Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Withdrawal of inhaled corticosteroids in COPD patients: rationale and algorithms
Authors Avdeev S , Aisanov Z , Arkhipov V, Belevskiy A , Leshchenko I, Ovcharenko S , Shmelev E , Miravitlles M
Received 6 March 2019
Accepted for publication 17 May 2019
Published 10 June 2019 Volume 2019:14 Pages 1267—1280
DOI https://doi.org/10.2147/COPD.S207775
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Sergey Avdeev,1,2 Zaurbek Aisanov,3 Vladimir Arkhipov,4 Andrey Belevskiy,3 Igor Leshchenko,5 Svetlana Ovcharenko,6 Evgeny Shmelev,7 Marc Miravitlles8
1Department of Pulmonology, I.M. Sechenov First Moscow State Medical University, Moscow, Russian Federation; 2Clinical Department, Federal Pulmonology Research Institute, Federal Medical and Biological Agency of Russia, Moscow, Russian Federation; 3Department of Pulmonology, N.I. Pirogov Russian State National Research Medical University, Moscow, Russian Federation; 4Department of Clinical Pharmacology and Therapy, Russian Medical Academy of Continuous Professional Education, Moscow, Russian Federation; 5Department of Phthisiology, Pulmonology and Thoracic Surgery, Ural State Medical University, Ekaterinburg, Russian Federation; 6Department of Internal Diseases No.1, I.M. Sechenov First Moscow State Medical University, Moscow, Russian Federation; 7Department of Differential Diagnostics, Federal Central Research Institute of Tuberculosis, Moscow, Russian Federation; 8Pneumology Department, University Hospital Vall d’Hebron/Vall d’Hebron Research Institute (VHIR), Ciber de Enfermedades Respiratorias (CIBERES), Barcelona, Spain
Abstract: Observational studies indicate that overutilization of inhaled corticosteroids (ICS) is common in patients with chronic obstructive pulmonary disease (COPD). Overprescription and the high risk of serious ICS-related adverse events make withdrawal of this treatment necessary in patients for whom the treatment-related risks outweigh the expected benefits. Elaboration of an optimal, universal, user-friendly algorithm for withdrawal of ICS therapy has been identified as an important clinical need. This article reviews the available evidence on the efficacy, risks, and indications of ICS in COPD, as well as the benefits of ICS treatment withdrawal in patients for whom its use is not recommended by current guidelines. After discussing proposed approaches to ICS withdrawal published by professional associations and individual authors, we present a new algorithm developed by consensus of an international group of experts in the field of COPD. This relatively simple algorithm is based on consideration and integrated assessment of the most relevant factors (markers) influencing decision-making, such a history of exacerbations, peripheral blood eosinophil count, presence of infection, and risk of community-acquired pneumonia.
Keywords: COPD, exacerbation, inhaled corticosteroid, patient follow-up, guideline adherence, treatment algorithm
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive chronic disease characterized by persistent bronchial obstruction and respiratory symptoms such as dyspnea, chronic cough, and sputum production.1 Chronic airway inflammation plays a key role in the pathogenesis of COPD, causing narrowing of the small airways, mucus hypersecretion, and destruction of the lung parenchyma.2
At present, there are considerable changes in approaches to prescription of inhaled corticosteroids (ICS) in COPD.3 As has been shown in clinical trials, an ICS, usually prescribed in a fixed-dose combination with a long-acting β2-agonist (LABA), reduces the frequency of COPD exacerbations by an average of 25%,4 and may delay COPD progression5 and improve disease prognosis in certain subgroups of patients.6
However, it is important to note that a significant proportion of COPD patients will not benefit from ICS therapy. For example, Lee et al7 tested the hypothesis that different COPD subtypes respond differently to ICS/LABA therapy, and they found that ICS/LABA therapy provides no significant functional or clinical benefits in comparison with LABA alone in emphysema-dominant subtype patients. Airway inflammation in COPD is primarily neutrophil-driven,8,9 and does not respond to ICS. Moreover, the combination of neutrophil-driven inflammation, bacterial infection, and ICS use can lead to adverse consequences.10,11
In agreement with current evidence, the Global Initiative for Chronic Obstructive Lung Disease (GOLD) strategy reserves ICS use as part of initial treatment for COPD group D patients with two or more moderate exacerbations or one exacerbation leading to hospitalization in the preceding year and a blood eosinophil count greater than 300 cells/µL, or as part of follow-up treatment in COPD patients with persistent exacerbations on long-acting bronchodilator monotherapy or LABA/LAMA combination therapy and a blood eosinophil count greater than 100 cells/µL.1
In the real-world environment, doctors frequently ignore clinical guidelines and, as shown in a number of observational studies conducted in different countries, overutilization of ICS is common, with up to 70% of all COPD patients receiving ICS therapy, including patients in GOLD groups A and B (up to 35% of COPD patients).12 The recent SUPPORT study conducted in Russia also found that the number of ICS prescriptions in real-life practice is too high, with 33% of ICS users among GOLD group B patients.13
The observation that ICS are overprescribed in COPD has led to the suggestion that ICS therapy should be withdrawn in certain patients in order to decrease the risk of adverse effects while also reducing treatment costs.3,11,14 In this article, we provide a rationale for the withdrawal of unnecessary ICS treatment, and suggest an algorithm to achieve this objective.
In this paper we present a new algorithm developed by consensus of an international group of experts. Expert selection for the inclusion into consensus panel was based on their research activities in COPD and their previous publications in peer reviewed journals. First of all, an expert group identified areas of uncertainty about the efficacy of ICS, possible adverse effects associated with their use, and criteria for the withdrawal of ICS. The meeting of the expert group was held three times in a 6 -month period to discuss different publications on ICS withdrawal that were considered by all members of the committee, in order to evaluate whether these published data could have an impact on the development of the algorithm. Then the expert group reached a consensus on whether to take into account the appropriate approach or if the data presented in the analyzed article could serve as a reference supporting this approach. In the absence of consensus it was agreed to make a final solution by a simple majority. Finally, the expert group summarized the available evidence in response to the aforementioned questions.
Efficacy of ICS in COPD – what is the evidence?
Early studies on ICS therapy in COPD patients failed to demonstrate any beneficial effects on lung function and symptom severity.15 However, an assessment of secondary endpoints in the ISOLDE study16 showed that fluticasone propionate did reduce the number of exacerbations in COPD patients with severe airflow limitation (forced expiratory volume in 1 second (FEV1) of less than 50% predicted).17 Subsequently, this ability to prevent COPD exacerbations was demonstrated for a number of other ICS,18 and it was found that these drugs were effective not only in high doses, as believed initially, but also in moderate doses.19
A number of studies were conducted to test the hypothesis that ICS therapy not only decreases the number of exacerbations, but can also reduce mortality and delay COPD progression. Despite a considerable body of data accumulated over up to 3 years of follow-up, no statistically significant results have been obtained in favor of the hypothesis, although the studies revealed a slight tendency for increased survival in ICS (combined with LABA) vs placebo.20,21
As ICS do not have the bronchodilator properties that are necessary to alleviate COPD symptoms, further studies evaluated fixed-dose ICS/LABA combinations. This treatment combination was assumed to have a good bronchodilator effect and to be superior to bronchodilator therapy alone in terms of exacerbation prevention. However, evidence in favor of this assumption has not been demonstrated in all studies. For example, in the 3-year TORCH study, the combination of fluticasone propionate and salmeterol was superior to salmeterol alone (risk ratio [RR]=0.88) in preventing moderate and severe exacerbations in COPD patients (with a prebronchodilator FEV1 of less than 60% predicted), but provided no benefit when only severe exacerbations were included in the analysis.20 Furthermore, in the INSTEAD trial, no difference in the number of exacerbations was found between fluticasone/salmeterol and indacaterol in COPD patients with moderate airflow limitation and a history of no exacerbations in the previous year.22 Finally, another fixed combination (budesonide/formoterol) significantly decreased the number of exacerbations compared to formoterol, but the effect of formoterol on the risk of exacerbations differed little compared to placebo in these studies (all patients had a FEV1 of less than 50% predicted and ≥1 exacerbation within 2–12 months before the study).23,24 A meta-analysis of 18 randomized clinical trials found no significant superiority of ICS/LABA combinations over treatment with LABA alone in reducing the risk of severe exacerbations.25
Fewer clinical studies have compared the efficacy of ICS/LABA combinations and long-acting muscarinic agonists (LAMA). Once again, no difference was found in exacerbation rates between patients who received fluticasone/salmeterol and those who were treated with tiotropium bromide over a 2-year period (COPD patients had FEV1 of less than 50% predicted and a clinical history of COPD exacerbations).26 However, significantly more exacerbations related to an acute respiratory infection were observed in the fluticasone/salmeterol group.
Two possible explanations may be suggested for the inconsistent results of direct comparisons of ICS/LABA with bronchodilator monotherapies. First, some COPD patients may develop resistance to ICS,27 and, second, ICS efficacy may be strongly influenced by the nature of inflammation in the airways. A post-hoc analysis of data obtained in a number of studies revealed a correlation between the efficacy of ICS and eosinophil count in the mucus and blood of COPD patients.28 In particular, it was found that efficacy of the fluticasone furoate/vilanterol combination was no different from that of vilanterol therapy alone when eosinophil count is low and increases on a pro rata basis as the blood eosinophil count rises.28 A similar tendency was observed when the extrafine beclamethasone dipropionate/formoterol combination was compared to formoterol monotherapy.29 The FLAME study, which compared the number of exacerbations in COPD patients receiving either fluticasone/salmeterol or glycopirronium/indacaterol for 1 year, showed that the combination of two bronchodilator drugs was generally more effective in decreasing the number of exacerbations (RR=0.88, intention-to-treat analysis).30 However, a post hoc analysis based on blood eosinophil count found that the effect of the ICS-containing combination on the number of exacerbations was no different from that of the combination of two bronchodilator drugs in patients with high eosinophilia (≥3% or ≥150 cells/μL).31
Although ICS have been shown conclusively in many studies to reduce the number of exacerbations compared to placebo, the advantage of ICS/LABA combinations over bronchodilator drugs used as monotherapy cannot be easily demonstrated. First, ICS are more effective in patients with more severe lung function impairment. Second, the efficacy of ICS in reducing the risk of exacerbations is higher on a pro rata basis as mucus secretion and blood eosinophil count increase and, although ICS-containing combinations more effectively prevent exacerbations requiring ICS prescription, these are a priori inferior to bronchodilator therapies in preventing exacerbations associated with a respiratory infection.26
Risks associated with ICS therapy in COPD patients
Use of ICS in COPD patients is associated with a number of adverse events.32 Important adverse effects of ICS therapy include increased risks of pneumonia,33 tuberculosis (TB), and non-tuberculous mycobacterial infections,34 osteoporosis and bone fracture,35 as well as poor diabetes control36 and local reactions (such as oral candidiasis, hoarse voice, cough, etc.) (Table 1).
 | Table 1 Side-effects of ICS in COPD and the type of studies |
Pneumonia is the most frequently reported problem in both randomized controlled trials (RCTs) and observational studies. In a meta-analysis, Horita et al37 reported a 43% increased risk of pneumonia in COPD patients treated with an ICS/LABA combination compared to LABA/LAMA (hazard ratio [HR]=0.57; 95% confidence interval [CI]=0.42–0.79). The increased risk of pneumonia in patients treated with an ICS, such as fluticasone and budesonide, has attracted the attention of regulatory authorities on both sides of the Atlantic.
In May 2015, the European Medicines Agency (EMA) issued a statement declaring that a thorough analysis of the risk of pneumonia in ICS-treated COPD patients needed to be performed based on recent studies and meta-analyses of earlier trials.38 This task was assigned to the Pharmacovigilance Risk Assessment Committee (PRAC). In March 2016, the PRAC published its report, stating that COPD patients on ICS treatment are indeed at increased risk of pneumonia.39 However, the PRAC experts noted that the benefits of ICS outweighed the related risks. Furthermore, the PRAC did not find conclusive evidence of different risks of pneumonia associated with different types of ICS.39
It is generally accepted that long-term therapy with ICS leads to suppression of the cellular component of the immune system and increases susceptibility to intracellular infections of all types. Therefore, it is interesting to assess whether ICS use is associated with an increased risk of non-tuberculous mycobacterial pulmonary disease (NTM-PD) or tuberculosis. Brode et al34 showed that the use of ICS at moderate and high daily doses for 1 year was associated with an increased risk of NTM-PD, but not TB. However, other studies have found a significant association between ICS use and TB.34
Prospective studies on the impact of ICS on bone mineral density in COPD patients have yielded inconsistent results. A meta-analysis by Yang et al40 failed to demonstrate any difference in bone fracture rate in patients treated with ICS compared to placebo. In an earlier meta-analysis, Loke et al35showed that ICS use led to a 19% increased risk of bone fracture compared to placebo; however, this difference was not statistically significant.
In a large cohort study, ICS therapy was associated with an increased risk of diabetes mellitus (HR=1.34; 95% CI=1.29–1.39) and of diabetes progression (HR=1.34; 95% CI=1.17–1.53).36 The risk increased in a dose-dependent manner and was highest in patients who received ICS doses equivalent to fluticasone doses of 1,000 μg/day or higher.
The existence of a moderate risk for cataract has been shown in open-label studies in patients treated with ICS, especially at high doses and for a long time.41 A meta-analysis showed that the number needed to harm is 16, that is to say that 16 patients need to be treated with ICS to cause this adverse event in one patient [95% CI=13–19].42 Also of note is an increased risk of recurrent peptic ulcer hemorrhages in COPD patients on ICS therapy.43
Special caution should be taken when prescribing ICS to older COPD patients. The expected benefits should be carefully weighed against the increased risk of adverse events such as cataract, diabetes, pneumonia, osteoporosis, and bone fracture.44 Furthermore, it should be kept in mind that elderly patients can suffer from vision, hearing, and/or coordination disturbances that may preclude proper use of ICS-containing inhalation devices.44 Therefore, a benefit-risk analysis is strongly recommended prior to prescribing ICS to COPD patients, especially in those who have risk factors for ICS-related complications.
Effects of ICS withdrawal in COPD patients: evidence from clinical trials
Overprescription of ICS to COPD patients and the high risk of serious ICS-related adverse events makes withdrawal of this treatment necessary in patients for whom the treatment-related risks overweigh expected benefits. The effects of ICS withdrawal have been studied in several randomized and non-randomized trials (Table 2).
 | Table 2 Trials investigating the effects of ICS in COPD patients |
In two randomized controlled trials (COPE and WISP), withdrawal of fluticasone propionate led to an increase in exacerbation risk by approximately 50% compared to COPD patients continuing ICS treatment.45,46 However, it should be noted that in these studies patients only received placebo following ICS withdrawal. In all subsequent studies, COPD patients were switched to long-acting bronchodilator drugs post-ICS withdrawal.
In the COSMIC trial, the effects of ICS withdrawal were studied in 373 patients with moderate-to-severe COPD with frequent exacerbations (two or more over the preceding year). Following treatment with fluticasone/salmeterol for 3 months, one group of patients continued the treatment while the second group was switched to salmeterol alone.47 For 1 year, no significant difference in the number of moderate-to-severe exacerbations was observed between the study groups. However, lung function was found to be better in the ICS group (FEV1 difference=4.1%, 95% CI=1.6–6.6%).47
The INSTEAD trial included COPD patients with no indication for ICS prescription, ie, having no severe airflow limitation (FEV1>50% predicted) and without a history of exacerbations.22 Included patients must have been on treatment with fluticasone/salmeterol for at least 3 months before ramdomization. Patients were randomized to two groups and received either continuation with fluticasone/salmeterol or indacaterol for 26 weeks. The study failed to find any difference in the number of COPD exacerbations (RR=0.86; 95% CI=0.62–1.20) or FEV1 (the difference was 9 mL) between the groups.22 Interestingly, no deterioration in quality-of-life or symptom aggravation was seen in COPD patients following ICS withdrawal.
The INSTEAD results were reproduced in the OPTIMO and DACCORD trials, which were conducted in the real-world environment. The OPTIMO trial included COPD patients with FEV1>50%, and with a history of less than two exacerbations over the preceding year.48 The decision to discontinue ICS therapy was made by the treating physician at enrolment. No difference was found between the study groups in the number of exacerbations, symptom severity, or decline of lung function after 6 months.
In DACCORD, which included 1,258 COPD patients (mean FEV1 60% predicted), the decision to discontinue ICS therapy was also made by the treating doctor at study enrolment. ICS was discontinued in 19% of the study population. The study failed to show any difference between ICS users and non-users in the number of exacerbations and symptom severity over 2 years of observation.49 Moreover, by the end of the 2-year period, the risk of exacerbations was found to be lower in ICS non-users compared to patients who were still on ICS therapy.49
The largest trial conducted thus far, WISDOM, which included 2,485 COPD patients, compared a continued treatment regimen with tiotropium, salmeterol, and fluticasone propionate to a treatment regimen which included tiotropium, salmeterol, and a stepwise ICS dose de-escalation (every 6 weeks) up to full withdrawal.50 All COPD patients had severe airflow limitation (FEV1<50% predicted) and a history of at least one exacerbation during the year prior to enrolment. ICS withdrawal did not lead to an increased number of COPD exacerbations compared to continued ICS users (RR=1.06; 95% CI=0.94–1.19). Nevertheless, ICS withdrawal led to a statistically significant deterioration of lung function (mean FEV1 difference=38 mL), but this was not clinically relevant. A subsequent post-hoc analysis of WISDOM data showed that the rate of decline of lung function in COPD patients who discontinued ICS was no different from that in patients who continued ICS use.51 The post-hoc analysis of the WISDOM trial found that patients with higher blood eosinophil counts at screening were more likely to develop exacerbations after ICS treatment was withdrawn.52 Continuation of ICS was associated with a reduced rate of moderate or severe exacerbations in the smaller subgroups of patients with eosinophil counts of 300 cells/μL or more or 4% or greater, whereas there was no difference in exacerbation rate after discontinuation of ICS in patients with counts of less than 4% or less than 300 cells/μL.
In the open-label, randomized CRYSTAL trial, patients with moderate COPD and pronounced symptoms (mMRC dyspnea score ≥1) were switched from ICS/LABA to a fixed-dose combination of indacaterol/glycopyrronium for 12 weeks. This resulted in an improvement of FEV1 (by 71 mL, 95% CI=36–107 mL) and a decrease in dyspnea severity (improvement of TDI score=1.10 units, 95% CI=0.65–1.55 units).53
The 26-week, randomized SUNSET trial, which involved 1,053 COPD patients, studied the effects of ICS withdrawal in COPD patients who received triple therapy for at least 6 months.53 COPD patients (FEV1 50–80% predicted) with a history of at least one exacerbation over the preceding year were randomly assigned to either tiotropium plus salmeterol/fluticasone or indacaterol/glycopyrronium. Over the study period, no difference was seen between study groups in the number of exacerbations (RR=1.08; 95% CI=0.83–1.40), with a minor, clinically irrelevant reduction of FEV1 (−26 mL; 95% CI=−53–1 mL) observed in the ICS withdrawal group. In a subgroup of COPD patients with peripheral blood eosinophil count ≥300 cells/μL (approximately 25%), the number of moderate and severe COPD exacerbations was significantly higher following ICS withdrawal (RR=1.86; 95% CI=1.06–3.29), which is consistent with earlier data indicative of a higher ICS efficacy in this patient category.
A recently published study on the efficacy and safety of the direct switch to indacaterol/glycopyrronium from salmeterol/fluticasone in non-frequently exacerbating COPD patients (the FLASH trial) provided additional evidence on abrupt withdrawal of ICS therapy in COPD patients.55 In this 12-week double-blind study of 502 COPD patients it has been observed that a direct switch from ICS/LABA to LAMA/LABA, without a washout period, improved lung function (pre-dose FEV1 and FVC) in patients with moderate-to-severe COPD with up to one exacerbation in the previous year, without new safety signals. These results add new evidence that dual bronchodilation is a preferable way of treatment compared with ICS/LABA in symptomatic COPD patients with low risk of exacerbations.
ICS withdrawal can also result in a reduction in the risk of adverse events, particularly pneumonia. In a population-based cohort trial, ICS withdrawal was associated with a reduction of the risk of clinically relevant pneumonia by 20% at 1 month and by 37% during the subsequent 3 years.56 Reduction of the risk of pneumonia was higher in patients treated with fluticasone as compared to budesonide-treated patients.54 These data show that beneficial effects on selected outcomes (eg, pneumonia) can be expected soon after ICS withdrawal.
Based on the results of the studies described above (see Table 2), it can be concluded that ICS withdrawal does not lead to a significant increase in the risk of exacerbations in COPD patients with no prior history of frequent exacerbations, regardless of their lung function (ie, both in those with FEV1>50% and <50% predicted). Moreover, ICS withdrawal results in a reduction of the risk of adverse events, particularly pneumonia, in the short-term. However, ICS withdrawal can have unfavorable effects in COPD patients who suffer from frequent exacerbations and who have elevated eosinophil levels in the peripheral blood.
Algorithms for ICS withdrawal in COPD patients
As a result of overutilization of ICS in real-world practice and with the advent of effective combined bronchodilator therapies to treat COPD, elaboration of an optimal, universal, user-friendly algorithm for withdrawal of ICS therapy appears to be important.
In recent years, some professional associations and individual authors have designed approaches to discontinue ICS. One of the first approaches for stepwise ICS withdrawal was proposed by Kaplan.57 The proposed algorithm was built on five steps and took into account:
- reviewing current management of COPD;
- evaluating the risk-benefit profile of continuing ICS therapy, which needs considering patient history and clinical characteristics, spirometry, sputum/blood eosinophil, and FeNO levels for answering the questions about presence of ACO, frequent exacerbations, and markers of eosinophilic inflammation;
- initiating stepwise ICS withdrawal based on the patient’s current ICS dose;
- optimization of bronchodilation with LABA/LAMA combination; and
- following-up patient every 3 months during the first year and then annually if COPD stable and exacerbation free.
The discussion and proposition of differential stepwise, “hit hard”, step-up, and ICS withdrawal therapeutic approaches for COPD patients based on their GOLD category and sub-categories was made by a group of Portuguese experts.58 They concluded that all approaches are feasible and the choice of the best approach should be based on patient careful selection and sub-categorizing, taking into account different characteristics: initial FEV1 and worsening of FEV1 in annual assessment for category A (AX1: FEV1>80%; no worsening of FEV1 in annual assessment; AX2: 50%<FEV1<80%; and/or worsening of FEV1 in annual assessment); lung function, mMRC-score, and cardiovascular comorbidities (category B); lung function and exacerbation risk for categories C and D.
This approach is based to a certain extent on WISDOM study and seems to be complicated for practical use. Moreover, the authors emphasized that the proposed algorithm will need to be validated, particularly in the real-life setting.
Another strategy has been proposed by a group of Spanish authors.59 The first and essential condition for its implementation is an established COPD diagnosis (criteria for COPD diagnosis: age ≥35 years, current or past smoking history ≥10 pack-years, post-bronchodilation FEV1/FVC <0.7) with or without asthma. A concomitant diagnosis of asthma and COPD is made when the key diagnostic criteria for both diseases are met. Asthma–COPD overlap can also be diagnosed in patients without diagnostic criteria for asthma, but who have a positive bronchodilation test ≥400 mL and 15% FEV1 improvement and/or eosinophil count ≥300 cells/μL.60 Patients with non-fully reversible airflow obstruction, but with a smoking history of fewer than 10 pack-years are classified and treated as asthma patients.
The proposed algorithm distinguishes two categories on opposite ends of the spectrum: patients without asthma, with preserved lung function (FEV1>50% predicted) and no history of exacerbation; and patients with concomitant asthma and a history of exacerbations. ICS therapy should definitely be discontinued in the first group and continued in the second. Intermediate categories, which include patients without asthma, with FEV1>50% and exacerbations, and patients with FEV1<50%, concomitant asthma, and no history of exacerbations, require close follow-up and the use of a differentiated approach to ICS withdrawal. ICS withdrawal is not indicated for patients with asthma-COPD overlap and exacerbations.
A group of authors from the UK proposed a protocol for ICS withdrawal in COPD patients intended for primary care physicians.61 The authors recommend caution in applying the protocol to patients with a concomitant diagnosis of COPD and asthma. The protocol uses severity of lung function impairment as its starting point. For patients with relatively preserved lung function (FEV1≥50% predicted) and a history of frequent exacerbations (≥2 exacerbations over 1 year), the protocol recommends reducing ICS dose, with due consideration for concomitant diseases (asthma, bronchiectasis), as well as prescription of pulmonary rehabilitation programs and monitoring of inhaler technique. For patients with preserved lung function (FEV1≥50% predicted) and less than two exacerbations during the past year, an immediate switch to a LAMA/LABA combination is recommended.
In patients with a pronounced decrease of FEV1 (<50% predicted) and a history of exacerbations, ICS therapy should be continued, and a reduction of the ICS dose should be considered. For patients with a 50% decrease in lung function and less than two exacerbations during the past year, a switch to a fixed-dose bronchodilator drug combination is recommended.
In order to prepare a new algorithm for ICS withdrawal, the international expert group reviewed a number of significant issues related to ICS withdrawal in COPD patients raised by Cataldo et al3 in a recent publication:
- Only a minority of COPD patients derive benefit from ICS therapy. These are patients with a high benefit-to-risk ratio, including patients with a concomitant diagnosis of COPD and asthma, as well as patients suffering from frequent exacerbations despite optimal bronchodilator therapy.
- ICS therapy is not indicated in COPD patients:
- with a newly-diagnosed disease,
- who have developed severe adverse reactions as a consequence of ICS therapy, and
- who have no history of exacerbations.
- ICS withdrawal can be considered in patients who had no apparent indication for ICS therapy in the past. Generally, these are patients whose treatment was initiated prior to the publication of the current guidelines and before the advent of fixed-dose LABA/LAMA combinations.
- Current evidence on the topic is limited, and the available studies are heterogeneous in terms of study designs (randomized, controlled, or observational) and study populations (variable disease severity and exacerbation frequency).
- It should be remembered that no substantiated, practical guidelines on the strategy for ICS withdrawal, either immediate or stepwise, are available at present. Instead, there are limited data indicating systemic symptoms that may develop as a result of depression of endogenous steroid production, such as those observed after ICS withdrawal in the run-in period in the ISOLDE study.62 On the other hand, the level of patient acceptance of ICS therapy is low in the real-world environment, resulting in poor adherence to the prescribed treatment.11,61,63
Cataldo et al3 proposed a fairly simple algorithm based on exacerbation history. The authors suggest that patients with no history of exacerbations during the preceding year (approximately 75% of the DACCORD study population)64 who are treated with either an ICS/LABA/LAMA or an ICS/LABA combination may benefit from ICS withdrawal and a switch to a double bronchodilator therapy.
A different approach should be applied in patients with a history of exacerbations during the previous 12 months (25% of the DACCORD study population).64 If the patient was on triple therapy, prescription of an add-on therapy and ICS dose correction should be considered. If the patient was receiving an ICS/LABA combination, modification of the treatment regimen is recommended by either adding a LAMA or by switching the patient to a LAMA/LABA combination.
Finally, the new GOLD 2017 classification has not included the FEV1 in the initial classification of symptoms and risk, and, therefore, a significant number of patients originally classified as D according to GOLD 2013 will now be classified as B, indicating low level of risk. In this case, patients newly classified as B according to GOLD 2017 should be discontinued from ICS if they have less than 300 eosinophils/μL.65
This is a selective and far from complete list of approaches to withdrawal of unnecessary ICS, as defined in currently available clinical guidelines. The expert group held several working group meetings in order to formulate approaches to ICS withdrawal in COPD patients.66–68 The expert team reviewed available approaches to (and algorithms for) ICS withdrawal and switching to treatment regimens without ICS. It was emphasized that any attempt to include more factors than are considered in the currently available protocols would inevitably lead to an oversophisticated algorithm that would be difficult to use in everyday clinical practice. On the other hand, oversimplification of the approach by minimizing the number of the factors would not allow for all possible clinical situations to be covered. In view of the above, the working group tried to use an integrated approach to formulate a relatively simple algorithm that is based on consideration and integrated assessment of the most relevant factors (markers) influencing decision-making, such as a history of exacerbations, peripheral blood eosinophil count, presence of infection, and risk of community-acquired pneumonia.66–68
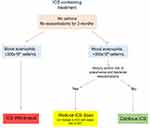
In the algorithm proposed (see Figure 1), stable COPD, ie, the absence of exacerbations in the previous 3 months, is the essential prerequisite for further therapy revision in COPD patients without concomitant asthma. Once the above requirement is met, blood eosinophil count is used as the main marker to guide decision-making. If the blood eosinophil count is below a cut-off value of 300 cells/μL, ICS withdrawal is appropriate for patients treated with an ICS/LABA, ICS/LAMA, or ICS/LABA/LAMA triple combination.
For patients with a blood eosinophil count above a cut-off value of 300 cells/μL, the treatment strategy should be based on whether the patient has a history and risk of infectious exacerbations and/or community-acquired pneumonia, which is indicative of a high risk of occurrence of these conditions in the future.56 Factors associated with a significant increase in the risk of ICS associated pneumonia could be defined as: being a current smoker, having prior pneumonia, body mass index <25 kg/m2, and severe airflow limitation (FEV1<50%).69 A large population-based cohort trial demonstrated that reduction of the risk of pneumonia can be expected soon after ICS withdrawal.56
Patients with persistent exacerbations despite ICS treatment and features of so called “infective phenotype“ (frequent bacterial exacerbations, chronic bacterial colonization, bronchiectasis)70 can also require ICS reduction or withdrawal, and additional use of long-term antibiotics has been advocated in these patients.71
ICS dose reduction as a way of treatment modification (rather than withdrawal) would be a reasonable therapeutic option for many COPD patients, for example, with high eosinophil counts and a history/risk of pneumonia and infectious exacerbations.
This ICS reduction step of the decision-making tree can be justified by previously shown predominantly anti-inflammatory effects related to modulation of the humoral components of the innate immune response, which may lead to reduced airway bacterial load and lower risk of pneumonia.10,72
The concept of switching to ICS with lower risk of side-effects (SE) is based on data which demonstrated substantially increased risk of pneumonia with using of more potent and lipophilic ICS such as fluticasone propionate or fluticasone furoate, but not beclometasone dipropionate or budesonide.73,74 It is possible that the differences observed in the incidence of pneumonia reflect the dose of ICS used. Almost all salmeterol/fluticasone trials used fluticasone 500 μg bid, whereas all the budesonide/formoterol trials used budesonide 320 μg bid. (Dose equivalences used are beclomethasone 1,000 μg, budesonide 800 μg, fluticasone propionate 500 μg, and fluticasone furoate 100 μg).
So, in COPD patients with history and/or increased risk of pneumonia and/or infectious exacerbations, it’s reasonable to change ICS to a molecule with lower risk of side-effects (eg, from fluticasone to budesonide or beclomethasone).74,75
ICS withdrawal is not recommended for COPD patients with high eosinophil counts without a risk of infectious exacerbations and/or community-acquired pneumonia.
Conclusion
Common overutilization and the high risk of serious treatment-related adverse events make withdrawal of this ICS therapy necessary in COPD patients for whom its use is not recommended by current guidelines. Practical implementation of ICS withdrawal in everyday clinical practice for clinicians is needed in the management of COPD patients. The ICS withdrawal algorithm developed by an expert group provides a user-friendly yet comprehensive integrated assessment of the most relevant factors influencing decision-making, such as a history of exacerbations, peripheral blood eosinophil count, presence of infection, and risk of community-acquired pneumonia.
Disclosure
Professor Dr Sergey Avdeev has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, Teva, and Novartis; and research grants from AstraZeneca, GlaxoSmithKline, and Novartis, outside the submitted work. Professor Dr Zaurbek Aisanov has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, Teva, Novartis, and GlaxoSmithKline; and research grants from AstraZeneca, GlaxoSmithKline, and Novartis, outside the submitted work. Professor Dr Vladimir Arkhipov has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, Teva, and Astrllas; non-financial support from Boehringer Ingelheim; and research grants from AstraZeneca, outside the submitted work. Professor Dr Andrey Belevskiy has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, and Novartis, outside the submitted work. Professor Dr Igor Leshchenko has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, Teva, and Novartis, outside the submitted work. Professor Dr Svetlana Ovcharenkohas received speaker/consultancy fees from AstraZeneca, Boehringer Ingelheim, Chiesi, and Novartis, outside the submitted work. Professor Dr Evgeny Shmelev has received speaker/consultancy fees from Boehringer Ingelheim, AstraZeneca, Chiesi, Roche, Teva, and Novartis, outside the submitted work. Dr Marc Miravitlles has received speaker fees from Boehringer Ingelheim, Chiesi, Cipla, Menarini, Rovi, Bial, CSL Behring, Grifols, and Novartis; consultancy fees from Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Bial, Gebro Pharma, CSL Behring, Laboratories Esteve, Mereo BioPharm, Verona Pharma, pH Pharma, Novartis, and Grifols; and research grants from GlaxoSmithKline and Grifols, outside the submitted work.
References
1.
2. Barnes PJ. Cellular and molecular mechanisms of chronic obstructive pulmonary disease. Clin Chest Med. 2014;35:71–86. doi:10.1016/j.ccm.2013.10.004
3. Cataldo D, Derom E, Liistro G, et al. Overuse of inhaled corticosteroids in COPD: five questions for withdrawal in daily practice. Int J Chron Obstruct Pulmon Dis. 2018;13:2089–2099. doi:10.2147/COPD.S164259
4. Nannini LJ, Lasserson TJ, Poole P. Combined corticosteroid and long-acting beta(2)-agonist in one inhaler versus long-acting beta(2)-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012; 9: CD006829.
5. Celli BR, Thomas NE, Anderson JA, et al. Effect of pharmacotherapy on rate of decline of lung function in chronic obstructive pulmonary disease: results from the TORCH study. Am J Respir Crit Care Med. 2008;178:332–338. doi:10.1164/rccm.200712-1869OC
6. Gershon AS, Campitelli MA, Croxford R, et al. Combination long-acting beta-agonists and inhaled corticosteroids compared with long-acting beta-agonists alone in older adults with chronic obstructive pulmonary disease. JAMA. 2014;312:1114–1121. doi:10.1001/jama.2014.11432
7. Lee JH, Lee YK, Kim EK, et al. Responses to inhaled long-acting beta-agonist and corticosteroid according to COPD subtype. Respir Med. 2010;104:542–549. doi:10.1016/j.rmed.2009.10.024
8. Agusti A, Bel E, Thomas M, et al. Treatable traits: toward precision medicine of chronic airway diseases. Eur Respir J. 2016;47:410–419. doi:10.1183/13993003.01359-2015
9. Dicker AJ, Crichton ML, Pumphrey EG, et al. Neutrophil extracellular traps are associated with disease severity and microbiota diversity in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2018;141:117–127. doi:10.1016/j.jaci.2017.04.022
10. Contoli M, Pauletti A, Rossi MR, et al. Long-term effects of inhaled corticosteroids on sputum bacterial and viral loads in COPD. Eur Respir J. 2017;50:1700451. doi:10.1183/13993003.00711-2017
11. Chalmers JD. POINT: should an attempt be made to withdraw inhaled corticosteroids in all patients with stable GOLD 3 (30%</= FEV1<50% predicted) COPD? Yes. Chest. 2018;153:778–782. doi:10.1016/j.chest.2018.01.029
12. Yawn BP, Li Y, Tian H, Zhang J, Arcona S, Kahler KH. Inhaled corticosteroid use in patients with chronic obstructive pulmonary disease and the risk of pneumonia: a retrospective claims data analysis. Int J Chron Obstruct Pulmon Dis. 2013;8:295–304. doi:10.2147/COPD.S42366
13. Arkhipov V, Arkhipova D, Miravitlles M, Lazarev A, Stukalina E. Characteristics of COPD patients according to GOLD classification and clinical phenotypes in the Russian Federation: the SUPPORT trial. Int J Chron Obstruct Pulmon Dis. 2017;12:3255–3262. doi:10.2147/COPD.S142997
14. Price D, Yawn B, Brusselle G, Rossi A. Risk-to-benefit ratio of inhaled corticosteroids in patients with COPD. Prim Care Respir J. 2013;22:92–100. doi:10.4104/pcrj.2012.00092
15. Pauwels RA, Lofdahl CG, Laitinen LA, et al. Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European respiratory society study on chronic obstructive pulmonary disease. N Engl J Med. 1999;340:1948–1953. doi:10.1056/NEJM199906243402503
16. Burge PS, Calverley PM, Jones PW, Spencer S, Anderson JA, Maslen TK. Randomised, double blind, placebo controlled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trial. Bmj. 2000;320:1297–1303. doi:10.1136/bmj.320.7245.1297
17. Jones PW, Willits LR, Burge PS, Calverley PM. Disease severity and the effect of fluticasone propionate on chronic obstructive pulmonary disease exacerbations. Eur Respir J. 2003;21:68–73.
18. Zervas E, Samitas K, Gaga M, Beghe B, Fabbri LM. Inhaled corticosteroids in COPD: pros and cons. Curr Drug Targets. 2013;14:192–224.
19. Cheng SL, Su KC, Wang HC, Perng DW, Yang PC. Chronic obstructive pulmonary disease treated with inhaled medium- or high-dose corticosteroids: a prospective and randomized study focusing on clinical efficacy and the risk of pneumonia. Drug Des Devel Ther. 2014;8:601–607. doi:10.2147/DDDT.S63100
20. Calverley PM, Anderson JA, Celli B, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775–789. doi:10.1056/NEJMoa063070
21. Vestbo J, Anderson JA, Brook RD, et al. Fluticasone furoate and vilanterol and survival in chronic obstructive pulmonary disease with heightened cardiovascular risk (SUMMIT): a double-blind randomised controlled trial. Lancet. 2016;387:1817–1826. doi:10.1016/S0140-6736(16)30069-1
22. Rossi A, van der Molen T, Del Olmo R, et al. INSTEAD: a randomised switch trial of indacaterol versus salmeterol/fluticasone in moderate COPD. Eur Respir J. 2014;44:1548–1556. doi:10.1183/09031936.00126814
23. Szafranski W, Cukier A, Ramirez A, et al. Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease. Eur Respir J. 2003;21:74–81.
24. Calverley PM, Boonsawat W, Cseke Z, Zhong N, Peterson S, Olsson H. Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease. Eur Respir J. 2003;22:912–919.
25. Rodrigo GJ, Castro-Rodriguez JA, Plaza V. Safety and efficacy of combined long-acting beta-agonists and inhaled corticosteroids vs long-acting beta-agonists monotherapy for stable COPD: a systematic review. Chest. 2009;136:1029–1038. doi:10.1378/chest.09-0821
26. Wedzicha JA, Calverley PM, Seemungal TA, Hagan G, Ansari Z, Stockley RA. The prevention of chronic obstructive pulmonary disease exacerbations by salmeterol/fluticasone propionate or tiotropium bromide. Am J Respir Crit Care Med. 2008;177:19–26. doi:10.1164/rccm.200707-973OC
27. Barnes PJ. Corticosteroid resistance in patients with asthma and chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2013;131:636–645. doi:10.1016/j.jaci.2012.12.1564
28. Pascoe S, Locantore N, Dransfield MT, Barnes NC, Pavord ID. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomised controlled trials. Lancet Respir Med. 2015;3:435–442. doi:10.1016/S2213-2600(15)00106-X
29. Siddiqui SH, Guasconi A, Vestbo J, et al. Blood eosinophils: a biomarker of response to extrafine beclomethasone/formoterol in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;192:523–525. doi:10.1164/rccm.201502-0235LE
30. Wedzicha JA, Banerji D, Chapman KR, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. N Engl J Med. 2016;374:2222–2234. doi:10.1056/NEJMoa1516385
31. Roche N, Chapman KR, Vogelmeier CF, et al. Blood eosinophils and response to maintenance chronic obstructive pulmonary disease treatment. data from the FLAME trial. Am J Respir Crit Care Med. 2017;195:1189–1197. doi:10.1164/rccm.201701-0193OC
32. Ernst P, Saad N, Suissa S. Inhaled corticosteroids in COPD: the clinical evidence. Eur Respir J. 2015;45:525–537. doi:10.1183/09031936.00128914
33. Kew KM, Seniukovich A. Inhaled steroids and risk of pneumonia for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014; 3: CD010115.
34. Brode SK, Campitelli MA, Kwong JC, et al. The risk of mycobacterial infections associated with inhaled corticosteroid use. Eur Respir J. 2017;50:1700037. doi:10.1183/13993003.00711-2017
35. Loke YK, Cavallazzi R, Singh S. Risk of fractures with inhaled corticosteroids in COPD: systematic review and meta-analysis of randomised controlled trials and observational studies. Thorax. 2011;66:699–708. doi:10.1136/thx.2011.160028
36. Suissa S, Kezouh A, Ernst P. Inhaled corticosteroids and the risks of diabetes onset and progression. Am J Med. 2010;123:1001–1006. doi:10.1016/j.amjmed.2010.06.019
37. Horita N, Goto A, Shibata Y, et al. Long-acting muscarinic antagonist (LAMA) plus long-acting beta-agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev. 2017;2:CD012066.
38.
39.
40. Yang IA, Clarke MS, Sim EH, Fong KM. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012; 2: CD002991.
41. Weatherall M, Clay J, James K, Perrin K, Shirtcliffe P, Beasley R. Dose-response relationship of inhaled corticosteroids and cataracts: a systematic review and meta-analysis. Respirology. 2009;14:983–990. doi:10.1111/j.1440-1843.2009.01589.x
42. Uboweja A, Malhotra S, Pandhi P. Effect of inhaled corticosteroids on risk of development of cataract: a meta-analysis. Fundam Clin Pharmacol. 2006;20:305–309. doi:10.1111/j.1472-8206.2006.00397.x
43. Huang KW, Kuan YC, Chi NF, Huang YH, Luo JC, Chien LN. Chronic obstructive pulmonary disease is associated with increased recurrent peptic ulcer bleeding risk. Eur J Intern Med. 2017;37:75–82. doi:10.1016/j.ejim.2016.09.020
44. Rossi AP, Zanardi E, Zamboni M, Rossi A. Optimizing treatment of elderly COPD patients: what role for inhaled corticosteroids? Drugs Aging. 2015;32:679–687. doi:10.1007/s40266-015-0291-8
45. van der Valk P, Monninkhof E, van der Palen J, Zielhuis G, van Herwaarden C. Effect of discontinuation of inhaled corticosteroids in patients with chronic obstructive pulmonary disease: the COPE study. Am J Respir Crit Care Med. 2002;166:1358–1363. doi:10.1164/rccm.200206-512OC
46. Choudhury AB, Dawson CM, Kilvington HE, et al. Withdrawal of inhaled corticosteroids in people with COPD in primary care: a randomised controlled trial. Respir Res. 2007;8:93. doi:10.1186/1465-9921-8-91
47. Wouters EF, Postma DS, Fokkens B, et al. Withdrawal of fluticasone propionate from combined salmeterol/fluticasone treatment in patients with COPD causes immediate and sustained disease deterioration: a randomised controlled trial. Thorax. 2005;60:480–487. doi:10.1136/thx.2004.034280
48. Rossi A, Guerriero M, Corrado A. Withdrawal of inhaled corticosteroids can be safe in COPD patients at low risk of exacerbation: a real-life study on the appropriateness of treatment in moderate COPD patients (OPTIMO). Respir Res. 2014;15:77. doi:10.1186/1465-9921-15-77
49. Vogelmeier C, Worth H, Buhl R, et al. “Real-life” inhaled corticosteroid withdrawal in COPD: a subgroup analysis of DACCORD. Int J Chron Obstruct Pulmon Dis. 2017;12:487–494. doi:10.2147/COPD.S125616
50. Magnussen H, Disse B, Rodriguez-Roisin R, et al. Withdrawal of inhaled glucocorticoids and exacerbations of COPD. N Engl J Med. 2014;371:1285–1294. doi:10.1056/NEJMoa1407154
51. Magnussen H, Tetzlaff K, Bateman ED, et al. Lung function changes over time following withdrawal of inhaled corticosteroids in patients with severe COPD. Eur Respir J. 2016;47:651–654. doi:10.1183/13993003.01644-2015
52. Watz H, Tetzlaff K, Wouters EF, et al. Blood eosinophil count and exacerbations in severe chronic obstructive pulmonary disease after withdrawal of inhaled corticosteroids: a post-hoc analysis of the WISDOM trial. Lancet Respir Med. 2016;4:390–398. doi:10.1016/S2213-2600(16)00100-4
53. Vogelmeier CF, Gaga M, Aalamian-Mattheis M, et al. Efficacy and safety of direct switch to indacaterol/glycopyrronium in patients with moderate COPD: the CRYSTAL open-label randomised trial. Respir Res. 2017;18:140. doi:10.1186/s12931-017-0622-x
54. Chapman KR, Hurst JR, Frent SM, et al. Long-term triple therapy de-escalation to indacaterol/glycopyrronium in patients with chronic obstructive pulmonary disease (SUNSET): a randomized, double-blind, triple-dummy clinical trial. Am J Respir Crit Care Med. 2018;198:329–339. doi:10.1164/rccm.201803-0405OC
55. Frith PA, Ashmawi S, Krishnamurthy S, et al. Efficacy and safety of the direct switch to indacaterol/glycopyrronium from salmeterol/fluticasone in non-frequently exacerbating COPD patients: the FLASH randomized controlled trial. Respirology. 2018;23:1152–1159. doi:10.1111/resp.13374
56. Suissa S, Coulombe J, Ernst P. Discontinuation of inhaled corticosteroids in COPD and the risk reduction of pneumonia. Chest. 2015;148:1177–1183. doi:10.1378/chest.15-0627
57. Kaplan AG. Applying the wisdom of stepping down inhaled corticosteroids in patients with COPD: a proposed algorithm for clinical practice. Int J COPD. 2015;10:2535–2548. doi:10.2147/COPD.S93321
58. Ferreira AJ, Reis A, Marçal N, Pinto P, Bárbara C. COPD A stepwise or a hit hard approach? Rev Port Pneumol. 2016;22:214–221.
59. Miravitlles M, Cosio BG, Arnedillo A, et al. A proposal for the withdrawal of inhaled corticosteroids in the clinical practice of chronic obstructive pulmonary disease. Respir Res. 2017;18:198. doi:10.1186/s12931-017-0682-y
60. Miravitlles M, Alvarez-Gutierrez FJ, Calle M, et al. Algorithm for identification of asthma-COPD overlap: consensus between the Spanish COPD and asthma guidelines. Eur Respir J. 2017;49:1700068. doi:10.1183/13993003.00068-2017
61. Chalmers JD, Bobak A, Scullion J, Murphy A. Withdrawal of ICS treatment in primary care: a practical guide. Pract Nurs. 2017;28:22–27. doi:10.12968/pnur.2017.28.1.22
62. Jarad NA, Wedzicha JA, Burge PS, Calverley PM. An observational study of inhaled corticosteroid withdrawal in stable chronic obstructive pulmonary disease. ISOLDE study group. Respir Med. 1999;93:161–166.
63. Sulaiman I, Cushen B, Greene G, et al. Objective assessment of adherence to inhalers by patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2017;195:1333–1343. doi:10.1164/rccm.201604-0733OC
64. Kardos P, Vogelmeier C, Worth H, et al. A two-year evaluation of the ‘real life’ impact of COPD on patients in Germany: the DACCORD observational study. Respir Med. 2017;124:57–64. doi:10.1016/j.rmed.2017.02.007
65. Harlander M, Barrecheguren M, Turel M, Miravitlles M. Should patients switched from D to B in the GOLD 2017 classification be discontinued from inhaled corticosteroids? COPD. 2017;14:465–468. doi:10.1080/15412555.2017.1342233
66. Avdeev SN, Aisanov ZR, Belevskii AS, et al. Prospects for pharmacotherapy of chronic obstructive pulmonary disease: capabilities of combined bronchodilator drugs and the place of inhalation corticosteroids. Expert panel report [Article in Russian]. Pulmonologiya. 2016;26:65–72. doi:10.18093/0869-0189-2016-26-1-65-72
67. Avdeev SN, Aisanov ZR, Belevskii AS, et al. New options for prevention of exacerbations of chronic obstructive pulmonary disease. Report of the expert group of the Russian respiratory society [Article in Russian]. Pulmonologiya. 2017;27:108–113. doi:10.18093/0869-0189-2017-27-1-108-113
68. Avdeev SN, Belevskii AS, Aisanov ZR, et al. Capabilities of inhalation therapy for prevention of exacerbations of chronic obstructive pulmonary disease. Report of the expert panel of the Russian respiratory society [Article in Russian]. Pulmonologiya. 2018;28:368–380. doi:10.18093/0869-0189-2018-28-3-368-380
69. Crim C, Dransfield MT, Bourbeau J, et al. Pneumonia risk with inhaled fluticasone furoate and vilanterol compared with vilanterol alone in patients with COPD. Ann Amer Thorac Soc. 2015;12:27–34. doi:10.1513/AnnalsATS.201409-413OC
70. Matkovic Z, Miravitlles M. Chronic bronchial infection in COPD Is There an Infective Phenotype?. Respir Med. 2013;107:10–22.
71. Miravitlles M, Anzueto A. Antibiotic prophylaxis in COPD: why, when, and for whom? Pulm Pharmacol Ther. 2015;32:119–123. doi:10.1016/j.pupt.2014.05.002
72. Izquierdo JL, Cosio BG. The dose of inhaled corticosteroids in patients with COPD: when less is better. Int J Chron Obstruct Pulmon Dis. 2018;13:3539–3547. doi:10.2147/COPD.S175047
73. Suissa S, Patenaude V, Lapi F, Ernst P. Inhaled corticosteroids in COPD and the risk of serious pneumonia. Thorax. 2013;68:1029–1036. doi:10.1136/thoraxjnl-2012-202872
74. Lipworth B, Kuo CRW, Jabbal S. Current appraisal of single inhaler triple therapy in COPD. Int J Chron Obstruct Pulmon Dis. 2018;13:3003–3009. doi:10.2147/COPD.S177333
75. Janson C, Stratelis G, Miller-Larsson A, Harrison TW, Larsson K. Scientific rationale for the possible inhaled corticosteroid intraclass difference in the risk of pneumonia in COPD. Int J Chron Obstruct Pulmon Dis. 2017;19(12):3055–3064. doi:10.2147/COPD.S143656
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.