Back to Journals » Clinical Ophthalmology » Volume 9
Visual outcome and efficacy of conjunctival autograft, harvested from the body of pterygium in pterygium excision
Authors Bhandari V, Roa CL, Ganesh S, Brar S
Received 2 August 2015
Accepted for publication 26 September 2015
Published 3 December 2015 Volume 2015:9 Pages 2285—2290
DOI https://doi.org/10.2147/OPTH.S93580
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Vipul Bhandari, Chandan Latha Rao, Sri Ganesh, Sheetal Brar
Department of Cornea, Nethradhama Superspeciality Eye Hospital, Bangalore, India
Purpose: To evaluate the efficacy of conjunctival autograft after the pterygium excision with fibrin adhesive using conjunctiva over the pterygium.
Patients and methods: This prospective study included 25 eyes of 25 patients with a mean age of 40±10 years, who underwent the pterygium excision with conjunctival autograft derived from the body of the pterygium and attached using fibrin glue. The mean follow-up period was 6 months. On all postoperative visits, changes in uncorrected visual acuity, corrected distance visual acuity, astigmatism, complications, and the evidence of recurrence were recorded.
Results: At the end of mean follow-up, uncorrected visual acuity and corrected distance visual acuity improved by one or two lines in all eyes treated. Mean astigmatism reduced significantly from a preoperative value from 2.308D to 1.248D postoperatively (P<0.026). Minor postoperative complications such as congestion, chemosis, and subconjunctival hemorrhage were seen, which resolved with time. No major sight-threatening or graft-related complications were detected. There was no evidence of recurrence during a follow-up period of 6 months.
Conclusion: Self-conjunctival autograft following the pterygium excision appears to be a feasible, safe, and effective alternative method for management of pterygium. It also preserves the superior conjunctiva for future surgeries. However, longer follow-up is required to study the long-term outcomes, especially the incidence of recurrence.
Keywords: Pterygium, self-conjunctival graft, body, autograft
Introduction
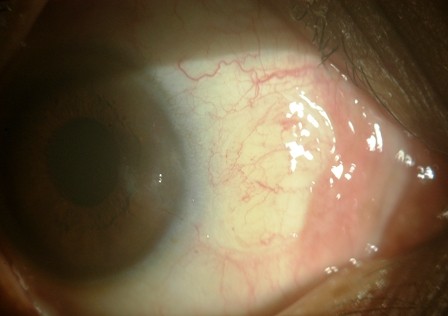
Pterygium, a word derived from “pterygion” (ancient Greek for a wing) is a wing-shaped, fibro-vascular overgrowth arising from subconjunctiva tissue extending across the limbus onto the cornea. It is a degenerative condition of the subconjunctival tissue, which proliferates as a vascularized granulation tissue to invade the cornea, destroying superficial layers of stroma and Bowman’s membrane.1
Several theories have been proposed to explain the pathogenesis of the pterygium such as inflammatory theory (Kamel), neoplastic theory (McReynolds and Schreiter), degenerative theory (Fuchs and Schoinger), and immunologic theory. The most accepted theory in pathogenesis is Kamel’s inflammatory theory. Various methods of surgical management of pterygia are excision, avulsion, and excision with primary closure, transplantation of the head of the pterygium, conjunctival autograft, limbal conjunctival autograft, and amniotic membrane graft. The pterygium surgery with conjunctival autograft remains the gold standard with less incidence of recurrence after its initial description in 1985.1 Recurrence rates consistently reported are in the range of 5%–10%.2 The recent technique is to use tissue fibrin glue for anchoring the graft to the bed.
The favored site to harvest the autograft is superotemporal2 because this location in an oblique quadrant will avoid inadvertent injury to extraocular muscle, while its position under the upper lid will aid with patient comfort and healing of the donor site.
However, in this study, we report outcomes with a novel technique of the pterygium management using conjunctival autograft harvested from the body of the pterygium. We also discuss our experience regarding the patient acceptability, visual results, and potential complications in a 6-month follow-up period with this new technique.
Patients and methods
The study was approved by the institutional review board of Nethradhama Superspeciality Eye Hospital, Bangalore and adhered to the tenets of the Declaration of Helsinki. Informed consent was obtained from all the patients participating in the study.
In the above review of literature, the sample size for similar studies was as low as 20 and as high as 88 eyes. In view of 6-month follow-up required in this study and an average pterygium surgeries of eleven per month in the hospital set up the sample size was selected as 25 eyes.
A total of 25 eyes of 25 patients with the primary pterygium diagnosed on slit-lamp were selected for the study. However, recurrent pterygia, ocular surface disorders, severe dry eye, and the patients already on antimetabolites were excluded from the study.
A detailed preoperative clinical examination was conducted on all patients which included recording of uncorrected visual acuity (UCVA) and corrected distance visual acuity (CDVA) using Snellen’s test charts and LogMAR, slit-lamp examination, keratometry readings using Orbscan II (Technolas, Munich, Germany), dry eye assessment using Schirmer’s 1 and 2 tests, dilated fundus examination with pupillary dilatation using 90D and indirect ophthalmoscopy was done. B-scan ultrasonography was performed whenever the fundus details were not clearly visible.
The pterygium was classified into three types depending on the extent of corneal involvement:3
Type 1: extends less than 2 mm on the cornea
Type 2: involves up to 4 mm on the cornea
Type 3: encroaches onto more than 4 mm of the cornea and involves the visual axis.
Surgical technique
All the patients were operated by a single, experienced corneal surgeon (VB) under topical anesthesia using an operating microscope. Proparacaine 0.5% (Sunways India Pvt Ltd., Mumbai, India), eye drops were instilled in the eye twice at an interval of 10 minutes before surgery and intraoperatively as required.
After instillation of topical anesthesia, the conjunctival tissue over the body of the pterygium was marked using a marker. It was then isolated from the underlying degenerative tissue using blunt dissection. Using an atraumatic conjunctival forceps and Vannas scissors, the conjunctiva was carefully dissected and isolated from subconjunctival tissue (Figure 1). In addition, care was taken to prevent buttonholing during dissection. The dissected graft was flipped over the cornea.
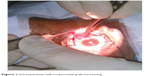  | Figure 1 Intraoperative self-conjunctival graft harvesting. |
The pterygium was excised in a circular fashion similar to performing a capsulorhexis. Blunt and sharp dissection was performed to separate the pterygium from underlying sclera and surrounding conjunctiva. The body of the pterygium, along with underlying tenons was excised using Westcott scissors. The wound bed was smoothened by scraping and bleeding vessels cauterized using wet field cautery. The harvested graft was then placed over the bare scleral bed, while maintaining the orientation that is epithelium side up and limbal edge toward limbus. In addition, care was taken to prevent the graft from rolling over.
Fibrin glue (Tisseel fibrin sealant Baxter AG, Vienna, Austria) was used to attach the autograft in place. The mixing of the component was done as per the manufacturer’s directions. Two to three drops of the thrombin solution were applied on the scleral bed followed by the fibrinogen component. The conjunctival autograft was then immediately flipped over the area of conjunctival defect and smoothened out with a non-toothed forceps, while maintaining proper alignment of the edges (Figure 2). It was ensured that the bare sclera was completely covered. In certain situations where the graft was insufficient, an amniotic membrane was used to cover the remaining bare sclera using fibrin adhesive. The graft was dried and allowed to adhere to the bed for 3 minutes, following which the lid speculum was removed.
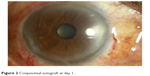  | Figure 2 Conjunctival autograft at day 1. |
Postoperatively, patients were prescribed tapering doses of Zylopred (Gatifloxacin 0.3% and Loteprednol 0.5%, Allergan, Inc., Irvine, CA, USA) and lubricants for a period of 6 weeks.
All patients were followed-up for a minimum period of 6 months. On all postoperative visits, slit-lamp photography (Figure 3; UCVA, best-corrected visual acuity [BCVA]), astigmatism, and complications were recorded and subjective questionnaire regarding patient comfort was taken from all patients.
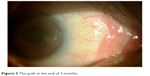  | Figure 3 The graft at the end of 3 months. |
Statistical analysis
The data were collected and tabulated using Microsoft Excel and Word. Descriptive and analysis of variance statistical analysis were carried out in this study. The results of continuous measurements were presented on mean ± standard deviation (SD) and results of categorical measurements were presented in number (%). Significance was assessed at 5% level of significance. Student’s t-test (two-tailed, dependent) was used to find the significance of study parameters on a continuous scale within each group.
Results
A total of 25 eyes of 25 patients underwent the pterygium excision with this new technique. Mean age was 40±10 years. The number of eyes classified preoperatively as grade 1, 2, or 3 were 10, 11, and 4 eyes.
There were no intraoperative complications encountered. The procedure was well tolerated and the patients did not report any significant subjective complaints of excessive pain, irritation, or discomfort both intra as well as postoperatively.
Changes in keratometry
A significant flattening in mean simulated keratometry (sim K) was observed over time. A significant reduction in mean sim K was seen from a preoperative value of 2.308±2.25 SD to 1.248±0.99 SD at 1 month postoperatively, which was statistically significant (P-value <0.026). Keratometry remained stable and no significant changes in sim K observed between 1 month and subsequent follow-up visits (Table 1).
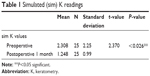  | Table 1 Simulated (sim) K readings |
Changes in visual acuity
The UCVA improved in all the three types of pterygium. In type 2, the preoperative mean was 0.350±0.20 SD, which significantly improved to 0.20±0.14 SD at 1-month postoperation (P-value =0.005).
Similarly, in type 3 preoperative mean improved from 0.53±0.18 SD to 0.275±0.05 SD on 1 month postoperatively. The results were also statistically significant (P-value =0.039). Further on, in both type 2 and 3 pterygium groups, UCVA remained stable at the end of the 3rd month and until the last follow-up (P-value <0.05) (Table 2). Out of the total 25 patients, one case had cataract due to which there was not much improvement in the UCVA.
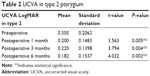  | Table 2 UCVA in type 2 pterygium |
Preoperative BCVA in type 1 remained same but in types 2 and 3, there was a significant improvement. In type 2, preoperative mean LogMAR of 0.109±0.15 SD improved to 0.018±0.075 SD at postoperative 1 month (Tables 3 and 4), which was statistically significant (P=0.03). However, in type 3, though there was improvement in BCVA postoperative, it was not statistically significant attributing to significant corneal scarring. Two patients required an amniotic membrane grafting (AMG) to cover the bare sclera as the conjunctiva harvested was smaller but did not alter the visual outcome.
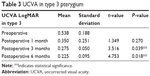  | Table 3 UCVA in type 3 pterygium |
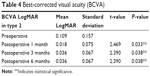  | Table 4 Best-corrected visual acuity (BCVA) |
Postoperative complications
No major complications were observed. Minor complications like congestion of 21 eyes (84%) on day 1, chemosis in five eyes (20%) on day 1 and subconjunctival hemorrhage in six eyes (24%). Discomfort was experienced in 13 eyes (52%), foreign body sensation in eight eyes (32%) and tearing in four eyes (16%) on postoperative day 1. All these subjective complaints that improved in due course of time and all patients were symptomatically comfortable by 2 weeks (Table 5) shows complications after surgery (Table 6).
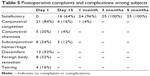  | Table 5 Postoperative complaints and complications among subjects |
  | Table 6 Graft-related complications postoperatively |
There was no incidence of graft dislocation, infection, necrosis, or other graft-related complications during the follow-up period of 6 months (Table 5). Slit-lamp examinations showed clear cornea with well-apposed graft during the 6 months of follow-up in all the cases (Figure 2 and Table 5). None of the eyes showed any evidence of recurrence of the pterygium at the end of the follow-up period.
Discussion
Till date, the pterygium remains to be an enigma for the ophthalmic world. Although extensive studies on the pterygium have unraveled the etiopathogenesis of this benign looking entity, the attempts to get rid of it have proved futile. Furthermore, the phenomenon of recurrence with its devastating effect on vision has seriously questioned the utility of simple excision as a mode of management of pterygium. Various researchers have reported a recurrence rate anywhere ranging from 30% to 80%.4–6 The existence of multiple adjuvant therapies for the pterygium in the present era, substantiate the fact that there is no single satisfactory and universally acceptable treatment modality for treating it.
This study was contemplated with the objective to study the results of the pterygium excision with self-conjunctival autograft of the pterygium using fibrin glue in the surgical management of pterygium. Since the pathology in the pterygium is subconjunctival degeneration, while the overlying conjunctiva being healthy, it may be unnecessary to sacrifice the healthy conjunctiva which itself may be utilized to cover the bare sclera after the excision of pterygium, instead of harvesting conjunctival tissue from another site. It has been found the conjunctival deficiency or scarring in the superior or superotemporal part due to the earlier pterygium surgery poses a surgical challenge in subsequent glaucoma or other ocular procedures. Hence, this technique would also preserve the conjunctiva for surgeries such as glaucoma procedures, if the need arises in the future.
In this study, we observed a statistically significant reduction in overall mean astigmatism postoperatively by Orbscan, which indicates that as the type of the pterygium increases, the amount of astigmatism also increases in the same proportion. A similar observation was found by earlier researchers.7,8 They also found that the size of the pterygium was directly proportional to the amount of the astigmatism induced by it. Successful pterygium surgery reduced the pterygium induced refractive astigmatism and improved the visual acuity. They also suggested early surgical intervention reduces the mean induced corneal astigmatism.8
We observed significant improvement in the UCVA after successful pterygium surgery that was significant in type 2 and type 3 pterygium. Though in type 1, there was an improvement in the UCVA, it was not statistically significant. Type 1 pterygium patients showed improvement in the UCVA postoperative 1 month and were constant till 6 months. These observations were similar to the study carried out by Maheshwari.7 Lindsay and Sullivan also found a similar significant correlation between successful pterygium surgery and improvement in visual acuity.9
Allan et al compared preoperative unaided visual acuity with 3-month postoperative unaided visual acuity on Snellen’s chart in 93 eyes. They found unaided visual acuity was either unchanged or improved in 86 out of 93 cases, while seven eyes showed the decline in visual acuity on Snellen’s chart. The decline in visual acuity was not related to surgery but was either due to astigmatism, cataract, or retinal pathology.10
This observation is also similar to our study. We compared preoperative unaided visual acuity with postoperative visual acuity and found that 16 cases (64%) reported improved visual acuity, eight cases (32%) showed unaltered visual acuity while one case (4%) showed deterioration in visual acuity. This decline was unrelated to the surgery, but it was because of progression of senile cataract.
We also studied the complication profile with this new technique, which was found to be favorable with only minor postoperative issues such as congestion, graft edema, subconjunctival hemorrhage etc. However, there were no graft-related or serious vision related complications, no graft inversion or dislocation was noticed. Also, there was no incidence of recurrence found at the end of mean follow-up of 6 months.
However, with the conventional technique of the pterygium excision with conjunctival autografting, some complications have been reported in literature. Allan et al in their study, encountered three cases of wound dehiscence, one case of conjunctival cyst and one case of Tenon’s granuloma.10 Similarly, Ma et al in a series of 80 eyes treated with the pterygium excision with conjunctival autografting had two cases of pyogenic granuloma and four cases of conjunctival inclusion cysts. All these complications were amenable to correction with minor surgical revision and did not have any sequelae.11
Hence, our technique appears to be equally safer compared to the conventional technique in terms of postoperative complications.
Conclusion
The conjunctiva harvested from the body of the pterygium after dissecting it from underlying subconjunctival tissue may offer a simple and effective way to treat pterygium. This technique also has the added advantage as the superior conjunctiva is untouched and can be used for future surgeries if required. The recurrence rate is not expected to be any different from the conventional technique, since the only difference exists in the method and site of harvesting the conjunctiva while the rest of the procedure being essentially the same. However, longer follow-up is required to study the long-term safety, efficacy, complications, and the incidence of recurrence with this technique.
Disclosure
The authors report no conflicts of interest in this work.
References
Lin A, Stern G. Correlation between pterygium size and induced corneal astigmatism. Cornea. 1998;17:28–30. | ||
Kenyon KR, Wagoner MD, Hettinger ME. Conjunctival autograft transplantation for advanced and recurrent pterygium. Ophthalmology. 1985;92:1461–1470. | ||
Frutch-Perry J, Charalambos SS, Isar M. Intraoperative application of topical mitomycin C for pterygium surgery. Ophthalmology. 1996;103:674–677. | ||
Lam DS, Wong AK, Fan DS, et al. Intraoperative mitomycin C to prevent recurrence of pterygium after excision. A 30-month follow up study. Ophthalmology. 1998;105:901–904. | ||
Gupta VP, Saxena T. Comparison of single-drop mitomycin C regime with other mitomycin C regimes in pterygium surgery. Indian J Ophthalmol. 2003;51:59–65. | ||
Kanski JJ, Bowling B. Clinical Ophthalmology. 7th ed. Sydney: Elsevier; 2011:220. | ||
Maheshwari S. Effect of pterygium excision on pterygium induced astigmatism. Indian J Ophthalmol. 2003;51:187–188. | ||
Avisar R, Loya N, Yassur Y, Weinberger D. Pterygium induced corneal astigmatism. Isr Med Assoc J. 2000;2(1):14–15. | ||
Lindsay RG, Sullivan L. Pterygium-induced corneal astigmatism. Clin Exp Optom. 2001;84:200–203. | ||
Allan BD, Short P, Crawford GJ, Barrett GD, Constable IJ. Pterygium excision with conjunctival autografting: an effective and safe technique. Br J Ophthalmol. 1993;77(11):698–701. | ||
Ma DH, See LC, Liau SB, Tsai RJ. Amniotic membrane graft for primary pterygium: comparison with conjunctival autograft and topical mitomycin C treatment. Br J Ophthalmol. 2000;(84):973–978. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
