Back to Journals » OncoTargets and Therapy » Volume 13
The Role of DLLs in Cancer: A Novel Therapeutic Target
Authors Xiu MX, Liu YM, Kuang B
Received 5 January 2020
Accepted for publication 6 April 2020
Published 7 May 2020 Volume 2020:13 Pages 3881—3901
DOI https://doi.org/10.2147/OTT.S244860
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
Meng-Xi Xiu, Yuan-Meng Liu, Bo-hai Kuang
Medical School of Nanchang University, Nanchang, People’s Republic of China
Correspondence: Bo-hai Kuang
Medical School of Nanchang University, Nanchang, People’s Republic of China
Tel +86 (0791)-86360580
Email [email protected]
Abstract: Delta-like ligands (DLLs) control Notch signaling. DLL1, DLL3 and DLL4 are frequently deregulated in cancer and influence tumor growth, the tumor vasculature and tumor immunity, which play different roles in cancer progression. DLLs have attracted intense research interest as anti-cancer therapeutics. In this review, we discuss the role of DLLs in cancer and summarize the emerging DLL-relevant targeting methods to aid future studies.
Keywords: delta-like ligands, notch signaling, cancer, therapy
Introduction
Notch is an evolutionary-conserved signaling system that regulates cell fate through local cell–cell interactions. In mammals, four Notch receptors (Notch1–4) and five Notch ligands (Delta-like 1, 3, 4 and Jagged 1–2) are expressed (Figure 1A). Notch receptors are cleaved (S1 cleavage) in the Golgi apparatus and traffic to the cell surface as a transmembrane heterodimeric protein. When interacting with Notch ligands in adjacent cells, the Notch receptor undergoes proteolytic cleavage by a-disintegrin-and-metalloproteinase 10/17 (ADAM10/17) (S2 cleavage) and the γ-secretase complex (S3 cleavage), causing the release of the Notch intracellular domain (NICD). NICD then enters the nucleus and binds to the DNA-binding protein CSL (CBF-1 (RBPJ)/suppressor of hairless/Lag1), which recruits mastermind-like protein (MAML) to activate the transcription of Notch target genes including Hes and Hey families.1,2
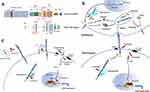 |
Figure 1 (A) Structure of DLL and the Notch receptor. The full-length Notch receptor consists of epidermal growth factor (EGF) repeats, a negative regulatory region (NRR), a transmembrane domain (TM), an RBPJ-association module (RAM) domain, seven ankyrin (ANK) repeats, two nuclear localization signals (NLS), a trans-activation domain (TAD) and a C-terminal proline, glutamate, serine, and threonine-rich (PEST) domain. The full-length DLL consists of an N-terminal domain of Notch ligand (MNNL), a Delta/Serrate/LAG-2 (DSL) domain, 6–8 EGF repeats, a TM and a post-synaptic density protein ligand (PDZ) domain (excluding DLL3) . Adapted with permission form Xiu, M. X. & Liu, Y. M. The role of oncogenic Notch2 signaling in cancer: a novel therapeutic target. Am J Cancer Res. 2019;9(5):837–854.3 The core interactions between DLL and the Notch receptor are DSL-EGF11 and MNNL-EGF12. The full-length DLL can undergo juxtamembrane and intramembrane cleavage, releasing DICD. The proteolytic cleavage sites of DLL and the Notch receptor are marked. (B) DLLs activate Notch signaling. (C) DLLs inhibit Notch signaling. ① DLL3 inhibits Notch activation in the Golgi apparatus. ② Extracellular vesicle-containing DLL4 binds to Notch receptors and inhibits Notch signaling. ③ DLLs bind to Notch receptors at the cell surface and inhibit Notch signaling “in cis”. ④ DICD prevents the formation of NICD-CSL-MAML complex. |
Three members of the Delta family (DLL1, DLL3, DLL4) are encoded on chromosome 6q27 (6: 170,282,200–170,291,078), 19q13.2 (19: 39,498,947–39,508,469) and 15q15.1 (15: 40,929,340–40,939,073), respectively. Prior to trafficking to the cell surface, DLLs undergo O-fucosylation mediated by Lunatic Fringe (Lfng) in the Golgi apparatus, which enhances4,5 or attenuates6,7 DLL-Notch signaling. Membrane DLLs bind to the EGF repeats of Notch receptors, subsequently triggering Notch signaling “in trans” through endocytic “pulling”. After endocytosis, internalized DLLs can undergo proteasomal/lysosome degradation or can be recycled back to the plasma membrane.2,8 The intracellular endocytosis/recycling events of DLLs are dependent on the ubiquitination of the intracellular domain of DLL (DICD).9,10 The Notch regulator Numb prevents DLL degradation, promoting their recycling back to the cell surface11 (Figure 1B).
In addition to activating Notch signaling, DLLs negatively regulate Notch signaling through a variety of mechanisms (Figure 1C). Like Notch receptors, DLLs undergo proteolytic cleavage by ADAMs within their juxtamembrane (the cleavage site is 10 aa N-terminal to the transmembrane domain) and subsequently by the γ-secretase complex within their transmembrane regions (the cleavage site is unknown), releasing DTMIC (delta-like transmembrane and intracellular domain) and DICD.12–14 DICD is 100–150 aa in length and harbors a putative PDZ domain, predicted to couple DICD to membrane-bound proteins for DLL trafficking10 and to the JUN proto-oncogene and NICD-CSL-MAML complex for the nuclear regulation of target gene transcription.15–17
Notch signaling in cancer can be oncogenic or tumor-suppressive.3–20 The roles of DLLs as an important family of Notch ligands, have not been systematically elucidated. In this review, we provide a comprehensive overview of the role of DLLs in cancer and propose novel safe and effective DLL-targeted treatment strategies.
Delta-Like Ligands in Cancer
DLLs are aberrantly expressed in a range of human cancers and influence cancer progression. To date, the roles of DLLs have been extensively studied in lung cancer, liver cancer, gliomas and breast cancer. These tumors have special properties such as neuroendocrine activity, viral infection or hormone secretion, which are closely related to DLLs. Furthermore, several DLL-targeted therapy strategies for the treatment of these cancer types have been proposed and are currently in pre- or clinical trials. We herein review recent progress in our understanding of DLLs in these four cancer types, and discuss the major roles of DLLs in other cancers.
Delta-Like Ligands in Lung Cancer
DLL1 plays a tumor-suppressive role in lung cancer. In the bone marrow of lung cancer patients, DLL1 is poorly expressed, whilst another Notch ligand Jagged1 is overexpressed in dendritic cells.21 Through the interactions between dendritic cells and CD8+ T-cells, Jagged1-Notch signaling activates PD-1, impairing the activation of CD8+ T-cells. The overexpression of DLL1 by clustered DLL1 downregulates Jagged1-Notch signaling and decreases PD-1 expression, leading to a recovery in anti-tumor T-cell responses and increased T-cell infiltration.21 T-cell responses supported by DLL1 inhibit the growth and vascularization of lung tumors, revealing new avenues to modulate tumor immunity.21–23 In addition, DLL1 overexpression in small cell lung cancer (SCLC) cells improves the efficacy of chemotherapeutic and oncogene-targeted agents, leading to cell apoptosis and cell cycle arrest.24
The majority of non-small cell lung cancer (NSCLC) tumors (69/84 in one study) exhibit DLL3 overexpression.25 High DLL3 expression predicts poor overall survival (OS) in lung adenocarcinoma, playing an opposing prognostic role to DLL1.26 In high-grade neuroendocrine lung tumors (including small cell lung cancer (SCLC) and large-cell neuroendocrine cancer (LCNEC)), DLL3 is overactive at the tumor cell surface and positively correlates with achaete-scute homolog-1 (ASCL1), a member of the BHLH family of transcription factors that is involved in neuroendocrine cell fate decisions.27–29 ASCL1 activates DLL3 by binding to special E-box sites in the DLL3 promoter, as observed in the dorsal neural tube.30 In a recent study, LCNEC was divided into two molecular phenotypes based on the expression of DLL3 and ASCL1.31 Type I LCNEC with high ASCL1 and DLL3 expression exhibited a neuroendocrine profile with similarity to SCLC tumors, whilst type II LCNEC with low levels of ASCL1 and DLL3 exhibited reduced levels of neuroendocrine markers and markedly differed from SCLC tumors. The expression of DLL3 and ASCL1 may form a precise delineation of the molecular boundaries between different neuroendocrine tumors, aiding the identification of the respective molecular subtypes.29,31 In addition to ASCL1, thyroid transcription factor-1 (TTF1), a target of ASCL1, is overexpressed in SCLC tissue and positively correlates with DLL3 expression.32,33 The combination of DLL3 and TTF1 overexpression can predict poorer OS in SCLC patients.32 Further studies highlighted TTF1 as a surrogate DLL3 immunohistochemistry marker to identify DLL3-positive SCLC tissues. This may improve the precise identification of DLL3-responsive tumors for precision therapy approaches.33
Recently, DLL3 has been detected in circulating tumor cells (CTCs) and in blood samples from SCLC patients.34,35 SCLC patients with DLL3-positive CTCs show decreased OS and progression-free survival (PFS) compared to DLL3-negative patients. The percentage of DLL3-positive CTCs is a liable dynamic real-time biomarker that predicts cancer progression. It significantly declines after chemotherapy but increases in cases of tumor relapse.35
Due to the strong expression of DLL3 on the surface of SCLC cells, the DLL3-targeted investigational product Rova-T was designed to bind to DLL3-positive SCLC cells, exerting further cytotoxic effects (see details in 4.4.).36 Other DLL3-targeted products include AMG 119 (HLE BiTE® antibody) and AMG 757 (chimeric antigen receptor (CAR)), designed to activate and redirect CD3-positive T cells to DLL3-expressing tumors to induce cell apoptosis.36 Two Phase I clinical trials NCT03392064 and NCT03319940 have evaluated the safety, tolerability and efficacy of AMG 119 and AMG 757 for SCLC. In addition to targeted drugs, near infrared photoimmunotherapy (NIR-PIT) is a novel DLL3-targeted strategy for SCLC treatment that employs an antibody-photosensitiser conjugate followed by NIR light exposure to damage DLL3-positive tumor cells. In vivo experiments have confirmed that DLL3-overexpressing SCLC tumors are immediately destroyed upon NIR-light exposure.37
Immunohistochemistry is used to assess DLL3 expression from SCLC biopsies and predict the effects of DLL3-targeted agents; however, several practical limitations remain.38 The application of immuno-positron emission tomography (immunoPET) can non-invasively provide real-time information regarding the status of in vivo DLL3 expression in tumors, facilitating the selection of patients for treatment with DLL3-targeted agents. An 89Zr-labeled, DLL3-targeted immunoconjugate leveraging the humanized antibody, SC16, has been developed to serve as a companion diagnostic immunoPET agent, with a promising performance in preclinical mouse models of SCLC.38
The role of DLL4 expression in lung cancer is controversial. DLL4 expression is accompanied by elevated hypoxia inducible factor-1α (HIF1α), microvessel density (MVD) and vascular endothelial growth factor (VEGF), promoting tumor angiogenesis.39–41 Conversely, endothelial DLL4 overexpression in Lewis lung cancer (LLC)-bearing mice decreases the expression of VEGF-R2, which reduces the vascular sensitivity to VEGF-A, leading to decreased endothelial density. The tumor vasculature is more mature and stable upon DLL4 stimulation, enhancing the delivery of chemotherapy drugs to the tumor site.42 In addition, DLL4-expressing endothelial cells (ECs) can inhibit the proliferation of neighboring NSCLC cells through the activation of Notch1/PTEN signaling in NSCLC cells, suggesting endothelial DLL4 has a tumor-suppressive role in lung cancer.43
Delta-Like Ligands in Liver Cancer
Activated DLL1-Notch signaling in HCC is maintained through the high expression of Pofut1, a glycosyltransferase that enhances the interaction between DLL1 and Notch receptors. Pofut1 silencing is a promising strategy for inhibiting DLL1/Notch signaling, which substantially represses the migration and invasion of HCC cells.44
Hepatitis viruses play an important role in the regulation of DLL expression. DLL3 is expressed in the cytoplasm of normal hepatocytes, but is silenced in HCC by DNA methylation and histone acetylation induced by the hepatitis B virus (HBV).45 Histone deacetylase (HDAC) inhibitors such as TSA effectively restore DLL3 expression in HCC cells. The reactivation of DLL3 inhibits HCC cell growth and induces cell apoptosis independently of Notch1, suggesting that DLL3 has a tumor-suppressive role through Notch-independent mechanisms.45–47 In contrast, DLL3 gene expression is 7.22-fold higher in circulating CD133+ cells from hepatitis C virus (HCV)-infected HCC patients compared to cells from control patients, however the effects of DLL3 overexpression on HCV-associated HCC tumors remain undefined.48
Unlike DLL3, DLL4 is upregulated in HCC by HBV infection and activates oncogenic Notch1-Hes1 signaling. DLL4 silencing has been shown to reduce HCC cell viability, leading to G1-phase cell cycle arrest, with no effects on viral replication.49 Conversely, Kunanopparat et al showed that DLL4 is able to suppress HBV replication by an unknown mechanism, independent of anti-viral cytokine production or enhanced T cell immunity.50 Further investigations should focus on the association between DLLs and hepatitis viruses, to improve our understanding of hepatitis virus-associated HCC and the development of novel targeted therapies.
Delta-Like Ligands in Gliomas
Overexpressed DLL1 inhibits neural differentiation and contributes to the development of medulloblastoma tumors.51 In glioma cells, DLL1 forms a positive feedback loop with Notch1 to promote cell survival and growth.52 DLL1 prevents glioma cells from transdifferentiating into vascular ECs, furtherly maintaining their population.53 DLL1 or Notch1 silencing inhibits the proliferation and survival of glioma cells, and significantly prolongs the survival of glioma-bearing mice.52,54
DLL3 expression in IDH mutant gliomas, particularly in 1p/19q co-deleted subsets is higher than that of IDH wild-type glioblastoma.55 The treatment of IDH mutant glioma cell lines with Rova-T can effectively induce the cytotoxicity of DLL3-expressing cells.55 In IDH wild-type glioblastoma, DLL3 is mainly expressed in the tumor lesions contacting the subventricular zone (SVZ) but not involving the cortex, which is a prognostic marker of poor PFS.56 However, in high-grade gliomas, DLL3 is defined as a proneural signature gene that is associated with longer survival, with DLL3/Notch signaling a major determinant of tumor growth.57 In vitro experiments indicate that DLL3 expression is downregulated in glioma cells, the recovery of which can inhibit the survival, proliferation and invasiveness of glioma cells.58,59 The pro-tumoral and tumor suppressor effects of DLL3 may be dependent on its distribution in tumor cells (see in 3.2.).
DLL4 localizes to the cytoplasm and membranes of gliomas ECs but rarely distributes in glioma cells or normal brain tissue.60–63 Increased DLL4/Notch expression in the large and mature vessels of gliomas promotes a quiescent vascular phenotype that reduces the density of the tumor vasculature.61,64–66 However, DLL4 in some cases is associated with high MVD and is expressed in some microvascular formations of gliomas, including delicate capillary-like,67,68 garland-like,68 sprouted and clustered60,69 and glomeruloid cells.62,68,69 These findings indicate a complex relationship between DLL4 and tumor angiogenesis. Its potential involvement mechanisms are discussed in 3.3.2.
Delta-Like Ligands in Breast Cancer
DLL1 is overexpressed in breast cancer (BC) and is associated with poorer prognosis, particularly in the ERα+ luminal subtype.70 Estrogen stabilizes DLL1 expression by inhibiting the proteasomal and lysosomal degradation of DLL1, which promotes the growth and angiogenesis of ERα+ luminal tumors. Silencing ERα expression in ERα+ BC cells significantly decreases DLL1 expression and prevents Notch activation, suggesting that blocking the estrogen/DLL1/Notch axis is a potential targeting strategy for ERα+ BC.70 Recently, in vitro experiments demonstrated the anti-tumor effects of DLL1 knockdown on human BC cell lines were mediated through inhibiting the proliferation and survival of luminal A cells, the clonogenic growth of luminal B cells, and the migration and invasion of triple-negative, claudin-low cell lines.71 miRNA-130b was identified as a potential inhibitor of DLL1 in BC through binding to its 3ʹUTR region (217–224 bp) suppressing its translation. The inhibition of DLL1 by miR-130b mimics effectively reduces the migration and invasion of BC cells.72
DLL4 is overexpressed in the plasma and tumor tissue of BC patients and is associated with a poor outcome, metastasis and drug resistance.73–77 The inhibition of DLL4 by RGD peptide-modified lipid nanoparticles (RGD-LNPs) encapsulating siRNA prolongs the OS of mouse models of BC with lung metastasis.78 In addition, lung metastasis in BC can be inhibited by the anti-cancer therapeutic peptides AD-01 and ALM201, which downregulate DLL4 and Notch4.74
Preventing DLL4 activation in BC using antibody-based drugs represents another potential DLL4-targeting strategy, exhibiting potent anti-tumor activity in pre-clinical studies.79,80 In addition, a combination of anti-DLL4 and anti-VEGF treatment in BC using bispecific antibodies not only induces tumor cell apoptosis, but also interferes with tumor angiogenesis, thus inhibiting BC progression in vivo.81,82
Delta-Like Ligands in Other Cancers
In addition to the above four cancers, the role of DLLs in other cancer types is shown in Table 1. Overexpressed DLL4 is the main ligand that activates oncogenic Notch signaling and is wildly reported to predict a poor clinical outcome in pancreatic cancer (PC), gastric cancer (GC) and clear cell renal cell cancer (ccRCC).83–92 Pre-clinical studies show that blockade of endothelial DLL4 (mDLL4) by anti-mouse DLL4 antibodies (anti-mDLL4) HMD4-2 or 21R30 inhibits neovascularization and the growth of PC in vivo, suggesting DLL4-Notch signaling is a potential target for PC treatment.93,94 In GC, DLL4 is mainly expressed in the membranes of tumor cells as opposed to the tumor stroma.89 DLL4/Notch signaling maintains the self-renewal and invasion ability of GC cells, which can be inhibited by DLL4 knockdown.88,95,96 In contrast, DLL4 is expressed in the vascular endothelium of ccRCC. Endothelial DLL4 in ccRCC can on one hand promote tumor angiogenesis through VEGF activation,92,97–99 or activate Notch signaling in tumor cells, thus inducing hematogenous metastasis.97 In Ewing’s sarcoma, DLL4/Notch signaling is activated in the perivascular stroma of tumors derived from bone marrow (BM), inducing the differentiation of BM cells into pericytes/vascular smooth muscle cells (vSMCs) that provide structural support to the vessels, permitting tumor vasculature maturation and functionality.100–102 Blocking tumor DLL4 (hLL4) with YW152F, an anti-human DLL4 antibody, blocks pericyte/vSMC formation, negatively regulating vessel functionality and vascular expansion.101
 |
Table 1 The Role of DLLs in Other Cancers |
Different Ligands Act Through Alternate Mechanisms
The roles of the three Notch DLLs in cancer are variable. Through complex mechanisms, DLLs can activate or inactivate Notch signaling under different conditions, which influences cancer progression. Prior to the identification of appropriate DLL-targeted therapies, it is necessary to understand the specific functions of different DLL ligands in cancer.
Dll1
As a membrane ligand, DLL1 binds to the Notch receptor and activates Notch signaling in cancer cells “in trans”, enhancing tumorigenesis.54,70,103 Prior to its translocation to the plasma membrane of cancer cells, cytoplasmic DLL1 undergoes Pofut1-dependent O-fucosylation modulation,44 and is transported through vesicular trafficking, supported by the Actin-related protein2/3 complex (ArpC), a regulator of the actin cytoskeleton.104 ArpC knockdown decreases DLL1 expression at the cell surface, impairing the stem-cell phenotype of cancer cells and abolishing their tumorigenicity.104
Notch1 signaling is silenced by the hypermethylation of the DLL1 promoter in GC samples (particularly in H. pylori-positive tissue), which constitute specialized characteristics of the diffuse histotype.105–107 The role of DLL1 methylation-induced Notch1 inactivation in GC is uncertain, and the overexpression of N1ICD maintains a cancer stem cell-like phenotype of GC cells.107
Although DLL1 plays an oncogenic role in cancer, its potential tumor-suppressive effects have been identified. With DLL1 interventions, tumor neovascularization is inhibited by the loss of tumor ECs derived from cancer stem cells (CSCs),53 the inhibition of VEGF-R2-mediated VEGF signaling108,109 and increased hypoxia and tumor cell necrosis.108,109 In the hematopoietic environment of tumors, the downregulation of DLL1 impairs T cell development, leading to a loss of anti-tumor T cell responses.22 Clustered DLL1 increases tumor infiltration by immune cells, but inhibits tumor neovascularization. Importantly, multivalent DLL1 therapy is a safe therapeutic intervention that does not stimulate tumor growth or organ abnormalities, the benefits of which now warrant further investigation.22,23
Dll3
Unlike other ligands, DLL3 is considered as an inhibitor of Notch signaling. On one hand, in a cell autonomous manner, DLL3 localizes to the Golgi apparatus and binds to DLL1 and/or the full-length Notch1 receptor, preventing their modification and promoting their degradation.6,110 On the other hand, membrane DLL3 binds to Notch receptors in the same cell and prevents their activation “in cis”.16 Notably, the inhibitory effects of DLL3 are dependent on the presence of Lfng, which mediates the O-fucosylation of DLL3 EGF-like repeats 2 and 5.6,7
In tumors, a range of DLL3 functions have been described (Figure 2). DLL3 is not expressed on the surface of normal cells, but is highly expressed on the surface of tumor cells, particularly those with histopathologic features of neuro- or neuro-endocrine origin, including IDH wild-type and mutant gliomas,55,56 neuroendocrine lung cancer,28 neuroendocrine prostate cancer (NEPC),111 gastrointestinal neuroendocrine cancer (GI-NEC)112 and small cell bladder cancer (SCBC).113 DLL3 is upregulated at the transcriptional level in neuroendocrine tumors by the ASCL1 oncogenic driver, and is predominantly expressed in metastatic and aggressive disease phenotypes.31,111,113 mRNA microarray analysis revealed that membrane DLL3 can activate Notch signaling “in trans” like other ligands in IDH wild-type gliomas.56 In GI-NEC, SCLC and SCBC, DLL3 shows a cytoplasmic distribution in tumor cells (reported as Golgi apparatus-localized in a single SCLC study114) but how this influences Notch signaling is unknown.27,28,112,113,115–117 In LLC cells, DLL3 localizes to the cell nuclei, inhibiting Notch signaling at the transcriptional level. Akt signaling is activated by DLL3, which promotes LLC cell survival and reduces cell apoptosis.25
Dll4
DLL4 in Regulation of Cell Behaviors
DLL4 overexpression in cancer leads to anti-angiogenic effects that restrain excessive vascular sprouting, through triggering a negative feedback with VEGF.118,119 The effects of DLL4 are profound, not only affecting tumor angiogenesis, but also regulating cell behavior. DLL4 permits sustained Notch activation and promotes the crosstalk between tumor cells (T-T),49,73,88,120–125 ECs and ECs (E-E),98,126–128 tumor cells and ECs (T-E)43,97,129–132 (Figure 3).
The three main oncogenic pathways including NF-κB, PI3K/Akt and MMP-2/9 signaling are potential therapy targets that are activated by DLL4/Notch signaling through T-T and T-E interactions. In SCLC, blocking DLL4 expression prevents the interaction of N1ICD with the p65, p50, and RelB subunits of NF-κB, thus inhibiting the liver micro-metastasis of SCLC cells.124 In T-cell acute lymphoblastic leukemia (T-ALL) and colorectal cancer cells, Notch3 signaling is triggered by endothelial DLL4 and increases NF-κB DNA-binding, promoting tumor growth.131
DLL4/Notch1 signaling enhances Akt phosphorylation and the formation of the Akt/IKK-α complex in glioma cells, thus activating NF-κB and β-catenin signaling. β-catenin silencing and/or Akt inhibitor treatment abrogates glioma cell migration and invasion.122 In addition, the knockdown of DLL4 in esophageal cancer cells attenuates Akt phosphorylation and downregulates E‑cadherin expression, abolishing cell growth and metastasis.125
Through T-T interactions, the expression and secretion of MMP2 and MMP9 are upregulated in response to DLL4/Notch signaling, which contributes to cancer cell migration and invasion.121,123 Through T-E interactions, endothelial DLL4 activates Notch1-MMP2/9 signaling in cancer cells, promoting cell proliferation, migration and angiogenesis.130
DLL4 in Tumor Vasculature
Endothelial DLL4 is expressed in VEGF-sensitive tip cells and activates Notch signaling in adjacent VEGF-insensitive stalk cells, which in turn restricts VEGF-dependent neoangiogenesis, suppresses the tip cell fate and facilitates the conversion of tip cells to stalk cells.119,133 DLL4-overexpressing tumors exhibit unique vascular characteristics, including fewer but larger vessels, improved vessel perfusion and decreased tumor hypoxia and necrosis.42,68,134,135 MVD, as an indicator of the degree of tumor angiogenesis and the proliferation state of tumor ECs, negatively correlates with the expression of DLL4 in tumors.60,61,67,68,120,131,135–137 However, in several cases, MVD is positively associated with DLL4 expression, which contradicts the effects of DLL4 on inhibiting neovascularization.39–41,50,63,75,83,92,97,98,138,139 Herein, we discuss the possible mechanisms responsible for DLL4-induced functional and productive angiogenesis, and for the first time describe the potential effects of DLL4 on promoting tumor neovascularization.
Through methylation of the VEGF-R2 promoter, DLL4 directly inhibits VEGF-R2 expression in downstream stalk cells, which blocks VEGF-A/VEGF-R2 signaling and suppresses tumor angiogenesis.140 Surprisingly, in some tumors, a positive correlation between the expression of DLL4 and VEGF-R2 exists, indicating that DLL4 can also activate the VEGF-A/VEGF-R2 axis.39,50,87,91,130 VEGF-R1, which lacks potent signaling activity in ECs, can be upregulated by DLL4.39,42,141 In addition, VEGF-R3 can be activated42 or inhibited141,142 by DLL4 in tumors, which has potential effects on VEGF-C/VEGF-R3-mediated angiogenesis.
In addition to VEGF signaling, DLL4 affects tumor endothelial function by activating Ephrin-B2/EphB4 signaling.126,127 Ephrin-B2 is directly downstream of VEGF and DLL4, and the VEGF/DLL4/Notch4 axis activates Ephrin-B2 reverse signaling to induce abnormal vessel remodeling in liver tumors through elevated Ephrin-B2.127 In U87-DLL4 tumors, Ephrin-B2 reverse signaling is activated by elevated EphB4 in response to anti-VEGF therapy.141 The inhibition of Ephrin-B2/EphB4 signaling through anti-Ephrin-B2 antibodies, soluble Ephrin-B2 or soluble EphB4 leads to increased EC proliferation, reduced vessel size and a loss of tumor growth, which mimics the non-productive angiogenesis induced by DLL4 blockade.128,141 However, Djokovic et al found that soluble EphB4 therapy in RIP1-Tag2 tumors leads to reduced tumor vessel density, suggesting the existence of a positive feedback loop between Ephrin-B2/EphB4 and VEGF signaling.142
Intratumor hypoxia is a major driver of tumor angiogenesis,143 which can be inhibited by DLL4 stimulation.68,100,101,134,135,141,144 However, a positive correlation between DLL4/Notch signaling and intratumor hypoxia also has been found in clinical cases and in vitro, which may promote tumor angiogenesis.39,42,63,145 Notably, because hypoxia is a critical inducer of VEGF, DLL4 indirectly determines changes in VEGF expression through its effects on HIF1α.42,142
The crosstalk of tumor cells and tumor ECs mediated by DLL4 is not only dependent on cell-cell interactions, but also on vesicles. In HCC, DLL4 and Ephrin-B2 are secreted by HCC cells and are transported by micro-vesicles and exosomes to ECs, enhancing migration and angiogenesis.129 In U87 tumors, DLL4-containing exosomes are incorporated into the plasma membrane of ECs and Notch receptors are downregulated. The inhibition of Notch signaling in ECs leads to nonproductive angiogenesis and increased MVD, suggesting that tumor cell-derived vesicles containing DLL4 have a positive effect on tumor neovascularization.146
Targeted DLL Therapy: Is It Effective and Safe?
According to the characteristics of each DLL ligand, an array of DLL-targeting strategies have been proposed. Recombinant DLL proteins can be used to exert tumor-suppression, whilst a large number of pre- or clinical studies using DLL-targeting drugs have been reported (Table 2). However, some non-negligible problems regarding drug safety have been encountered, highlighting how both the safety and effectiveness of DLL-targeted strategies must be considered during tumor therapy.
 |
Table 2 Multiple miRNAs Affect Cancer Progression by Regulating DLLs |
Recombinant Proteins
The majority of soluble recombinant DLL proteins are formed from specific Notch-receptor binding domains (DSL/DSL~EGF2-3/extracellular domain) fused to IgG Fc- or His-tags.21,66,68,109,147–149 Due to the lack of multivalent interactions between Notch receptors and their ligands, and their inability to induce Notch ligand endocytosis, these monovalent soluble forms of DLL1/4 can be classed as Notch inhibitors.21,66,124,149 However, soluble DLL1/4 can also activate Notch signaling, indicating their uncertain effects on the Notch signaling axis.68,118,148
To exert a positive role in anti-tumor T cell immunity, a multivalent clustered DLL1 was designed consisting of the extracellular domain of DLL1, IgG Fc-tags, biotinylated anti-Fc antibodies, and NeutrAvidin, which triggers Notch signaling in T cells.22,23 A soluble DLL1 protein containing a DSL domain and an arginine-glycineaspartate (RGD) motif (CRGDCGVRY) can also be targeted to Notch receptors on tumor EC cells, triggering DLL1-Notch signaling in the tumor vasculature, inhibiting angiogenesis.109
MiRNAs
MicroRNAs (miRNAs) are small non-coding RNAs that directly bind to the 3ʹ-UTR of target mRNAs, leading to gene silencing.150 Several miRNAs have been shown to be involved in the regulation of DLL with a range of multifaceted functions reported (Table 3).
 |  |  |
Table 3 The DLL-Targeted Drugs |
MiRNA-34a is an important antagonist of DLL1 in tumors, which can be directly activated by p53.53,151 The positive effects of miRNA-34a include its inhibitory effects on the stem cell-like self-renewal, invasion and migration of tumor cells induced by DLL1/Notch signaling.51,151,152 However, miRNA-34a also reverses the anti-tumorigenic functions of DLL1/Notch signaling, inducing the trans-differentiation and chemoresistance of tumor cells.53,153 In addition, due to the different effects of DLL3 on Notch signaling and cancer progression, two DLL3 antagonists miRNA-518d and miRNA-18 have been shown as anti-tumorigenic and tumorigenic, respectively.58,154 This highlights the need to accurately define the functions of DLL prior to the use of miRNA-targeting strategies.
Antibodies
Humanized anti-DLL4 (anti-hDLL4) IgG antibodies are commonly used in pre- or clinical studies to inhibit tumorigenicity by reducing CSC frequency79,94,155,156 and to disrupt the tumor vasculature by inducing the production of immature non-functional blood vessels.79,132,136,137,157 However, despite its potent anti-tumor activity, anti-DLL4 antibodies show high levels of toxicity.158 Upon the assessment of anti-hDLL4 IgG antibodies in athymic nude mice, anti-DLL4 antibodies led to nonlinear pharmacokinetics (PK) and rapidly distributed to several normal tissues including lung and liver. At doses greater than 10 mg/kg, its clearance decreased.159 DLL4 blockade in normal tissues can remove the inhibition of VEGF-R2, which perturbs Notch/VEGF signaling, alters vascular homeostasis, and promotes the pathological activation of ECs.160,161 In the livers of mice, rats and cynomolgus monkeys, anti-DLL4 treatment leads to pathological changes including sinusoidal dilatation (SD), centrilobular hepatic cord atrophy, bile ductular proliferation and abnormal liver function. In the skin, heart and lungs of rats, vascular lesions have also been observed.160 The functional angiogenesis and growth of early tumors can be achieved by lower levels of DLL4, as observed in pre-cancerous skin papillomas.161 To reduce the DLL4 blockade-associated toxicity of full-length IgG, anti-DLL4 F(ab’)2 antibodies have been developed, which have a shorter half-life and are more rapidly cleared compared than anti-hDLL4 IgG antibodies, but maintaining robust anti-tumor activity.162 However, although general toxicity is reduced, the liver toxicity of anti-DLL4 F(ab’)2 at the genetic level is unavoidable.162,163 The anti-DLL4 nanobody 3Nb3, has a smaller size (15 KD) and higher affinity and specificity to the DLL4 antigen on the surface of tumor cells and ECs.164,165 As nanobodies generally have a reduced half-life and higher clearance compared to antibodies, they are speculated to reduce DLL4 blockade-associated toxicity, although this has not been confirmed experimentally. In addition to nanobodies, a DLL4-targeted nanomedicine GD16-PTX-NP has been developed and exhibits antiangiogenic effects on subcutaneous FaDu xenografts in pre-clinical studies.166 As a nanoparticulate drug delivery systems (nano-DDS), GD16-PTX-NP controls the release and long-circulating features of the antiangiogenic model drug paclitaxel and is dependent on a peptide GD16 (H2N-GRCTNFHNFIYICFPD-CONH2) to bind tumor endothelial DLL4.166
DLL4 mediates tumor resistance to the VEGF inhibitor bevacizumab, leading to tumor regrowth and the formation of residual well-organized large vessels.68,141 The combined inhibition of DLL4 and VEGF signaling can re-sensitize tumors to VEGF inhibition, increasing tumor hypoxia and markedly reducing tumor growth and angiogenesis.141,167 Recently, several bispecific monoclonal antibodies targeting both hDLL4 and hVEGF have been established and exhibit inhibitory effects on tumor cells and the vasculature. Based on the different structural designs, anti-hDLL4 and anti-hVEGF bispecific antibodies include Navicixizumab (knob-in-hole),168 HD-105 (scFv2-Fc),169 HB-32 (CrossMAb)81 and ABT-165 (DVD-Ig).82 Toxicity studies in cynomolgus monkeys indicate that ABT-165 lacks antidrug antibody responses and is well-tolerated at doses up to 200 mg/kg with non-adverse effects in the liver and thymus. This demonstrates more favorable pharmacokinetic and safety profiles than anti-hDLL4 IgG antibodies.82
Antibody-Drug Conjugates
Antibody-drug conjugates (ADCs) use antibody binding to specific antigens on the tumor cell surface to deliver small-molecule chemotherapeutic agents into the tumor cells to promote cell killing. Rovalpituzumab tesirine/SC16LD6.5/Rova-T is a DLL3-targeted ADC that contains an anti-hDLL3 IgG1 antibody, a linker and a pyrrolobenzodiazepine dimer toxin.36 In pre-clinical studies, Rova-T effectively targeted DLL3 and inhibited tumor growth in “patient-derived xenograft” mouse models of neuroendocrine tumors including SCBC, NEPC, SCLC and LCNEC.28,111,113 In a phase I clinical study (NCT01901653) of 72 SCLC and 8 LCNEC USA patients, Rova-T produced an improved overall response (objective response rate (ORR) 38%) in DLL3-high patients (≥50%) compared to patients with low DLL3 expression (<50%) (ORR 0%).170 In a recent phase I clinical study (NCT03086239) of 63 Japanese SCLC patients treated with Rova-T, 17% with DLL3-high expression (≥75%) had an objective response, with 56% achieving disease control.171 In addition, the median overall survival of patients with DLL3-high expression (7.4 months) was longer compared to those with DLL3-low expression (5.1 months).171 In a single-arm, Phase II TRINITY trial (NCT02674568) which assessed the safety and efficacy in a third-line treatment setting for 261 patients with SCLC, Rova-T treatment showed modest anti-tumor effects.172 ORR was 16% in the DLL3-high expression (≥75%) group and the disease control rate (DCR) was 24%. Unfortunately, grade 3–5 treatment-emergent adverse event (TEAE) occurred in 213 (63%) patients and drug-related serious TEAEs occurred in 100 (30%) patients. The most common TEAE was fatigue (38%) (n=130 patients), with the most common drug-related serious TEAE being photosensitivity reactions (42%) (n=143 patients).172 The TAHOE Phase III trial NCT03061812 compared Rova-T to topotecan as second-line therapy for SCLC, but was recently halted due to a shorter OS in the Rova-T arm. An additional MERU phase III trial NCT03033511 evaluated the efficacy of Rova-T as a first-line maintenance therapy following first-line platinum-based chemotherapy for advanced SCLC. However, the trial has been terminated as Rova-T treatment had no survival benefit at a pre-planned interim analysis. The clinical results suggest that the exploration and modification of the dose and schedule of DLL3-targeted drugs for SCLC treatment are necessary to reduce toxicity and improve efficacy. Despite the cessation of phase II and III trials, DLL3-expressing tumor cells remain a high-value target, and an additional phase I clinical trial NCT03000257 is recruiting participants to evaluate the safety and tolerability of Rova-T in combination with ABBV-181 for advanced solid tumors.
MvM03 and MGD03 are two novel DLL4-targeting ADCs containing an anti-hDLL4 IgG2a antibody MMGZ01, a linker, and a cytotoxic agent (MMAE and Doxorubicin respectively). In vitro and in vivo experiments in breast cancer models highlight the potent anti-tumor activity of MvM03 and MGD03, suggesting the potential therapeutic value of anti-DLL4 ADCs.173
Conclusion
When the Notch receptor is activated, NICD is released following proteolytic cleavage, and DLLs determine the activation of downstream signaling pathways that can be oncogenic or tumor-suppressive. DLLs act as ligands at the cell surface, cytoplasm and nucleus, regulating the behavior of tumor cells, tumor EC cells and tumor-infiltrating immune cells.
Membrane DLL1 expression in cancer cells plays an oncogenic role through its activation of Notch signaling. However, active DLL1 can prevent tumor neovascularization and is critical for anti-tumor T cell immunity in the tumor hematopoietic environment.21–23,53,108,109 DLL3 localizes to the plasma membrane of tumor cells and acts as a reliable biomarker to predict cancer progression and a poor clinical outcome. Pre- and clinical trial results indicate that membrane DLL3 is a potential target for preventing tumor growth.28,113,170 DLL4 plays an oncogenic role by regulating cell behavior and tumor angiogenesis. DLL4 antagonizes VEGF-mediated neovascularization and promotes the formation of large, mature and well-perfused vessels, all of which are beneficial to tumor growth.68,118,134 However, a positive correlation between DLL4 and tumor angiogenesis were also identified, though the mechanisms are complicated.
To promote the tumor-suppressive functions of DLLs and to avoid oncogenicity, numerous DLL-targeting strategies have been proposed. These include (1) DLL mimics that can activate or inactivate Notch signaling, including DLL recombinant proteins; (2) DLL binding molecules that inhibit their activity, including anti-DLL antibodies, bispecific antibodies and nanobodies; (3) DLL binding molecules that deliver cytotoxic agents or recruit CD3-positive T cells to kill DLL-positive cells, including nanomedicines, ADCs, HLE BiTE® antibodies and CARs; (4) Methods to inhibit the expression of DLLs, including miRNAs and therapeutic peptides. (5) Blocking other oncogenic pathways that are activated by DLL/Notch signaling, such as NF-κB, PI3K/Akt and MMP-2/9 signaling.
In conclusion, DLLs play complex roles in the regulation of cancer progression through a variety of pathways, but are not restricted to cell-surface Notch-signaling. Prior to the clinical application of DLL-targeted therapies, their precise roles in a range of cancers must be fully understood. In addition, DLL-targeted drugs exhibit non-negligible side effects, and their effectiveness and safety now require improvement.
Abbreviations
ADAM, A Disintegrin and metalloproteinase domain-containing protein; Hes, hairy enhance of split; Hey, Hairy/Enhancer of spit related with YRPW motif; PDZ, Post-synaptic density protein 95 (PSD-95), Discs large (DLG), Zonula occludens 1 (ZO-1); PD-1, programmed cell death protein 1; BHLH, basic helix-loop-helix; PTEN, phosphatase and tensin homologue; Pofut1, protein o-fucosyltransferase 1; IDH, isocitrate dehydrogenase; Akt, protein kinase B; PI3K, phosphatidylinositol 3-kinase; NF-κB, nuclear factor-κ-gene binding; MMP, matrix metalloproteinase.
Data Sharing Statement
All data generated or analyzed during this study are included in this published article.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81760328).
Disclosure
The authors declare that they have no competing interests.
References
1. Wang MM. Notch signaling and Notch signaling modifiers. Int J Biochem Cell Biol. 2011;43(11):1550–1562. doi:10.1016/j.biocel.2011.08.005
2. D’Souza B, Miyamoto A, Weinmaster G. The many facets of Notch ligands. Oncogene. 2008;27(38):5148–5167. doi:10.1038/onc.2008.229
3. Xiu MX, Liu YM. The role of oncogenic Notch2 signaling in cancer: a novel therapeutic target. Am J Cancer Res. 2019;9(5):837–854.
4. Shi X, Wang R. Glioma cell fate decisions mediated by Dll1-Jag1-Fringe in Notch1 signaling pathway. BMC Syst Biol. 2017;11(Suppl 4):84. doi:10.1186/s12918-017-0457-6
5. Yang LT, Nichols JT, Yao C, Manilay JO, Robey EA, Weinmaster G. Fringe glycosyltransferases differentially modulate Notch1 proteolysis induced by Delta1 and Jagged1. Mol Biol Cell. 2005;16(2):927. doi:10.1091/mbc.e04-07-0614
6. Katrin S, Karin SG, Elisabeth K, Birte H, Britta MKH, Achim G. O-fucosylation of DLL3 is required for its function during somitogenesis. PLoS One. 2015;10(4):e0123776. doi:10.1371/journal.pone.0123776
7. Okubo Y, Sugawara T, Abe-Koduka N, Kanno J, Kimura A, Saga Y. Lfng regulates the synchronized oscillation of the mouse segmentation clock via trans-repression of Notch signalling. Nat Commun. 2012;3(4):1141. doi:10.1038/ncomms2133
8. Musse AA, Meloty-Kapella L, Weinmaster G. Notch ligand endocytosis: mechanistic basis of signaling activity. Semin Cell Dev Biol. 2012;23(4):429–436.
9. Sara Farrah H, Delphine NL, Six EM, Alain IL, Frédérique L. The intracellular region of notch ligands Dll1 and Dll3 regulates their trafficking and signaling activity. Proc Natl Acad Sci U S A. 2008;105(32):11212–11217.
10. Pintar A, Biasio AD, Popovic M, Ivanova N. The intracellular region of Notch ligands: does the tail make the difference? Biol Direct. 2007;2(1):19.
11. Shao X, Ding Z, Zhao M, et al. Mammalian Numb protein antagonizes Notch by controlling postendocytic trafficking of the Notch ligand Delta-like 4. J Biol Chem. 2017;292(50):
12. Six E, Ndiaye D, Laabi Y, et al. The Notch ligand Delta1 is sequentially cleaved by an ADAM protease and gamma-secretase. Proc Natl Acad Sci U S A. 2003;100(13):7638–7643. doi:10.1073/pnas.1230693100
13. Ikeuchi T, Sisodia SS. The notch ligands, delta1 and jagged2, are substrates for presenilin-dependent “gamma-secretase” cleavage. J Biol Chem. 2003;278(10):7751–7754. doi:10.1074/jbc.C200711200
14. Emilia D, Danqiong S, Haiqing Y, Atsuko SF, Blobel CP, Anna Z. Proteolytic processing of delta-like 1 by ADAM proteases. J Biol Chem. 2007;282(1):436–444. doi:10.1074/jbc.M605451200
15. Forghany Z, Robertson F, Lundby A, Olsen JV, Baker DA. Control of endothelial cell tube formation by Notch ligand intracellular domain interactions with activator protein 1 (AP-1). J Biol Chem. 2017;293(4):
16. Jung J, Mo JS, Kim MY, Ann EJ, Yoon JH, Park HS. Regulation of Notch1 signaling by Delta-like ligand 1 intracellular domain through physical interaction. Mol Cells. 2011;32(2):161–165. doi:10.1007/s10059-011-1046-y
17. Redeker C, Schuster-Gossler K, Kremmer E, Gossler A. Normal development in mice over-expressing the intracellular domain of DLL1 argues against reverse signaling by DLL1 in vivo. PLoS One. 2013;8(10):e79050. doi:10.1371/journal.pone.0079050
18. Aburjania Z, Jang S, Whitt J, Jaskula-Stzul R, Chen H, Rose JB. The role of notch3 in cancer. Oncologist. 2018;23(8):900–911. doi:10.1634/theoncologist.2017-0677
19. Aster JC, Pear WS, Blacklow SC. The varied roles of notch in cancer. Annu Rev Pathol. 2017;12(1):245. doi:10.1146/annurev-pathol-052016-100127
20. Nowell CS, Radtke F. Notch as a tumour suppressor. Nat Rev Cancer. 2017;17(3):145–159. doi:10.1038/nrc.2016.145
21. Tchekneva EE, Goruganthu MUL, Uzhachenko RV, et al. Determinant roles of dendritic cell-expressed notch Delta-like and Jagged ligands on anti-tumor T cell immunity. J Immunother Cancer. 2019;7(1):95. doi:10.1186/s40425-019-0566-4
22. Huang Y, Lin L, Shanker A, et al. Resuscitating cancer immunosurveillance: selective stimulation of DLL1-Notch signaling in T cells rescues T-cell function and inhibits tumor growth. Cancer Res. 2011;71(19):6122–6131. doi:10.1158/0008-5472.CAN-10-4366
23. Biktasova AK, Dudimah DF, Uzhachenko RV, et al. Multivalent forms of the notch ligand DLL-1 enhance antitumor T-cell Immunity in Lung Cancer and Improve Efficacy of EGFR-Targeted Therapy. Cancer Res. 2015;75(22):4728–4741. doi:10.1158/0008-5472.CAN-14-1154
24. Liu H, Peng J, Bai Y, Guo L. [Up-regulation of DLL1 may promote the chemotherapeutic sensitivity in small cell lung cancer]. Zhongguo Fei Ai Za Zhi. 2013;16(6):282–288. Chinese. doi:10.3779/j.issn.1009-3419.2013.06.02
25. Deng SM, Yan XC, Liang L, et al. The Notch ligand delta-like 3 promotes tumor growth and inhibits Notch signaling in lung cancer cells in mice. Biochem Biophys Res Commun. 2017;483(1):488–494. doi:10.1016/j.bbrc.2016.12.117
26. Liu ZY, Wu T, Li Q, et al. Notch Signaling Components: diverging Prognostic Indicators in Lung Adenocarcinoma. Medicine (Baltimore). 2016;95(20):e3715. doi:10.1097/MD.0000000000003715
27. Furuta M, Sakakibara-Konishi J, Kikuchi H, et al. Analysis of DLL3 and ASCL1 in Surgically Resected Small Cell Lung Cancer (HOT1702). Oncologist. 2019;24(11). doi:10.1634/theoncologist.2018-0676.
28. Saunders LR, Bankovich AJ, Anderson WC, et al. A DLL3-targeted antibody-drug conjugate eradicates high-grade pulmonary neuroendocrine tumor-initiating cells in vivo. Sci Transl Med. 2015;7(302):302ra136. doi:10.1126/scitranslmed.aac9459
29. Hermans BCM, Derks JL, Thunnissen E, et al. DLL3 expression in large cell neuroendocrine carcinoma (LCNEC) and association with molecular subtypes and neuroendocrine profile. Lung Cancer. 2019;138:102–108. doi:10.1016/j.lungcan.2019.10.010
30. Henke RM, Meredith DM, Borromeo MD, Savage TK, Johnson JE. Ascl1 and Neurog2 form novel complexes and regulate Delta-like3 (Dll3) expression in the neural tube. Dev Biol. 2009;328(2):529–540. doi:10.1016/j.ydbio.2009.01.007
31. George J, Walter V, Peifer M, et al. Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. Nat Commun. 2018;9(1):1048. doi:10.1038/s41467-018-03099-x
32. Yan LX, Liu YH, Li Z, et al. Prognostic value of delta-like protein 3 combined with thyroid transcription factor-1 in small-cell lung cancer. Oncol Lett. 2019;18(3):2254–2261. doi:10.3892/ol.2019.10538
33. Cardnell RJ, Li L, Sen T, et al. Protein expression of TTF1 and cMYC define distinct molecular subgroups of small cell lung cancer with unique vulnerabilities to aurora kinase inhibition, DLL3 targeting, and other targeted therapies. Oncotarget. 2017;8(43):73419–73432. doi:10.18632/oncotarget.20621
34. Obermayr E, Agreiter C, Schuster E, et al. Molecular Characterization of Circulating Tumor Cells Enriched by A Microfluidic Platform in Patients with Small-Cell Lung Cancer. Cells. 2019;8(8):880. doi:10.3390/cells8080880
35. Messaritakis I, Nikolaou M, Koinis F, et al. Characterization of DLL3-positive circulating tumor cells (CTCs) in patients with small cell lung cancer (SCLC) and evaluation of their clinical relevance during front-line treatment. Lung Cancer. 2019;135:33–39. doi:10.1016/j.lungcan.2019.06.025
36. Owen DH, Giffin MJ, Bailis JM, Smit MD, Carbone DP, He K. DLL3: an emerging target in small cell lung cancer. J Hematol Oncol. 2019;12(1):61. doi:10.1186/s13045-019-0745-2
37. Isobe Y, Sato K, Nishinaga Y, et al. Near infrared photoimmunotherapy targeting DLL3 for small cell lung cancer. EBioMedicine. 2020;52:102632. doi:10.1016/j.ebiom.2020.102632
38. Sharma SK, Pourat J, Abdel-Atti D, et al. Noninvasive Interrogation of DLL3 Expression in Metastatic Small Cell Lung Cancer. Cancer Res. 2017;77(14):3931–3941. doi:10.1158/0008-5472.CAN-17-0299
39. Yu S, Sun J, Zhang J, et al. Aberrant expression and association of VEGF and Dll4/Notch pathway molecules under hypoxia in patients with lung cancer. Histol Histopathol. 2013;28(2):277–284. doi:10.14670/HH-28.277
40. Li X, Zhang Q, Lu B, et al. [Expression of DLL4 and VEGF in Lung Adenocarcinoma and their Relationship with Angiogenesis in Tumor.]. Zhongguo Fei Ai Za Zhi. 2009;12(2):117–121. Chinese. doi:10.3779/j.issn.1009-3419.2009.02.04
41. Maeda A, Nakata M, Yasuda K, et al. Influence of vascular endothelial growth factor single nucleotide polymorphisms on non-small cell lung cancer tumor angiogenesis. Oncol Rep. 2013;29(1):39–44. doi:10.3892/or.2012.2075
42. Trindade A, Djokovic D, Gigante J, Mendonca L, Duarte A. Endothelial Dll4 overexpression reduces vascular response and inhibits tumor growth and metastasization in vivo. BMC Cancer. 2017;17(1):189. doi:10.1186/s12885-017-3171-2
43. Ding XY, Ding J, Wu K, et al. Cross-talk between endothelial cells and tumor via delta-like ligand 4/Notch/PTEN signaling inhibits lung cancer growth. Oncogene. 2012;31(23):2899–2906. doi:10.1038/onc.2011.467
44. Ma L, Dong P, Liu L, et al. Overexpression of protein O-fucosyltransferase 1 accelerates hepatocellular carcinoma progression via the Notch signaling pathway. Biochem Biophys Res Commun. 2016;473(2):503–510. doi:10.1016/j.bbrc.2016.03.062
45. Hamamoto H, Maemura K, Matsuo K, et al. Delta-like 3 is silenced by HBx via histone acetylation in HBV-associated HCCs. Sci Rep. 2018;8(1):4842. doi:10.1038/s41598-018-23318-1
46. Maemura K, Yoshikawa H, Yokoyama K, et al. Delta-like 3 is silenced by methylation and induces apoptosis in human hepatocellular carcinoma. Int J Oncol. 2013;42(3):817–822. doi:10.3892/ijo.2013.1778
47. Mizuno Y, Maemura K, Tanaka Y, Hirata A, Kondo Y. Expression of delta-like 3 is downregulated by aberrant DNA methylation and histone modification in hepatocellular carcinoma. Oncol Rep. 2018;39(5):2209–2216.
48. Zekri AN, El-Sisi ER, Abdallah ZF, Ismail A, Barakat Barakat A. Gene expression profiling of circulating CD133(+) cells of hepatocellular carcinoma patients associated with HCV infection. J Egypt Natl Canc Inst. 2017;29(1):19–24. doi:10.1016/j.jnci.2016.12.002
49. Kongkavitoon P, Tangkijvanich P, Hirankarn N, Palaga T. Hepatitis B Virus HBx Activates Notch Signaling via Delta-Like 4/Notch1 in Hepatocellular Carcinoma. PLoS One. 2016;11(1):e0146696. doi:10.1371/journal.pone.0146696
50. Kunanopparat A, Issara-Amphorn J, Leelahavanichkul A, et al. Delta-like ligand 4 in hepatocellular carcinoma intrinsically promotes tumour growth and suppresses hepatitis B virus replication. World J Gastroenterol. 2018;24(34):3861–3870. doi:10.3748/wjg.v24.i34.3861
51. de Antonellis P, Medaglia C, Cusanelli E, et al. MiR-34a targeting of Notch ligand delta-like 1 impairs CD15+/CD133+ tumor-propagating cells and supports neural differentiation in medulloblastoma. PLoS One. 2011;6(9):e24584. doi:10.1371/journal.pone.0024584
52. Qian CF, Yan W, Zhang JX, et al. Notch1 induces enhanced expression of Delta-like-1 in the U251MG glioma cell line. Int J Mol Med. 2009;24(4):445–451. doi:10.3892/ijmm_00000251
53. Jin Z, Zhan T, Tao J, et al. MicroRNA-34a induces transdifferentiation of glioma stem cells into vascular endothelial cells by targeting Notch pathway. Biosci Biotechnol Biochem. 2017;81(10):1899–1907. doi:10.1080/09168451.2017.1364965
54. Purow BW, Haque RM, Noel MW, et al. Expression of Notch-1 and its ligands, Delta-like-1 and Jagged-1, is critical for glioma cell survival and proliferation. Cancer Res. 2005;65(6):2353–2363. doi:10.1158/0008-5472.CAN-04-1890
55. Spino M, Kurz SC, Chiriboga L, et al. Cell Surface Notch Ligand DLL3 is a Therapeutic Target in Isocitrate Dehydrogenase-mutant Glioma. Clin Cancer Res. 2019;25(4):1261–1271. doi:10.1158/1078-0432.CCR-18-2312
56. Jungk C, Mock A, Exner J, et al. Spatial transcriptome analysis reveals Notch pathway-associated prognostic markers in IDH1 wild-type glioblastoma involving the subventricular zone. BMC Med. 2016;14(1):170. doi:10.1186/s12916-016-0710-7
57. Phillips HS, Kharbanda S, Chen R, et al. Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell. 2006;9(3):157–173. doi:10.1016/j.ccr.2006.02.019
58. Turchi L, Debruyne DN, Almairac F, et al. Tumorigenic potential of miR-18A* in glioma initiating cells requires NOTCH-1 signaling. Stem Cells. 2013;31(7):1252–1265. doi:10.1002/stem.1373
59. Hu B, Nandhu MS, Sim H, et al. Fibulin-3 promotes glioma growth and resistance through a novel paracrine regulation of Notch signaling. Cancer Res. 2012;72(15):3873–3885. doi:10.1158/0008-5472.CAN-12-1060
60. Qiu XX, Chen L, Wang CH, et al. The Vascular notch ligands delta-like ligand 4 (DLL4) and Jagged1 (JAG1) have opposing correlations with microvascularization but a uniform prognostic effect in primary glioblastoma: a preliminary study. World Neurosurg. 2016;88:447–458. doi:10.1016/j.wneu.2015.10.058
61. Zhang JF, Chen Y, Qiu XX, et al. The vascular delta-like ligand-4 (DLL4)-Notch4 signaling correlates with angiogenesis in primary glioblastoma: an immunohistochemical study. Tumour Biol. 2016;37(3):3797–3805. doi:10.1007/s13277-015-4202-8
62. Jubb AM, Lisa B, Leticia C, et al. Expression of vascular Notch ligands Delta-like 4 and Jagged-1 in glioblastoma. Histopathology. 2012;60(5):740–747. doi:10.1111/j.1365-2559.2011.04138.x
63. Li Z, Wang J, Gong L, Wen Z, Xu C, Huang X. Correlation of Delta-like ligand 4 (DLL4) with VEGF and HIF-1alpha expression in human glioma. Asian Pac J Cancer Prev. 2011;12(1):215–218.
64. Payton M, Jun T, Wayne W, et al. Antagonism of Ang-Tie2 and Dll4-Notch signaling has opposing effects on tumor endothelial cell proliferation, evidenced by a new flow cytometry method. Lab Invest. 2014;94(11):1296–1308. doi:10.1038/labinvest.2014.116
65. Reis M, Czupalla CJ, Ziegler N, et al. Endothelial Wnt/beta-catenin signaling inhibits glioma angiogenesis and normalizes tumor blood vessels by inducing PDGF-B expression. J Exp Med. 2012;209(9):1611–1627.
66. Noguera-Troise I, Daly C, Papadopoulos NJ, et al. Blockade of Dll4 inhibits tumour growth by promoting non-productive angiogenesis. Nature. 2006;444(7122):1032–1037.
67. El Hindy N, Keyvani K, Pagenstecher A, et al. Implications of Dll4-Notch signaling activation in primary glioblastoma multiforme. Neuro Oncol. 2013;15(10):1366–1378.
68. Li JL, Sainson RC, Shi W, et al. Delta-like 4 Notch ligand regulates tumor angiogenesis, improves tumor vascular function, and promotes tumor growth in vivo. Cancer Res. 2007;67(23):11244–11253.
69. Qiu XX, Chen L, Wang CH, et al. High delta-like ligand 4 (DLL4) is correlated with peritumoral brain edema and predicts poor prognosis in primary glioblastoma. Medicine (Baltimore). 2014;93(8):e57.
70. Kumar S, Srivastav RK, Wilkes DW, et al. Estrogen-dependent DLL1-mediated Notch signaling promotes luminal breast cancer. Oncogene. 2019;38(12):2092–2107.
71. Sales-Dias J, Silva G, Lamy M, Ferreira A, Barbas A. The Notch ligand DLL1 exerts carcinogenic features in human breast cancer cells. PLoS One. 2019;14(5):e0217002.
72. Shui Y, Yu X, Duan R, et al. miR-130b-3p inhibits cell invasion and migration by targeting the Notch ligand Delta-like 1 in breast carcinoma. Gene. 2017;609:80–87.
73. Wang Q, Shi Y, Butler HJ, et al. Role of delta-like ligand-4 in chemoresistance against docetaxel in MCF-7 cells. Hum Exp Toxicol. 2017;36(4):328–338.
74. McClements L, Annett S, Yakkundi A, et al. FKBPL and its peptide derivatives inhibit endocrine therapy resistant cancer stem cells and breast cancer metastasis by downregulating DLL4 and Notch4. BMC Cancer. 2019;19(1):351.
75. Jubb AM, Soilleux EJ, Turley H, et al. Expression of Vascular Notch Ligand Delta-Like 4 and Inflammatory Markers in Breast Cancer. Am J Pathol. 2010;176(4):2019–2028.
76. Kontomanolis E, Panteliadou M, Giatromanolaki A, et al. Delta-like ligand 4 (DLL4) in the plasma and neoplastic tissues from breast cancer patients: correlation with metastasis. Med Oncol. 2014;31(5):1–6.
77. Min X, Shanshan Y, Xiaoming N, Yuanxi H. Aberrant expression of δ-like ligand 4 contributes significantly to axillary lymph node metastasis and predicts postoperative outcome in breast cancer. Hum Pathol. 2014;45(11):2302–2310.
78. Sakurai Y, Hada T, Kato A, Hagino Y, Mizumura W, Harashima H. Effective Therapy Using a Liposomal siRNA that Targets the Tumor Vasculature in a Model Murine Breast Cancer with Lung Metastasis. Mol Ther Oncolytics. 2018;11:102–108.
79. Xu Z, Wang Z, Jia X, et al. MMGZ01, an anti-DLL4 monoclonal antibody, promotes nonfunctional vessels and inhibits breast tumor growth. Cancer Lett. 2016;372(1):118–127.
80. Jia X, Wang W, Xu Z, et al. A humanized anti-DLL4 antibody promotes dysfunctional angiogenesis and inhibits breast tumor growth. Sci Rep. 2016;6(1):27985.
81. Zhou R, Wang S, Wen H, Wang M, Wu M. The bispecific antibody HB-32, blockade of both VEGF and DLL4 shows potent anti-angiogenic activity in vitro and anti-tumor activity in breast cancer xenograft models. Exp Cell Res. 2019;380(2):141–148.
82. Li Y, Hickson JA, Ambrosi DJ, et al. ABT-165, a Dual Variable Domain Immunoglobulin (DVD-Ig) Targeting DLL4 and VEGF, Demonstrates Superior Efficacy and Favorable Safety Profiles in Preclinical Models. Mol Cancer Ther. 2018;17(5):
83. Zhou L, Yu L, Ding G, Chen W, Zheng S, Cao L. Overexpressions of DLL4 and CD105 are Associated with Poor Prognosis of Patients with Pancreatic Ductal Adenocarcinoma. Pathol Oncol Res Por. 2015;21(4):1141.
84. Huang SF, Yang ZL, Li DQ, et al. Jagged1 and DLL4 expressions in benign and malignant pancreatic lesions and their clinicopathological significance. Hepatobiliary Pancreat Dis Int. 2016;15(6):640–646.
85. Ye J, Wen J, Ning Y, Li Y. Higher notch expression implies poor survival in pancreatic ductal adenocarcinoma: a systematic review and meta-analysis. Pancreatology. 2018;18(8):954–961.
86. Drouillard A, Puleo F, Bachet JB, et al. DLL4 expression is a prognostic marker and may predict gemcitabine benefit in resected pancreatic cancer. Br J Cancer. 2016;115(10):1245–1252.
87. Hai-Tao Chen MD, Quan-Cai Cai MD, Jian-Ming Zheng MD, et al. High Expression of Delta-Like Ligand 4 Predicts Poor Prognosis After Curative Resection for Pancreatic Cancer. Ann Surg Oncol. 2012;19(3):464–474.
88. Miao ZF, Xu H, Xu HM, et al. DLL4 overexpression increases gastric cancer stem/progenitor cell self-renewal ability and correlates with poor clinical outcome via Notch-1 signaling pathway activation. Cancer Med. 2017;6(1):245–257.
89. Ishigami S, Arigami T, Uenosono Y, et al. Clinical implications of DLL4 expression in gastric cancer. J Exp Clin Cancer Res. 2013;32(1):46.
90. Wang X, Zhang J, Wang Y, Tu M, Wang Y, Shi G. Upregulated VEGFA and DLL4 act as potential prognostic genes for clear cell renal cell carcinoma. Onco Targets Ther. 2018;11:1697–1706.
91. Wang W, Yi YU, Wang YA, et al. Delta-like ligand 4: a predictor of poor prognosis in clear cell renal cell carcinoma. Oncol Lett. 2014;8(6):2627–2633.
92. Guang-Hui H, Huan L, Peng L, et al. Delta-like ligand 4 (Dll4) predicts the prognosis of clear cell renal cell carcinoma, and anti-Dll4 suppresses tumor growth in vivo. Int J Clin Exp Pathol. 2014;7(5):2143–2152.
93. Hidekazu O, Makoto S, Shinichi E, et al. Blockade of delta-like ligand 4 signaling inhibits both growth and angiogenesis of pancreatic cancer. Pancreas. 2010;39(6):897–903.
94. Yen WC, Fischer MM, Hynes M, et al. Anti-DLL4 has broad spectrum activity in pancreatic cancer dependent on targeting DLL4-Notch signaling in both tumor and vasculature cells. Clin Cancer Res. 2012;18(19):5374–5386.
95. Sun HW, Wu C, Tan HY, Wang QS. Combination DLL4 with Jagged1-siRNA can enhance inhibition of the proliferation and invasiveness activity of human gastric carcinoma by Notch1/VEGF pathway. Hepatogastroenterology. 2012;59(115):924–929.
96. Kang M, Zhang Y, Jin X, et al. Concurrent Treatment with Anti-DLL4 Enhances Antitumor and Proapoptotic Efficacy of a gamma-Secretase Inhibitor in Gastric Cancer. Transl Oncol. 2018;11(3):599–608.
97. Huang QB, Ma X, Li HZ, et al. Endothelial Delta-like 4 (DLL4) promotes renal cell carcinoma hematogenous metastasis. Oncotarget. 2014;5(10):3066–3075.
98. Huang QB, Ma X, Zhang X, et al. Down-Regulated miR-30a in Clear Cell Renal Cell Carcinoma Correlated with Tumor Hematogenous Metastasis by Targeting Angiogenesis-Specific DLL4. PLoS One. 2013;8(6):e67294.
99. Patel NS, Ji-Liang L, Daniele G, Richard P, Cranston DW, Harris AL. Up-regulation of delta-like 4 ligand in human tumor vasculature and the role of basal expression in endothelial cell function. Cancer Res. 2005;65(19):8690–8697.
100. Schadler KL, Zweidler-McKay PA, Guan H, Kleinerman ES. Delta-Like Ligand 4 Plays a Critical Role in Pericyte/Vascular Smooth Muscle Cell Formation during Vasculogenesis and Tumor Vessel Expansion in Ewing\”s Sarcoma. Clin Cancer Res an off J Am Assoc Cancer Res. 2010;16(3):848–856. doi:10.1158/1078-0432.CCR-09-1299
101. Stewart KS, Zhou Z, Zweidlermckay P, Kleinerman ES. Delta-like ligand 4–Notch signaling regulates bone marrow–derived pericyte/vascular smooth muscle cell formation. Blood. 2011;117(2):719. doi:10.1182/blood-2010-05-284869
102. Zhichao Z, Ling Y, Kleinerman ES. EWS-FLI-1 regulates the neuronal repressor gene REST, which controls Ewing sarcoma growth and vascular morphology. Cancer. 2014;120(4):579–588. doi:10.1002/cncr.28555
103. Zhang JP, Li N, Bai WZ, et al. Notch ligand Delta-like 1 promotes the metastasis of melanoma by enhancing tumor adhesion. Braz J Med Biol Res. 2014;47(4):299–306. doi:10.1590/1414-431X20143368
104. Zhang C, Hai L, Zhu M, et al. Actin cytoskeleton regulator Arp2/3 complex is required for DLL1 activating Notch1 signaling to maintain the stem cell phenotype of glioma initiating cells. Oncotarget. 2017;8(20):33353–33364. doi:10.18632/oncotarget.16495
105. Piazzi G, Fini L, Selgrad M, et al. Epigenetic regulation of Delta-Like1 controls Notch1 activation in gastric cancer. Oncotarget. 2011;2(12):1291–1301. doi:10.18632/oncotarget.414
106. Piazzi G, Bazzoli F, Ricciardiello L. Epigenetic silencing of Notch signaling in gastrointestinal cancers. Cell Cycle. 2012;11(23):4323–4327. doi:10.4161/cc.22388
107. Shen CH, Tung SY, Tseng MJ, Leu YW. Inverse Correlation between Methylation and Expression of the Delta-like Ligand 1 Gene in Gastric Cancer. Chin J Physiol. 2018;61(2):65–74. doi:10.4077/CJP.2018.BAG513
108. Zhang JP, Qin HY, Wang L, et al. Overexpression of Notch ligand Dll1 in B16 melanoma cells leads to reduced tumor growth due to attenuated vascularization. Cancer Lett. 2011;309(2):220–227. doi:10.1016/j.canlet.2011.06.008
109. Zhao XC, Dou GR, Wang L, et al. Inhibition of tumor angiogenesis and tumor growth by the DSL domain of human Delta-like 1 targeted to vascular endothelial cells. Neoplasia. 2013;15(7):815–825. doi:10.1593/neo.13550
110. Gavin C, Sparrow DB, Elisabeth K, Dunwoodie SL. Notch inhibition by the ligand DELTA-LIKE 3 defines the mechanism of abnormal vertebral segmentation in spondylocostal dysostosis. Hum Mol Genet. 2011;20(5):905. doi:10.1093/hmg/ddq529
111. Puca L, Gavyert K, Sailer V, et al. Delta-like protein 3 expression and therapeutic targeting in neuroendocrine prostate cancer. Sci Transl Med. 2019;11(484):484. doi:10.1126/scitranslmed.aav0891
112. Matsuo K, Taniguchi K, Hamamoto H, et al. Delta-like 3 localizes to neuroendocrine cells and plays a pivotal role in gastrointestinal neuroendocrine malignancy. Cancer Sci. 2019;110(10):3122–3131. doi:10.1111/cas.14157
113. Koshkin VS, Garcia JA, Reynolds J, et al. Transcriptomic and Protein Analysis of Small-cell Bladder Cancer (SCBC) Identifies Prognostic Biomarkers and DLL3 as a Relevant Therapeutic Target. Clin Cancer Res. 2019;25(1):210–221. doi:10.1158/1078-0432.CCR-18-1278
114. Tanaka K, Isse K, Fujihira T, et al. Prevalence of Delta-like protein 3 expression in patients with small cell lung cancer. Lung Cancer. 2018;115:116–120. doi:10.1016/j.lungcan.2017.11.018
115. Brcic L, Kuchler C, Eidenhammer S, et al. Comparison of four DLL3 antibodies performance in high grade neuroendocrine lung tumor samples and cell cultures. Diagn Pathol. 2019;14(1):47. doi:10.1186/s13000-019-0827-z
116. Huang RSP, Holmes BF, Powell C, et al. Delta-like Protein 3 Prevalence in Small Cell Lung Cancer and DLL3 (SP347) Assay Characteristics. Arch Pathol Lab Med. 2019;143(11):1373–1377. doi:10.5858/arpa.2018-0497-OA
117. Regzedmaa O, Li Y, Li Y, et al. Prevalence of DLL3, CTLA-4 and MSTN Expression in Patients with Small Cell Lung Cancer. Onco Targets Ther. 2019;12:10043–10055. doi:10.2147/OTT.S216362
118. Williams CK, Li JL, Murga M, Harris AL, Tosato G. Up-regulation of the Notch ligand Delta-like 4 inhibits VEGF-induced endothelial cell function. Blood. 2006;107(3):931–939. doi:10.1182/blood-2005-03-1000
119. Thurston G, Kitajewski J. VEGF and Delta-Notch: interacting signalling pathways in tumour angiogenesis. Br J Cancer. 2008;99(8):1204–1209. doi:10.1038/sj.bjc.6604484
120. Kang M, Jiang B, Xu B, et al. Delta like ligand 4 induces impaired chemo-drug delivery and enhanced chemoresistance in pancreatic cancer. Cancer Lett. 2013;330(1):11–21. doi:10.1016/j.canlet.2012.11.015
121. Li GG, Li L, Li C, et al. Influence of up-regulation of Notch ligand DLL4 on biological behaviors of human gastric cancer cells. World J Gastroenterol Wjg. 2013;19(28):4486–4494. doi:10.3748/wjg.v19.i28.4486
122. Xiaohua Z, Tao C, Jiannan Z, et al. Notch1 promotes glioma cell migration and invasion by stimulating β-catenin and NF-κB signaling via AKT activation. Cancer Sci. 2012;103(2):181–190. doi:10.1111/j.1349-7006.2011.02154.x
123. Wei L, Yuepeng Z, Yuting S, et al. IL-23 promotes invasion of esophageal squamous cell carcinoma cells by activating DLL4/Notch1 signaling pathway. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2015;31(6):812.
124. Kuramoto T, Goto H, Mitsuhashi A, et al. Dll4-Fc, an inhibitor of Dll4-notch signaling, suppresses liver metastasis of small cell lung cancer cells through the downregulation of the NF-kappaB activity. Mol Cancer Ther. 2012;11(12):2578–2587. doi:10.1158/1535-7163.MCT-12-0640
125. Guo X, Duan Y, Ye X, et al. Stable silencing of dll4 gene suppresses the growth and metastasis of esophagus cancer cells by attenuating Akt phosphorylation. Oncol Rep. 2018;40(1):495–503. doi:10.3892/or.2018.6427
126. Cassin Kimmel W, Marta S, Sierra MDLL, Sainson RCA, Giovanna T, Harris AL. Regulation of CXCR4 by the Notch ligand delta-like 4 in endothelial cells. Cancer Res. 2008;68(6):1889–1895. doi:10.1158/0008-5472.CAN-07-2181
127. Hainaud P, Contreres JO, Villemain A, et al. The role of the vascular endothelial growth factor-Delta-like 4 ligand/Notch4-ephrin B2 cascade in tumor vessel remodeling and endothelial cell functions. Cancer Res. 2006;66(17):8501–8510. doi:10.1158/0008-5472.CAN-05-4226
128. Yamanda S, Ebihara S, Asada M, et al. Role of ephrinB2 in nonproductive angiogenesis induced by Delta-like 4 blockade. Blood. 2009;113(15):3631–3639. doi:10.1182/blood-2008-07-170381
129. Jamshidi-Parsian A, Griffin RJ, Kore RA, Todorova VK, Makhoul I. Tumor-endothelial cell interaction in an experimental model of human hepatocellular carcinoma. Exp Cell Res. 2018;372(1):16–24. doi:10.1016/j.yexcr.2018.09.001
130. Zhang J, Ye J, Ma D, et al. Cross-talk between leukemic and endothelial cells promotes angiogenesis by VEGF activation of the Notch/Dll4 pathway. Carcinogenesis. 2013;34(3):667–677. doi:10.1093/carcin/bgs386
131. Indraccolo S, Minuzzo S, Masiero M, et al. Cross-talk between tumor and endothelial cells involving the notch3-dll4 interaction marks escape from tumor dormancy. Cancer Res. 2009;69(4):1314. doi:10.1158/0008-5472.CAN-08-2791
132. Kuhnert F, Chen G, Coetzee S, et al. Dll4 blockade in stromal cells mediates antitumor effects in preclinical models of ovarian cancer. Cancer Res. 2015;75(19):4086–4096. doi:10.1158/0008-5472.CAN-14-3773
133. Gridley T. Notch signaling in the vasculature. Curr Top Dev Biol. 2010;92(C):277–309.
134. Oon CE, Bridges E, Sheldon H, et al. Role of Delta-like 4 in Jagged1-induced tumour angiogenesis and tumour growth. Oncotarget. 2017;8(25):40115–40131. doi:10.18632/oncotarget.16969
135. Liu SK, Bham SAS, Emmanouil F, et al. Delta-like ligand 4-notch blockade and tumor radiation response. J Natl Cancer Inst. 2011;103(23):1778. doi:10.1093/jnci/djr419
136. Huang J, Hu W, Hu L, et al. Dll4 inhibition plus aflibercept markedly reduces ovarian tumor growth. Mol Cancer Ther. 2016;15(6):1344–1352. doi:10.1158/1535-7163.MCT-15-0144
137. John R, Gu Z, Yan W, et al. Inhibition of Dll4 signalling inhibits tumour growth by deregulating angiogenesis. Nature. 2006;444(7122):1083. doi:10.1038/nature05313
138. Zhi-Qiang L, Ling-Ling G, Zhi-Hua W, Jiang W, Cheng-Shi X, Xiao-Dong H. Delta-like ligand 4 correlates with endothelial proliferation and vessel maturation in human malignant glioma. Onkologie. 2012;35(12):763–768. doi:10.1159/000345116
139. Patel NS, Dobbie MM, Steers G, et al. Up-regulation of endothelial delta-like 4 expression correlates with vessel maturation in bladder cancer. Clin Cancer Res. 2006;12(16):4836–4844. doi:10.1158/1078-0432.CCR-06-0285
140. Wei H, Chunhua L, Han Hee D, et al. Biological roles of the Delta family Notch ligand Dll4 in tumor and endothelial cells in ovarian cancer. Cancer Res. 2011;71(18):6030–6039. doi:10.1158/0008-5472.CAN-10-2719
141. Li JL, Sainson RC, Oon CE, et al. DLL4-Notch signaling mediates tumor resistance to anti-VEGF therapy in vivo. Cancer Res. 2011;71(18):6073–6083. doi:10.1158/0008-5472.CAN-11-1704
142. Djokovic D, Trindade A, Gigante J, et al. Combination of Dll4/Notch and Ephrin-B2/EphB4 targeted therapy is highly effective in disrupting tumor angiogenesis. BMC Cancer. 2010;10(1):641. doi:10.1186/1471-2407-10-641
143. Wong BW, Marsch E, Treps L, Baes M, Carmeliet P. Endothelial cell metabolism in health and disease: impact of hypoxia. EMBO J. 2017;36(15):e201696150. doi:10.15252/embj.201696150
144. Iwamoto H, Zhang Y, Seki T, et al. PlGF-induced VEGFR1-dependent vascular remodeling determines opposing antitumor effects and drug resistance to Dll4-Notch inhibitors. Sci Adv. 2015;1(3):e1400244. doi:10.1126/sciadv.1400244
145. Pistollato F, Rampazzo E, Persano L, et al. Interaction of hypoxia-inducible factor-1α and Notch signaling regulates medulloblastoma precursor proliferation and fate. Stem Cells. 2010;28(11):1918–1929. doi:10.1002/stem.518
146. Sheldon H, Heikamp E, Turley H, Dragovic R, Harris AL. New mechanism for Notch signaling to endothelium at a distance by Delta-like 4 incorporation into exosomes. Blood. 2010;116(13):2385. doi:10.1182/blood-2009-08-239228
147. Klose R, Berger C, Moll I, et al. Soluble Notch ligand and receptor peptides act antagonistically during angiogenesis. Cardiovasc Res. 2015;107(1):153–163. doi:10.1093/cvr/cvv151
148. Ferreira A, Lamy M, Margarida Rocha M, Silva G, Bandeiras TM, Barbas A. Production and characterization of a novel Delta-like 1 functional unit as a tool for Notch pathway activation and generation of a specific antibody. Protein Expr Purif. 2018;146:8–16. doi:10.1016/j.pep.2018.01.008
149. Scehnet JS, Weidong J, Ram SK, et al. Inhibition of Dll4-mediated signaling induces proliferation of immature vessels and results in poor tissue perfusion. Blood. 2007;109(11):4753–4760. doi:10.1182/blood-2006-12-063933
150. Lu TX, Rothenberg ME. MicroRNA. J Allergy Clin Immunol. 2018;141(4):1202–1207. doi:10.1016/j.jaci.2017.08.034
151. Pang RT, Leung CO, Lee CL, et al. MicroRNA-34a is a tumor suppressor in choriocarcinoma via regulation of Delta-like1. BMC Cancer. 2013;13(1):25. doi:10.1186/1471-2407-13-25
152. Bettinsoli P, Ferrari-Toninelli G, Bonini SA, Prandelli C, Memo M. Notch ligand Delta-like 1 as a novel molecular target in childhood neuroblastoma. BMC Cancer. 2017;17(1):352. doi:10.1186/s12885-017-3340-3
153. Pu Y, Zhao F, Wang H, Cai S. MiR-34a-5p promotes multi-chemoresistance of osteosarcoma through down-regulation of the DLL1 gene. Sci Rep. 2017;7(1):44218. doi:10.1038/srep44218
154. Huang J, Cao D, Sha J, Zhu X, Han S. DLL3 is regulated by LIN28B and miR-518d-5p and regulates cell proliferation, migration and chemotherapy response in advanced small cell lung cancer. Biochem Biophys Res Commun. 2019;514(3):853–860. doi:10.1016/j.bbrc.2019.04.130
155. Hoey T, Yen WC, Axelrod F, et al. DLL4 Blockade inhibits tumor growth and reduces tumor-initiating cell frequency. Cell Stem Cell. 2009;5(2):168–177. doi:10.1016/j.stem.2009.05.019
156. Marcus F, Wan-Ching Y, Kapoun AM, et al. Anti-DLL4 inhibits growth and reduces tumor-initiating cell frequency in colorectal tumors with oncogenic KRAS mutations. Cancer Res. 2011;71(5):1520–1525. doi:10.1158/0008-5472.CAN-10-2817
157. Jenkins DW, Ross S, Veldman-Jones M, et al. MEDI0639: a novel therapeutic antibody targeting Dll4 modulates endothelial cell function and angiogenesis in vivo. Mol Cancer Ther. 2012;11(8):1650–1660. doi:10.1158/1535-7163.MCT-11-1027
158. Ji-Liang L, Jubb AM, Harris AL. Targeting DLL4 in tumors shows preclinical activity but potentially significant toxicity. Future Oncol. 2010;6(7):1099–1103. doi:10.2217/fon.10.62
159. Kamath AV, Victor Y, Priyanka G, et al. Dose dependent pharmacokinetics, tissue distribution, and anti-tumor efficacy of a humanized monoclonal antibody against DLL4 in mice. Mabs. 2014;6(6):1631–1637. doi:10.4161/mabs.36107
160. Minhong Y, Callahan CA, Beyer JC, et al. Chronic DLL4 blockade induces vascular neoplasms. Nature. 2010;463(7282):6–7. doi:10.1038/nature08751
161. Djokovic D, Trindade A, Gigante J, Pinho M, Harris AL, Duarte A. Incomplete Dll4/Notch signaling inhibition promotes functional angiogenesis supporting the growth of skin papillomas. BMC Cancer. 2015;15(1):608. doi:10.1186/s12885-015-1605-2
162. Couch J, Zhang G, Beyer J, et al. Balancing efficacy and safety of an anti-DLL4 antibody through pharmacokinetic modulation. Clin Cancer Res an off J Am Assoc Cancer Res. 2015;22(6):1469. doi:10.1158/1078-0432.CCR-15-1380
163. Jarzabek MA, Proctor WR, Vogt J, et al. Interrogation of transcriptomic changes associated with drug-induced hepatic sinusoidal dilatation in colorectal cancer. PLoS One. 2018;13(6):e0198099. doi:10.1371/journal.pone.0198099
164. Baharlou R, Tajik N, Habibi-Anbouhi M, et al. Generation and characterization of an anti-delta like ligand-4 Nanobody to induce non-productive angiogenesis. Anal Biochem. 2017;544:34–41. doi:10.1016/j.ab.2017.12.014
165. Baharlou R, Tajik N, Behdani M, et al. An antibody fragment against human delta-like ligand-4 for inhibition of cell proliferation and neovascularization. Immunopharmacol Immunotoxicol. 2018;40(5):368–374. doi:10.1080/08923973.2018.1505907
166. Liu YR, Guan YY, Luan X, et al. Delta-like ligand 4-targeted nanomedicine for antiangiogenic cancer therapy. Biomaterials. 2015;42(42):161–171. doi:10.1016/j.biomaterials.2014.11.039
167. Miles KM, Seshadri M, Ciamporcero E, et al. Dll4 blockade potentiates the anti-tumor effects of VEGF inhibition in renal cell carcinoma patient-derived xenografts. PLoS One. 2014;9(11):e112371. doi:10.1371/journal.pone.0112371
168. Yen W-C, Axelrod F, Bond C, et al. Dual targeting of DLL4 and VEGF signaling by a novel bispecific antibody inhibits tumor growth and reduces cancer stem cell frequency. AACR Ann Meeting. 2014;454:656–660.
169. Lee D, Kim D, Choi YB, et al. Simultaneous blockade of VEGF and Dll4 by HD105, a bispecific antibody, inhibits tumor progression and angiogenesis. MAbs. 2016;8(5):892–904. doi:10.1080/19420862.2016.1171432
170. Rudin CM, Pietanza MC, Bauer TM, et al. Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, Phase 1 study. Lancet Oncol. 2016;18(1):42. doi:10.1016/S1470-2045(16)30565-4
171. Udagawa H, Akamatsu H, Tanaka K, et al. Phase I safety and pharmacokinetics study of rovalpituzumab tesirine in Japanese patients with advanced, recurrent small cell lung cancer. Lung Cancer. 2019;135:145–150. doi:10.1016/j.lungcan.2019.07.025
172. Morgensztern D, Besse B, Greillier L, et al. Efficacy and safety of rovalpituzumab tesirine in third-line and beyond patients with DLL3-expressing, relapsed/refractory small-cell lung cancer: results from the phase II TRINITY Study. Clin Cancer Res. 2019;25(23):6958–6966. doi:10.1158/1078-0432.CCR-19-1133
173. Wang S, Zhou R, Sun F, Li R, Wang M, Wu M. The two novel DLL4-targeting antibody-drug conjugates MvM03 and MGD03 show potent anti-tumour activity in breast cancer xenograft models. Cancer Lett. 2017;409:125. doi:10.1016/j.canlet.2017.09.004
174. Rajamani D, Bhasin MK. Identification of key regulators of pancreatic cancer progression through multidimensional systems-level analysis. Genome Med. 2016;8(1):38. doi:10.1186/s13073-016-0282-3
175. Mullendore ME, Koorstra JB, Li YM, et al. Ligand-dependent Notch signaling is involved in tumor initiation and tumor maintenance in pancreatic cancer. Clin Cancer Res. 2009;15(7):2291–2301. doi:10.1158/1078-0432.CCR-08-2004
176. Ding X, Li F, Zhang L. Knockdown of Delta-like 3 restricts lipopolysaccharide-induced inflammation, migration and invasion of A2058 melanoma cells via blocking Twist1-mediated epithelial-mesenchymal transition. Life Sci. 2019;226:149–155. doi:10.1016/j.lfs.2019.04.024
177. Konstantakou EG, Velentzas AD, Anagnostopoulos AK, et al. Deep-proteome mapping of WM-266-4 human metastatic melanoma cells: from oncogenic addiction to druggable targets. PLoS One. 2017;12(2):e0171512. doi:10.1371/journal.pone.0171512
178. Kiniwa Y, Ogawa E, Uchiyama A, Kumagai N, Okuyama R. Delta-like protein 3 promotes proliferation and migration of melanoma via MAPK activation. J Dermatol Sci. 2016;84(1):e176. doi:10.1016/j.jdermsci.2016.08.519
179. Wang J, Zhang K, Liu Z, Wang T, Jia Y. Upregulated delta-like protein 3 expression is a diagnostic and prognostic marker in endometrial cancer: a retrospective study. Medicine. 2018;97(51):e13442. doi:10.1097/MD.0000000000013442
180. Jia D, Underwood J, Xu Q, Xie Q. NOTCH2/NOTCH3/DLL3/MAML1/ADAM17 signaling network is associated with ovarian cancer. Oncol Lett. 2019;17(6):4914–4920. doi:10.3892/ol.2019.10170
181. Yu L, Jiao YJ, Zhou L, Song WQ, Wu SW, Wang DN. [Expressions of OCT4, Notch1 and DLL4 and their clinical implications in epithelial ovarian cancer]. Nan Fang Yi Ke Da Xue Xue Bao. 2016;37(4):444–450.
182. Minuzzo S, Agnusdei V, Pusceddu I, et al. DLL4 regulates NOTCH signaling and growth of T acute lymphoblastic leukemia cells in NOD/SCID mice. Carcinogenesis. 2015;36(1):115–121. doi:10.1093/carcin/bgu223
183. Yang S, Liu Y, Xia B, et al. DLL4 as a predictor of pelvic lymph node metastasis and a novel prognostic biomarker in patients with early-stage cervical cancer. Tumour Biol. 2016;37(4):5063–5074. doi:10.1007/s13277-015-4312-3
184. Lv JY, Hu TY, Wang RY, Zhu JM, Wang G. Deciphering the anti-angiogenic effect of endostatin/cyclophosphamide to normalize tumor micrangium through notch signaling pathway in colon cancer. World J Surg Oncol. 2016;14(1):10. doi:10.1186/s12957-015-0761-9
185. Yoon HA, Noh MH, Kim BG, et al. Clinicopathological significance of altered Notch signaling in extrahepatic cholangiocarcinoma and gallbladder carcinoma. World J Gastroenterol. 2011;17(35):4023–4030. doi:10.3748/wjg.v17.i35.4023
186. Luo Y, Yang ZL, Wang C, et al. The clinicopathological significance of Jagged1 and DLL4 in gallbladder cancer. Tumori. 2017;103(6):557–565. doi:10.5301/tj.5000519
187. Koukourakis MI, Giatromanolaki A, Sivridis E, Gatter KC, Harris AL. High DLL4 expression in tumour-associated vessels predicts for favorable radiotherapy outcome in locally advanced squamous cell head-neck cancer (HNSCC). Angiogenesis. 2013;16(2):343–351. doi:10.1007/s10456-012-9318-5
188. Zhang J-X, Cai MB, Wang XP, et al. Elevated DLL4 expression is correlated with VEGF and predicts poor;prognosis of nasopharyngeal carcinoma. Med Oncol. 2013;30(1):1–10. doi:10.1007/s12032-012-0390-x
189. Wang W, Hu W, Wang Y, et al. Long non-coding RNA UCA1 promotes malignant phenotypes of renal cancer cells by modulating the miR-182-5p/DLL4 axis as a ceRNA. Mol Cancer. 2020;19(1):18. doi:10.1186/s12943-020-1132-x
190. Jurcevic S, Klinga-Levan K, Olsson B, Ejeskar K. Verification of microRNA expression in human endometrial adenocarcinoma. BMC Cancer. 2016;16(1):261. doi:10.1186/s12885-016-2296-z
191. Veliceasa D, Biyashev D, Qin G, et al. Therapeutic manipulation of angiogenesis with miR-27b. Vasc Cell. 2015;7(1):6. doi:10.1186/s13221-015-0031-1
192. El KA, Nofiele J, Giles A, Cho S, Liu SK, Czarnota GJ. Dll4-notch signalling blockade synergizes combined ultrasound-stimulated microbubble and radiation therapy in human colon cancer xenografts. PLoS One. 2014;9(4):e93888. doi:10.1371/journal.pone.0093888
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.