Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 17
Ultrasound Assessment of the Rectus Femoris in Patients with Chronic Obstructive Pulmonary Disease Predicts Sarcopenia
Authors Deng M, Yan L , Tong R, Zhao J, Li Y, Yin Y , Zhang Q, Gao J, Wang Q, Hou G, Zhou X
Received 15 August 2022
Accepted for publication 25 October 2022
Published 2 November 2022 Volume 2022:17 Pages 2801—2810
DOI https://doi.org/10.2147/COPD.S386278
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Min Zhang
Mingming Deng1– 4*, Liming Yan5 *, Run Tong1– 4*, Joseph Zhao,6 Yanxia Li,7 Yan Yin,8 Qin Zhang,8 Jinghan Gao,8 Qiuyue Wang,8 Gang Hou,1– 4 Xiaoming Zhou9,10
1Department of Pulmonary and Critical Care Medicine, Center of Respiratory Medicine, China-Japan Friendship Hospital, Beijing, People’s Republic of China; 2National Center for Respiratory Medicine, Beijing, People’s Republic of China; 3Institute of Respiratory Medicine, Chinese Academy of Medical Sciences, Beijing, People’s Republic of China; 4National Clinical Research Center for Respiratory Diseases, Beijing, People’s Republic of China; 5Department of Pulmonary and Critical Care Medicine, Fourth Hospital of China Medical University, Shenyang, People’s Republic of China; 6Sage Hill High School, Newport Coast, CA, USA; 7Institute of Respiratory Diseases, Department of Hematology, The First Affiliated Hospital of Dalian Medical University, Dalian, People’s Republic of China; 8Department of Pulmonary and Critical Care Medicine, First Hospital of China Medical University, Shenyang, People’s Republic of China; 9Respiratory Department, Center for Pulmonary Vascular Diseases, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China; 10Department of Pulmonary and Critical Care Medicine, Shengjing Hospital of China Medical University, Shenyang, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Gang Hou, Department of Pulmonary and Critical Care Medicine, Centre of Respiratory Medicine, China-Japan Friendship Hospital, Beijing, People’s Republic of China, Email [email protected] Xiaoming Zhou, Respiratory Department, Center for Pulmonary Vascular Diseases, Fuwai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China, Email [email protected]
Purpose: Sarcopenia, an age-related loss of skeletal muscle mass and function, is frequent in patients with chronic obstructive pulmonary disease (COPD) and is linked to a poor prognosis. The diagnosis of sarcopenia requires specific equipment and is inconvenient to introduce into clinical practice. Ultrasound is an innovative method to assess muscle quantity. The objective of this research was to evaluate the use of ultrasound for prospectively screening for sarcopenia in COPD patients.
Patients and Methods: A total of 235 stable patients with COPD were included in this observational study and divided into development and validation sets. The Asian Working Group for Sarcopenia standards were used to define sarcopenia. The thickness (RFthick) and cross-sectional area (RFcsa) of the rectus femoris were measured using ultrasound. Receiver operating characteristic curve analysis of RFthick and RFcsa was used to predict sarcopenia. Nomogram models were constructed based on RFthick, RFcsa, age and body mass index to identify sarcopenia.
Results: Sarcopenia was present in 83 (35.32%) patients. Patients with sarcopenia had advanced age, decreased pulmonary function, decreased physical function and poor clinical outcomes. RFthick and RFcsa showed good predictive ability for sarcopenia in the development and validation sets. The nomogram based on RFthick and RFcsa could detect sarcopenia in COPD patients, and all had significant predictive performance in the development and validation sets. The calibration plot showed good agreement between the nomogram predictions and actual observations.
Conclusion: The ultrasound measurement of the rectus femoris has potential for the clinical assessment of sarcopenia in patients with COPD.
Keywords: chronic obstructive pulmonary disease, sarcopenia, ultrasound, rectus femoris, nomogram
Introduction
Chronic airway inflammation and extrapulmonary changes are hallmarks of the systemic illness chronic obstructive pulmonary disease (COPD).1 One of the significant systemic symptoms of COPD patients is sarcopenia, which is an age-related loss of skeletal muscle mass and function, and the prevalence rate can reach 152–55%.3 The major symptom of sarcopenia is a gradual and severe loss of muscular function. It leads to decreased exercise tolerance and quality of life in COPD patients and is a separate risk factor for a higher risk of mortality.4 Therefore, early identification and diagnosis of sarcopenia would enhance prognosis and support medical professionals in clinical therapy.
The European Working Group of Sarcopenia in Older People (EWGSOP)5 and the Asian Working Group for Sarcopenia (AWGS)6 suggested that the diagnosis of sarcopenia should involve combining measurements of skeletal muscle quantity (by bioimpedance analysis or dual-energy X-rays) and skeletal muscular function (walking speed and handgrip strength). Sarcopenia diagnosis requires specialized tools and expert training and is generally challenging to carry out in underdeveloped nations and primary healthcare settings. Therefore, it is important to establish a convenient method of sarcopenia screening.
A crucial component of sarcopenia diagnosis is the evaluation of muscle quantity. Ultrasound (US) has been demonstrated to be precise, dependable, not involve exposure to ionizing radiation, has high repeatability for measuring muscle quantity in different populations and is available at the bedside.7–10 Importantly, ultrasound offers extremely strong reliability and repeatability when performed by ultrasonographers who received formal training [ICC (intraclass correlation coefficient) of 0.97–0.99 for muscle thickness parameter].11 Skeletal muscle dysfunction in COPD patients can manifest as abnormal muscle mass of the lower extremities.12 Therefore, ultrasound-measured muscles of the lower limb may have potential clinical relevance in sarcopenia prediction in COPD patients. Several studies in patients with COPD13–15 have demonstrated that rectus femoris size measured by ultrasound is related to fat-free mass, muscle function (quadriceps strength), and physical performance. However, the ability of ultrasound measurement of the rectus femoris to anticipate sarcopenia in COPD patients is still unclear.
In this study, we first analyzed the prevalence and clinical impact of sarcopenia in Chinese COPD patients. Next, we tried to evaluate the usefulness of ultrasonography in prospectively evaluating sarcopenia in COPD patients and to select the cut-off values that would be employed in clinical practice. Finally, a nomogram model was created to enhance the predictive ability of sarcopenia.
Materials and Methods
Study Design and Patients
A total of 235 patients (≥40 years old) with stable COPD from the First Hospital of China Medical University (Shenyang, China) and the First Hospital of Dalian Medical University (Dalian, China) participated in the prospective trial between August 2018 and December 2019.
The inclusion criteria were as follows: diagnosis of stable COPD determined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria; age ≥ 40.
The exclusion criteria were as follows: COPD exacerbation during the previous month, active pulmonary disease, serious cardiovascular illness, concurrent musculoskeletal disease, long-term systemic steroid medication, and an inability to comprehend informed consent documents. The study was approved by the research ethics committees of the First Hospital of China Medical University [No. 2018–144-2] and ethics committees at the First Hospital of Dalian Medical University. This study complied with the Declaration of Helsinki. All patients signed informed consent. Clinical characteristics were gleaned from medical records, including age, sex, weight and height.
A total of 117 COPD patients from the First Hospital of China Medical University were recruited for the development set to evaluate the clinical usefulness of ultrasound and set the cut-off values. The validation group included an additional 118 COPD patients from the First Hospital of Dalian Medical University.
Pulmonary Function and Assessment by the Modified British Medical Research Council (mMRC) Score and the COPD Assessment Test (CAT)
A Jaeger MasterScreen system (Jaeger, Viasys Healthcare GmbH, Hochberg, Germany) was used to carry out spirometry measures in line with the recommendations of the American Thoracic Society and the European Respiratory Society. The Chinese version of the mMRC dyspnea score16 and the CAT17 were used to assess dyspnoea symptoms and health status, respectively.
Five-Repetition Sit-to-Stand Test (5STS)
The participant is seated on an armless, 48 cm-high chairs with feet on the floor, backs supporting the back of the chair, and arms crossed in front of chests. The patient was instructed to carry out 5 standing and sitting motions as quickly as they could after hearing the test start order, and the time was recorded. Throughout the test, hands must be crossed on the chest and cannot be separated, and the knee joint must be completely even when standing. During the test, the participants were provided verbal support. The patients completed three 5STSs with the requisite intervals (>5 minutes). Participants were given a score based on the minimum value of the 5STS after three trials.
6MWT
An enclosed, straight, and linear 30 m corridor was selected indoors based on the recommendations from the 2002 American Thoracic Society (ATS).18 Before the test, the patients were informed of the test procedure and instructed to walk as much as possible. If they experience shortness of breath, chest pain, or dizziness, they can slow down and stop to rest. The test should be stopped immediately under the experimenter’s supervision, and standard language should be used to reassure the patient if the aforementioned symptoms persist even after rest. The patient is given the “time is up” order to halt after 6 minutes, and the test staff records the distance travelled in meters.
Health-Related Quality of Life (HRQoL) Measurement
The HRQoL was captured by three instruments: St George’s Respiratory Questionnaire (SGRQ), and BODE (Body-Mass Index, airway obstruction, dyspnea, and exercise capacity) index. All measurements were applied by the same researcher.
The St. George’s Respiratory Questionnaire is a self-administered or face-to-face or telephone-administered instrument, consists of 50 items divided into three components—symptoms (distress caused by respiratory symptoms), activity (disturbance in daily activities), and impact (psychosocial function)—summed to give a total score of overall health status.19 Overall scores range from 0–100, where a higher score represents a poorer quality of life.
The BODE index is a composite value of the Body-Mass Index (BMI), airflow Obstruction (assessed by FEV1% (forced expiratory volume in 1 s)), Dyspnoea (mMRC) and Exercise capacity (6MWT). Its value ranges from 0 to 10 points where 10 indicates maximum mortality risk and an increase in the BODE Index by one point is associated with a significant rise in all-cause mortality.20,21
Measurement Thickness and Cross-Sectional Area of the Rectus Femoris
The thickness and cross-sectional area of the quadriceps rectus femoris were measured following previous studies.22 A 4- to 15-MHz linear-array transducer (SuperSonic Imagine, Aix-en-Provence, France) was employed with greyscale ultrasound. The ultrasound tests were carried out by two ultrasonography doctors.
The patient was asked to refrain from any vigorous exercise within 72 hours. To lessen muscle distortion brought on by outside pressures, the researchers fixed the ultrasound probe to a bracket and positioned it perpendicular to the dominant leg of patients. The transducer was placed with its long axis perpendicular to the dominant leg (exactly at a distance of 3/5 of the distance between the anterior superior iliac spine and the superior patellar border). The femur’s orientational ability was taken into consideration when setting the scanning depth. To identify muscle septa before image capture, gentle contraction-relaxation manoeuvres were performed. After a moving cursor on a frozen picture traced the inner echogenic line of the rectus femoris, RFthick and RFcsa were determined. Three consecutive readings within 10% were averaged to determine RFthick and RFcsa.
Assessment of Sarcopenia
The definition of sarcopenia adhered to the AWGS recommendations:6 low muscle mass (bioelectrical impedance (M: <7.0 kg/m2, F: <5.7 kg/m2), low muscle strength (hand grip strength (M: <28 kg, F: <18 kg)) and/or poor physical performance (5-time chair stand test: ≥12 s). The measurement of muscle mass was accomplished by implementing bioelectrical impedance analysis (BIA) (InBody770; InBody, Seoul, Korea). Hand grip test (HGS) was assessed by handgrip strength using a JAMAR®Plus+ hand dynamometer (Sammons Preston, Bolingbrook, IL, USA). HGS assessment was performed using a standard technique,23,24 with the patient in the sitting position, the elbow at 90° flexion and the wrist in a neutral position. The subjects were instructed to apply the maximum HGS 3 times with both hands, with 30s of rest allowed between each measurement. Meanwhile, physical performance was evaluated using the 5-time chair stand test.
Construction of the Nomogram and Decision Curve Analysis (DCA)
A nomogram constructed following our previous study,25 using the R package “rms” is a convenient and powerful way to illustrate the outcomes of a risk model. To determine if the prediction nomogram was clinically beneficial, DCA was utilized.
Statistical Analyses
SPSS 13.0 software was used to conduct statistical analysis (IBM, Armonk, NY, USA). The relationship between continuous variables was examined by using Pearson’s correlation coefficient. Analysis of associations between categorical variables was performed using the chi-square test. One-way analysis of variance (ANOVA) with the post hoc Tukey multiple comparison test (for normally distributed data) or Kruskal‒Wallis test (for nonnormal distribution) was used to evaluate differences between three or more groups for continuous variables. The t-test (normally distributed data) or Mann‒Whitney test (nonnormal distribution) was utilized to contrast the variations between the two groups. p values less than 0.05 were regarded as statistically significant.
Results
Baseline Characteristics of the Subjects
A total of 235 patients were enrolled in the final analysis. Table 1 provides a list of baseline characteristics of the patients. The patients were 64.4±10.7 (range 43–81) years old, and 68.01% (160/235) were male. Sarcopenia was present in 83 (35.32%) patients. Sarcopenic patients were noticeably older. Sarcopenia patients with COPD showed markedly decreased pulmonary function (FEV1, FEV1%predicted, FVC, FVC% predicted, and FEV1/FVC), lower physical function (6MWD and 5STS) and notably advanced GOLD stages. Sarcopenic patients exhibited considerably lower body composition measures such as BMI, FFM, FFMI, SMM, SMMI, and HGS.
 |
Table 1 Baseline Characteristics of Subjects |
Respiratory Symptoms and Health-Related Quality of Life Associated with Sarcopenia in Patients with COPD
Next, the relationship between respiratory symptoms, health-related quality of life and sarcopenia in patients with COPD was analysed (Table 2). The clinical respiratory symptoms of dyspnea in COPD patients are frequently evaluated using the CAT and the mMRC score.26 Patients with sarcopenia reported considerably worse dyspnea symptoms and higher mMRC and CAT scores than those without the condition. The St. George’s Respiratory Questionnaire (SGRQ) is a scale widely used to assess the quality of life and health impairment of patients with COPD,27 of which a higher score denotes a lower quality of life. According to our research, patients with sarcopenia experienced considerably higher SGRQ symptoms, activity, impact, and total scores than patients without sarcopenia. The SGRQ scores between the two groups, however, showed no appreciable variation. Finally, we analyzed the difference in the BODE index, a multidimensional scoring system designed as a prognostic indicator for COPD patients. We found that the median score on the BODE index was also significantly higher in patients with sarcopenia. These results suggest that patients with sarcopenia show poor clinical outcomes.
 |
Table 2 Clinical Outcome According to Presence or Absence of Sarcopenia |
Both univariate and multivariate analyses were performed to investigate the clinical variables connected to sarcopenia (Table 3). Age (odds ratio [OR]: 1.084, 95% CI: 1.028–1.144, p=0.003), BMI (OR: 0.844, 95% CI: 0.752–0.948, p=0.004), and GOLD stage (OR: 1.746, 95% CI: 1.076–2.835, p=0.024) were related to sarcopenia in COPD patients, in line with the outcomes of the univariate analysis. Meanwhile, old age (OR: 1.125, 95% CI: 1.036–1.222, p= 0.005) and BMI (OR: 0.800, 95% CI: 0.683–0.937, p=0.006) were shown to be independent factors in multivariate analysis.
 |
Table 3 Clinical Factors Associated with Sarcopenia in Patients with COPD |
Development and Validation of RFthick and RFcsa for Predicting Sarcopenia in Patients with COPD
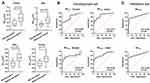
Next, we aimed to assess and evaluate the effectiveness of ultrasound for screening sarcopenia in COPD patients. Regardless of sex, patients with sarcopenia had much lower RFcsa and RFthick values than those without the condition (Figure 1A).
A total of 235 patients were split into a development set (117 cases) and a validation set (118 cases) (Table 4). The ROC curves from the development set that demonstrate that skeletal muscle ultrasonography can predict sarcopenia based on RFthick and RFcsa (Figure 1B). In male COPD patients for predicting sarcopenia, RFthick had a sensitivity and specificity of 76.36% and 75% (the cut-off point was 5.22 cm and the AUC value was 0.839), respectively, whereas RFcsa had a sensitivity and specificity of 90.91% and 67.86%, respectively (the cut-off point was 6.805 cm2 and the AUC value was 0.872). When predicting sarcopenia in female patients, RFcsa had a sensitivity of 89.47% and a specificity of 60% (the cut-off point was 4.99 cm, and the AUC value was 0.805), whereas RFcsa had a sensitivity of 73.68% and a specificity of 86.67% (the cut-off point was 6.996 cm2, and the AUC value was 0.791). The results from the validation set (Figure 1C) revealed that the AUC value for predicting sarcopenia with the RFthick was 0.835 (p<0.001), whereas the AUC value for predicting sarcopenia based on RFcsa was 0.849 (p<0.001).
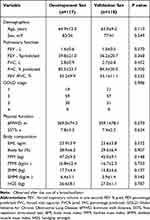 |
Table 4 Patient Characteristics of Development Set and Validation Set |
To further determine the potential of RFthick and RFcsa as predictive biomarkers for sarcopenia, we next performed a multivariate logistic analysis (Table 3). Notably, decreased RFthick (OR: 0.216, 95% CI: 0.146–0.493, p=0.016) and RFcsa (OR: 0.132, 95% CI: 0.071–0.198, p=0.007) levels emerged as independent factors for sarcopenia.
Construction of a Nomogram to Predict Sarcopenia
The joint evaluation of multiple independent predictive factors can help doctors better predict sarcopenia. The nomogram could integrate multiple predictive indicators to provide a personalized predictive model. Next, we aimed to develop a nomogram model combining ultrasound measurements of the rectus femoris (RFthick, RFcsa) and clinical features to predict sarcopenia. Age and BMI were also taken into consideration while creating the nomogram model, as they were independent risk factors. First, a nomogram model combining US, age and BMI was constructed using a development set (Figure 2A). The estimates with the nomogram and real measurements revealed good agreement in the calibration curve (Figure 2B). The ROC analysis findings showed that the nomogram model’s AUC value was 0.952 (Figure 2C). The results were validated using decision curve analysis (DCA). The combined nomogram model had the best effectiveness for sarcopenia predictions when compared to a single clinical variable (Figure 2D). Additionally, the calibration curve (Figure 2E) and ROC analysis (AUC=0.956) dependent on the validation cohort both support the capacity to predict outcomes (Figure 2F). Overall, the nomogram models based on clinical features, RFthick, and RFcsa could identify sarcopenia in COPD patients.
Discussion
The prevalence of sarcopenia in patients with stable COPD in this study was 35.32%, which was in line with prevalence estimates of 15–55% reported in previous studies from other countries.2,3 However, the result of this study cannot represent the prevalence of sarcopenia in COPD patients in China, which may need a national cross-sectional study using multistage stratified cluster sampling procedure. In our study, patients with sarcopenia tended to have advanced age, lower physical function, and poor clinical outcomes. Sarcopenia was also significantly higher in patients with more severe diseases than in those with less severe diseases. In our study, the prevalence of sarcopenia in male and female patients was 35.3% (53/150) and 40% (30/75), respectively, and there was no significant difference. In addition, the number of men with COPD in our study was significantly greater than that of women. This composition does not affect our research results because the result from a national cross-sectional study of China28 showed that the prevalence of COPD was higher in men (11.9%) than in women (5.4%).
The clinical diagnosis of sarcopenia still heavily relies on the assessment of muscle mass, which can be acquired by dual X-ray absorptiometry (DXA) or multifrequency bioelectrical impedance.5,6 Ultrasound (US) has proven to be an accurate, reliable, noninvasive technique to measure muscle mass in different populations, including patients with COPD. Most importantly, ultrasound offers extremely strong repeatability and dependability when carried out by adequately qualified professionals.11 In this study, our results indicate that ultrasound measurement of the rectus femoris has good clinical application value in predicting sarcopenia in COPD patients. Through our study, the cut-off value for an ultrasound examination was established, which will aid in the use of the clinical application of skeletal muscle ultrasound in the screening of sarcopenia.
Compared to single biomarkers, multiple biomarkers might considerably increase the prediction strength of predictive models. A nomogram, a statistical modelling method that comprehensively incorporates the impact of diverse clinical variables,29 has been used for the prediction of low muscle mass or sarcopenia in patients with cirrhosis30 and gastric cancer.31 However, there is currently no nomogram for predicting sarcopenia in COPD patients. In this research, the nomograms based on RFthick, RFcsa (measured by ultrasound) and clinical features could identify sarcopenia in COPD patients and demonstrated a strong propensity for prediction (AUC>0.92) in both the development and validation sets. The calibration plot also showed good agreement between the nomogram predictions and actual observations. Overall, for the first time, we constructed nomogram models for patients with COPD based on ultrasound to predict sarcopenia.
This study also has several limitations. First, although BIA is frequently used to diagnose sarcopenia, it is not a reliable substitute for measuring skeletal muscle mass. Second, an important flaw in this study is the absence of a healthy control group. Additionally, the trial was restricted to those with stable COPD, so the applicability of the results to COPD patients experiencing acute exacerbations and those undertaking pulmonary rehabilitation is still uncertain. These issues will be further covered in subsequent research.
Conclusion
The clinical examination of sarcopenia in COPD patients may benefit from the ultrasonic measurement of the rectus femoris.
Acknowledgments
This research was supported by National High Level Hospital Clinical Research Funding (2022-NHLHCRF-LX-01), the Elite Medical Professionals project of China-Japan Friendship Hospital (No.ZRJY2021-BJ08), the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (No. 2020-PT320-001), National Natural Science Foundation of China (No. 81900040), Liaoning Education Ministry Supporting Foundation (No. QN2019014), and Liaoning Science and Technology Ministry Supporting Foundation (No. 2019-ZD-0766).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Vanfleteren LEGW, Spruit MA, Wouters EFM, Franssen FME. Management of chronic obstructive pulmonary disease beyond the lungs. Lancet Respir Med. 2016;4(11):911–924. doi:10.1016/S2213-2600(16)00097-7
2. Jones SE, Maddocks M, Kon SSC, et al. Sarcopenia in COPD: prevalence, clinical correlates and response to pulmonary rehabilitation. Thorax. 2015;70(3):213–218. doi:10.1136/thoraxjnl-2014-206440
3. Cebron Lipovec N, Schols AM, van den Borst B, et al. Sarcopenia in advanced COPD affects cardiometabolic risk reduction by short-term high-intensity pulmonary rehabilitation. J Am Med Dir Assoc. 2016;17(9):814–820. doi:10.1016/j.jamda.2016.05.002
4. Sepúlveda-Loyola W, Osadnik C, Phu S, Morita AA, Duque G, Probst VS. Diagnosis, prevalence, and clinical impact of sarcopenia in COPD: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2020;11(5):1164–1176. doi:10.1002/jcsm.12600
5. Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31. doi:10.1093/ageing/afy169
6. Chen L-K, Woo J, Assantachai P, et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21(3):300–307.e2. doi:10.1016/j.jamda.2019.12.012
7. Nijholt W, Scafoglieri A, Jager-Wittenaar H, Hobbelen JSM, van der Schans CP. The reliability and validity of ultrasound to quantify muscles in older adults: a systematic review. J Cachexia Sarcopenia Muscle. 2017;8(5):702–712. doi:10.1002/jcsm.12210
8. Tillquist M, Kutsogiannis DJ, Wischmeyer PE, et al. Bedside ultrasound is a practical and reliable measurement tool for assessing quadriceps muscle layer thickness. JPEN J Parenter Enteral Nutr. 2014;38(7):886–890. doi:10.1177/0148607113501327
9. Perkisas S, Bastijns S, Baudry S, et al. Application of ultrasound for muscle assessment in sarcopenia: 2020 SARCUS update. Eur Geriatr Med. 2021;12(1):45–59. doi:10.1007/s41999-020-00433-9
10. Liu J, Yang P, Tian H, et al. Right ventricle remodeling in chronic thromboembolic pulmonary hypertension. J Transl Int Med. 2022;10(2):125–133. doi:10.2478/jtim-2022-0027
11. Hammond K, Mampilly J, Laghi FA, et al. Validity and reliability of rectus femoris ultrasound measurements: comparison of curved-array and linear-array transducers. J Rehabil Res Dev. 2014;51(7):1155–1164. doi:10.1682/JRRD.2013.08.0187
12. Man WDC, Soliman MGG, Nikoletou D, et al. Non-volitional assessment of skeletal muscle strength in patients with chronic obstructive pulmonary disease. Thorax. 2003;58(8):665–669. doi:10.1136/thorax.58.8.665
13. Seymour JM, Ward K, Sidhu PS, et al. Ultrasound measurement of rectus femoris cross-sectional area and the relationship with quadriceps strength in COPD. Thorax. 2009;64(5):418–423. doi:10.1136/thx.2008.103986
14. Shrikrishna D, Patel M, Tanner RJ, et al. Quadriceps wasting and physical inactivity in patients with COPD. Eur Respir J. 2012;40(5):1115–1122. doi:10.1183/09031936.00170111
15. Cruz-Montecinos C, Guajardo-Rojas C, Montt E, et al. Sonographic measurement of the quadriceps muscle in patients with chronic obstructive pulmonary disease: functional and clinical implications. J Ultrasound Med. 2016;35(11):2405–2412. doi:10.7863/ultra.15.11032
16. Bestall JC, Paul EA, Garrod R, Garnham R, Jones PW, Wedzicha JA. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax. 1999;54(7):581–586. doi:10.1136/thx.54.7.581
17. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD assessment test. Eur Respir J. 2009;34(3):648–654. doi:10.1183/09031936.00102509
18. ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166(1):111–117. doi:10.1164/ajrccm.166.1.at1102
19. Jones PW. Quality of life measurement for patients with diseases of the airways. Thorax. 1991;46(9):676–682. doi:10.1136/thx.46.9.676
20. Celli BR, Cote CG, Marin JM, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(10):1005–1012. doi:10.1056/NEJMoa021322
21. Araujo ZT, Holanda G. Does the BODE index correlate with quality of life in patients with COPD? J Bras Pneumol. 2010;36(4):447–452. doi:10.1590/s1806-37132010000400009
22. Maynard-Paquette AC, Poirier C, Chartrand-Lefebvre C, Dubé BP. Ultrasound evaluation of the quadriceps muscle contractile index in patients with stable chronic obstructive pulmonary disease: relationships with clinical symptoms, disease severity and diaphragm contractility. Int J Chron Obstruct Pulmon Dis. 2020;15:79–88. doi:10.2147/copd.S222945
23. Bahat G, Kilic C, Ozkok S, Ozturk S, Karan MA. Associations of sarcopenic obesity versus sarcopenia alone with functionality. Clin Nutr. 2021;40(5):2851–2859. doi:10.1016/j.clnu.2021.04.002
24. Massy-Westropp NM, Gill TK, Taylor AW, Bohannon RW, Hill CL. Hand grip strength: age and gender stratified normative data in a population-based study. BMC Res Notes. 2011;4:127. doi:10.1186/1756-0500-4-127
25. Li XL, Wu C, Xie JG, et al. Development and validation of a nomogram for predicting the disease progression of nonsevere coronavirus disease 2019. J Transl Int Med. 2021;9(2):131–142. doi:10.2478/jtim-2021-0030
26. Zhou Z, Zhou A, Zhao Y, Chen P. Evaluating the clinical COPD questionnaire: a systematic review. Respirology. 2017;22(2):251–262. doi:10.1111/resp.12970
27. Martin AL, Marvel J, Fahrbach K, Cadarette SM, Wilcox TK, Donohue JF. The association of lung function and St. George’s respiratory questionnaire with exacerbations in COPD: a systematic literature review and regression analysis. Respir Res. 2016;17:40. doi:10.1186/s12931-016-0356-1
28. Wang C, Xu J, Yang L, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study. Lancet. 2018;391(10131):1706–1717. doi:10.1016/S0140-6736(18)30841-9
29. Liu P, Zhu H, Zhu H, et al. Predicting survival for hepatic arterial infusion chemotherapy of unresectable colorectal liver metastases: radiomics analysis of pretreatment computed tomography. J Transl Int Med. 2022;10(1):56–64. doi:10.2478/jtim-2022-0004
30. Tandon P, Low G, Mourtzakis M, et al. A model to identify sarcopenia in patients with cirrhosis. Clin Gastroenterol Hepatol. 2016;14(10):1473–1480.e3. doi:10.1016/j.cgh.2016.04.040
31. Zhang F-M, Chen X-L, Wu Q, et al. Development and validation of nomograms for the prediction of low muscle mass and radiodensity in gastric cancer patients. Am J Clin Nutr. 2021;113(2):348–358. doi:10.1093/ajcn/nqaa305
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.