Back to Journals » Degenerative Neurological and Neuromuscular Disease » Volume 5
The role of cell transplantation in modifying the course of limb girdle muscular dystrophy: a longitudinal 5-year study
Authors Sharma A, Sane H, Gokulchandran N, Gandhi S, Bhovad P, Khopkar D, Paranjape A, Bhagwanani K, Badhe P
Received 25 July 2014
Accepted for publication 23 September 2014
Published 14 September 2015 Volume 2015:5 Pages 93—102
DOI https://doi.org/10.2147/DNND.S71775
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Thomas Müller
Alok Sharma,1 Hemangi Sane,2 Nandini Gokulchandran,1 Sushant Gandhi,3 Pradnya Bhovad,3 Dipti Khopkar,2 Amruta Paranjape,2 Khushboo Bhagwanani,3 Prerna Badhe1
1Department of Medical Services and Clinical Research, 2Department of Research and Development, 3Department of Neurorehabilitation, NeuroGen Brain and Spine Institute, Stemasia Hospital and Research Centre, Nerul, Navi Mumbai, Maharashtra, India
Abstract: Limb girdle muscular dystrophy (LGMD), a group of progressive degenerative disorders, causes functional limitation affecting the quality of life. Cell therapy is being widely explored and preliminary studies have shown beneficial effects. Cell therapy induces trophic-factors release, angiogenesis, anti-inflammation, and protein synthesis, which helps in the reparative process at the microcellular level. In this 5-year longitudinal study, the effect of autologous bone marrow mononuclear cells is studied on the natural course of 65 patients with LGMD. Functional Independence Measure and manual muscle testing showed statistically significant improvement, post-cell transplantation. The key finding of this study was demonstration of a plateau phase in the disease progression of the patients. No adverse events were noted. Autologous bone marrow mononuclear cells may be a novel, safe, and effective treatment approach to control the rate of progression of LGMD, thus improving the functional outcomes. Further randomized controlled trials are required.
Keywords: cell therapy, autologous, bone marrow
Introduction
Limb girdle muscular dystrophy (LGMD) is a group of heterogeneous disorders that are caused by gene mutations, thereby resulting in progressive dystrophic muscular weakness.1 LGMD patients present with weakness in both upper and lower limbs, with more weakness in proximal limbs than in distal limbs, depicting a typical involvement of the shoulder and hip girdles. Varying degrees of muscle degeneration on muscle biopsy are noted. Due to the lack of diagnostic specificity and diversity, the prevalence rates for all forms of LGMD are variable.2
At present, no definitive treatment for LGMD exists. Current management aims at improving the overall functional status of the patient, thus enhancing their quality of life. These may include a multidisciplinary rehabilitation management to promote mobility, weight control to reduce obesity, surgical interventions for orthopedic complications, usage of respiratory aids, and monitoring for cardiomyopathy.3 Cell therapy has recently gained attention as a novel treatment option.4 Autologous bone marrow mononuclear cells (BMMNCs) have shown to be a promising approach for therapeutic applications in view of their capacity for self-renewal and differentiation. Mononuclear cells (MNCs) from the adult bone marrow can be easily obtained, show a high degree of genomic stability, and have no ethical concerns, unlike embryonic stem cells. These cells have the ability to maintain, regenerate, and replace terminally differentiated cells within their specific tissue for cell turnover or recovery from tissue injury. They can participate in the regeneration of more than just their specific tissue type. Myogenic differentiation of the MNCs has also been proved in the previous study.5 Thus, autologous BMMNCs have been postulated to promote muscle regeneration, promote repopulation of muscle by host cells, and improve muscle function and pathology, thus translating into enhanced functional outcomes.6–8 Though the genetic defect in patients with LGMD cannot be repaired, the resultant muscle damage, which is progressive with time, can be repaired and the progression may be arrested or slowed down with the cell therapy, thus altering the course of the disease.
To study the benefits of cell therapy, 59 patients of LGMD were administered autologous BMMNCs, intrathecally and intramuscularly.
Materials and methods
Study design
An open label study was carried out on a nonrandomized sample of LGMD patients which spanned across 5 years (ClinicalTrials.gov identifier: NCT02050776). The objective of this study was to analyze if introduction of BMMNCs along with neurorehabilitation helps in halting or slowing down the progressive nature of LGMD. The patients in this study served as self-controls as all the patients showed progressive deterioration in the disease process before cell therapy. The patients underwent intrathecal and intramuscular transplantation of autologous BMMNCs. The protocol of the study was approved by The Institutional Committee for Stem Cell Research and Therapy (IC-SCRT) in accordance with the Indian Council of Medical Research (ICMR) guidelines. A total of 65 patients with LGMD were included in this study. Out of these, six patients were lost to follow-up and therefore they were not included in the analysis (Figure 1).
  | Figure 1 Procedure for patient selection. |
Patient selection
Patients were selected based on the World Medical Association Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects.9 The protocol of the study was approved by IC-SCRT in accordance with ICMR guidelines. The inclusion criteria were patients of both sexes, in the age group of 15 years and above, with LGMD diagnosed on the basis of clinical presentation, electromyographic, and nerve conduction velocity findings. The exclusion criteria were: presence of respiratory distress; presence of acute infections such as HIV/hepatitis B virus/hepatitis C virus; malignancies; and other acute medical conditions such as respiratory infection, fever, hemoglobin less than 8 g/dL, bleeding tendency, bone marrow disorder, left ventricular ejection fraction <30%, and pregnancy or breastfeeding. The intervention was performed only after receiving an informed consent from all the patients and parents/guardians.
Preintervention assessment
Before cell therapy intervention, the patients underwent a comprehensive evaluation consisting of neurological examination, manual muscle testing (MMT), and functional evaluation on the Functional Independence Measure (FIM) scale.10–12 Motor points were identified and plotted for the muscles to be injected by experienced physiotherapists. Motor points are the points in the muscle belly where the nerve enters the muscle for innervation. All the patients underwent routine biochemical, serological, and hematological tests for medical fitness, along with magnetic resonance imaging (MRI), musculoskeletal, and electromyography/nerve conduction velocity studies.
Isolation of BMMNCs
All the patients were administered granulocyte colony-stimulating factor injection 48 hours and 24 hours prior to the procedure, to mobilize the cells and to enhance their numbers.13 Bone marrow aspiration procedure was carried out under local anesthesia with or without sedation depending on the individual case. Approximately 100 mL of bone marrow was aspirated from the anterior superior iliac crest bone using a bone marrow aspiration needle and was collected in heparinized tubes. MNCs were obtained by density gradient separation. The isolated MNCs were checked for viability manually as well as confirmed on TALI machine using propidium iodide. The average viability count was 96%. The MNCs were checked for CD34+ by fluorescence-activated cell sorting using CD34 PE antibody.
Administration of BMMNCs
The separated MNCs (body weight ×106) were administered immediately after separation. Out of these, 50% of the cells were injected intrathecally in the L4–L5 using a lumbar puncture needle. The other 50% of the cells were diluted in the patient’s own cerebrospinal fluid obtained during the lumbar puncture. These were then injected intramuscularly, bilaterally in the motor points of hip extensors, knee extensors, ankle peronei, tibialis anterior, upper abdominals, lower abdominals, back extensors, deltoid, biceps, and triceps. The patients were also on intravenous methylprednisolone (200 mg/kg body weight) in 500 mL Ringer lactate over 1 hour during the procedure of introduction of the cells, to avoid immediate local reaction to the cells.
Neurorehabilitation
After cell transplantation, every patient underwent an individualized neurorehabilitation program designed according to each patient’s requirements, consisting of physiotherapy, occupational therapy, and psychological interventions, which was continued at home.
Monitoring and follow-up
Patients were monitored regularly for any immediate adverse effects in the hospital for 4 days after the cell therapy. The patients were advised to have regular follow-up at 3 months and 6 months, followed by yearly follow-up. During each follow-up, the patients underwent complete neurological assessment and were monitored for any long-term adverse effects. This was a longitudinal study, with follow-ups ranging from minimum of 9 months to maximum of 4.5 years.
Outcome measures
A comprehensive neurological evaluation was carried out for every patient to note any symptomatic changes. The outcome measures used were FIM and MMT.
Repeat doses
After the first treatment of cell therapy, some patients showed mild deterioration after 6 months of improvement. These patients opted for repeat doses of cell therapy. Twenty-four patients underwent second treatments of cell therapy 6–24 months after the first treatment. There were four patients who showed mild deterioration after 6–12 months post-second treatment of cell therapy, thus requiring the third treatment. They underwent the same procedure for cell therapy followed by rehabilitation.
Results
Fifty-nine patients diagnosed with LGMD were included in this longitudinal study, based on the set inclusion and exclusion criteria. These patients underwent autologous BMMNC transplantation intrathecally and intramuscularly. Following cell therapy, the patients showed minor procedure-related adverse events as shown in Table 1. These side effects were self-limiting and subsided in a few days. There were no cell-transplantation-related adverse events.
  | Table 1 Adverse events monitored over a period of 5 years of the study |
The patients were divided into three groups, depending on the number of cell therapy treatments administered. Thirty-one patients were in group 1 and underwent one treatment of cell therapy. Group two included 24 patients administered two treatments, while group 3 included four patients who underwent three transplantation treatments. The results of these patients are analyzed separately. Mean of left and right side of the muscles was taken for each muscle. For statistical analysis, 6-months’ follow-up data after every treatment of cell therapy was used.
Statistical tests used
Descriptive statistics were used to analyze the age, sex, and age of onset of the disease. Wilcoxon signed rank test was used to compare the data before and after the initial and repeated cell therapy treatment. SPSS 20.0 version and Microsoft Word and Excel were used for the computation of data.
Statistical analysis
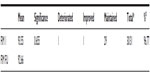
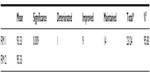
Descriptive analysis was done to analyze the demographic data. Mean age was 32 years with minimum of 16 years and maximum of 57 years, as in Table 2. Sex distribution was calculated as a percentage, with 67% males and 33% females, as described in Table 2. Mean age of onset was 18 years with minimum age of onset of 3 years and maximum of 36 years (Table 2). These results show that the age of onset of disease may vary widely in this population.
  | Table 1 Demographic characteristics |
Results of group 1
MMT
The patients in group 1 (n=31) underwent one treatment of cell therapy with neurorehabilitation. Measurements were taken before the cell therapy and after 6 months of the intervention during the follow-up. The means of pre-cell therapy and post-6-months of cell therapy, significance value between the groups, and the number of muscles improved, deteriorated, and maintained at 6 months are shown in Table 3. Since LGMD is a progressive disease with reduction in muscle strength and mass over time, maintenance of muscle strength is also considered as a marker of improvement in both the groups. Number of muscles improved with percentage of improvement is also shown in Table 3. There was a statistically significant difference (P<0.05) seen in the hip extensors, knee extensors, peronei, tibialis anterior, upper abdominals, and deltoid, indicating improved muscle strength after 6 months of cell therapy. The statistical difference seen can be attributed to the positive changes in the muscle strength. A small number of muscles showed statistically insignificant difference owing to a greater number of muscles maintaining their strength at 6 months. These muscles included lower abdominals, back extensors, biceps, and triceps. Considering the muscle strength maintained at 6 months as a favorable outcome in patients with LGMD, the total percentage of improvement ranged from 84% to 100% in all the muscles.
FIM
Thirty-one patients were assessed on FIM before the transplantation and 6 months following the cell therapy. Throughout this time, all patients were on rehabilitation in the home environment. The analysis showed statistically insignificant difference between the groups as the majority of the patients showed stability in their FIM scores. As the stabilized FIM scores are considered to be a positive therapeutic response, 97% of patients showed better functional status (Table 4).
Results of group 2
MMT
The patients in group 2 (n=24) underwent two treatments of cell therapy with neurorehabilitation. The average time between the two cell therapies treatments ranged from 6 to 12 months. Measurements were taken before the first cell transplantation and 6 months after the second. The means of pre-cell therapy post-6-months of second transplantation, significance value between the groups, and number of muscles improved, deteriorated, and maintained are shown in Table 5. Number of muscles improved with percentage of improvement is also shown in Table 5. There was a statistically significant difference (P<0.05) seen in hip extensors, knee extensors, peronei, tibialis anterior, lower abdominals, deltoid, biceps, and triceps. The significance can be attributed to the greater number of muscles that improved in strength. A small number of muscles showed statistically insignificant difference owing to a greater number of muscles maintaining their strength at 6 months after second treatment of cell therapy, which included upper abdominals and back extensors. Considering the muscle strength maintained at 6 months after the second treatment as a positive outcome, the total percentage of improvement ranged from 90% to 100%.
FIM
All 24 patients were assessed on FIM before the first transplantation and 6 months after the second. Throughout this time, all patients were on rehabilitation in the home environment. There was a statistically significant difference between the groups. Out of 24 patients, one patient showed decreased FIM score, whereas nine patients showed improvement, and 14 showed stability in the scores. Considering the stabilized FIM scores as improvement, 96% of patients improved in their functional status and maintained the same as seen in Table 6.
Results of group 3
Group 3: patients with three treatments of cell therapy
There were four patients who underwent three treatments of cell therapy. Of the four patients, one patient deteriorated in his FIM score, whereas two patients improved, and one maintained functional status. With respect to muscle strength, most of the patients were stabilized. Statistical significance could not be calculated due to the very small number of patients. One patient, who deteriorated on FIM score, also showed deterioration in muscle strength. This patient had come for the cell therapy at an advanced stage of the disease, where the muscle strength was minimal and he was completely dependent for all his activities of daily living. Thus, based on the results obtained, it may be assumed that early intervention may be beneficial in LGMD. Intervention, when targeted before the muscles reach a nonfunctional level, may yield a better outcome.
Discussion
LGMD is a group of muscular disorders which are characterized by degeneration of muscles as a result of a defect in specific skeletal muscle proteins.14 One of the prominent features of LGMD is the involvement of the limb girdles.14 These muscle proteins are responsible for maintaining the integrity of the muscles. Wide genetic and phenotypic inter- and intrafamily heterogeneity is presented by this group of diseases. The absence of production of these muscle proteins leads to mechanical fragility, causing alterations in the muscle fiber contraction.15,16 These alterations at the level of muscle fibers lead to infiltration of inflammatory cells and muscle damage.17 The satellite cells, present beneath the muscle membrane, are in a quiescent state until the presence of muscle injury. The activation of satellite cells promotes muscle regeneration. Thus, there is a continuous cycle of muscle degeneration and regeneration until the later stage of the disease, where the pool of satellite cells gets exhausted. Thus, the process of degeneration surpasses the process of regeneration, causing replacement of muscle fibers by fibrotic and adipose tissue, thus altering the structural and functional components of skeletal muscles.18
The natural course of LGMD is progressive, with deterioration of muscle strength, directly affecting the functional status of the patients. Clinically, these sets of patients exhibit wide variability in symptoms, even between individuals with the same type of LGMD. The age of onset varies from infancy to adulthood. Onset usually begins as muscle weakness around hips and shoulders, causing difficulties in rising from a chair, climbing stairs, lifting heavy objects, holding arms outstretched, and overhead activities. Sometimes, trunk muscles can undergo weakness, causing scoliosis. As the disease progresses, waddling gait is evidently seen. Enlargement or decrease in size of muscles is seen as the disease progresses. Eventually, walking may become impossible, leading to the patient becoming wheelchair bound for mobility. Some individuals develop joint pain and muscle soreness as a result of reduced mobility, leading to joint contractures and muscle cramps. LGMD is also associated with occasional weakening of heart muscles and/or respiratory muscles leading to difficulty in breathing. Cardiomyopathy may be seen in the later stages of the disease.19
Currently, there are no effective treatment approaches developed, which can alter the disease process by either halting the progression or improving the functional outcomes in these sets of patients. Numerous studies have been conducted in the past, trying to unravel the potential of cell-based therapies in promoting muscle regeneration. Preliminary studies in the field of cell therapy for dystrophy involved use of adult myoblasts derived from satellite cells in an attempt to develop new or hybrid muscle fibers.20 However, transplantation of these adult myoblasts has failed to show remarkable results owing to the reduced survival and limited migratory capacity of the transplanted cells.21 BMMNCs appear to contribute to the regeneration of skeletal muscles which is demonstrated in the literature as they possess myogenic properties.8 Cell therapy, though not a cure, as it does not repair the underlying genetic mutation, definitely has potential to repair the subsequent muscle damage. Therefore, the capacity of cell therapy to alter the course of the disease needs to be studied.
Studies have also demonstrated the efficacy of autologous bone-marrow derived MNCs in improving the quality of life of patients with muscular dystrophy.6,7,22 The main aim of cell therapy in patients with muscular dystrophy is to directly aim at regeneration of wasted adult muscle fibers through systemic and targeted injection of cells. These cells work toward blocking the deleterious process of muscle loss, thereby restoring, at least partially, the normal muscle function.23 The MNCs exert reparative effects at the site of injury. They express paracrine effects by signaling factors including cytokines and other growth factors which act as catalysts for the stem cell-driven process by increasing angiogenesis, decreasing inflammation, preventing apoptosis, releasing chemotactic factors, assisting in remodeling of extracellular matrix, and activating satellite cells.24
Here, we discuss the rationale behind the selection of the particular source of cells and route of administration. Autologous stem cells were the adopted source, as they would contribute to muscle regeneration and reconstitute the pool of satellite cells without causing any immunologic reaction or chances of tumor formation. The MNCs derived from the bone marrow are the most easily accessible and most studied source of stem cell. Initially, the bone marrow was thought to contain only hematopoietic type of stem cells. However, an increasing amount of evidence has shed light on this concept. The bone marrow cells comprise hematopoietic stem cells, tissue-specific progenitor cells, stromal cells, and specialized blood cells in different stages of development. These cells differ in their potential to differentiate and form cells, giving rise to tissues which are different from the main stem cell.24 In an animal study, it has been demonstrated that intramuscular injection of myoblasts into muscular dystrophy (mdx) mice, which lack dystrophin, resulted in fusion with host fibers and extensive dystrophin production.25 There are evidences seen in the past for involvement of neural structures in muscular dystrophy. The motor end plates terminating in the healthy muscles in dystrophic mice were abnormal. They were characterized by low densities of synaptic vesicles at the nerve endings and other related findings.26–28 Thus, intrathecal route was the choice, as it is the closest environment to the nervous system. Intramuscular injections are administered at the motor points plotted bilaterally on muscles by physiotherapists experienced in the field. As motor points are the points where the innervating nerve enters the muscle, delivery of cells at this point is facilitated and further implantation of these cells in the muscle is more efficient. Thus, implantation of cells in the muscles aids in enhancing the effect of these cells on the degenerating muscles.
The objective of this study was to analyze if the introduction of stem cells along with neurorehabilitation helped in halting the progression of the disease, and if it could improve the structure and function of the skeletal muscles in all 59 patients. These patients served as self-controls, as they exhibited progressive disease, with respect to constantly decreasing muscle strength and deteriorating function. In our study, we have considered three groups, as mentioned, depending on the number of cell therapy transplantations. Each of the patients in every group followed up at 3 months, 6 months, and yearly thereafter. The duration between two treatments of cell therapy was 6–12 months.
Group 1: patients with one treatment of cell therapy
The muscles of patients undergoing one treatment of cell therapy showed statistically significant improvement in six out of ten muscles injected, whereas in the other four muscles, the strength was stabilized. Clinically, stabilized muscle strength is also considered as an improvement due to the progressive nature of the disease. Over the time of 6 months, the percentage of muscles improved in their strength, including stabilization, ranged from 83.87% to 100%. As a result, the FIM scores remained stable after 6 months of cell therapy. In the patients who did not respond (four out of 31) to cell therapy, one of the possibilities could be noncompliance to the home program. Experimental studies show that a combination of various therapies such as cellular therapies along with exercise or including neurofacilitation techniques together yield a better outcome as opposed to single strategies used independently.29 The other patients have achieved a plateau phase in the course of their condition and have stabilized functional status, with no reports of deterioration.
Group 2: patients with two treatments of cell therapy
The patients in group 2, where all the candidates underwent two treatments of stem cell therapy, opted for the second treatment, as there was a mild deterioration observed 6 months after the first cell therapy. In these patients, the strength of muscles showed statistically significant improvement in eight out of ten muscles injected, whereas in the other two muscles, the strength was stabilized. The FIM scores showed statistically significant difference at 6 months post-second cell therapy. Clinically, overall the patients maintained a plateau stage at a mean of 3 years after the second treatment with cell therapy. One patient showed deterioration as a result of overexerting the muscles during the rehabilitation in the home environment, causing faster deterioration of muscles. Thus, the regime of muscle strengthening in these groups of patients has to be incorporated with care. The strengthening program should focus on low-intensity, high-frequency exercises, with adequate rest periods.
Patients in groups 1 and 2 showed a plateau phase in their disease process until 3 years after their cell therapy.
Group 3: patients with three treatments of cell therapy
There were four patients who underwent three treatments of cell therapy. Of the four patients, one patient deteriorated in his FIM score, whereas two patients improved, and one maintained the functional status. With respect to muscle strength, most of the patients were stabilized. Statistical significance could not be calculated due to the very small number of patients. One patient, who deteriorated on FIM score, also showed deterioration in muscle strength. This patient had come for the cell therapy at an advanced stage of the disease, where the muscle strength was minimal and he was completely dependent for all his activities of daily living. Thus, based on the results obtained, it may be assumed that early intervention may be beneficial in LGMD. Intervention, when targeted before the muscles reach a nonfunctional level may yield a better outcome.
Out of 24 patients who underwent two treatments of cell therapy, MRI/musculoskeletal imaging was available for 15 patients. Out of these, two patients deteriorated in their subsequent MRI, suggesting increased fatty infiltration in a few muscle groups. One patient showed improvement, whereas 12 patients remained stable and there were no changes seen in the subsequent scans. Though MRI/musculoskeletal imaging is an emerging tool in the assessment of the muscle structure, it gives us an insight only at the macrocellular level, which is structural, and not of the microcellular level, which is more physiological. Thus, the changes happening at the physiological level remain less understood. Further research needs to be done to get an insight into the changes happening at the physiological level.
The clinical observations of the study are as follows. Recovery of the patients with respect to maintenance and improvement depends on a few factors which directly affect the impact of cell therapy in patients with LGMD. These factors include the stage at which the patients present to us, the overall health status of the patient, factors influencing the effect of cell therapy like fever and infection occurring naturally to the patient, and so on. Cell therapy showed a major amount of improvement in muscles that scored higher on manual muscle testing and helped them to push toward a better functional level. It is probably unable to improve the functional status of those muscles that are quite weak and are in the late stages of the disease, where the muscle tissue is replaced with fibrous and adipose tissue and has lost the property of regeneration completely. Thus, targeting the muscles that are just below the functional level has a better possibility of improving function. Cell therapy demonstrated improved muscle strength of the muscles, leading to better functional outcomes.
Overall, patients who underwent two treatments of cell therapy had better outcomes with respect to muscle strength and functional status. Almost all patients in this group have maintained their functional status 3 years after the second cell therapy, and have achieved a plateau in their disease process. These results can be compared with the deteriorating status of these patients before the cell therapy, as they were their own self-controls. The small number of patients in group 3, who underwent three transplantations of cell therapy, shows that very few patients need to opt in for the third treatment. Also, the patients in this group were in the advanced stages of the disease.
Back pain is a common factor experienced by these sets of patients. The pain emerges as a result of imbalance between abdominals and back extensors, mainly the paraspinal muscles, causing the load to shift on the facet joints. Overloading of these facet joints causes these patients to experience back pain especially in antigravity postures and activities, including prolonged standing and walking long distances. One of the important findings of this study was the reduced perception of fatigue and pain, leading to improved stamina of the patients during prolonged standing and walking as well as being able to perform activities for prolonged periods of time.
With respect to the intramuscular injections, the amount of stem cells to be injected is limited. Selection of near-functional-level muscles rather than extremely weak muscles should be considered, while at the same time keeping in mind the targeting of more of the girdle muscles for better outcomes with respect to the strength of the muscles and functionally. Thus, careful selection of the number of muscles and their motor points is of paramount importance. MRI/musculoskeletal imaging is a good technique to assess the structural changes happening, but finding an investigation technique where changes happening at the microcellular level can be visualized is the need of the hour.
The limitations of this study are that it is a single-center study. It is a nonrandomized study without a control group. Also, objective imaging along with the assessment of growth factors which would have given more information about the physiological process occurring at cellular level could not be done.
Conclusion
Autologous BMMNC transplantation in combination with a comprehensive rehabilitation program is a safe and feasible treatment option for LGMD. It may help to control the disease progression along with providing functional improvements, thereby enhancing quality of life. Further large randomized controlled trials are needed for conclusive findings.
Disclosure
The authors report no conflicts of interest in this work.
References
Mitsuhashi S, Kang PB. Update on the genetics of limb girdle muscular dystrophy. Semin Pediatr Neurol. 2012;19:211–218. | |
Zatz M, Vainzof M, Passos-Bueno MR. Limb-girdle muscular dystrophy: one gene with different phenotypes, one phenotype with different genes. Curr Opin Neurol. 2000;13(5):511–517. | |
Pegoraro E, Hoffman EP. Limb-Girdle Muscular Dystrophy Overview. In: Pagon RA, Adam MP, Ardinger HH, et al, editors. GeneReviews® [Internet]. Seattle: University of Washington, Seattle: 2000 [updated August 30, 2012]. | |
Meregalli M, Farini A, Parolini D, Maciotta S, Torrente Y. Stem cell therapies to treat muscular dystrophy: progress to date. BioDrugs. 2010;24(4):237–247. | |
Bittner RE, Schöfer C, Weipoltshammer K, et al. Recruitment of bone-marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anat Embryol (Berl). 1999;199:391–396. | |
Sharma A, Gokulchandran N, Chopra G, et al. Administration of autologous bone marrow-derived mononuclear cells in children with incurable neurological disorders and injury is safe and improves their quality of life. Cell Transplant. 2012;21 Suppl 1:S79–S90. | |
Sharma A, Paranjape A, Sane H, Bhagawanani K, Gokulchandran N, Badhe P. Cellular transplantation alters the disease progression in Becker’s muscular dystrophy. Case Rep Transplant. 2013;2013:909328. | |
Luth ES, Jun SJ, Wessen MK, Liadaki K, Gussoni E, Kunkel LM. Bone marrow side population cells are enriched for progenitors capable of myogenic differentiation. J Cell Sci. 2008;121:1426–1434. | |
Carlson RV, Boyd KM, Webb DJ. The revision of the Declaration of Helsinki: past, present and future. Br J Clin Pharmacol. 2004;57: 695–713. | |
Rider LG, Koziol D, Giannini EH, et al. Validation of manual muscle testing and a subset of eight muscles for adult and juvenile idiopathic inflammatory myopathies. Arthritis Care Res (Hoboken). 2010;62(4):465–472. | |
Ottenbacher KJ1, Hsu Y, Granger CV, Fiedler RC. The reliability of the functional independence measure: A quantitative review. Arch Phys Med Rehabil. 1996;77(12):1226–1232. | |
Stineman MG, Shea JA, Jette A, et al. The Functional Independence Measure: tests of scaling assumptions, structure, and reliability across 20 diverse impairment categories. Arch Phys Med Rehabil. 1996;77:1101–1108. | |
Urdzíková L1, Jendelová P, Glogarová K, Burian M, Hájek M, Syková E. Transplantation of bone marrow stem cells as well as mobilization by granulocyte-colony stimulating factor promotes recovery after spinal cord injury in rats. J Neurotrauma. 2006;23(9):1379–1391. | |
Salani S, Donadoni C, Rizzo F, Bresolin N, Comi GP, Corti S. Generation of skeletal muscle cells from embryonic and induced pluripotent stem cells as an in vitro model and for therapy of muscular dystrophies. J Cell Mol Med. 2012;16:1353–1364. | |
McArdle A, Edwards RH, Jackson MJ. How does dystrophin deficiency lead to muscle degeneration? – evidence from the mdx mouse. Neuromuscul Disorder. 1995;5:445–456. | |
Tanabe Y, Esaki K, Nomura T. Skeletal muscle pathology in X chromosome-linked muscular dystrophy (mdx) mouse. Acta Neuropathol. 1986:69:91–95. | |
Watkins SC, Cullen MJ. A quantitative study of myonuclear and satellite cell nuclear size in Duchenne muscular dystrophy, polymyositis and normal human skeletal muscle. Anat Rec. 1988;222:6–11. | |
Vainzof M, Passos-Bueno MR, Pavanello RC, Marie SK, Oliveira AS, Zatz M. Sarcoglycanopathies are responsible for 68% of severe autosomal recessive limb-girdle muscular dystrophy in the Brazilian population. J Neurol Sci. 1999;164:44–49. | |
Alok S, Prerna B, Nandini G, et al. Stem cell therapy and other advances in muscular dystrophy. In: Stem Cell Therapy in Muscular Dystrophy. NeuroGen Publication. 2014;(1):1–13. | |
Brussee V, Tardif F, Roy B, Goulet M, Sebille A, Tremblay JP. Successful myoblast transplantation in fibrotic muscles: no increased impairment by the connective tissue. Transplantation. 1999;67:1618–1622. | |
Vilquin JT. Myoblast transplantation: clinical trials and perspectives. Mini-review. Acta Myol. 2005;24:119–127. | |
Sharma A, Kulkarni P, Chopra G, Gokulchandran N, Lohia M, Badhe P. autologous bone marrow-derived mononuclear cell transplantation in duchenne muscular dystrophy. Indian Journal of Clinical Practice. 2012;23:169–172. | |
Orlic D, Kajstura J, Chimenti S, Bodine DM, Leri A, Anversa P. Bone marrow stem cells regenerate infracted myocardium. Pediatr Transplant. 2003;7:86–88. | |
Alok S, Nandini G. Stem cell therapy in neurological disorders. In: Sharma A, Gokulchnadran N, editors. Mechanism of Action. Mumbai: Surekha Press; 2010:61–69. | |
Skuk D, Roy B, Goulet M, et al. Dystrophin expression in myofibers of Duchenne muscular dystrophy patients following intramuscular injections of normal myogenic cells. Mol Ther. 2004;9:475–482. | |
Blitzblau R, Storer E, Jacob M. Dystrophin and utrophin isoforms are expressed in glia, but not neurons, of the avian parasympathetic ciliary ganglion. Brain Res. 2008;1218:21–34. | |
Luo L, Zhou HY. [Co-transplantation of myoblasts and schwann cells in the therapy of Duchenne muscular dystrophy]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2011;42(1):101–105. Chinese. | |
Ragab AH. Motor end plate changes in mouse muscular dystrophy. Lancet. 1971;2:815–816. | |
Wahl P, Brixius K, Bloch W. Exercise-induced stem cell activation and its implication for cardiovascular and skeletal muscle regeneration. Minim Invasive Ther Allied Technol. 2008;17:91–99. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.