Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
The Screening Value Of ESS, SACS, BQ, And SBQ On Obstructive Sleep Apnea In Patients With Chronic Obstructive Pulmonary Disease
Authors Xiong M, Hu W, Dong M, Wang M , Chen J, Xiong H, Zhong M, Jiang Y, Liu D, Hu K
Received 18 July 2019
Accepted for publication 9 October 2019
Published 13 November 2019 Volume 2019:14 Pages 2497—2505
DOI https://doi.org/10.2147/COPD.S223354
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Chunxue Bai
Mengqing Xiong,1 Weihua Hu,1 Minglin Dong,1 Meifang Wang,2 Junwen Chen,3 Hanzhong Xiong,4 Minhua Zhong,5 Yan Jiang,6 Dan Liu,6 Ke Hu1
1Department of Respiratory and Critical Care Medicine, Renmin Hospital of Wuhan University, Wuhan 430060, People’s Republic of China; 2Department of Respiratory and Critical Care Medicine, Taihe Hospital, Hubei University of Medicine, Shiyan 442000, People’s Republic of China; 3Department of Respiratory and Critical Care Medicine, Xiangyang Hospital, Hubei University of Medicine, Xiangyang 441000, People’s Republic of China; 4Department of Respiratory and Critical Care Medicine, Ezhou Central Hospital, Ezhou 436000, People’s Republic of China; 5Department of Respiratory and Critical Care Medicine, Xiaogan Central Hospital, Wuhan University of Technology, Xiaogan 432000, People’s Republic of China; 6Division of Respiratory Disease, The Fifth Hospital of Wuhan City, Wuhan 430050, People’s Republic of China
Correspondence: Ke Hu
Department of Respiratory and Critical Care Medicine, Renmin Hospital of Wuhan University, Zhangzhidong Road, No. 99, Wuhan 430060, People’s Republic of China
Tel +86 189 7103 5988
Fax +86 27 8804 2292
Email [email protected]
Objective: To compare the performance of Epworth sleepiness scale (ESS), sleep apnea clinical score (SACS), Berlin questionnaire (BQ), and STOP-BANG questionnaire (SBQ) in screening for obstructive sleep apnea (OSA) in patients with chronic obstructive pulmonary disease (COPD).
Methods: A total of 431 patients were analyzed. All subjects completed lung function test, ESS, SACS, BQ, and SBQ survey and overnight polysomnography (PSG). According to lung function and PSG results, participants were divided into COPD with OSA group (OVS, AHI ≥5) and without OSA group (AHI <5). The value of ESS, SACS, BQ, and SBQ was compared in predicting OSA in patients with COPD by receiver-operating characteristic (ROC) curve statistics.
Results: Of the 431 subjects, there were 96 cases in COPD without OSA group, and 335 cases in OVS group including 183, 96, and 56 cases of COPD combined with mild, moderate or severe OSA. In predicting different degrees of severity of OSA in patients with COPD, the value of ESS was poor with all the values of area under the curve (AUC) < 0.7. SACS and BQ had moderate predictive value in screening for severe OSA with the value of AUC of 0.750, 0.735 respectively. However, the SBQ performed best in predicting various degrees of OSA. For screening mild OSA (AHI ≥5), the ROC statistics recommended the cut-off score of SBQ >2 was considered high risk of OSA; the sensitivity, specificity, and AUC were 92.8%, 40.6%, and 0.723 respectively, the odds ratio (OR) was 2.161. When AHI ≥15, AUC for SBQ was 0.737. In predicting severe OSA (AHI ≥30), the ROC curve showed cut-off point, sensitivity, specificity, and AUC for SBQ was >4, 66.1%, 82.1%, and 0.824 respectively; the positive and negative likelihood ratio was 3.70, 0.41 separately, the OR was 2.977.
Conclusion: SBQ performed better than ESS, SACS, and BQ in predicting OSA in patients with COPD.
Keywords: Berlin questionnaire, STOP-BANG questionnaire
Introduction
Chronic obstructive pulmonary disease (COPD) is a common lung disease and an important cause of impaired health-related quality of life, disability, and death worldwide.1 Similarly, the effects of obstructive sleep apnea (OSA) on health are increasingly being recognized.2 OSA has been listed as one of the major comorbidities of COPD1 and the term overlap syndrome (OVS) is used to describe the coexistence of the two conditions.3 Overlapping of the two conditions makes the pathophysiological changes more complex and causes more serious hypoxia and hypercapnia, which may promote cardio-cerebrovascular events, even pulmonary hypertension.4,5 Ignoring treatment for OSA in patients with OVS may contribute to increasing number of acute exacerbations, hospitalization, and mortality rate, as well as the financial burden of COPD patients.1,5,6 Therefore, identification of co-existing OSA in patients with COPD is of great significance.2
Diagnosing COPD is relatively simple and cheap,1 however, as the gold standard for diagnosis of OSA, in-laboratory polysomnography (PSG) is high-cost and time-consuming,3,7 more often than not, it is difficult for some patients with moderate to severe COPD to perform a full night PSG. Given that, some simple and efficient clinical screening tools were developed to prioritize the suitable high-risk cases for further overnight PSG study. Although several clinical questionnaires have demonstrated clinical utility for OSA evaluation in various populations,8–13 there are limited data regarding these tests in patients with COPD. In this study, we aimed to compare the performance of the Epworth sleepiness scale (ESS), sleep apnea clinical score (SACS), Berlin questionnaire (BQ), and STOP- BANG questionnaire (SBQ) in predicting OSA in patients with COPD (Supplementary materials show ESS, SACS, BQ, and SBQ questionnaires).
Methods
Design
This was one part of a prospective, observational project entitled “Study on the diagnosis and treatment of obstructive sleep apnea in patients with chronic obstructive pulmonary disease”, which was supported by the National Key Research and Development Program of China (project number: 2016YFC1304403). Primary and secondary study endpoints were the number of acute exacerbations and all-cause mortality in COPD patients with or without OSA respectively. The present study analyzed baseline data for eligible consecutive participants in order to compare the value of the ESS, SACS, BQ, and SBQ in screening OSA in patients with COPD.
Participants
From Dec 2016 to Dec 2018, a total of 476 consecutive patients with suspected COPD were enrolled as the study candidates, those who met the inclusion criteria and exclusion criteria were enrolled as study participants. The inclusion criteria were: subjects aged at least 40 years old with a diagnosis of COPD conforming to GOLD guideline.14 Candidates were excluded based on the following: 1) those were less than 40 years old or pregnant; 2) patients with evidence of bronchial asthma, bronchiectasis, pulmonary fibrosis, intratracheal neoplasms, destructive sequelae of tuberculosis, etc; 3) patients with combined other diseases affecting survival, such as neoplastic diseases, renal insufficiency, or acute myocardial infarction; 4) those with history of stroke, heart failure, neuromuscular, cognitive impairment or other mental and psychological diseases that would prevent completion of pulmonary function test, questionnaire or PSG; and 5) those who had other sleep disorders such as obesity hypoventilation syndrome.
The study flow diagram was shown in Figure 1. 45 cases dropped out of the study, of which 22 were unable to perform lung function test because of severe condition, 19 did not tolerate or refused to perform PSG (6 did not complete lung function test also), and 4 for had incomplete PSG data. Finally, a total of 431 stable subjects were included. The study procedure was approved by the Medical Ethics Committee of Renmin Hospital of Wuhan University and was registered in ClinicalTrials.gov (Clinical Trials ID: NCT 03182309). This trial was conducted in accordance with the Declaration of Helsinki and written informed consent was obtained from all the study participants.
 |
Figure 1 Study flow diagram.Abbreviation: COPD, chronic obstructive pulmonary disease. |
Study Protocol
Patients were initially eligible to take part in the study if they had suspected COPD (having history of exposure to risk factors and characteristic symptoms of COPD). At the first interview, each potential subject underwent a spirometry measurement and completed a comprehensive questionnaire involving demographic and anthropometric data. If the patients met the diagnostic criteria for lung function, a survey of four questionnaires (ESS, SACS, BQ, SBQ) was conducted and an overnight PSG examination was scheduled for each patient. Subjects with obstructive events and apnea-hypopnea index (AHI) ≥5 were considered to have OSA.
Lung Function Test
The completion of lung function test was guided by professional technicians by using a spirometer (MasterScreenBody, Jaeger, Germany). The diagnosis and severity of COPD were in accordance with GOLD guideline.14 Those with a ratio of FEV1/FVC of post-bronchodilator <70% was diagnosed as having COPD. The stratification of airflow limitation was based on post-bronchodilator FEV1%.
Questionnaires
Epworth sleepiness scale (ESS) is used to measure drowsiness of subjects in different situations during the day.15 In China, a subject with a score of ≥9 is considered at high risk of excessive daytime sleepiness.16 Sleep apnea clinical score (SACS) is used to assess sleep apnea,17 those with a score ≥15 are assumed to be at high risk of OSA. Berlin questionnaire (BQ) comprises three categories including 10 questions, high risk of OSA is defined as ≥ two positive results of the three categories.18 STOP-BANG questionnaire (SBQ) is a tool involving 4 dichotomous items and 4 clinical parameter items, a score≥ 3 is regarded as having a moderate to severe risk of OSA.19
PSG And OSA Diagnostic Criteria
All subjects with confirmed COPD underwent assessment of sleep events with a multichannel sleep diagnostic system (SOMNOscreen Plus Tele PSG, SOMNOmedics GmbH, Germany) in the sleep laboratory for no less than 7 hrs monitoring at night. All tracings were manually scored according to the American Academy of Sleep Medicine criteria.20 Subjects who experienced AHI ≥5 events/hour during sleep were considered to have OSA. Depending on the AHI, OSA severity is divided into mild (5–14.9), moderate (15–29.9), or severe (≥30).20
Statistical Analysis
Descriptive analysis was performed to compare COPD without OSA and OVS groups. Measurement data were presented as mean ± standard deviation (SD), and enumeration data were expressed by frequencies or percentages. For continuous variables, comparisons between OVS group and no OSA group were made using independent t-tests for normal distribution variables and the Mann–Whitney U-test for non-normal variables. The chi-squared test or Fisher’s exact test was used for categorical variables. All statistical analyses were performed using IBM SPSS Statistics software package (version 23). Receiver-operating characteristic (ROC) curves were constructed to assess the performance of ESS, SACS, BQ, and SBQ regarding their value in predicting OSA in patients with COPD. The optimal cut-off values of the four questionnaires in each OSA severity group were determined. Sensitivities, specificities, positive and negative likelihood ratios, and odds ratios were calculated for the optimal cut-off scores at various levels of severity of OSA. The response was dichotomized using all OSA (AHI ≥5), moderate to severe OSA (AHI ≥15), and severe OSA (AHI ≥30) as the thresholds. Differences in area under the curve (AUC) were calculated using the method of DeLong. The analyses were performed using MedCalc software (version 11.4.2.0). All tests were two sided. Statistical significance was considered when P-value was less than 0.05.
Results
Demographic And Anthropometric Data
Of the 431 patients with confirmed COPD, 388 (90.0%) were male and 43 (10.0%) were female, with an average age of 67.4 (±8.9) years. There were 96 (22.3%) cases of COPD alone and 335 (77.7%) cases complicated with OSA. Compared with COPD alone group, the patients with OVS had higher age, body mass index (BMI), neck circumference, smoking index, FEV1, FEV1%, AHI, and percentage of total sleep time with pulse oxygen saturation of < 90% (TS90%); however, the lowest pulse oxygen saturation (LSaO2) was lower (Table 1).
 |
Table 1 Characteristics Of COPD Cohort And Comparison With OVS |
Predictive Value Of ESS, SACS, BQ, And SBQ In Detecting OSA
In the OVS group, there were 183 (54.6%), 96 (28.7%), and 56 (16.7%) cases combined with mild, moderate or severe OSA, respectively. Comparisons of the related parameters and scores of the four questionnaires were shown in Table 2. In the eight questions of SBQ, the rate of habitual snoring was > 60% and the proportion of patients > 50 years or male gender was more than 90%, and there was no significant difference among the three groups (P>0.05). However, significant differences were found in the ratio of daytime fatigue, witnessed apnea, hypertension, and neck circumference > 40cm (P<0.05). In COPD with severe OSA group, the rates of witnessed apnea and hypertension were 66.1% and 50% respectively; moreover, no subjects had BMI of > 35kg/m2. As for BQ, the rate of positive response to question of category 1 or category 2 was 63.9% and 18.8% respectively in OVS group. For COPD with severe OSA group, the proportion of positive response to question of category 3 was 50%.
 |
Table 2 Descriptive Characteristics Of The Study Population For ESS, SACS, BQ And SBQ |
On the basis of recommendations,16–19 the cutoff value of four questionnaires for predicting high risk of OSA was used to calculate the cases of COPD complicated with OSA. The proportion of SACS score ≥ 15 was less than 30% in COPD patients with different degrees of OSA. The percentage of ESS score ≥ 9 points and BQ ≥ 2 points in COPD complicated with OSA were similar but both were low (36.1% and 35.7%, respectively), and slightly higher in severe OSA group (57.1% and 64.3%, respectively). As to SBQ, the ratio of score ≥ 3 in COPD combined with mild, moderate or severe OSA was 89.6%, 94.8%, and 100%, respectively.
Figures 2–4 showed the ROC curves of the four questionnaires in screening different degrees of severity of OSA. The optimal cut-off points, sensitivities, specificities, positive or negative likelihood ratios, and AUCs were compared in Table 3. In screening all OSA (AHI ≥ 5 events/h), ESS, SACS or BQ were poor with AUC < 0.7, while SBQ had a delightful effect. The optimal cut-off point for SBQ was > 2, with sensitivity of 92.8% and specificity of 40.6%, and the AUC and odds ratio were 0.723, 2.163 respectively. At threshold of AHI ≥ 15 events/h, the AUC of SBQ was 0.737, whereas that for ESS, SACS or BQ were all less than 0.7. To detect severe OSA (AHI ≥ 30 events/h), the ROC curve recommended optimal cut-off point of SBQ was > 4, with good sensitivity, specificity, and AUC of 66.1%, 82.1%, 0.824 respectively, and kept the highest positive and lowest negative likelihood ratio (3.70, 0.41) and high odds ratio of 2.977. SACS and BQ had decent predictive value with AUC of 0.750 and 0.735, respectively.
 |
Table 3 Predictive Parameters Of ESS, SACS, BQ, And SBQ |
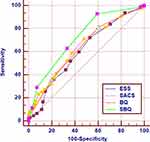 |
Figure 2 ROC curves for ESS, SACS, BQ, and SBQ in predicting mild-moderate-severe OSA (AHI ≥5 events/h) in patients with COPD. |
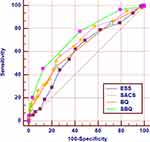 |
Figure 3 ROC curves for ESS, SACS, BQ, and SBQ in predicting moderate-to-severe OSA (AHI ≥15 events/h) in patients with COPD. |
 |
Figure 4 ROC curves for ESS, SACS, BQ, and SBQ in detecting severe OSA (AHI ≥30 events/h) in patients with COPD. |
Pairwise Comparison Of ROC Curves Of ESS, SACS, BQ, And SBQ
The results of AUC pairwise comparison among the questionnaires were shown in Table 4. SBQ held bigger AUC than SACS, BQ, and ESS in predicting various degrees of severity of OSA in patients with COPD (P<0.05). The AUC of SACS was relatively larger than that of BQ (P = 0.067) and larger than ESS (P = 0.049) in predicting moderate - severe OSA. In screening different degrees of severity of OSA, the AUC of BQ was bigger than ESS but there was no significant difference (P>0.05).
 |
Table 4 Pairwise Comparison Of ROC Curves Of ESS, SACS, BQ, And SBQ |
Discussion
Diagnosing OSA in COPD is clinically relevant because untreated OSA has been associated with poor health outcomes,3 however, it remains underdiagnosed and undertreated. Easy diagnosis of OSA in COPD patients is a challenge for clinicians. Although screening tools have been widely used to detect OSA, the findings about their diagnostic accuracy were controversial.8–13 A meta-analysis11 of 26 studies suggested that the BQ had the highest diagnostic odds ratio among different screening tools. Another bivariate meta-analysis of 108 studies including a total of 47,989 subjects recommended the SBQ as a screening tool for OSA because the SBQ is a more accurate tool for detecting mild, moderate, and severe OSA when compared with the BQ, STOP, and ESS.21 In a study on comparing nine screening questionnaires in prediction of OSA,22 the SACS and the SBQ were found to have the best positive and negative likelihood ratios, respectively. However, predictors of OSA among patients with different degrees of severity of COPD may be distinct from OSA in the general population. It is less clear whether the screening tools have parallel applicability in COPD patients with OSA. Although a study suggested that SACS had more screening value than BQ and ESS for OSA in patients with COPD, its shortcomings lied in small sample size and no SBQ was included; furthermore, only 24 out of 91 COPD patients underwent PSG.23 In the present study, we evaluated the performance characteristics of ESS, SACS, BQ, and SBQ as screening tools for the likelihood of OSA in patients with COPD. We found that SBQ performed better than ESS, SACS, and BQ in predicting various degrees of severity of OSA.
Our results showed that the value of ESS in predicting OSA in patients with COPD was unsatisfactory, which is in line with results of other studies.21,24 In screening severe OSA, the value of AUC was only 0.676 and the proportion of ESS ≥ 9 points was only 57.1%. Relatively, the effect of SACS was decent. In predicting the likelihood of severe OSA in COPD patients, the ROC curve indicated that the sensitivity, specificity, AUC, and positive likelihood ratio of SACS was 69.6%, 71.7%, 0.750, and 2.46, respectively, similar to the results of Grover et al's study about primary care populations.25 Flemons et al suggested that a score of SACS ≥ 15 had a likelihood ratio of 5.17 in moderate and severe sleep apnea (AHI≥10) in the general population;17 while Faria et al proposed that SACS had more screening value than BQ and ESS for OSA in patients with COPD.23 However, in this study, the percentage of SACS score ≥ 15 in COPD patients combined with severe OSA group was only 28.6% and the optimal cut-off point of SACS was small (>7). Therefore, the practical application of SACS in COPD patients was limited, the low SACS scale may be attributed to the small neck circumference and relatively low incidence of hypertension. In this OVS group, the ratio of neck circumference > 40 cm was only 17.6% and the incidence of hypertension was 37.9%. Soler et al found that traditional OSA predictors (age, gender, neck circumference, and ESS) did not perform well in patients with more advanced COPD.24 Our results also showed that the value of BQ in screening for OSA in patients with COPD was similar to that of SACS. For severe OSA, its optimal cut-off point, sensitivity, specificity, and AUC was >1, 64.3%, 72.5%, and 0.735, respectively.
The application of SBQ to screen for OSA has been recommended for its better predictive effect in different degrees of severity of OSA and clinical application compared to other questionnaires,26–29 however, there has been no comparison of SBQ with other tools in COPD patients. We found that the effect of SBQ was better than that of SACS, BQ, and ESS in screening for mild, moderate, and severe OSA. For predicting all mild-moderate-severe OSA, the sensitivity, AUC of SBQ was 92.8%, 0.723 respectively. At the threshold of AHI ≥ 15, SBQ had the AUC of 0.737. In predicting severe OSA (AHI ≥30), the ROC curve of SBQ indicated that the specificity and AUC was 82.1%, 0.824 respectively, along with the positive likelihood ratio of 3.70, negative likelihood ratio of 0.41, and OR of 2.977, which suggested that SBQ had good performance in screening different grades of OSA in COPD patients.
In this cohort of COPD patients, the optimal cut-off point of SBQ for predicting mild OSA (AHI ≥5) was >2 with a sensitivity of 92.8%; for moderate OSA (AHI ≥15), it was >3; for severe OSA (AHI ≥30), it was >4 with a specificity of 82.1%, and the proportion of patients with SBQ score ≥ 3 was 100%. It was reported that SBQ score ≥ 3 had a sensitivity of 92.9% and 100% for detecting moderate to severe OSA and severe OSA respectively in preoperative patients.19 In obese and morbidly obese surgical patients, SBQ score of 4 had the sensitivity of 88% for identifying severe OSA.30 A review by Chung et al summarized the application of SBQ in various populations and concluded that patients with SBQ score of 5–8 can be stratified as high risk for moderate to severe OSA.26 Because of the heterogeneity of populations and methods, SBQ showed various effects. The high sensitivity in screening all OSA may be helpful to avoid missing cases that may lead to advanced health consequence, which may be more useful for detecting OSA in patients with COPD. We might conclude that in this study of COPD patients, a score of SBQ ≥3 could detect OSA and a score of ≥4 could identify high risk for severe OSA. This study proposed SBQ as a preferable epidemiological tool and clinical screening test in patients with COPD.
The advantage of this study lies in the fact that the accuracy of the four questionnaires was validated by an overnight PSG – the gold standard for diagnosing OSA. Also, some limitations should be acknowledged. First, the prevalence of OVS was 77.7%, which was higher than some reports3,31 and might be associated with the different population and different diagnostic methods.32 Second, the enrolled subjects were from hospitals rather than the community population and therefore, there was likely a population selective bias. Finally, symptoms of patients affected by COPD itself, for example, patients with severe COPD would suffer from depression or anxiety, insomnia, poor sleep quality or daytime sleepiness.33,34 Therefore, more research is needed to validate the utility of SBQ in various COPD patients.
In conclusion, the present study showed that SBQ performed better in detecting different grades of OSA in patients with COPD when compared with ESS, SACS or BQ. A score of SBQ ≥3 could detect OSA and a score of ≥4 could identify high risk for severe OSA, which indicated that SBQ may be used as a preferential tool by clinicians to screen for OSA in patients with COPD.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Montes de Oca M, Pérez-Padilla R. Global Initiative For Chronic Obstructive Lung Disease (GOLD)-2017: Thealat perspective. Arch Bronconeumol. 2017;53(3):87–88. doi:10.1016/j.arbres.2017.01.002
2. Shawon MS, Perret JL, Senaratna CV, et al. Current evidence on prevalence and clinical outcomes of co-morbid obstructive sleep apnea and chronic obstructive pulmonary disease: a systematic review. Sleep Med Rev. 2017;32:58–68. doi:10.1016/j.smrv.2016.02.007
3. Malhotra A, Schwartz AR, Schneider H; on behalf of the ATS Assembly on Sleep and Respiratory Neurobiology, et al. Research priorities in pathophysiology for sleep-disordered breathing in patients with chronic obstructive pulmonary disease. An Official American Thoracic Society Research Statement. Am J Respir Crit Care Med. 2018;197(3):289–299. doi:10.1164/rccm.201712-2510ST
4. Soler X, Gaio E, Powell FL, et al. High Prevalence of obstructive sleep apnea in patients with moderate to severe chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2015;12(8):1219–1225. doi:10.1513/AnnalsATS.201506-379LE
5. Marin JM, Soriano JB, Carrizo SJ, et al. Outcomes in patients with chronic obstructive pulmonary disease and obstructive sleep apnea: the overlap syndrome. Am J Respir Crit Care Med. 2010;182(3):325–331. doi:10.1164/rccm.200912-1869OC
6. Machado MC, Vollmer WM, Togeiro SM, et al. CPAP and survival in moderate-to-severe obstructive sleep apnoea syndrome and hypoxaemic COPD. Eur Respir J. 2010;35(1):132–137. doi:10.1183/09031936.00192008
7. Epstein LJ, Kristo D, Strollo PJ, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263–276.
8. Luo J, Huang R, Zhong X, Xiao Y, Zhou J. STOP-Bang questionnaire is superior to Epworth sleepiness scales, Berlin questionnaire, and STOP questionnaire in screening obstructive sleep apnea hypopnea syndrome patients. Chin Med J. 2014;127:3065e70.
9. El-Sayed IH. Comparison of four sleep questionnaires for screening obstructive sleep apnea. Egypt J Chest Dis Tuberc. 2012;61:433e41. doi:10.1016/j.ejcdt.2012.07.003
10. Pataka A, Daskalopoulou E, Kalamaras G, Fekete Passa K, Argyropoulou P. Evaluation of five different questionnaires for assessing sleep apnea syndrome in a sleep clinic. Sleep Med. 2014;15:776e81. doi:10.1016/j.sleep.2014.03.012
11. Ramachandran SK, Josephs LA. A meta-analysis of clinical screening tests for obstructive sleep apnea. Anesthesiology. 2009;110:928e39. doi:10.1097/ALN.0b013e31819c47b6
12. Abrishami A, Khajehdehi A, Chung F. A systematic review of screening questionnaires for obstructive sleep apnea. Can J Anaesth. 2010;57:423e38. doi:10.1007/s12630-010-9280-x
13. Nagappa M, Liao P, Wong J, et al. Validation of the STOP-Bang questionnaire as a screening tool for obstructive sleep apnea among different populations: a systematic review and meta-analysis. PLoS One. 2015;10(12):e0143697. doi:10.1371/journal.pone.0143697
14. Vogelmeier CF, Criner GJ, Martínez FJ, et al. Global strategy for the diagnosis, management and prevention of chronic obstructive lung disease 2017 report: GOLD executive summary. Respirology. 2017;22(3):575–601.
15. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–545. doi:10.1093/sleep/14.6.540
16. He QY, Chen BY. The interpretation of the guideline of obstructive sleep apnea hypopnea syndrome. Zhonghua Jie He He Hu Xi Za Zhi. 2012;35(1):7–8. Chinese.
17. Flemons WW, Whitelaw WA, Brant R, et al. Likelihood ratios for a sleep apnea clinical prediction rule. Am J Respir Crit Care Med. 1994;150(5 Pt 1):1279–1285. doi:10.1164/ajrccm.150.5.7952553
18. Netzer NC, Stoohs RA, Netzer CM, et al. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med. 1999;131(7):485–491. doi:10.7326/0003-4819-131-7-199910050-00002
19. Chung F, Yegneswaran B, Liao P, et al. STOP questionnaire: a tool to screen patients for obstructive sleep apnea. Anesthesiology. 2008;108(5):812–821. doi:10.1097/ALN.0b013e31816d83e4
20. Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. Deliberations of the sleep apnea definitions task force of the American Academy of Sleep Medicine. J Clin Sleep Med. 2012;8(5):597–619. doi:10.5664/jcsm.2172
21. Chiu HY, Chen PY, Chuang LP, et al. Diagnostic accuracy of the Berlin questionnaire, STOP-BANG, STOP, and Epworth sleepiness scale in detecting obstructive sleep apnea: a bivariate meta-analysis. Sleep Med Rev. 2017;36:57–70. doi:10.1016/j.smrv.2016.10.004
22. Prasad KT, Sehgal IS, Agarwal R, Nath Aggarwal A, Behera D, Dhooria S. Assessing the likelihood of obstructive sleep apnea: a comparison of nine screeningquestionnaires. SleepBreath. 2017;21(4):909–917.
23. Faria AC, da Costa CH, Rufino R. Sleep Apnea Clinical Score, Berlin questionnaire, or Epworth sleepiness scale: which is the best obstructive sleep apnea predictor in patients with COPD? Int J Gen Med. 2015;8:275–281. doi:10.2147/IJGM.S86479
24. Soler X, Liao S, Marin JM, et al. Age, gender, neck circumference, and Epworth sleepiness scale do not predict obstructive sleep apnea (OSA) in moderate to severe chronic obstructive pulmonary disease (COPD): the challenge to predict OSA in advanced COPD. PLoS One. 2017;12(5):e177289. doi:10.1371/journal.pone.0177289
25. Grover M, Mookadam M, Chang YH, et al. Validating the diagnostic accuracy of the sleep apnea clinical score for use in primary care populations. Mayo Clin Proc. 2016;91(4):469–476. doi:10.1016/j.mayocp.2016.01.022
26. Chung F, Abdullah HR, Liao P. STOP-Bang Questionnaire: a practical approach to screen for obstructive sleep apnea. Chest. 2016;149(3):631–638. doi:10.1378/chest.15-0903
27. Chung F, Yang Y, Brown R, et al. Alternative scoring models of STOP-Bang questionnaire improve specificity to detect undiagnosed obstructive sleep apnea. J Clin Sleep Med. 2014;10(9):951–958. doi:10.5664/jcsm.4022
28. Silva GE, Vana KD, Goodwin JL, et al. Identification of patients with sleep disordered breathing: comparing the four-variable screening tool, STOP, STOP-Bang, and Epworth Sleepiness Scales. J Clin Sleep Med. 2011;7(5):467–472. doi:10.5664/JCSM.1308
29. Ong TH, Raudha S, Fook-Chong S, et al. Simplifying STOP-BANG: use of a simple questionnaire to screen for OSA in an Asian population. Sleep Breath. 2010;14(4):371–376. doi:10.1007/s11325-010-0350-7
30. Chung F, Yang Y, Liao P. Predictive performance of the STOP-Bang score for identifying obstructive sleep apnea in obese patients. Obes Surg. 2013;23(12):2050–2057. doi:10.1007/s11695-013-1006-z
31. McNicholas WT. COPD-OSA Overlap syndrome: evolvingevidence regarding epidemiology, clinical consequences, and management. Chest. 2017;152(6):1318–1326. doi:10.1016/j.chest.2017.04.160
32. Senaratna CV, Perret JL, Matheson MC, et al. Validity of the Berlin questionnaire in detecting obstructive sleep apnea: a systematic review and meta-analysis. Sleep Med Rev. 2017;36:116–124. doi:10.1016/j.smrv.2017.04.001
33. McNicholas WT. Impact of sleep in COPD. Chest. 2000;117(2Suppl):48S–53S. doi:10.1378/chest.117.2_suppl.48S
34. Budhiraja R, Siddiqi TA, Quan SF. Sleep disorders in chronic obstructive pulmonary disease: etiology, impact, and management. J Clin Sleep Med. 2015;11(3):259–270. doi:10.5664/jcsm.4540
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
