Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Variants in the Upstream Region of the Insulin Receptor Substrate-1 Gene Is Associated with Major Depressive Disorder in the Han Chinese Population
Authors Wang F, Yu S , Zhou R , Mao R , Zhao G , Guo X, Xu Q, Chen J , Zhang C, Fang Y
Received 12 July 2019
Accepted for publication 21 January 2020
Published 20 February 2020 Volume 2020:16 Pages 501—507
DOI https://doi.org/10.2147/NDT.S222906
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Yuping Ning
Fan Wang,1,2 Shunying Yu,3 Rubai Zhou,1 Ruizhi Mao,1 Guoqing Zhao,1,4 Xiaoyun Guo,1 Qingqing Xu,3 Jun Chen,1 Chen Zhang,1 Yiru Fang1,5,6
1Division of Mood Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai 200030, People’s Republic of China; 2Department of Psychiatry and Cellular & Molecular Medicine, University of Ottawa Institute of Mental Health Research at the Royal, Ottawa, ON, Canada; 3Department of Genetics, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai 200030, People’s Republic of China; 4Department of Psychology, Provincial Hospital Affiliated to Shandong University, Jinan 250021, People’s Republic of China; 5Shanghai Key Laboratory of Psychotic Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai 200030, People’s Republic of China; 6Brain Science and Technology Research Center, Shanghai Jiao Tong University, Shanghai 200240, People’s Republic of China
Correspondence: Yiru Fang
Clinical Research Center & Division of Mood Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, CAS Center for Excellence in Brain Science and Intelligence Technology Shanghai Key Laboratory of Psychotic Disorders, 600 South Wanping Road, Shanghai 200030, People’s Republic of China
Tel +86-21-3428 9888 Ext 3529
Fax +86-21-64387986
Email [email protected]
Chen Zhang
Division of Mood Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, 600 South Wanping Road, Shanghai 200030, People’s Republic of China
Email [email protected]
Introduction: Major depressive disorder (MDD) is one of the most prevalent and disabling mental disorders, although its underlying genetic mechanism remains unknown. Insulin receptor substrate-1 (IRS-1) is one of the critical downstream molecules in the insulin resistance signaling pathway, linking depression and diabetes. Therefore, we hypothesized that IRS-1 would be a susceptible gene for MDD, and we aimed to examine the genetic association between IRS-1 and MDD.
Methods: This case-control study included 583 patients with MDD and 564 controls, and the genotypic and allelic distributions of the IRS-1 gene’s four single nucleotide polymorphisms (SNPs) were detected by TaqMan SNP genotyping technology. Of the 583 patients, 191 underwent a further detailed interview about symptom severity and family history of mental illness. The chi-square or t test was used to analyze the data, and analyses were performed using SPSS19.0 software.
Results: A haplotype in the 5ʹ-upstream region of IRS-1 consisting of rs13411764 and rs3820926 was a risk factor of MDD. Patients with a family history of mental illness were more likely to have a GG genotype in rs13411764 and a G-T haplotype containing rs13411714-rs3820926.
Discussion: The findings imply that the haplotype consisting of rs13411764 and rs3820926 in the upstream of IRS-1 is a risk factor for MDD. This haplotype could affect IRS-1 expression levels, and it is mostly inherited from parents. Thus, the presence of variants in the upstream region of IRS-1 is a risk factor of MDD, and this study could serve as a convincing reference for further studies.
Keywords: major depressive disorder, IRS-1, single nucleotide polymorphisms, haplotype, family history
Introduction
Major depressive disorder (MDD) is one of the most prevalent and disabling neuropsychiatric diseases, affecting more than 15% of the population worldwide. Epidemiological data indicated that genetic factors play essential roles in the pathophysiology of MDD. However, its genetic mechanism is still unclear. Recent data showed that metabolic disturbances might be involved in the pathogenesis of depression, and simultaneously, there seems to be a link between depression and some metabolic illness, especially diabetes.20 Accumulating evidence has demonstrated that patients with depression have a higher risk of developing type II diabetes. A meta-analysis reported that the odds ratio of depression in the diabetic group was twice that of the nondiabetic group (odds ratio [OR] = 2.0, 95% confidence interval [CI] 1.8–2.2).2 In an animal study, experimental data showed that diabetic mice and rats presented with more pronounced depressive-like behavior when subjected to the forced swimming test.32 In an animal study, experimental data showed that diabetic mice and rats presented with more pronounced depressive-like behavior when subjected to the forced swimming test.9 All this evidence leads to exploration of the possible underlying pathogenesis shared by depression and diabetes.
Insulin resistance, which is the core feature of diabetes, is drawing significant attention for its correlation with depression. Reports showed a positive association between insulin resistance and depressive symptoms.25,31 Insulin resistance was also regarded as a risk factor for onset of post-stroke depression.26 Moreover, use of insulin-sensitizing agents, such as the PPAR-γ agonist pioglitazone could improve mood in patients with depression.18 Thus, insulin resistance has been widely accepted as an essential mechanism linking depression and diabetes.
Insulin receptor substrate (IRS) docking proteins are the key downstream signaling molecules in the insulin receptor (IR) signaling pathways. The IRS is spread widely in the brain and present in neurons and glial cells,15,16 and it possibly affects central nervous system development and function. Recent evidence indicates that insulin can act as a growth factor in neuronal growth and differentiation, synapse density, synaptic transmission and plasticity,and cognitive function through the regulation of neuronal signaling.11,12 Insulin receptor substrate-1 (IRS-1) encodes a protein that is bound to the intracellular domain of insulin receptors and phosphorylated by the insulin receptor tyrosine kinase.30 Tyrosine phosphorylation of IRS-1 can regulate theactivation or inhibition of proteins involved in the IR signaling pathway.33 A line of evidence from genetic and animal studies5,7,19 showed that IRS-1 is closely associated with insulin resistance. IRS-1 was also found to be highly expressed in the rodent brain;33 thus, disturbances in IRS-1 phosphorylation may contribute to the behavioral changes observed in depression.3
In regard to the critical roles of IRS-1 in the mechanism of insulin function, which is highly related to depression and neurological functions, we hypothesized that IRS-1 would be a susceptible gene for MDD. In this study, we aimed to examine the genetic association between IRS-1 and MDD.
Materials and Methods
Ethical Statements
The Ethics Committee of the Shanghai Mental Health Centerapproved this study protocol (2012-42). The experimental methods were carried out after the protocol was approved. Written informed consent conforming to the tenets of the Declaration of Helsinki was obtained from each participant before any study-related procedures were performed.
Participants
Five hundred eighty-three patients who met the Diagnostic and Statistical Manual of Mental Disorders, 4th edition Text Revision criteria for MDD were recruited from the outpatient department of Shanghai Mental Health Center and enrolled in this study. The diagnosis of MDD was made based on clinical interviews and medical records. At least two experienced psychiatrists independently assessed each patient. All diagnoses were further confirmed by an independent research psychiatrist who has received consistent training and used the Minnesota International Neuropsychiatric Interview. The selection criteria for MDD patients were as follows: (1) outpatients of Shanghai Mental Health Center from April 2014 to March 2016 met the DSM-TR diagnostic criteria for MDD; (2) 17-item Hamilton Rating Scale for Depression (HAMD) score≥17 points, and Young Manic Rating Scale (YMRS) total score≤10; (3) the routine test, glucose, lipids, liver function and kidney function in blood were normal; (4) the informed consent of patients or their families has been obtained. Exclusion criterias were (1) having ever been diagnosed with other axis I psychiatric disorders, such as schizophrenia, schizoaffective disorder, or bipolar disorder; (2) current diagnosis of nicotine dependence and use, alcohol dependence and other substance abuse; (3) depressive syndrome due to organic factors including severe medical illness and organic brain disease; (4) pregnancy or breast-feeding.Of 583 patients, 191underwent a detailed interview in which symptom severity was assessed using the Hamilton Depression Scale-17 for Depression (HAMD-17). Severe symptoms were defined as HAMD-17 sores ≥24. Mental disorders relating to patients’ family history were also collected.
The control group consisted of 564 healthy participants who were recruited via advertisement in the outpatient department and from a group of blood donors in the Shanghai Health Physical Examination Center. They were screened to ensure that they were free from any psychiatric disorders, alcohol dependence, and drug abuse. All participants were of Han Chinese origin andfrom the same region; thereby, they were considered as ethnically homogeneous.
Selection of SNPs and Genotyping
In order to select the SNPs, we previously retrieved genotypic data of the Han Chinese population from the HapMap database (http://hapmap.ncbi.nlm.nih.gov/). The gene region and potential regulatory sequences were considered during the selection of tagSNPs. Under the following criteria: minor allele frequency (MAF) ≥ 0.2 and r2 ≥ 0.8. We selected three tagSNPs (rs10203363, rs13411714, and rs3820926) and one functional variant (rs1522813) in the upstream of the gene. Genotyping of each gene was performed by using an optical 384-well plate with the TaqMan probe technique and the ABI PRISM 7900 HT Fast Real-Time instrument (Applied Biosystems, Foster City, CA, USA).
Brain eQTL Analysis
As MDD is considered to originate from aberrant brain functions, we used the expression quantitative trait loci (eQTL) database (http://www.braineac.org/), which is a large exon-specific eQTL dataset covering 10 human brainregions,27 for eQTL analysis of risk SNP(s).
Statistical Analysis
Demographic data were analyzed using the chi-square or t test as appropriate using SPSS19.0 (SPSSInc.Chicago, IL,USA). Correlation and risk were determined using the chi-square test.The Hardy-Weinberg equilibrium analysis and allele and genotype comparisons between the case and control groups for each SNP were performed using SHEsis online software.37 For individual SNP analysis, multiple testing corrections were made with Bonferroni tests. The corrected P-values were set at an uncorrected P-value multiplied by independent significance tests. SHEsis online software was also used to perform pairwise linkage disequilibrium (LD) estimation and haplotype analysis.21 The pairwise LD analysis was used to detect the intermarker relationship in the case-control samples. The significance threshold for the P-values was set at 0.05, and all P-values were two-tailed. The statistical power, which was calculated by Quanto (Jim Gauderman, et al, Department of Preventive Medicine, University of Southern California, Los Angeles, CA, US), was sufficient for detecting an association between the SNPs and MDD in this sample size. The OR and 95% CI was calculated by using a risk estimate.
Results
This study included 583 patients with MDD and 564 healthy controls. Demographic characteristics of our participants are presented in Table S1. As shown in Table S1, female participants were about twice as likely as male participants to develop depression (n=195/388), and the different prevalence of depression by sex was well documented.10,28 The genotype and allele frequencies of the selected SNPs between the case and control groups are displayed in Table S2. Genotypic distributions were in the Hardy-Weinberg equilibrium for all SNPs in the controls. The initial analysis showed statistically significant differences for 2 SNPs (rs1522813 and 13411764) (P=0.03) in the allelic distribution; neither of these differences was significant after multiple testing corrections.
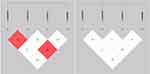
We found two pairwise LDs among the 4 studied SNPs (Figure 1). High LD was observed between rs10203363 and rs13411714 (D’0.83, r2 0.33), and between rs13411714 and rs3820926 (D’0.84, r2 0.25), which consisted of two haplotypes, rs10203363-rs13411714 and rs13411714-rs3820926. Table 1 presents all P-values corresponding to the two haplotypes with a haplotype frequency of less than 5% being dropped. The G-T haplotype consisted of rs13411714, and rs3820926 was statistically significantly associated with MDD (P=0.01, OR[95% CI]=1.33[1.07–1.65]) (Table 1). Overall, 22.7% of patients with MDD carried this haplotype, and this proportion was higher than that of the control (18.1%). The OR of 1.33 suggested that Han Chinese individuals with the rs13411714-rs3820926 G-T haplotype are more likely to have MDD.
 |
Table 1 Results of the Pairwise Haplotype Test Between MDD and Control Groups |
Among 191 patients with MDD who underwent a detailed interview, those with a family history of mental illness were 5 times more likely to carry the GG genotype in rs13411764 (P=0.02, OR[95%]=5.03 [1.75–14.50]) (Table 2). Because of the different prevalence of depression by sex, we further divided the MDD group into two subgroups by gender and observe the correlation between gene and family history. We found that female patients were 6 times more likely to carry the GG genotype in rs13411764 (P=0.04, OR[95%]=6.44 [1.78–23.26]) (Table S5). Whereas no correlation was found in male patients. In Additionally, patients with a family history of mental illness were more likely to carry a G-T haplotype containing rs13411714-rs3820926(P=0.003, OR[95%]=3.14[1.45–6.80]) (Table 3). We further observe the correlation between the G-T haplotype and family history, and found the correlation only existed in female patients (P=0.02, OR[95%]=2.81 [1.12–7.10]) (Table S6). This finding suggested that either rs13411764 alone or the rs13411714-rs3820926 haplotype was highly associated with a family history of mental illness.
 |
Table 2 Association Between Selected SNPs and a Family History of Mental Illness |
 |
Table 3 Association Between the rs13411764-rs3820926 G-T Haplotype and a Family History of Mental Illness |
No correlation was observed between symptom severity and the 4 SNP genotypes (Table S3), and no correlation was observed between symptom severity and the rs13411714-rs3820926 haplotype (Table S4). This finding suggested that the 4 SNPs and the haplotype were not associated with symptom severity.
Subsequently, we performed an eQTL analysis to explore the effects of rs13411714 and rs3820926 on IRS-1 expression in the brain. As shown in Figure S1, the SNP rs13411764 was significantly associated with IRS-1 expression in the intralobular white matter (P=0.042). Albeit this significance was not present after Bonferroni correction, individuals with the GG genotype had a lower level of IRS-1 expression. No association was found between rs3820926 and IRS-1 expressed in the 10 brain regions (Figure S2).
Discussion
IRS-1 is the first direct substrate for the insulin receptor kinase, which occupies a key position in the insulin signaling pathway. After insulin binds to the insulin receptor, the β-subunit of the receptor undergoes autophosphorylation, and in turn, it phosphorylates other endogenous protein substrates in the insulin cascade.30,34 Thus, abnormality of the structural or expression level of IRS-1 may affect the insulin receptor function. A previous study showed that a genetic variant of glycine to arginine change in the IRS-1 codon is a risk factor for postpartum depression.17 A decreasedIRS-1 level accompanied by an increase in phosphorylation of IRS-1 was observed in the brains of patients with Alzheimer disease.23 Nevertheless, no studies have been conducted yet to explore the variants in the upstream of IRS-1 in patients with MDD,as this factor could affect its expression level. However, no previous studies focused on the impact of MDD on IRS-1. Our study, a pioneer research study on exploring the association between IRS-1 and MDD, found that a haplotype in the 5ʹ-upstream region of IRS-1could be a risk factor for MDD, it affects IRS-1 expression levels, and it is mostly inherited from parents.
Our study showed that a G-T haplotype consisting of rs13411764 and rs3820926 is a risk factor for MDD, but there was no association between all 4 SNPs and MDD. For the SNP rs13411764, no statistical significance existed after multiple corrections, but the data showed a mild correlation between the allele G and MDD(p=0.03). Additionally, our eQTL analysis showed a mild correlation between the SNP rs13411764 and IRS-1 expression level. This finding may be due to the fact that a single SNP is not sufficient to affect the expression of a gene, but two SNPs, rs13411764 and rs3820926, acting simultaneously are sufficient to affect gene expression. SNPs rs13411764 and rs3820926 are located in the 272kb and 263kb upstreams from the transcription start site of IRS-1, respectively. This haplotype could increase the risk of MDD. Genetic mechanisms suggest that the promoter region of SNPs may change transcription factor binding sites and influence transcriptional levels of gene expression.6 Thus, altered IRS-1 expression may be involved in the pathology of mental disorders, such as MDD. Therefore, we infer that the G-T haplotype may disturb the expression of IRS-1 and impair the function of IR, which may induce the development of MDD. This idea requires further investigation and validation.
The correlation study also showed that the rs13411714-rs3820926 haplotype was a risk factor of having a family history of mental llness (OR[95%]=3.14[1.45~6.80]), which means that patients with a G-T rs13411714-rs3820926 haplotype are more likely to have a family history of mental illness. This evidence further explains that these variants had increased vulnerability to MDD, which was more likely to be obtained through ancestral inheritance, and IRS-1 may be a susceptible gene for MDD. Further lineage study is needed to validate this assumption. What’s more, recent studies showed many more genes associated with MDD, which could be our future work.35 Genome-wide association analyses identifed 44 risk variants were associatedwith MDD.36 Novel strategis were applied, and rare variants was identified to be associated with major depression.38 Whole-genome sequencing also found rare variants and loci were associated with MDD.1,29
Our data showed that all 4 SNPs and the rs13411714-rs3820926 haplotype were not associated with symptom severity. This result suggests that the expression level of IRS-1, which could be affected by the mutations in our study, may not affect the symptom severity of the depressive episode, but it might be related to the treatment efficacy and duration of the present depressive episode. Glombik et al study showed that the favorable effect of antidepressants on insulin receptor phosphorylation was mainly related to IRS-1 phosphorylation.13 A study also suggested a longer remission time of depression in diabetic patients,22 which indicates that insulin resistance may affect the treatment efficacy and prolong the course of the disease. Cognitive change was found in insulin-resistant rodents.22,24 IRS-1 is also believed to be capable of promoting neuroprotective effects in the brain.14 Therefore, IRS-1 is more likely to be related to the treatment efficacy and duration of the present depressive episode.
Our results should be interpreted with caution because MDD is a heterogeneous disorder based on multiple factors including socioeconomic status, family support, biological and genetic aspects.4,8 Our study had several limitations that should be addressed. First, our eQTL analysis was performed in the BRAINEAC database derived from Caucasian brain samples. We did not evaluate the IRS-1 expression level or collect any metabolic related datas in this study. It is unclear whether SNP rs13411764 affects IRS-1 expression in the brains of Han Chinese individuals. So further research focusing on metabolic ralated datas including insulin level, glucose level and bodyweight need to be done. Secondly, the sample size was small. Hence, further studies with a larger sample size are still required to confirm our findings.
Conclusions
Our study suggests that parental hereditary upstream variants of IRS-1 could lead to altered IRS-1 expression, and the presence of such variants is a risk factor for developing MDD. This preliminary finding may provide a reference for further studies on validating IRS-1 as a susceptible gene for MDD.
Abbreviations
MDD, major depressive disorder; IRS-1, insulin receptor substrate-1; SNP, single nucleotide polymorphism; IR, insulin receptor; HAMD-17, Hamilton Depression Scale-17 for Depression; LD, linkage disequilibrium; OR, odds ratio; CI, confidence interval; eQTL, expression quantitative trait loci.
Acknowledgments
The work was supported by the National Key Research and Development Program of China (2016YFC1307100), the National Natural Science Foundation of China (81771465, 81930033), the Sanming Project of Medicine in Shenzhen, SZSM201612006, the National Key Technologies R&D Program of China (2012BAI01B04); and also supported by the Innovative Research Team of High-level Local Universities in Shanghai. We would like to thank Editage for English language editing.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
We declare that we have no competing financial interests in this work.
References
1. Amin N, Jovanova O, Adams HH, et al. Exome-sequencing in a large population-based study reveals a rare Asn396Ser variant in the LIPG gene associated with depressive symptoms. Mol Psychiatry. 2017;22(4):537–543. doi:10.1038/mp.2016.101.mp2016101[pii]
2. Anderson RJ, Freedland KE, Clouse RE, Lustman PJ. The prevalence of comorbid depression in adults with diabetes: a meta-analysis. Diabetes Care. 2001;24(6):1069–1078. doi:10.2337/diacare.24.6.1069
3. Basta-Kaim A, Szczesny E, Glombik K, et al. Prenatal stress affects insulin-like growth factor-1 (IGF-1) level and IGF-1 receptor phosphorylation in the brain of adult rats. Eur Neuropsychopharmacol. 2014;24(9):1546–1556. doi:10.1016/j.euroneuro.2014.07.002
4. Belmaker RH, Agam G. Major depressive disorder. N Engl J Med. 2008;358(1):55–68. doi:10.1056/NEJMra073096.358/1/55.[pii]
5. Bodhini D, Radha V, Mohan V. Association study of IRS1 gene polymorphisms with type 2 diabetes in south Indians. Diabetes Technol Ther. 2011;13(7):767–772. doi:10.1089/dia.2011.0017
6. Brancucci NM, Witmer K, Schmid C, Voss TS. A var gene upstream element controls protein synthesis at the level of translation initiation in Plasmodium falciparum. PLoS One. 2014;9(6):e100183. doi:10.1371/journal.pone.0100183
7. Carvalho E, Jansson PA, Axelsen M, et al. Low cellular IRS 1 gene and protein expression predict insulin resistance and NIDDM. FASEB J. 1999;13(15):2173–2178. doi:10.1096/fasebj.13.15.2173
8. Chirita AL, Gheorman V, Bondari D, Rogoveanu I. Current understanding of the neurobiology of major depressive disorder. Rom J Morphol Embryol. 2015;56(2 Suppl):651–658.
9. de Groot M, Anderson R, Freedland KE, Clouse RE, Lustman PJ. Association of depression and diabetes complications: a meta-analysis. Psychosom Med. 2001;63(4):619–630. doi:10.1097/00006842-200107000-00015
10. Fatimah A, Mdidris M, Romzi M. Perception of bodyweight status among office workers in two gorvernment departments in Kuala Lumpur. Malays J Nutr. 1995;1(1):11–19.
11. Ghasemi R, Dargahi L, Haeri A, Moosavi M, Mohamed Z, Ahmadiani A. Brain insulin dysregulation: implication for neurological and neuropsychiatric disorders. Mol Neurobiol. 2013;47(3):1045–1065. doi:10.1007/s12035-013-8404-z
12. Ghasemi R, Haeri A, Dargahi L, Mohamed Z, Ahmadiani A. Insulin in the brain: sources, localization and functions. Mol Neurobiol. 2013;47(1):145–171. doi:10.1007/s12035-012-8339-9
13. Głombik K, Ślusarczyk J, Trojan E, et al. Regulation of insulin receptor phosphorylation in the brains of prenatally stressed rats: new insight into the benefits of antidepressant drug treatment. Eur Neuropsychopharmacol. 2017;27(2):120–131. doi:10.1016/j.euroneuro.2016.12.005
14. Gonzalez C, Diaz F, Alonso A. Neuroprotective effects of estrogens: cross-talk between estrogen and intracellular insulin signalling. Infect Disord Drug Targets. 2008;8(1):65–67. doi:10.2174/187152608784139659
15. Havrankova J, Brownstein M, Roth J. Insulin and insulin receptors in rodent brain. Diabetologia. 1981;20(Suppl 1):268–273. doi:10.1007/BF00254492
16. Havrankova J, Roth J, Brownstein M. Insulin receptors are widely distributed in the central nervous system of the rat. Nature. 1978;272(5656):827–829. doi:10.1038/272827a0
17. Kawamura S, Maesawa C, Nakamura K, et al. Predisposition for borderline personality disorder with comorbid major depression is associated with that for polycystic ovary syndrome in female Japanese population. Neuropsychiatr Dis Treat. 2011;7:655. doi:10.2147/NDT.S25504
18. Kemp DE, Schinagle M, Gao K, et al. PPAR-γ agonism as a modulator of mood: proof-of-concept for pioglitazone in bipolar depression. CNS Drugs. 2014;28(6):571–581. doi:10.1007/s40263-014-0158-2
19. Kilpelainen TO, Zillikens MC, Stancakova A, et al. Genetic variation near IRS1 associates with reduced adiposity and an impaired metabolic profile. Nat Genet. 2011;43(8):753–760. doi:10.1038/ng.866ng.866[pii]
20. Lang UE, Borgwardt S. Molecular mechanisms of depression: perspectives on new treatment strategies. Cell Physiol Biochem. 2013;31(6):761–777. doi:10.1159/000350094
21. Li Z, Zhang Z, He Z, et al. A partition-ligation-combination-subdivision EM algorithm for haplotype inference with multiallelic markers: update of the SHEsis (http://analysis.bio-x.cn). Cell Res. 2009;19(4):519–523. doi:10.1038/cr.2009.33
22. Mielke JG, Taghibiglou C, Liu L, et al. A biochemical and functional characterization of diet-induced brain insulin resistance. J Neurochem. 2005;93(6):1568–1578. doi:10.1111/j.1471-4159.2005.03155.x
23. Moloney AM, Griffin RJ, Timmons S, O’Connor R, Ravid R, O’Neill C. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer’s disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol Aging. 2010;31(2):224–243. doi:10.1016/j.neurobiolaging.2008.04.002
24. Morris MC, Evans DA, Bienias JL, Tangney CC, Wilson RS. Dietary fat intake and 6-year cognitive change in an older biracial community population. Neurology. 2004;62(9):1573–1579. doi:10.1212/01.WNL.0000123250.82849.B6
25. Pan A, Ye X, Franco OH, et al. Insulin resistance and depressive symptoms in middle-aged and elderly Chinese: findings from the Nutrition and Health of Aging Population in China Study. J Affect Disord. 2008;109(1–2):75–82. doi:10.1016/j.jad.2007.11.002
26. Qiu H-C, Liu H-Z, Li X, Zeng X, Zhao J-Z. Insulin resistance as estimated by homeostasis model assessment predicts incident post-stroke depression in Chinese subjects from ischemic stroke. J Affect Disord. 2018;231:1–7. doi:10.1016/j.jad.2018.01.023
27. Ramasamy A, Trabzuni D, Guelfi S, et al. Genetic variability in the regulation of gene expression in ten regions of the human brain. Nat Neurosci. 2014;17(10):1418. doi:10.1038/nn.3801
28. Seedat S, Scott KM, Angermeyer MC, et al. Cross-national associations between gender and mental disorders in the World Health Organization World Mental Health Surveys. Arch Gen Psychiatry. 2009;66(7):785–795. doi:10.1001/archgenpsychiatry.2009.36
29. Sparse whole-genome sequencing identifies two loci for major depressive disorder. Nature. 2015;523(7562):588–591. doi:10.1038/nature14659
30. Sun XJ, Rothenberg P, Kahn CR, et al. Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein. Nature. 1991;352(6330):73–77. doi:10.1038/352073a0
31. Timonen M, Rajala U, Jokelainen J, Keinanen-Kiukaanniemi S, Meyer-Rochow VB, Rasanen P. Depressive symptoms and insulin resistance in young adult males: results from the Northern Finland 1966 birth cohort. Mol Psychiatry. 2006;11(10):929–933. doi:10.1038/sj.mp.4001838
32. Wayhs CA, Tortato C, Mescka CP, et al. The association effect of insulin and clonazepam on oxidative stress in liver of an experimental animal model of diabetes and depression. Pharm Biol. 2013;51(5):533–538. doi:10.3109/13880209.2012.747544
33. White MF. IRS proteins and the common path to diabetes. Am J Physiol Endocrinol Metab. 2002;283(3):E413–422. doi:10.1152/ajpendo.00514.2001
34. White MF, Maron R, Kahn CR. Insulin rapidly stimulates tyrosine phosphorylation of a Mr-185,000 protein in intact cells. Nature. 1985;318(6042):183–186. doi:10.1038/318183a0
35. Wong ML, Arcos-Burgos M, Liu S, et al. The PHF21B gene is associated with major depression and modulates the stress response. Mol Psychiatry. 2017;22(7):1015–1025. doi:10.1038/mp.2016.174.mp2016174[pii]
36. Wray NR, Ripke S, Mattheisen M, et al. Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat Genet. 2018;50(5):668–681. doi:10.1038/s41588-018-0090-3
37. Yong Y, Lin H. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. 2005;15(2):97. doi:10.1038/sj.cr.7290272
38. Yu C, Baune BT, Licinio J, Wong ML. A novel strategy for clustering major depression individuals using whole-genome sequencing variant data. Sci Rep. 2017;7:44389. doi:10.1038/srep44389
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.