Back to Journals » Drug Design, Development and Therapy » Volume 18
Natural and Synthetic Drugs Approached for the Treatment of Recurrent Aphthous Stomatitis Over the Last Decade
Authors Vitamia C, Iftinan GN , Latarissa IR, Wilar G , Cahyanto A , Elamin KM , Wathoni N
Received 14 November 2023
Accepted for publication 10 April 2024
Published 22 April 2024 Volume 2024:18 Pages 1297—1312
DOI https://doi.org/10.2147/DDDT.S449370
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Anastasios Lymperopoulos
Cszahreyloren Vitamia,1,2 Ghina Nadhifah Iftinan,3 Irma Rahayu Latarissa,4 Gofarana Wilar,4 Arief Cahyanto,5 Khaled M Elamin,6 Nasrul Wathoni1
1Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia; 2Departement of Pharmacy, Akademi Farmasi Bumi Siliwangi, Bandung, Indonesia; 3Center of Excellence for Pharmaceutical Care Innovation, Universitas Padjadjaran, Sumedang, Indonesia; 4Department of Pharmacology and Clinical Pharmacy, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia; 5Department of Restorative Dentistry, Faculty of Dentistry, Universiti Malaya, Kuala Lumpur, Malaysia; 6Graduate School of Pharmaceutical Sciences, Kumamoto University, Kumamoto, Japan
Correspondence: Nasrul Wathoni, Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, Universitas Padjadjaran, Jl. Raya Bandung Sumedang KM 21, Jatinangor, 45363, Indonesia, Tel +622 842 888888 3510, Fax +622 842 888888, Email [email protected]
Abstract: Recurrent aphthous stomatitis (RAS) refers to a sore and frequently recurring inflammation of the oral tissues, distinguished by the presence of small ulcers that cause significant discomfort and cannot be attributed to any underlying disease. Different treatments have been used for RAS. This review aims to provide a comprehensive overview of the treatment options over the past decade for recurrent aphthous stomatitis (RAS), encompassing both natural and synthetic treatments. It will utilize clinical efficacy studies conducted in vivo and in vitro, along with a focus on the pharmaceutical approach through advancements in drug delivery development. We conducted a thorough literature search from 2013 to 2023 in prominent databases such as PubMed, Scopus, and Cochrane, utilizing appropriate keywords of recurrent aphthous stomatitis, and treatment. A total of 53 clinical trials with 3022 patients were included, with 35 using natural materials in their research and a total of 16 articles discussing RAS treatment using synthetic materials. All the clinical trials showed that natural and synthetic medicines seemed to benefit RAS patients by reducing pain score, ulcer size, and number of ulcers and shortening the healing duration.
Keywords: recurrent aphthous stomatitis, drug therapy, natural products, treatment outcome, clinical trial
Introduction
RAS is an oral mucosa disease characterized by recurrent, painful, single or multiple well-demarcated ulcerations with peripheral red halo where healing occurs with or without scarring.1 In the adult population, 60–85% of patients experience their first ulceration before age thirty. Although the primary causes of RAS remain unknown, certain factors have been identified as triggers for RAS outbreaks, including mental stress, trauma, lack of sleep, iron and folic acid deficiencies, menstruation, anemia, and changes in women’s sex hormones.2,3
With a 39% frequency, children are more likely to experience it.4 The prevalence rises with the female gender and a higher socioeconomic level and falls with age.5 Numerous perspectives have proposed that the oral microbiota could be the cause of RAS. Among the possibly significant components of the core microbiota responsible for this condition are Streptococcus, Helicobacter pylori, CMV (Cytomegalovirus), and a host of other unknown microorganisms.6 The presence of ulcers as a significant phenotype in systemic illnesses such as Crohn’s disease, Behçet’s illness, herpes stomatitis, and ulcerative colitis further compounds the difficulty in determining the etiologic cause.7 Stress may contribute to the likelihood of developing RAS; hence, it is crucial to address stress management in patients as a means of controlling its recurrence.8
Numerous therapies for RAS have been studied, such as topical steroids, local anesthetics, topical antibiotics, antiseptics, and analgesic/anti-inflammatory medications. Not every patient will respond well to topical therapies, despite the fact that they can be useful.9 Several systemic drugs have been investigated to treat RAS, including colchicine, thalidomide, dapsone, and corticosteroid.10 The current therapeutic options can only lessen the frequency or severity of the lesions. In most cases, the primary goal of RAS treatment is to achieve pain reduction (Visual Analog Scale Score),11 decrease in ulcer size, decrease in ulcer count, and acceleration of ulcer healing duration.12
Herbal supplements and natural items have been used for a long time and are recommended as an alternative to systemic pharmaceuticals due to their potential side effects.13,14 For them to serve as a resource and be considered while developing a treatment for RAS, this literature review addresses the natural and synthetic drug-based therapies for RAS that have been demonstrated, as well as the results of their potential.
Methods
We searched journals with the keywords “Recurrent Aphthous Stomatitis”, and “Treatment”, in 3 journal sources: PubMed, Scopus, and Cochrane. This narrative review was prepared based on studies related to the implementation of natural or synthetic products for “Recurrent Aphthous Stomatitis” AND “Treatment”. The authors use all original research and clinical reports published from 2013 to September 2023 regarding the use of treatment in recurrent aphthous stomatitis. The authors exclude irrelevant articles, which are the results of clinical research, but the results do not evaluate the effect on the health of research subjects, RAS treatment with laser or other physical intervention, research articles that do not clearly state the interventions, and other sources such as book chapters, article abstracts only, conference reports, reviews, posters, discussion results, and articles that only contain research designs. Authors C.V. and G.N.I. examined all the articles identified for inclusion. The authors later discussed the findings to reach a consensus. The procedure for selecting research articles is shown in Figure 1.
Result
Numerous treatment methods have been reported to treat RAS. Natural and synthetic treatments are the most frequently reported among the various treatments. Other strategies involve physical interventions such as diode laser, Er, Cr:YSGG laser irradiation, and low laser therapy, to name a few.15–17 According to the results obtained from journal sources, treatment for RAS mainly uses topical medication in the form of gels, patches, ointments, and mouthwash.18 However, there are also therapies using oral drugs that work systemically. Data and explanations regarding natural and synthetic drug-based treatments used as RAS treatments are shown in Tables 1 and 2. The positive overall effect showed that the test materials produced a better outcome than the comparator or control material, as evidenced by the Negative overall impact, which indicates that the comparator or control materials outperformed the test materials, and not significant overall effect indicates that the materials indicated a better outcome with no significant difference with the comparator material.
 |
Table 1 Natural Materials |
 |
Table 2 Synthetic Materials |
 |
Figure 1 The Procedure for selecting research articles. |
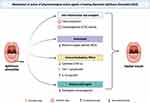 |
Figure 2 Mechanism of action for pharmacological active agents in RAS therapy. Created with Biorender.com. |
A total of 3072 RAS patients were studied in this review. The sample size ranged from 15 to 210 patients in each study. Twenty-one different types of formulations were used in the articles, such as pasta, tablet, injection, mouthwash, gel, nanomicelle gel, powder, pill, film, lozenges, patch, capsule, buccal film, liquid, ointment, membrane piece, oil, gelatin, orabase, and tincture. The experimental period ranged from 5 days to 12 months.
Natural Treatment
Natural treatment is a naturally occurring secondary metabolite with possible clinical activity that is taken from living things like fungi, bacteria, plants, and animals.72 It is remarked that natural treatments are excellent sources for creating and manufacturing anti-inflammatory, analgesic, antimicrobial, and immunomodulator agents.73 Medicinal plants and natural remedies have long been conventionally used for various pharmacological uses.74
Numerous varieties of natural substances are employed in the research article that has been compiled. Each of these materials possesses distinct components and activities in managing Recurrent Aphthous Stomatitis (RAS). Typically, medications for RAS treatment are formulated in topical forms, including gels, patches, pastes, or mouthwash.18 This formulation is selected due to its capacity to endure in the oral cavity for an extended period, offering ulcer protection to mitigate deterioration and facilitating a swifter wound healing process.75
The entire study incorporated pain reduction as one of its outcome measures. Out of the 37 studies, 25 demonstrated that natural substances yielded superior therapeutic effects compared to the control materials. Pain assessment was conducted using the Visual Analog Scale (VAS) score, where a score of 1 signified minimal pain, and a score of 10 indicated maximum pain.76 Three studies, however, reported negative results or lower therapeutic effects compared to the comparator. The remaining 9 articles indicated better therapeutic effects than the comparator, although the differences were not statistically significant.
The reduction in ulcer size represented the second most frequently measured outcome in the trials. Out of 28 articles reporting on ulcer size reduction, 18 trials yielded positive results, 7 articles showed not statistically significant results, and 3 articles reported negative outcomes compared to the reference materials. Only 7 articles assessed the difference in the number of ulcers in the trials, with 3 showing results that were not statistically significant and 4 articles demonstrating a positive effect for RAS compared to the control materials. As for the reduction in healing duration, it was evaluated in 16 articles, with 12 reporting positive results and 2 articles each indicating results that were not statistically significant and negative compared to the control.
Synthetic-Based Treatment
Synthetic pharmaceuticals are formulated to address symptoms associated with specific diseases based on the scientific understanding of pathology.77 Synthetic drugs are pharmaceuticals derived from synthetic compounds and are typically prescribed by medical professionals to treat specific diseases.78 These synthetic medications represent a modern approach, often synthesized from artificial or naturally occurring substances that have undergone contemporary processing techniques.79
Therapeutic strategies for RAS primarily focus on alleviating pain and expediting wound healing.8 The management of minor aphthous ulcers involves supportive care, such as topical analgesics and protective bio-adhesive agents, while topical corticosteroids have been employed for both major and minor RAS ulcers.80 However, the frequent use of corticosteroids is often limited due to topical side effects and the risk of systemic absorption, including oral candidiasis, mucosal atrophy, susceptibility to infections, and gastrointestinal complications.81
There are 16 articles discussing the use of synthetic drugs in the management of RAS. Fifteen of these articles evaluate the effects of drug utilization on pain control, with 12 articles reporting positive outcomes, 1 article yielding negative results, and 3 articles indicating superior effects, albeit with non-significant differences.
Discussion
RAS is an oral mucosa disease characterized by recurrent, painful, single or multiple well-demarcated ulcerations with peripheral red halo where healing occurs with or without scarring.1 In the adult population, 60–85% of patients experience their first ulceration before age thirty.7 Although the primary causes of RAS remain unknown, certain factors have been identified as triggers for RAS outbreaks, including mental stress, trauma, lack of sleep, iron and folic acid deficiencies, menstruation, anemia, and changes in women’s sex hormones.2,3 The current therapeutic options can only lessen the frequency or severity of the lesions. In most cases, the primary goal of RAS treatment is to achieve pain reduction, decrease in ulcer size, decrease in ulcer count, and acceleration of ulcer healing duration.12
Various medications, whether derived from natural or synthetic sources, are employed as therapies for RAS, each with diverse mechanisms of action, including anti-inflammatory, antioxidant, immunomodulatory, analgesic, wound healing, antiulcer, antibacterial, antiviral, and antibiotic effects as depicted in Figure 2.73
Immune alterations are among the factors that contribute to the development of RAS. It starts with antigen secretion on keratinocytes that activates T lymphocytes, causing TNF-α secretion and leukocyte chemotaxis. Studies have demonstrated a 2–5-fold increase in the secretion of TNF-α in the saliva of individuals suffering from RAS.9 Therefore, anti-inflammatory agents are essential to cure RAS, such as thalidomide, which speeds up messenger RNA degradation to reduce TNF-α activity, and pentoxifylline, which inhibits TNF-α production.82 Another example is chitosan, which can suppress TNF-α production and inhibit cytokine synthesis. These actions trigger re-epithelialization and contribute to the rejuvenation of the mucous layer.83
The treatment of choice for RAS often includes natural immunomodulatory agents like Lactobacillus acidophilus and Lactobacillus reuteri and synthetic options like Levamisole. These drugs regulate both cellular and humoral immunity in individuals with RAS.41,59 Furthermore, levamisole exerts effects on T suppressor cells in vitro. Suppressing specific T-cell responses can result in immunosuppression, which is advantageous in managing RAS. This drug’s mechanism of action helps normalize the CD4+/CD8+ cell ratio and enhances IgA and IgM levels.59
Antioxidants also have a crucial role in the treatment of RAS. This condition can arise due to an elevated level of reactive oxygen species (ROS) that suppresses the activity of the immune system and induces cellular damage.84 Sage extract, a natural antioxidant, has been employed as a treatment for RAS. The phenolic and flavonoid compounds present in sage extract can enhance blood oxygen levels and protect the body against oxidative stress and free radicals that can cause cell damage, thus protecting against various types of ulcers.85 N-acetylcysteine (NAC), recognized for its potent antioxidant properties, is utilized as a therapy for RAS. In clinical scenarios characterized by glutathione deficiency and/or oxidative stress, the use of NAC has demonstrated effectiveness.86
The clinical presentation of RAS involves inflammation in the oral mucosa, manifesting as yellowish-white ulcers with clear boundaries and surrounded by an erythematous halo.87 Hence, substances with antiulcerogenic properties represent a viable therapeutic approach for RAS. Limonene, the primary component in citrus essential oil, exhibits antiulcerogenic activity.88 The antiulcerogenic effect of limonene acts as an immunomodulatory agent for oral aphthous ulcers.32 The mechanism of these properties is attributed to its capacity to enhance mucus secretion, heat shock protein-70, vasoactive intestinal peptide, and prostaglandin E2.88 In addition, rebamipide, which has activity as a gastro protective agent, is also recommended as RAS therapy, especially to improve ulcer conditions. It works by diminishing oxygen radicals, enhancing blood flow, and promoting the production of protective prostaglandins in the ulcerated mucosa, thereby expediting the healing process.89
In the management of RAS, supportive treatment is often advised, which aims to reduce pain, accelerate healing, and prevent recurrence.90 To reduce pain, it is essential to utilize medications with analgesic properties. Diphenhydramine and aluminum magnesium simethicone (AlMgS) syrup are often prescribed as an analgesic mouthwash to mitigate various symptoms of oral ulcers, including aphthous ulcers. Ibuprofen is a potent non-steroidal anti-inflammatory drug (NSAID) with significant anti-inflammatory capabilities and is frequently recommended in dental care.67 Moreover, natural substances like cannabidiol, aloe vera, fenugreek seed, and chamomile are known to possess analgesic properties and can be employed as therapeutic options for RAS.40,47,52,54
Supportive treatment to accelerate wound healing is important, and substances like alum and Ageratina pichinchensis can be valuable. Alum, for instance, induces tissue contraction, which reduces mucous membrane inflammation and accelerates wound healing.91 Ageratina pichinchensis also exhibits wound healing properties. In vitro studies have shown that the compound 7-O-(β-D-glucopyranosyl) galactin, found in Ageratina pichinchensis, can stimulate the proliferation of normal human skin cells (HFS-30), thereby promoting the acceleration of wound healing.39 Other natural compounds that significantly accelerate wound healing are propolis, honey, and aloe vera.33,38,47 Additionally, a synthetic compound known as topical insulin-liposomal has demonstrated its potential in this regard. Insulin, when applied topically, can enhance the healing process by accelerating re-epithelialization, promoting angiogenesis, and stimulating the secretion of extracellular matrix components in keratinocytes, endothelial cells, and fibroblasts.65,92
The antibacterial properties are also crucial for treating RAS because open sores on mucosal tissues make it easy for bacteria to colonize, especially Gram-positive bacteria, which can slow down the healing process.93 Bacterial infections disrupt the regeneration of the oral mucosa. Eradicating microbes within the oral cavity supports the normal wound healing process.94 The presence of catechins in gambier extract possesses the ability to reduce bacterial populations in oral ulcers. Catechins modify membrane fluidity, resulting in a loss of membrane integrity and, ultimately, bacterial cell death.95 Other studies have also indicated the effectiveness of gambier extract in reducing the levels of Streptococcus mutans in saliva.44 By maintaining a sterile wound environment, gambier extract expedites the wound healing process by promoting angiogenic responses.96
From the analysis of a total of 53 clinical trials conducted by the authors in the last 10 years, natural-based medicines have shown significant improvement in patients’ symptoms, including the reduction of pain scores, ulcer size, the number of ulcers, and a decrease in ulcer healing duration. However, when compared with the control group, natural-based medicines provided better therapeutic effects compared to negative control or placebo but did not demonstrate significantly different or superior therapeutic effects compared to positive control or triamcinolone acetonide.
The drug delivery system also plays a crucial role in influencing the effectiveness of RAS therapy.97 Typically, medications intended for RAS treatment are formulated in topical forms, such as gels, patches, pastes, or mouthwash.9 This choice of the formulation is made because it can persist in the oral cavity for an extended period, providing protection against ulcer deterioration and facilitating a more rapid wound healing process.98,99 For instance, in the case of curcumin nanomicelle gel, it has shown better therapeutic outcomes than curcumin gel.20 This is because nanomicelle gel does not readily dissolve in saliva, making it highly suitable for the physiological conditions of the oral cavity and facilitating easy absorption by the mucosa.100 Furthermore, the development of drug delivery systems for RAS is an ongoing endeavor, encompassing research conducted in both in vitro and in silico settings. These developments include muco-adhesive sponges containing tenoxicam and miconazole nitrate,101 hydrogel formulations with alpha mangostin,102 adhesive buccal films comprising alginate and ambroxol,103 prednisolone,104 and benzydamine hydrochloride,105 as well as mucoadhesive films containing betamethasone valerate,106 and clobetasol propionate.107
The trial duration also shows a rather significant difference, with synthetic-based drugs having trial periods ranging from 3 days to 12 months. Topical and systemic corticosteroids, antibiotics, and analgesics are highly recommended for RAS patients.80 However, extended treatment periods and frequent exposure to these medications can lead to fungal infections and drug resistance, which can subsequently result in more severe side effects or even life-threatening complications.75 Therefore, the long-term use of these medications for RAS management should be avoided.108
Finding effective treatment for oral disease is among the most critical challenges of oral medicine. Consequently, there is a demand for therapy that can be effectively administered to the oral mucosa, withstand the washout effect of saliva, maintain a good safety profile, and minimize adverse side effects.109 Research in RAS therapy is an ongoing endeavor encompassing various stages of development, starting with drug formulation, pre-clinical testing, and clinical trials. An example of such research is the study conducted by Milanda et al, in which the compound alpha mangostin was utilized as a hydrogel film formulation based on chitosan alginate. This formulation demonstrated in vivo efficacy in the context of RAS therapy in white mice. The efficacy was indicated by the swiftest healing process, as determined through slope calculations.110
In summary, current data indicate a favorable benefit from both synthetic and natural remedies for the treatment of recurrent acute stomatitis (RAS). These remedies have demonstrated effectiveness in reducing pain, the number of ulcers, ulcer size, and healing duration. Natural remedies are recognized for their therapeutic benefits in RAS treatment but have yet to surpass the effectiveness of corticosteroid therapy. Conversely, the use of synthetic remedies, such as corticosteroids, tends to be of longer duration and raises concerns regarding potential side effects. The development of RAS therapy, including active ingredients, formulations, and delivery systems, must continue to be pursued in order to obtain effective, safe, and minimally side-effect-inducing treatments from both natural and synthetic sources.
Conclusion
The utilization of natural and synthetic medicinal approaches for the treatment of recurrent aphthous stomatitis (RAS) has exhibited considerable success in enhancing the well-being of patients, as evidenced by the reduction in pain, ulcer size, ulcer count, and the duration required for complete healing. Nevertheless, natural-based medicines necessitate further investigation and research to establish their therapeutic effectiveness equivalent to or surpassing that of corticosteroids, intending to potentially be integrated as a standard treatment modality for RAS. The progression of RAS therapy, encompassing the exploration of active constituents, formulations, and delivery systems, must persist in attaining effective and safe treatments while inducing minimal side effects originating from both natural and synthetic sources.
Abbreviations
RAS, Recurrent Aphthous Stomatitis; CMV, Cytomegalovirus; NAC, N-Acetylcysteine; VAS, Visual Analog Scale; AlMgS, Aluminum Magnesium Simethicone; NSAID, Non-steroidal Anti-inflammatory Drug; RCT, Randomized Controlled Trial; QD, Once a day; BID, Twice a day; TID, Thrice a day; QID, 4 times a day.
Acknowledgments
We would like to thank The Rector of Universitas Padjadjaran for the APC.
Funding
Kedaireka Matching Fund, Ministry of Education and Culture, Republic of Indonesia (20/E1/PPK/KS.03.00/2023).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Jurge S, Kuffer R, Scully C, Porter SR. Mucosal disease series. Number VI. Recurrent aphthous stomatitis. Oral Dis. 2006;12(1):1–21. doi:10.1111/j.1601-0825.2005.01143.x
2. Cui RZ, Bruce AJ, Rogers RS. Recurrent aphthous stomatitis. Clin Dermatol. 2016;34(4):475–481. doi:10.1016/J.CLINDERMATOL.2016.02.020
3. Ślebioda Z, Szponar E, Kowalska A. Etiopathogenesis of recurrent aphthous stomatitis and the role of immunologic aspects: literature review. Arch Immunol Ther Exp. 2014;62(3):205–215. doi:10.1007/S00005-013-0261-Y/TABLES/3
4. Hussein A, Darwazeh Azmi MG, Al-Jundi S. Prevalence of oral lesions among Jordanian children. Saudi J Oral Sci. 2017;4(1):12. doi:10.4103/1658-6816.200135
5. Zhu Z, He Z, Xie G, Fan Y, Shao T. Altered oral microbiota composition associated with recurrent aphthous stomatitis in young females. Medicine. 2021;100(10):E24742. doi:10.1097/MD.0000000000024742
6. Bankvall M, Sjöberg F, Gale G, Wold A, Jontell M, Östman S. The oral microbiota of patients with recurrent aphthous stomatitis. J Oral Microbiol. 2014;6:1–11. doi:10.3402/jom.v6.25739
7. Gomes VB, Porter S. Review Ulcerative Lesions of the Mouth: an Update. Int J Med. 2007;62(6):769–780.
8. Mandasari M, Rahmayanti F. Identification of stress as a predisposing factor in minor recurrent aphthous stomatitis: a case report. J Case Rep Dental Med. 2022;4:56–59. doi:10.20956/jcrdm.v4i3.213
9. Belenguer-Guallar I, Jiménez-Soriano Y, Claramunt-Lozano A. Treatment of recurrent aphthous stomatitis. A literature review. J Clin Exp Dent. 2014;6(2):168–174. doi:10.4317/jced.51401
10. Vitamia C, Iftinan GN, Latarissa IR, et al. α-Mangostin hydrogel film with chitosan alginate base for recurrent aphthous stomatitis (RAS) treatment: study protocol for double-blind randomized controlled trial. Front Pharmacol. 2024:15. doi:10.3389/FPHAR.2024.1353503
11. Minervini G, Franco R, Marrapodi MM, et al. Correlation between Temporomandibular Disorders (TMD) and Posture Evaluated trough the Diagnostic Criteria for Temporomandibular Disorders (DC/TMD): a Systematic Review with Meta-Analysis. J Clin Med. 2023;12(7). doi:10.3390/jcm12072652
12. Messadi DV, Younai F. Aphthous ulcers. Dermatol Ther. 2010;23(3):281–290. doi:10.1111/j.1529-8019.2010.01324.x
13. Rattan J, Schneider M, Arber N, Gorsky M, Dayan D. Sucralfate suspension as a treatment of recurrent aphthous stomatitis. J Intern Med. 1994;236(3):341–343. doi:10.1111/j.1365-2796.1994.tb00805.x
14. Liu X, Guan X, Chen R, Hua H, Liu Y, Yan Z. Repurposing of Yunnan Baiyao as an alternative therapy for minor recurrent aphthous stomatitis. Evid Based Complement Alternat Med. 2012;2012:284620. doi:10.1155/2012/284620
15. Huo X, Han N, Liu L. Effect of different treatments on recurrent aphthous stomatitis: laser versus medication. Lasers Med Sci. 2021;36(5):1095–1100. doi:10.1007/s10103-020-03166-0
16. Yilmaz HG, Albaba MR, Caygur A, Cengiz E, Boke-Karacaoglu F, Tumer H. Treatment of recurrent aphthous stomatitis with Er, Cr:YSGG laser irradiation: a randomized controlled split mouth clinical study. J Photochem Photobiol B. 2017;170:1–5. doi:10.1016/j.jphotobiol.2017.03.011
17. Albrektson M, Hedström L, Bergh H. Recurrent aphthous stomatitis and pain management with low-level laser therapy: a randomized controlled trial. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117(5):590–594. doi:10.1016/j.oooo.2014.01.228
18. Barrons RW. Treatment strategies for recurrent oral aphthous ulcers. Am J Health Syst Pharm. 2001;58(1):41–43.
19. Lavaee F, Ghasemi M, Amiri MA, Farshidfar N. The Comparative Effects of Rhus Coriaria and Triamcinolone in Patients with Recurrent Aphthous Stomatitis: a Single-Blinded Randomized Controlled Clinical Trial. Biomed Res Int. 2022;2022:5067. doi:10.1155/2022/5855067
20. Bakhshi M, Mahboubi A, Jaafari MR, Ebrahimi F, Tofangchiha M, Alizadeh A. Comparative Efficacy of 1% Curcumin Nanomicelle Gel and 2% Curcumin Gel for Treatment of Recurrent Aphthous Stomatitis: a Double-Blind Randomized Clinical Trial. J Evidence-Based Dental Practice. 2022;22(2):101708. doi:10.1016/j.jebdp.2022.101708
21. Raman P, Pity R, Krithika CL, Anand SN, Subramani GP. Topical Curcumin and Triamcinolone Acetonide in Recurrent Minor Aphthous Ulcers: a Pilot Trial. Journal of Contemporary Dental Practice. 2020;21(8):884–890. doi:10.5005/JP-JOURNALS-10024-2901
22. Deshmukh R, Bagewadi A. Comparison of effectiveness of curcumin with triamcinolone acetonide in the gel form in treatment of minor recurrent aphthous stomatitis: a randomized clinical trial. Int J Pharm Investig. 2014;4(3):138. doi:10.4103/2230-973x.138346
23. Shao Y, Zhou H. Clinical evaluation of an oral mucoadhesive film containing chitosan for the treatment of recurrent aphthous stomatitis: a randomized, double-blind study. J Dermatological Treat. 2020;31(7):739–743. doi:10.1080/09546634.2019.1610548
24. Rahmani F, Moghadamnia AA, Kazemi S, Shirzad A, Motallebnejad M. Effect of 0.5% Chitosan mouthwash on recurrent aphthous stomatitis: a randomized double-blind crossover clinical trial. Electron Physician. 2018;10(6):6912–6919. doi:10.19082/6912
25. Abbasi F, Rasoulzadeh Z, Yavari A. The effect of sage (Salvizan gel) compared to triamcinolone acetonide on the treatment of recurrent aphthous stomatitis: a double-blinded randomized clinical trial. BMC Oral Health. 2023;23(1):1–5. doi:10.1186/s12903-023-02861-y
26. Aggour RL, Mahmoud SH, Abdelwhab A. Evaluation of the effect of probiotic lozenges in the treatment of recurrent aphthous stomatitis: a randomized, controlled clinical trial. Clin Oral Investig. 2021;25(4):2151–2158. doi:10.1007/s00784-020-03527-7
27. Molania T, Malekzadeh Shafaroudi A, Saeedi M, et al. Evaluation of cinnamaldehyde mucoadhesive patches on minor recurrent aphthous stomatitis: a randomized, double-blind, placebo-controlled clinical trial. BMC Oral Health. 2022;22(1):1–7. doi:10.1186/s12903-022-02248-5
28. Nosratzehi T, Akar A. Efficacy of Omega-3 in Treatment of Recurrent Aphthous Stomatitis: a Randomised, Double-blind, Placebo-controlled Study. Chin J Dent Res. 2016;19(3):159–164. doi:10.3290/j.cjdr.a36681
29. Hadian Z, Moghadamnia AA, Kazemi S, Shirzad A. Effect of Omega-3 on Recurrent Aphthous Stomatitis and Improvement Quality of Life. Int J Dent. 2021;2021:7575. doi:10.1155/2021/6617575
30. El Khouli AM, El-Gendy EA. Efficacy of omega-3 in treatment of recurrent aphthous stomatitis and improvement of quality of life: a randomized, double-blind, placebo-controlled study. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117(2):191–196. doi:10.1016/j.oooo.2013.09.003
31. Rogulj AA, Alajbeg IZ, Brailo V, et al. Topical NAVS naphthalan for the treatment of oral lichen planus and recurrent aphthous stomatitis: a double blind, randomized, parallel group study. PLoS One. 2021;16(4 April):1–14. doi:10.1371/journal.pone.0249862
32. Kürklü-Gürleyen E, Öğüt-Erişen M, Çakır O, Uysal Ö, Ak G. Quality of life in patients with recurrent aphthous stomatitis treated with a mucoadhesive patch containing citrus essential oil. Patient Prefer Adherence. 2016;10:967–973. doi:10.2147/PPA.S106530
33. Arafa MG, Ghalwash D, El-Kersh DM, Elmazar MM. Propolis-based niosomes as oromuco-adhesive films: a randomized clinical trial of a therapeutic drug delivery platform for the treatment of oral recurrent aphthous ulcers. Sci Rep. 2018;8(1):1–14. doi:10.1038/s41598-018-37157-7
34. Gavanji S, Larki B, Bakhtari A. The effect of extract of Punica granatum var. pleniflora for treatment of minor recurrent aphthous stomatitis. Integr Med Res. 2014;3(2):83–90. doi:10.1016/j.imr.2014.03.001
35. Darakhshan S, Malmir M, Bagheri F, et al. The effects of pomegranate peel extract on recurrent aphthous stomatitis. Curr Issues Pharm Med Sci. 2019;32(3):115–120. doi:10.2478/cipms-2019-0021
36. Tavangar A, Soleymani B, Ghalayani P, Zolfaghary B, Farhad A. The efficacy of Punica granatum extract in the management of recurrent aphthous stomatitis. J Res Pharm Pract. 2013;2(2):88. doi:10.4103/2279-042x.117389
37. Tavangar A, Aslani A, Nikbakht N. Comparative Study of Punica granatum Gel and Triadent Oral Paste Effect on Recurrent Aphthous Stomatitis, a Double Blind Clinical Trial. J Dent. 2019;20(3):184–189. doi:10.30476/DENTJODS.2019.44913
38. El-Haddad SA, Asiri FY, Al-Qahtani HH, Al-Ghmlas AS. Efficacy of honey in comparison to topical corticosteroid for treatment of recurrent minor aphthous ulceration: a randomized, blind, controlled, parallel, double-center clinical trial. Quintessence Int. 2014;45(8):691–701. doi:10.3290/j.qi.a32241
39. Romero-Cerecero O, Zamilpa A, Tortoriello J. Pilot study that evaluated the clinical effectiveness and safety of a phytopharmaceutical elaborated with an extract of Ageratina pichinchensis in patients with minor recurrent aphthous stomatitis. J Ethnopharmacol. 2015;173:225–230. doi:10.1016/j.jep.2015.06.021
40. Umpreecha C, Bhalang K, Charnvanich D, Luckanagul J. Efficacy and safety of topical 0.1% cannabidiol for managing recurrent aphthous ulcers: a randomized controlled trial. BMC Complement Med Ther. 2023;23(1):1–11. doi:10.1186/s12906-023-03886-0
41. Pedersen AML, Bukkehave KH, Bennett EP, Twetman S. Effect of Lozenges Containing Lactobacillus reuteri on the Severity of Recurrent Aphthous Ulcers: a Pilot Study. Probiotics Antimicrob Proteins. 2020;12(3):819–823. doi:10.1007/s12602-019-09586-x
42. Vaziri S, Mojarrab M, Osein FMH, Najafi F, Ghobadi A. Evaluation of anti-aphthous activity of decoction of Nicotiana tabacum leaves as a mouthwash: a placebo-controlled clinical study. Journal of traditional Chinese medicine = Chung i tsa chih ying wen pan / sponsored by All-China Association of Traditional Chinese Medicine. Acad Traditional Chine Med. 2016;36(2):160–164. doi:10.1016/s0254-6272(16)30022-x
43. Rafieian N, Abdolsamadi H, Moghadamnia A, et al. Efficacy of alum for treatment of recurrent aphthous stomatitis. Caspian J Intern Med. 2016;7(3):201–206.
44. Puspa Dewi SR, Karina Ginting MP, Anggraini NR, Parisa N, Handayani P, Chairani S. Evaluation of the effect of gambier (Uncaria gambier) extract for treatment of recurrent aphthous stomatitis. International Research Journal of Pharmacy. 2020;11(1):27–31. doi:10.7897/2230-8407.11015
45. Du Q, Ni S, Guo L, et al. Effects of Zingiber officinalis (WILLD.) ROSC. Membranes on minor recurrent aphthous stomatitis: a randomized pragmatic trial. J Traditional Chine Med Sci. 2018;5(1):58–63. doi:10.1016/j.jtcms.2018.02.004
46. Haghpanah P, Moghadamnia AA, Zarghami A, Motallebnejad M. Muco-bioadhesive containing ginger officinale extract in the management of recurrent aphthous stomatitis: a randomized clinical study. Caspian J Intern Med. 2015;6(1):3–8.
47. Yousef NJ, Aljoujou AA, Mashlah AM, Hajeer MY. Assessment of the Effectiveness of Aloe vera Versus Amlexanox in the Treatment of Recurrent Aphthous Ulcers: a Three-Arm Placebo-Controlled Randomized Clinical Trial. Cureus. 2022;14(10):1–8. doi:10.7759/cureus.30693
48. Mansour G, Ouda S, Shaker A, Abdallah HM. Clinical efficacy of new aloe vera- and myrrh-based oral mucoadhesive gels in the management of minor recurrent aphthous stomatitis: a randomized, double-blind, vehicle-controlled study. J Oral Pathol Med. 2014;43(6):405–409. doi:10.1111/jop.12130
49. Halim DS, Abdullah NA, Alam MK, Samsee SNB, May TS. Comparison of the effectiveness between virgin coconut oil (VCO) and triamcinolone for treatment of minor recurrent aphthous stomatitis (RAS). International Medical Journal. 2014;21(3):319–320.
50. Jiang XW, Zhang Y, Zhu YL, et al. Effects of berberine gelatin on recurrent aphthous stomatitis: a randomized, placebo-controlled, double-blind trial in a Chinese cohort. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;115(2):212–217. doi:10.1016/j.oooo.2012.09.009
51. Elamrousy WA, Mortada A, Shoukheba M. Evaluation of Novel Topical Camel Whey Protein Gel for the Treatment of Recurrent Aphthous Stomatitis: randomized Clinical Study. J Int Soc Prev Community Dent. 2021;11(5):574–581. doi:10.4103/jispcd.JISPCD_172_21
52. Ansari M, Hashemipour M, Farsinejad A, et al. Clinical efficacy of a buccoadhesive paste from Fenugreek seeds (Trigonella foenum graecum L.) on recurrent aphthous stomatitis: in-vitro assessment of non-toxic concentration and pilot trial. Adv Integr Med. 2022;9(1):17–21. doi:10.1016/j.aimed.2021.06.001
53. Raeesi V, Arbabi-Kalati F, Akbari N, Hamishekar H. Comparison effectiveness of the bioadhesive paste containing licorice 5% with bioadhesive paste without drug in the management of recurrent aphthous stomatitis. Acta Med Mediterr. 2015;31(7):1331–1335.
54. Seyyedi SA, Sanatkhani M, Pakfetrat A, Olyaee P. The therapeutic effects of chamomilla tincture mouthwash on oral aphthae: a randomized clinical trial. J Clin Exp Dent. 2014;6(5):e535–e538. doi:10.4317/jced.51472
55. Andishe Tadbir A, Pourshahidi S, Ebrahimi H, Hajipour Z, Memarzade MR, Shirazian S. The effect of Matricaria chamomilla (chamomile) extract in Orabase on minor aphthous stomatitis, a randomized clinical trial. J Herb Med. 2015;5(2):71–76. doi:10.1016/j.hermed.2015.05.001
56. Ghorbani A, Akbari J, Boorboor M, Nekoukar Z, Eslami G. Evaluation of zinc sulfate mucoadhesive formulation on recurrent aphthous stomatitis: a randomized double-blind, placebo-controlled clinical trial. BMC Oral Health. 2020;20(1):1–6. doi:10.1186/s12903-020-01194-4
57. Al-Oudah GA, AL-Ameedee AH, Shwailiya SAS. Activity of zinc oral dispersible tablet on marjory clinical type of recurrent aphthous stomatitis ulceration, a clinical trial human study. Ann Trop Med Public Health. 2020;23(12). doi:10.36295/ASRO.2020.231201
58. Habibzadeh S, Sheikh Rahimi M, Edalatkhah H, Piri H, Maleki N. Effectiveness of booster dose of tetanus and diphtheria toxoids (Td) vaccine in management of recurrent aphthous stomatitis: a prospective, randomized, triple-blind and placebo-controlled clinical trial. J Dermatological Treat. 2021;32(3):361–366. doi:10.1080/09546634.2019.1654597
59. Sharda N, Shashikanth MC, Kant P, Jain M. Levamisole and low-dose prednisolone in the treatment of recurrent aphthous stomatitis. J Oral Pathol Med. 2014;43(4):309–316. doi:10.1111/jop.12126
60. Vijayabala GS, Kalappanavar AN, Annigeri RG, Sudarshan R, Shettar SS. Single application of topical doxycycline hyclate in the management of recurrent aphthous stomatitis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;116(4):440–446. doi:10.1016/j.oooo.2013.06.015
61. Shrivastava K, Naidu G, Deshpande A, Handa H, Raghuvanshi V, Gupta M. Comparative evaluation of the efficacy of topical amlexanox 5% oral paste and triamcinolone acetonide 0.1% oral paste in the treatment of Recurrent Aphthous Stomatitis (RAS). J Indian Acad Oral Med Radiol. 2018;30(3):235–240. doi:10.4103/jiaomr.jiaomr_40_18
62. Alsahaf S, Alkurdi KA, Challacombe SJ, Tappuni AR. Topical betamethasone and systemic colchicine for treatment of recurrent aphthous stomatitis: a randomised clinical trial. BMC Oral Health. 2023;23(1):709. doi:10.1186/s12903-023-03335-x
63. Halboub E, Alkadasi B, Alakhali M, et al. N-acetylcysteine versus chlorhexidine in treatment of aphthous ulcers: a preliminary clinical trial. J Dermatological Treat. 2021;32(6):649–653. doi:10.1080/09546634.2019.1688231
64. Eslami G, Ghorbani A, Akbari J, Farmoudeh A, Faghih F, Moghimi M. Efficacy of Oral Mucoadhesive N-acetylcysteine Tablets in Treatment of Recurrent Aphthous Stomatitis: a Randomized Double-Blind, Placebo-Controlled Clinical Trial. Front Dent. 2023;20:1–8. doi:10.18502/fid.v20i18.12824
65. El-Wakeel NM, Dawoud MHS. Topical insulin-liposomal formulation in management of recurrent aphthous ulcers: a randomized placebo-controlled trial. J Investig Clin Dent. 2019;10(4):1–6. doi:10.1111/jicd.12437
66. Zeng Q, Shi X, Yang J, et al. The efficacy and safety of thalidomide on the recurrence interval of continuous recurrent aphthous ulceration: a randomized controlled clinical trial. J Oral Pathol Med. 2020;49(4):357–364. doi:10.1111/jop.12960
67. Borhan-Mojabi K, Mirmiran F, Nassiri-Asl M, Nazeman P, Jahanihashemi H. Therapeutic Effects of “Ibuprofen, Diphenhydramine and Aluminium MgS” on Recurrent Aphthous Stomatitis: a Randomized Controlled Trial. J Dent (Tehran). 2014;11(2):167–171.
68. Yarom N, Zelig K, Epstein JB, Gorsky M. The efficacy of minocycline mouth rinses on the symptoms associated with recurrent aphthous stomatitis: a randomized, double-blind, crossover study assessing different doses of oral rinse. Oral Surg Oral Med Oral Pathol Oral Radiol. 2017;123(6):675–679. doi:10.1016/j.oooo.2017.02.013
69. Ofluoglu D, Ergun S, Warnakulasuriya S, Namdar-Pekiner F, Tanyeri H. An evaluation of the efficacy of a topical gel with Triester Glycerol Oxide (TGO) in the treatment of minor recurrent aphthous stomatitis in a Turkish cohort: a randomized, double-blind, placebo-controlled clinical trial. Med Oral Patol Oral Cir Bucal. 2017;22(2):e159–e166. doi:10.4317/medoral.21469
70. Parvathi Devi MK, Dnsv R, Koppal S, Byatnal AR, Rukmangada T, Byatnal AA. Efficacy of rebamipide and levamisole in the treatment of patients with recurrent aphthous ulcer - A comparative study. Journal of Clinical and Diagnostic Research. 2014;8(11):ZC119–ZC122. doi:10.7860/JCDR/2014/10295.5202
71. Owlia M, Mirzadeh M, Mehrpoor G. Penicillin in oral aphthosis, new insight for an old drug: a randomized, double-blind, controlled clinical trial. J Res Med Sci. 2020;25:95. doi:10.4103/jrms.JRMS_748_19
72. Suhandi C, Alfathonah SS, Hasanah AN. Potency of Xanthone Derivatives from Garcinia mangostana L. for COVID-19 Treatment through Angiotensin-Converting Enzyme 2 and Main Protease Blockade: a Computational Study. Molecules. 2023;28(13). doi:10.3390/MOLECULES28135187
73. Dhama K, Karthik K, Khandia R, et al. Medicinal and Therapeutic Potential of Herbs and Plant Metabolites / Extracts Countering Viral Pathogens – current Knowledge and Future Prospects. Curr Drug Metab. 2018;19. doi:10.2174/1389200219666180129145252
74. Kaileh M, Vanden Berghe W, Boone E, Essawi T, Haegeman G. Screening of indigenous Palestinian medicinal plants for potential anti-inflammatory and cytotoxic activity. J Ethnopharmacol. 2007;113(3):510–516. doi:10.1016/j.jep.2007.07.008
75. Eisen D, Lynch DP. Selecting topical and systemic agents for recurrent aphthous stomatitis. Cutis. 2001;68(3):201–206.
76. Delgado DA, Lambert BS, Boutris N, et al. Validation of Digital Visual Analog Scale Pain Scoring With a Traditional Paper-based Visual Analog Scale in Adults. J Am Acad Orthop Surg Glob Res Rev. 2018;2(3):e088. doi:10.5435/JAAOSGlobal-D-17-00088
77. Karimi A, Majlesi M, Rafieian-Kopaei M. Herbal versus synthetic drugs; beliefs and facts. J Nephropharmacol. 2015;4(1):27–30.
78. Furnham AF. Alternative and Complementary Healing Practices. International Encyclopedia of the Social & Behavioral Sciences. 2001;404–407. doi:10.1016/B0-08-043076-7/03905-X
79. Lau SH, Herman H, Rahmat M. Studi perbandingan tingkat pengetahuan masyarakat tentang obat herbal dan obat sintetik di campagayya kelurahan panaikang Kota makassar. Jurnal Farmasi Sandi Karsa. 2019;5(1):33.
80. Femiano F, Lanza A, Buonaiuto C, et al. Guidelines for diagnosis and management of aphthous stomatitis. Pediatr Infect Dis J. 2007;26(8):728–732. doi:10.1097/INF.0b013e31806215f9
81. Messier C, Epifano F, Genovese S, Grenier D. Licorice and its potential beneficial effects in common oro-dental diseases. Oral Dis. 2012;18(1):32–39. doi:10.1111/j.1601-0825.2011.01842.x
82. Chaudhuri K, Nair KK, Ashok L. Salivary levels of TNF-α in patients with recurrent aphthous stomatitis: a cross-sectional study. J Dent Res Dent Clin Dent Prospects. 2018;12(1):45–48. doi:10.15171/joddd.2018.007
83. Kovalik AC, Bisetto P, Pochapski MT, Campagnoli EB, Pilatti GL, Santos FA. Effects of an orabase formulation with ethanolic extract of Malva sylvestris L. in oral wound healing in rats. J Med Food. 2014;17(5):618–624. doi:10.1089/jmf.2013.0001
84. Suhandi C, Wilar G, Lesmana R, et al. Propolis-Based Nanostructured Lipid Carriers for α-Mangostin Delivery: formulation, Characterization, and In Vitro Antioxidant Activity Evaluation. Molecules. 2023;28(16). doi:10.3390/MOLECULES28166057
85. Mohammed S, Abdulraheem S. Evaluate the efficiency of sage (Salvia officinalis) and curcumin mouthwash in the treatment of recurrent aphthous stomatitis)(comparative study). Al-Rafidain Dental Journal. 2021;48.
86. Abdulrab S, Halboub E, Barngkgei I, Al-hebshi N. N-Acetylcysteine as a Candidate Therapeutic for Recurrent Aphthous and Aphthous-Like Ulcers. Dent Hypotheses. 2017;8:17–22. doi:10.4103/2155-8213.202028
87. A M. Etiology and pathophysiology of recurrent aphthous stomatitis: a review. Int J Curr Res Rev. 2014;6:16–22.
88. Moraes TM, Kushima H, Moleiro FC, et al. Effects of limonene and essential oil from Citrus aurantium on gastric mucosa: role of prostaglandins and gastric mucus secretion. Chem Biol Interact. 2009;180(3):499–505. doi:10.1016/j.cbi.2009.04.006
89. Kudur MH, Hulmani M. Rebamipide: a Novel Agent in the Treatment of Recurrent Aphthous Ulcer and Behcet’s Syndrome. Indian J Dermatol. 2013;58(5):352–354. doi:10.4103/0019-5154.117298
90. Rodu B, Mattingly G. Oral mucosal ulcers: diagnosis and management. J Am Dent Assoc. 1992;123(10):83–86. doi:10.14219/jada.archive.1992.0268
91. ALtaei TS, AI-Jubouri RH. Evaluation of the efficacy of alum suspension in treatment of recurrent ulcerative ulceration. J Baghdad College Dentistry. 2005;17(2):45–48.
92. Attia EAS, Belal DMI, El Samahy MH, El Hamamsy MH. A pilot trial using topical regular crystalline insulin vs. aqueous zinc solution for uncomplicated cutaneous wound healing: impact on quality of life. Wound Repair Regen. 2014;22(1):52–57. doi:10.1111/wrr.12122
93. Negut I, Grumezescu V, Grumezescu AM. Treatment Strategies for Infected Wounds. Molecules. 2018;23(9). doi:10.3390/molecules23092392
94. Tarakji B, Gazal G, Al-Maweri SA, Azzeghaiby SN, Alaizari N. Guideline for the diagnosis and treatment of recurrent aphthous stomatitis for dental practitioners. J Int Oral Health. 2015;7(5):74–80.
95. Tsuchiya H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules. 2015;20(10):18923–18966. doi:10.3390/molecules201018923
96. Suhandi C, Mohammed AFA, Wilar G, El-Rayyes A, Wathoni N. Effectiveness of Mesenchymal Stem Cell Secretome on Wound Healing: a Systematic Review and Meta-analysis. Tissue Eng Regen Med. 2023;20(7):1053–1062. doi:10.1007/S13770-023-00570-9
97. Suharyani I, Fouad Abdelwahab Mohammed A, Muchtaridi M, Wathoni N, Abdassah M. Evolution of Drug Delivery Systems for Recurrent Aphthous Stomatitis. Drug Des Devel Ther. 2021;15:4071–4089. doi:10.2147/DDDT.S328371
98. Boddupalli BM, Mohammed ZNK, Nath RA, Banji D. Mucoadhesive drug delivery system: an overview. J Adv Pharm Technol Res. 2010;1(4):381–387. doi:10.4103/0110-5558.76436
99. Shaikh R, Raj Singh TR, Garland MJ, Woolfson AD, Donnelly RF. Mucoadhesive drug delivery systems. J Pharm Bioallied Sci. 2011;3(1):89–100. doi:10.4103/0975-7406.76478
100. Bakhshi M, Gholami S, Mahboubi A, Jaafari MR, Namdari M. Combination Therapy with 1% Nanocurcumin Gel and 0.1% Triamcinolone Acetonide Mouth Rinse for Oral Lichen Planus: a Randomized Double-Blind Placebo Controlled Clinical Trial. Dermatol Res Pract. 2020;2020:4298193. doi:10.1155/2020/4298193
101. Ossama M, Lamie C, Tarek M, Wagdy HA, Attia DA, Elmazar MM. Management of recurrent aphthous ulcers exploiting polymer-based Muco-adhesive sponges: in-vitro and in-vivo evaluation. Drug Deliv. 2021;28(1):87–99. doi:10.1080/10717544.2020.1858999
102. Wathoni N, Yuniarsih N, Cahyanto A, Muhctaridi M. Α-Mangostin Hydrogel Film Based Chitosan-Alginate for Recurrent Aphthous Stomatitis. Applied Sciences. 2019;9(23). doi:10.3390/app9235235
103. Laffleur F, Küppers P. Adhesive alginate for buccal delivery in aphthous stomatitis. Carbohydr Res. 2019;477:51–57. doi:10.1016/j.carres.2019.03.009
104. Farid RM, Wen MM. Promote Recurrent Aphthous Ulcer Healing with Low Dose Prednisolone Bilayer Mucoadhesive Buccal Film. Curr Drug Deliv. 2017;14(1):123–135. doi:10.2174/1567201813666160316122548
105. Sharma A, Yadav N, Ram Murti SS, Author C, Ram MS. Review On Mucoadhesive Buccal Drug Delivery For The Treatment Of Aphthous Ulcers. J Pharmaceutical Negative Results. 2022;13(9). doi:10.47750/pnr.2022.13.S09.1297
106. Sizílio RH, Galvão JG, Trindade GGG, et al. Chitosan/pvp-based mucoadhesive membranes as a promising delivery system of betamethasone-17-valerate for aphthous stomatitis. Carbohydr Polym. 2018;190:339–345. doi:10.1016/j.carbpol.2018.02.079
107. Colley HE, Said Z, Santocildes-Romero ME, et al. Pre-clinical evaluation of novel mucoadhesive bilayer patches for local delivery of clobetasol-17-propionate to the oral mucosa. Biomaterials. 2018;178:134–146. doi:10.1016/j.biomaterials.2018.06.009
108. Li CL, Huang HL, Wang WC, Hua H. Efficacy and safety of topical herbal medicine treatment on recurrent aphthous stomatitis: a systemic review. Drug Des Devel Ther. 2015;10:107–115. doi:10.2147/DDDT.S96589
109. Chinna Reddy P, Chaitanya KSC, Madhusudan Rao Y. A review on bioadhesive buccal drug delivery systems: current status of formulation and evaluation methods. Daru. 2011;19(6):385–403.
110. Milanda T, Cindana Mo’o FR, Mohammed AFA, et al. Alginate/Chitosan-Based Hydrogel Film Containing α-Mangostin for Recurrent Aphthous Stomatitis Therapy in Rats. Pharmaceutics. 2022;14(8). doi:10.3390/pharmaceutics14081709
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
