Back to Journals » Clinical Ophthalmology » Volume 14
Vitreous and Serum Concentrations of Vascular Endothelial Growth Factor and Platelet-Derived Growth Factor in Proliferative Diabetic Retinopathy
Authors Muhiddin HS, Kamaruddin MI , Ichsan AM, Budu
Received 7 February 2020
Accepted for publication 18 May 2020
Published 9 June 2020 Volume 2020:14 Pages 1547—1552
DOI https://doi.org/10.2147/OPTH.S248812
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Habibah Setyawati Muhiddin, Muhammad Irfan Kamaruddin, Andi Muhammad Ichsan, Budu
Department of Ophthalmology, Faculty of Medicine, Hasanuddin University, Makassar, South Sulawesi, Indonesia
Correspondence: Habibah Setyawati Muhiddin Email [email protected]
Purpose: This study aimed to investigate the concentrations of vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF) in vitreous and serum samples, analyze the ratio, and compare among proliferative diabetic retinopathy (PDR) subgroups.
Patients and Methods: This study included 17 eyes of patients with PDR, identified as the PDR group which was divided into three subgroups (vitreous hemorrhage [VH], VH with fibrotic tissues, and tractional retinal detachment), and five control eyes (nucleus and intraocular lens drop). Vitreous and serum samples were obtained on the same day. The VEGF-A and PDGF-AB concentrations were calculated by enzyme-linked immunosorbent assay.
Results: The VEGF-A and PDGF-AB concentrations in vitreous samples were significantly higher in the PDR group (630.72 ± 342.81 pg/mL) compared with those in the control group (153.58 ± 145.85 pg/mL); however, they were not detected in serum samples. The vitreous/serum ratio of the VEGF-A concentration in the PDR group (2.1 ± 1.8) was significantly higher compared with that in the control group (0.31 ± 0.33). The VEGF-A concentrations in vitreous samples were highest in the VH group and lowest in the VH with fibrotic tissue subgroup (mean difference 536.16 pg/mL). The vitreous VEGF-A/PDGF-AB concentration ratios were also significantly different among the PDR subgroups.
Conclusion: High concentrations of VEGF and PDGF in vitreous samples of PDR eyes indicate its local related activity in PDR pathology. There is a possibility of PDGF involvement in the pathogenesis of PDR. The VEGF/PDGF concentration ratios possibly play a significant role in the formation of fibrotic tissue in PDR.
Keywords: vascular endothelial growth factor, platelet-derived growth factor, proliferative diabetic retinopathy, fibrotic tissue
Introduction
The neovascularization process in the retina depends on the growth factor production induced by hypoxic condition.1 Vascular endothelial growth factor (VEGF) is a dominant proangiogenic factor in proliferative diabetic retinopathy (PDR), which triggers migration and proliferation of endothelial cells and blood flow increase, together with microvascular permeability.2–4 It was proven to have significant role on the retina of patients with PDR, especially on retinal pigment epithelial (RPE) cells, glial cells, vitreous fibroblast, ganglion cells, and Muller cells.1,5,6 The amount and duration of VEGF needed to pass through the blood–brain barrier are possibly less than the concentrations needed to induce neovascularization. Hence, monoclonal antibodies against VEGF were introduced and used widely in the form of anti-VEGF agents, such as pegaptanib, ranibizumab, and bevacizumab.5,7,8
Another growth factor found to contribute to the pathology of PDR is platelet-derived growth factor (PDGF).9–11 Both VEGF and PDGF have very similar characteristics.2,3 PDGF could directly trigger cellular proliferation and movement. It is an autocrine growth stimulator in the RPE cells, which participates in the retinal wound repair and epiretinal membrane formation.12 There are four different PDGF chains: PDGF-A, PDGF-B, PDGF-C, and PDGF-D. They later form five isoforms that bind to two different PDGF receptors (PDGFRs), PDGFRα and PDGFRβ, with different affinities.13 The three isoforms of PDGF (AA, AB, and BB) were proven not only to be related to the neovascularization processes in PDR but also to play important role in the formation of fibrotic tissues in the retina of patients with PDR.14 This study aimed to investigate the concentrations of VEGF and PDGF in vitreous and serum samples, analyze the ratio, and compare it among clinical PDR stage.
Patients and Methods
Study Design and Subjects
This is an analytical observational study with a cross-sectional design. We enrolled consecutively attending adults (>18 years old) who had a clinically confirmed diagnosis of PDR, nucleus drop, and intraocular lens (IOL) drop. Inclusion criterion for PDR group was patients with PDR who were candidates for vitreoretinal surgery for long-standing (>3 months) vitreous or preretinal hemorrhage or tractional retinal detachment (TRD). The PDR group was divided into three subgroups: subgroup I, vitreous hemorrhage (VH); subgroup II, VH with fibrotic tissues; subgroup III, TRD. The control group consisted of patients without diabetes who underwent pars plana vitrectomy due to a whole nucleus drop or IOL drop. Exclusion criteria for the PDR group were previous ocular surgery within the last 2 years, existing or a history of ocular inflammation, rubeosis iridis or neovascular glaucoma, and rhegmatogenous retinal detachment.
The study was conducted in August 2018–March 2019 and was approved by the Institutional Review Board of the Medical Faculty of Hasanuddin University of Makassar (ethics committee reference number 1106/H4.8.4.5.31/PP36-KOMETIK/2018). Written informed consent for surgery, blood sampling, and vitreous sampling was obtained from all participants in accordance with the Declaration of Helsinki.
Sample Collection and Measurement of VEGF-A and PDGF-AB Concentrations
Blood samples were collected from all participants on the day of surgery and were left for 2 h at room temperature until clotted. Serum was separated by centrifugation at 4°C for 15 min at 1000 × g, divided into aliquots, and then stored at −80°C until assayed.
At the onset of vitrectomy, 0.5–0.8 mL of undiluted vitreous samples was obtained by aspiration into a 1 mL sterile syringe attached to the vitreous cutter (Alcon Constellation) with the stopcock of the infusion closed. The samples were transferred into sterilized Corning microcentrifuge tubes (1.5 mL), placed immediately on ice, and centrifuged for 20 min at 4°C at 1000 × g. Supernatants without sediment were divided into aliquots and immediately frozen at −80°C until assayed.
The VEGF-A and PDGF-AB concentrations were measured in the vitreous and serum samples by enzyme-linked immunosorbent assay (ELISA) using the Elabscience Human VEGF-A and PDGF-AB ELISA Kit. Each assay was performed in duplicate according to the manufacturer’s instructions, using 100 μL aliquots of vitreous or serum samples, then diluted accordingly to comply with the detection range of the relevant assay, and eventually to contain the same amount of protein. The data presented are per milligram of protein. The optical density was determined at 450 and 570 nm using an absorption spectrophotometer.
Statistical Analyses
All values are expressed as mean ± standard deviation, and all data are presented in the tables and figures. Statistical analyses were conducted using Statistical Package for Social Science version 21.0. The differences between groups were tested using Mann–Whitney U-test and Kruskal–Wallis test. A value of p < 0.05 was considered statistically significant.
Results
Patient Characteristics
A total of 22 eyes were enrolled, 17 eyes of 17 patients with PDR and 5 eyes of 5 patients with nucleus and IOL drop as controls. Based on the PDR subgroups, subgroup I (VH) was diagnosed in six patients; whereas, subgroup II (VH with fibrotic tissues) was diagnosed in five patients and subgroup III (TRD) in six patients. Nineteen eyes had best-corrected visual acuity (BCVA) between 0–3/60 and three eyes had BCVA better than 3/60 (Table 1).
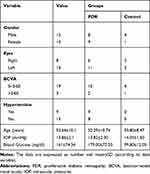 |
Table 1 Patients’ Characteristics |
VEGF-A and PDGF-AB Concentrations in Vitreous and Serum Samples
The mean vitreous VEGF-A concentrations in the PDR group (659.01 ± 332.92 pg/mL) were significantly higher compared with that in the control group (190.94 ± 138.05 pg/mL and 57.01 ± 20.83 pg/mL) (p < 0.05) (Table 2). The vitreous PDGF-AB concentrations in the PDR group (124.52 ± 53.87 pg/mL) were also significantly higher compared with that in the control group (57.01 ± 20.83 pg/mL) (p < 0.05) (Table 2).
 |
Table 2 The VEGF-A and PDGF-AB Concentrations |
The serum concentrations of both VEGF-A and PDGF-AB in the PDR group (551.38 ± 461.84 pg/mL and 98.69 ± 38.56 pg/mL) were not significantly different compared with those in the control group (577.85 ± 336.29 pg/mL and 140.13 ± 80.93 pg/mL) (p = 0.493 and p = 0.319) (Table 2). Interestingly, the concentrations of PDGF-AB in the control group were higher compared with that in the PDR group.
The VEGF-A/PDGF-AB concentration ratios in both vitreous and serum samples were not significantly higher in the diabetic patients compared with those in the control group (Table 3). The vitreous/serum ratio of the VEGF-A concentrations was significantly higher in the PDR group compared with that in the control group (p < 0.05), but not significant in the PDGF-AB concentrations (p = 0.058) (Table 3).
 |
Table 3 The VEGF-A and PDGF-AB Concentration Ratios on Vitreous and Serum |
VEGF-A and PDGF-AB Concentrations in Vitreous and Serum Samples of Each PDR Group
There was no significant difference in the serum concentrations of both VEGF-A (p = 0.586) and PDGF-AB (p = 0.265) among all PDR subgroups in our study. The vitreous VEGF-A concentrations were significantly different among all PDR subgroups (p < 0.05). Post hoc Bonferroni shows that there were significantly lower concentrations of vitreous VEGF-A in subgroup II compared with subgroup I (mean difference 536.16 pg/mL; *p < 0.05) (Figure 1).
There are interesting patterns of the VEGF-A and PDGF-AB concentrations in vitreous samples: in subgroup II, the vitreous VEGF-A concentrations were lowest, whereas the vitreous PDGF-AB concentrations were highest. There was no significant difference in the vitreous/serum ratio of both the VEGF-A and PDGF-AB concentrations among the PDR subgroups (p = 0.754 (VEGF = A); p = 0.482 (PDGF = AB)) (Table 4). The VEGF-A/PDGF-AB concentration ratios in vitreous samples were significantly different among each PDR subgroup (p < 0.01), but not in serum samples (p = 0.591) (Table 4). The vitreous VEGF-A/PDGF-AB concentration ratios were lowest in subgroup II (2.54) (Table 4).
 |
Table 4 The VEGF-A and PDGF-AB Concentration Ratios in Vitreous and Serum in Each PDR Group |
Discussion
In this study, we found that the vitreous VEGF-A concentrations in patients with PDR were significantly increased compared with those in controls, which is supported by the evidence that the vitreous/serum ratio of the VEGF-A concentrations was also significantly higher in the PDR group compared with that in the control group. Similar results have been reported by Praidou et al.10 Baharivand et al15 and Wang et al.16 Chernykh et al17 even reported that vitreous VEGF concentrations of patients with PDR were 17 times higher compared with those of controls. This result is supported by well-known theories about VEGF’s role in PDR pathology.
The concentrations of vitreous PDGF-AB in our study were also increased in PDR eyes compared with controls and even though its vitreous/serum concentration ratios were not significantly high, our findings with respect to ratios were complex. Freyberger et al9 reported that the PDGF-AB concentrations were significantly increased in patients with PDR, with a higher level in rubeosis iridis eyes. Praidou et al10 also reported that PDGF concentrations of all isoforms were increased significantly in patients with PDR. Our result not only suggests that PDGF has a role in the pathology of PDR; it also provides evidence supporting how anti-PDGF could possibly work on the neovascularization processes in PDR. Previously, Phase IIb study by Jaffe et al18 already reported how the combination of anti-VEGF and anti-PDGF was superior to anti-VEGF alone in the treatment of neovascular AMD.
In this study, we found that there was no significant difference in the serum VEGF-A concentrations between PDR and controls. Praidou et al10 also confirmed such findings in their study. This finding was contrary to the theory that diabetic patients have high serum VEGF levels which will lead to diabetes complications, not only retinopathy but also nephropathy, neuropathy, atherosclerosis and other vascular disturbances.19 Baharavind et al15 found in their study that the serum VEGF levels in patients with PDR were increased compared with those in controls. The difference in our result may be due to the fact that our control group was not free from other systemic conditions, which could cause increase in VEGF concentration. The reason was supported by the fact that in the PDR group, the vitreous VEGF-A concentrations were also not significantly different from the serum VEGF-A concentrations, meaning that our serum VEGF-A concentrations were possibly high.
In this study, we also found that the vitreous VEGF-A concentrations of the VH subgroup were significantly high compared with the VH with fibrotic tissue subgroup. The vitreous VEGF-A concentrations were highest in the VH subgroup and lowest in the VH with fibrotic tissue subgroup. Contrarily, the vitreous PDGF-AB concentrations were highest in the VH with fibrotic tissue subgroup and lowest in the VH subgroup. To the best of our knowledge, there are no studies yet that have similar results to ours. A study by Pennock et al20 found that VEGF-A was a competitor of PDGF toward PDGFRs. In 2014, Pennock et al21 even reported that VEGF-A was worked via PDGFRα to maintain the sustainability of cells that express both VEGF and PDGF receptors. A recent study in 2016 by Pennock et al22 reported that VEGF-A not only worked on endothelial cells to maintain and stimulate angiogenesis but also worked on PDGFRα to maintain the sustainability of cells enduring hypoxia. These studies’ results could explain our results, why when hypoxia happen (active PDR, VH group), the PDGF-AB concentrations decrease, possibly because high concentrations of VEGF-A at that stage act as a competitor of PDGF toward PDGFRα, indirectly decreasing the concentrations of PDGF in vitreous samples.
Another question is why the VEGF-A concentrations are low in the VH with fibrotic tissue subgroup. A study by Kuiper et al23 could explain the situation; they found that after the increase in the VEGF-A concentration in the VH stage, the level of connective tissue growth factor (CTGF) also increases and inactivates VEGF-A, so its concentrations decrease gradually. Van Geest RF et al24 even reported that the CTGF/VEGF ratio was a strong predictive factor in the formation of fibrotic tissue in PDR. These results further support that anti-VEGF therapy should not be given when there are already fibrovascular membranes present because it could possibly aggravate angiofibrotic switch that could accelerate TRD.
In this study, we also reported interesting results regarding the vitreous VEGF-A/PDGF-AB ratio in each PDR subgroup. To the best of our knowledge, this is the first study to report the vitreous VEGF-A/PDGF-AB concentration ratios in PDR group. The ratio of the VH with fibrotic tissue subgroup was lower than that of the VH subgroup, and surprisingly, the ratio was not only influenced by lower VEGF-A concentration but also by higher PDGF-AB concentration. Based on these findings, we assume that there is an indication that the vitreous VEGF-A/PDGF-AB ratio attempted to reach an equilibrium state in the VH with fibrotic tissue subgroup and that the ratio was possibly related to the process of fibrotic tissue formation.
The empirical results reported herein should be considered in light of some limitations. It is quite difficult to find truly healthy subjects to be included in the control group in our study; thus, they are subject to biases and confounding factors that may influence our results. However, we attempted to exclude any control eyes with conditions that could influence growth factor concentrations, including fragmented nucleus drop. Besides, for the patients with PDR in our study, advanced examinations, such as fundus fluorescein angiography and optical coherence tomography, were not performed to clearly state the ischemic condition of the retina. We also have a relatively small sample size compared with the other previous related studies, but we used proper statistical analyses to maintain good results.
Conclusion
High concentrations of VEGF and PDGF in vitreous samples of PDR eyes, but not in serum samples, indicate its local related activity in PDR pathology. The vitreous VEGF-A concentrations in the VH with fibrotic tissue subgroup was low in contrast to the PDGF-AB concentrations. The vitreous VEGF-A/PDGF-AB ratio is possibly associated with the formation of fibrotic tissue in PDR. Further investigations and experimental studies using animal models may be required in the future.
Acknowledgments
We would like to express our great appreciation to Dr Burhanuddin Bahar, MS for assistance with statistical analyses, and Dr Rizalinda Sjahril, MSc, PhD for her valuable and constructive suggestions during the planning and development of this research work. The authors also would like to thank Enago for the English language review.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Perrin R, Konopatskaya O, Qiu Y, et al. Diabetic retinopathy is associated with a switch in splicing from anti- to pro-angiogenic isoforms of vascular endothelial growth factor. Diabetologia. 2005;48:2422–2427. doi:10.1007/s00125-005-1951-8
2. Ferrara N. Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev. 2004;25:581–611.
3. Young T, Miller J. Intraocular neovascularization. In: Ryan SJ, Schachat AP, editors. Retina.
4. Frank R. Etiologic mechanisms in diabetic retinopathy. In: Ryan SJ, Schachat AP, editors. Retina.
5. Shams N, Ianchulaev T. Role of vascular endothelial growth factor in ocular angiogenesis. Ophthalmol Clin N Am. 2006;9:335–344.
6. Boulton M, Foreman D, Williams G. VEGF localization in diabetic retinopathy. Br J Ophthalmol. 1998;82:561–568. doi:10.1136/bjo.82.5.561
7. Jorge R, Costa RA, Calucci D, et al. Intravitreal bevacizumab for persistent new vessels in diabetic retinopathy (IBEPE Study). Retina. 2006;26:1006–1013. doi:10.1097/01.iae.0000246884.76018.63
8. Andreoli CM, Miller JW. Anti-vascular endothelial growth factor therapy for ocular neovascular disease. Curr Opin Ophthalmol. 2007;18(6):502–508. doi:10.1097/ICU.0b013e3282f0ca54
9. Freyberger H, Brocker M, Yakut H, et al. Increased levels of platelet-derived growth factor in the vitreous fluid of patients with proliferative diabetic retinopathy. Exp Clin Endocrinol Diabetes. 2000;108(02):106–109. doi:10.1055/s-2000-5803
10. Praidou A, Klangas I, Papakonstantinou E, et al. Vitreous and serum levels of platelet-derived growth factor and their correlation in patients with proliferative diabetic retinopathy. Curr Eye Res. 2009;34(2):152–161. doi:10.1080/02713680802585920
11. Praidou A, Papakonstantinou E, Andraudi S, et al. Vitreous and serum levels of vascular endothelial growth factor and platelet-derived growth factor and their correlation in patients with non-proliferative diabetic retinopathy and clinically significant macula oedema. Acta Ophthalmol. 2011;89(3):248–254. doi:10.1111/j.1755-3768.2009.01661.x
12. Cassidy L, Barry P, Shaw C, et al. Platelet derived growth factor and fibroblast growth factor basic levels in the vitreous of patients with vitreoretinal disorders. Br J Ophthalmol. 1998;82(2):181–185. doi:10.1136/bjo.82.2.181
13. Li X, Eriksson U. Novel PDGF family members: PDGF-C and PDGF-D. Cytokine Growth Factor Rev. 2003;14(2):91–98. doi:10.1016/S1359-6101(02)00090-4
14. Klaassen I, de Vries EW, Vogels Ilse MC, et al. Identification of proteins associated with clinical and pathological features of proliferative diabetic retinopathy in vitreous and fibrovascular membranes. PLoS One. 2017;12(11):e0187304. doi:10.1371/journal.pone.0187304
15. Baharivand N, Zarghami N, Panahi F, et al. Relationship between vitreous and serum vascular endothelial growth factor levels, control of diabetes and microalbuminuria in proliferative diabetic retinopathy. Clin Ophthalmol. 2012;6:185–191. doi:10.2147/OPTH.S27423
16. Wang J, Chen S, Jiang F, et al. Vitreous and plasma VEGF levels as predictive factors in the progression of proliferative diabetic retinopathy after vitrectomy. PLoS One. 2014;9(10):e110531. doi:10.1371/journal.pone.0110531
17. Chernykh VV, Varvarinsky EV, Smirnov EV, Chernykh DV, Trunov AN. Proliferative and inflammatory factors in the vitreous of patients with proliferative diabetic retinopathy. Indian J Ophthalmol. 2015;63(1):33–36. doi:10.4103/0301-4738.151464
18. Jaffe GJ, Ciulla TA, Ciardella AP, et al. Dual antagonism of PDGF and VEGF in neovascular age-related macular degeneration a phase IIb, multicenter, randomized controlled trial. Ophthalmology. 2017;124:224–234. doi:10.1016/j.ophtha.2016.10.010
19. Guo L, Jiang F, Tang Y-T, et al. The association of serum vascular endothelial growth factor and ferritin in diabetic microvascular disease. Diabetes Technol Ther. 2014;16(4):224–234. doi:10.1089/dia.2013.0181
20. Pennock S, Kazlauskas A. Vascular endothelial growth factor A competitively inhibits platelet-derived growth factor (PDGF)-dependent activation of PDGF receptor and subsequent signalling events and cellular responses. Mol Cell Bio. 2012:32:1955–1966.
21. Pennock S, Haddock LJ, Mukai S, Kazlauskas A. Vascular endothelial growth factor acts primarily via platelet-derived growth factor receptor α to promote proliferative vitreoretinopathy. Am J Pathol. 2014;184(11):3052–3068. doi:10.1016/j.ajpath.2014.07.026
22. Pennock S, Kim LA, Kazlauskas A. Vascular endothelial cell growth factor A acts via platelet-derived growth factor receptor α to promote viability of cells enduring hypoxia. Mol Cell Biol. 2016;36(18):2314–2327. doi:10.1128/MCB.01019-15
23. Kuiper EJ, Van Nieuwenhoven FA, de Smet MD, et al. The angio-fibrotic switch of VEGF and ctgf in proliferative diabetic retinopathy. PLoS One. 2008;3(7):e2675. doi:10.1371/journal.pone.0002675
24. Van Geest RJ, Lesnik-Oberstein SY, Tan HS, et al. A shift in the balance of vascular endothelial growth factor and connective tissue growth factor by bevacizumab causes the angiofibrotic switch in proliferative diabetic retinopathy. Br J Ophthalmol. 2012;96(4):587–590. doi:10.1136/bjophthalmol-2011-301005
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

