661–11666. | ||
Slovin SF, Keding SJ, Ragupathi G. Carbohydrate vaccines as immunotherapy for cancer. Immunol Cell Biol. 2005;83(4):418–428. |
Supplementary materials
  | Figure S1 Globo H mass spectroscopy. |
  | Figure S2 Protected Globo H 1H NMR. |
  | Figure S3 Protected Globo H 13C NMR. |
  | Figure S4 Globo H 1H NMR. |
  | Figure S5 Globo H 13C NMR. |
  | Figure S6 Globo H FTIR. |
  | Figure S7 (Globo H)3-DTPA-KLH HPLC. |
  | Figure S8 KLH 1H NMR. |
  | Figure S9 Globo H-KLH 1H NMR. |
  | Figure S10 (Globo H)3-DTPA-KLH 1H NMR. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

