Back to Journals » OncoTargets and Therapy » Volume 13
Three Novel EGFR Mutations (750_758del, I759S, T751_I759delinsS) in One Patient with Metastatic Non-Small Cell Lung Cancer Responding to Osimertinib: A Case Report
Authors Li H , Yu T, Lin Y, Xie Y, Feng J, Huang M, Guo A, Liu X, Yin Z
Received 23 April 2020
Accepted for publication 20 July 2020
Published 10 August 2020 Volume 2020:13 Pages 7941—7948
DOI https://doi.org/10.2147/OTT.S259616
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nicola Silvestris
Huiying Li,1 Tingting Yu,1 Yongjuan Lin,1 Yu Xie,1 Jie Feng,1 Mingmin Huang,1 Aibin Guo,1 Xiangyu Liu,2 Zhenyu Yin1
1Department of Geriatric Oncology, Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School, Nanjing, People’s Republic of China; 2Department of Neurosurgery, Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School, Nanjing, People’s Republic of China
Correspondence: Zhenyu Yin Department of Geriatric Oncology
Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School Tel +8613913904579
Email [email protected]
Xiangyu Liu
Department of Neurosurgery, Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School Tel +8613851672018
Email [email protected]
Abstract: Generations of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) can significantly improve the outcome of EGFR-positive NSCLC patients. However, acquired TKIs-resistant mutations are inevitable. Except the common EGFR alterations, more and more rare mutations are revealed by next-generation sequencing (NGS), the clinical significance of which are still unclear. Here, we report an advanced lung adenocarcinoma patient who harbored two novel EGFR exon 19 deletions (750_758del and I759S) at the beginning and exhibited a short response to icotinib for 7.0 months. Then, secondary resistance EGFR T751_I759delinsS occurred. Chemotherapy combined with bevacizumab and erlotinib was administered in turn but failed. Standard-dose osimertinib (80 mg daily) obtained durable clinical remission for 16 months, and high-dose osimertinib (160 mg daily) further prolonged the survival of 9 months after leptomeningeal metastases (LM) occurring. This study presented the first case of intractable terminal NSCLC in a patient with EGFR 750_758del, I759S and T751_I759delinsS mutations, who responded positively to osimertinib and achieved a prolonged OS of 52 months, providing a potential therapeutic option for the patients harboring these particular EGFR mutations.
Keywords: non-small cell lung cancer, EGFR, 750_758del, I759S, T751_I759delinsS, osimertinib
Introduction
About 48.0% of Asian patients with lung adenocarcinoma have active epidermal growth factor receptor (EGFR) mutations, which is higher than the population of Europe.1 Generations of EGFR tyrosine kinase inhibitors (TKIs) can significantly improve the prognosis of patients with EGFR-positive NSCLC.2 It has been reported that common EGFR alterations (ie, exon 19 deletion and exon 21 point mutation) approximately account for 85%, which can result in signal activation through modifying the spatial structure of enzyme functional domain.3 However, an increasing number of rare EGFR mutations are revealed by next-generation sequencing (NGS), the clinical significance of which is still unclear and deserves more attention so as to choose the optimal treatment.4 Herein, we report an advanced lung adenocarcinoma patient who harbored three novel EGFR exon 19 deletions (750_758del, I759S and T751_I759delinsS) and presented a durable response to osimertinib after the failure of icotinib and erlotinib treatment.
Case Presentation
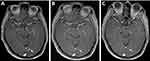
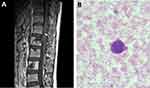
A 55-year-old Asian male with a smoking history was diagnosed with left upper lobe lung cancer by chest computed tomography (CT) in a regular medical examination in November 2015. The postsurgical pathology confirmed the stage IIIA (T3N1M0) lung adenocarcinoma. Adjuvant chemotherapy was applied with 4 cycles of gemcitabine and cisplatin. The disease was stable until July 2016, when positron emission tomography CT (PET-CT) scan revealed multiple enlarged right supraclavicular and mediastinal lymph nodes. Biopsy of the supraclavicular lymph node detected EGFR 750_758del and I759S in exon 19 with tumor protein tp53 gene (TP53) and AKT serine/threonine kinase 1 (AKT1) mutations (Figure 1). Icotinib 125mg three times daily was administrated followed by stereotactic radiotherapy to the mediastinum and supraclavicular (50 Gy in 2 Gy fractions). After 7 months, he experienced disease progression with intrapulmonary metastasis. NGS analysis of plasma samples was performed, and a rare mutation of EGFR T751_I759delinsS located in exon 19 was found (Figure 2), accompanied by TP53 and AKT1 mutation. Four cycles of pemetrexed, carboplatin and bevacizumab were administered, but multiple vertebral metastases were detected via MRI (Figure 3). So, the treatment with erlotinib (150 mg daily) was started in August 2017. However, brain metastasis was confirmed by MRI in January 2018 (Figure 4A). Repeating NGS with plasma detected EGFR T751_I759delinsS along with TP53 and AKT1 mutation. The therapy was switched to osimertinib at 80 mg daily, which decreased the levels of multi-tumor markers (Figure 5), and stabilized the lesions of brain (Figure 4B and C) and pulmonary (Figure 6) for 16 months. In June 2019, he showed symptoms of severe headache, nausea, weakness of the lower extremities and back pain, and leptomeningeal metastases (LM) was confirmed by the CSF cytology and MRI (Figure 7). Further NGS analysis of both CSF and plasma demonstrated EGFR T751_I759delinsS, TP53 and AKT1. Moreover, ataxia telangiectasia related-3 (ATR) mutation was found in the CSF sample. Osimertinib was increased to 160 mg daily, which alleviated neurological symptoms significantly without any notable side effects and maintained the remission for 9 months. The patient passed away in March 2020, with an overall survival (OS) time of 52 months. The patient had no obvious liver, kidney or cardiopulmonary dysfunction while hospitalized. The whole course of treatment was constructed as a timeline (Figure 8).
 |
Figure 1 Next-generation sequencing of tissue sample in July 2016. |
 |
Figure 2 Next-generation sequencing of plasma sample in February 2017. |
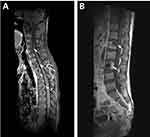 |
Figure 3 Sagittal T1‐weighted (T1w) spinal MRI showed multiple vertebral metastases in August 2017. Notes: (A) C2-7 and T1-6 (white arrows). (B) L1, L3 and S2-4 (white arrows). |
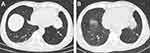 |
Figure 6 Chest CT showed stable lesions in the inferior lobe of left lung. Notes: (A) In February 2017 (white arrow). (B) In January 2019 (white arrow). |
 |
Figure 8 Treatment timeline. |
All of the samples were examined using NGS instrument Illumina HiSeq provided by Nanjing Shihe Jiyin Biotech Inc. A total of 139 lung cancer-related genes were detected, including exons, fusion-related introns, specific microsatellite site, and so on. The summary of genetic mutations detected by NGS and the corresponding treatment were shown in Table 1.
 |
Table 1 Summary of Genetic Mutations Detected by Next-Generation Sequencing and the Corresponding Treatment |
Discussion
The treatment of advanced NSCLC is intractable, especially for the patients with central nervous system (CNS) metastases. EGFR-TKIs are the first selection for the EGFR-mutated patients and have achieved significant survival benefits.5 However, drug resistance is inevitable, which can be classified into primary resistance and acquired resistance, and has largely restricted the clinical efficacy of TKIs. Acquired resistance always occurs in 9–13 months after the start of TKIs treatment, but the comprehension of primary resistance is limited.6
Among the multiple TKIs-resistant mutations, common alterations have been studied well, while the knowledge of uncommon alterations is restricted. For a number of uncommon mutations, first-generation EGFR-TKIs have limited efficacy, but second-generation TKI afatinib has been proved to have better activity against these mutations.4,7 Osimertinib (AZD9291), a third-generation TKI, also can effectively control some of the uncommon mutations,7 especially for CNS metastases thanks to its excellent blood–brain barrier (BBB) permeability and antitumour activity.5,8
In the present case, he harbored EGFR 750_758del and I759S mutations at first, and exhibited a short response to icotinib for 7.0 months. After that, secondary resistance EGFR T751_I759delinsS occurred without co-existing T790M mutation, and pemetrexed, carboplatin, bevacizumab and erlotinib were administered but all failed. Surprisingly, osimertinib at 80 mg daily obtained durable clinical remission for 16 months, indicating that osimertinib has antitumor activity toward acquired EGFR T751_I759delinsS mutation. Moreover, after LM appearing, osimertinib at 160 mg daily further reduced the allele frequency and prolonged the survival of 9 months, owing to that high-dose osimertinib ensured higher local drug level in the CNS and more remarkable curative effect regardless of T790M status.5,8 In view of the poor prognosis of patients with CNS metastases from NSCLC, a duration of 25 months survival was meaningful.
As far as we know, EGFR 750_758del and I759S were reported for the first time, and might be resistant to icotinib. T751_I759delinsS mutation was detected for only once in the previous studies, but unfortunately without the detailed information about the therapy.9 Our study showed that EGFR T751_I759delinsS mutation had better response to osimertinib compared to erlotinib, providing an important reference to the choice of medication. Due to the lack of NGS analysis of resected tumor sample and repeated tests of tissue samples after disease progression in this case, the above conclusions are limited and need more clinical cases and further researches to be determined.
The co-occurring TP53 mutation has been identified as a negative prognostic factor and indicates shorter progression-free survival (PFS) and OS. It could be due to TP53 mutation is involved in primary resistance to first- and second-generation TKIs in EGFR-positive NSCLC patients.10,11 In addition, the clinical outcome of patients treated with osimertinib could be affected by the TP53 gene mutation, exon 8 of which has the worse prognosis than other types.11 TP53 mutation of this case was located in exon 5, and its frequency showed a descending trend during the therapeutic process, which might contribute to his longer survival.
Another accompanying mutation, AKT1 is known to promote the invasion and metastasis of NSCLC cell. Activated AKT1 is overexpressed in NSCLC, and its inhibitors, such as A-674563, could be effective to reduce NSCLC cell survival.12 However, Rao et al found that inhibition of AKT1 is associated with the increased migration and invasion of EGFR+ NSCLC cells by regulating the AKT1- Myristoylated alanine-rich C-kinase substrate (MARCKS)-LAMC2 feedback loop.13 This study showed a conflicting role of AKT1 on metastatic NSCLC processes. Therefore, the accurate efficacy of AKT1 could not be evaluated in this report.
Moreover, ATR mutation was identified by NGS analysis of CSF after a period of osimertinib, which is a regulator involved in necessary DNA damage response. Research has shown that the selective ATR inhibitor, M6620, has preclinical antitumor activity and can increase the susceptibility of NSCLC cells to the chemotherapy of gemcitabine.14 Another study demonstrated that the inhibition of ATR could reverse the drug resistance of NSCLC cells caused by cisplatin or cisplatin plus gemcitabine via obstructing the repair of DNA interstrand crosslinks (ICLs) and double-strand breaks (DSBs).15 In the present case, no ATR inhibitor was implemented due to unknown effects on the patients with rare EGFR mutation.
In conclusion, this study presented the first case of intractable terminal NSCLC patient with EGFR 750_758del, I759S and T751_I759delinsS mutations, who responded well to osimertinib and achieved a prolonged OS, providing a feasible treatment option for such a patient. The underlying mechanisms need to be further investigated.
Statement of Ethics
Written informed consent was obtained from his next-of-kin following his death regarding the publication of the case details and associated images. This is a retrospective case report and institutional approval was not needed.
Acknowledgments
This work was supported by funding from the Medical Key Science and Technology Development Project of Nanjing (No. ZKX18014), the Cadre Health Care Project of Jiangsu Province (No. BJ18006), the Cadre Health Care Project of Jiangsu Province (No. BJ19001) and the Cancer Research Funding of CSCO-Hausen (No. Y-HS2019-5). We thank Nanjing Shihe Jiyin Biotech Inc for supplying the data.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Shi Y, Li J, Zhang S, et al. Molecular epidemiology of EGFR mutations in asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology - Mainland China subset analysis of the PIONEER study. PLoS One. 2015;10(11):e0143515. doi:10.1371/journal.pone.0143515
2. Greenhalgh J, Dwan K, Boland A, et al. First-line treatment of advanced epidermal growth factor receptor (EGFR) mutation positive non-squamous non-small cell lung cancer. Cochrane Database Syst Rev. 2016;5:Cd010383. doi:10.1002/14651858.CD010383.pub2
3. Pao W, Chmielecki J. Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer. 2010;10:760–774. doi:10.1038/nrc2947
4. Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015;16:830–838. doi:10.1016/S1470-2045(15)00026-1
5. Cheng H, Perez-Soler R. Leptomeningeal metastases in non-small cell lung cancer. Lancet Oncol. 2018;19:43–55. doi:10.1016/S14702045(17)30689-7
6. Shien K, Yamamoto H, Soh J, Miyoshi S, Toyooka S. Drug resistance to EGFR tyrosine kinase inhibitors for non-small cell lung cancer. Acta Med Okayama. 2014;68:191–200. doi:10.18926/AMO/52785
7. Kohsaka S, Nagano M, Ueno T, et al. A method of high-throughput functional evaluation of EGFR gene variants of unknown significance in cancer. Sci Transl Med. 2017;9:eaan6566. doi:10.1126/scitranslmed.aan6566
8. Yang JCH, Kim SW, Kim DW, et al. Osimertinib in patients with epidermal growth factor receptor mutation-positive non-small-cell lung cancer and leptomeningeal metastases: the BLOOM study. J Clin Oncol. 2020;38(6):538–547. doi:10.1200/JCO.19.00457
9. Nakajima E, Sugita M, Furukawa K, et al. Frequency and significance of epidermal growth factor receptor mutations detected by PCR methods in patients with non-small cell lung cancer. Oncol Lett. 2019;17(6):5125–5131. doi:10.3892/ol.2019.10157
10. Qin K, Hou H, Liang Y, et al. Prognostic value of TP53 concurrent mutations for EGFR- TKIs and ALK-TKIs based targeted therapy in advanced non-small cell lung cancer: a meta-analysis. BMC Cancer. 2020;20(1):328. doi:10.1186/s12885-020-06805-5
11. Canale M, Petracci E, Delmonte A, et al. Concomitant TP53 mutation confers worse prognosis in EGFR-mutated non-small cell lung cancer patients treated with TKIs. J Clin Med. 2020;9(4):1047. doi:10.3390/jcm9041047
12. Chorner PM, Moorehead RA. A-674563, a putative AKT1 inhibitor that also suppresses CDK2 activity, inhibits human NSCLC cell growth more effectively than the pan-AKT inhibitor, MK-2206. PLoS One. 2018;13(2):e0193344. doi:10.1371/journal.pone.0193344
13. Rao G, Pierobon M, Kim IK, et al. Inhibition of AKT1 signaling promotes invasion and metastasis of non-small cell lung cancer cells with K-RAS or EGFR mutations. Sci Rep. 2017;7(1):7066. doi:10.1038/s41598-017-06128-9
14. Plummer R, Cook N, Arkenau T, et al. Phase I dose expansion data for M6620 (formerly VX-970), a first-in-class ATR inhibitor, combined with gemcitabine (Gem) in patients (pts) with advanced non-small cell lung cancer (NSCLC). Ann Oncol. 2018;29 Suppl 8:viii519. doi:10.1093/annonc/mdy292.059
15. Li XQ, Ren J, Chen P, et al. Co-inhibition of Pol η and ATR sensitizes cisplatin-resistant non-small cell lung cancer cells to cisplatin by impeding DNA damage repair. Acta Pharmacol Sin. 2018;39(8):1359. doi:10.1038/aps.2017.187
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.