Back to Journals » Research and Reports in Neonatology » Volume 4
The potential of erythropoietin to treat asphyxia in newborns
Received 30 July 2014
Accepted for publication 30 September 2014
Published 18 November 2014 Volume 2014:4 Pages 195—207
DOI https://doi.org/10.2147/RRN.S52375
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Robert Schelonka
Gillian C Pet, Sandra E Juul
Department of Pediatrics, Division of Neonatology, University of Washington, Seattle, WA, USA
Abstract: Perinatal asphyxia is a cause of significant neonatal morbidity worldwide. Lack of oxygenation and perfusion to the neonatal brain leads to energy failure and cell death. Currently, therapeutic hypothermia is the standard of care for term infants with hypoxic-ischemic encephalopathy, but as it has shown only modest effects on survival and morbidity, additional neuroprotective agents are needed. Erythropoietin has been extensively studied as a neuroprotective agent for infants who suffer a hypoxic-ischemic brain injury. It has multiple mechanisms of action, in both preventing cell death and promoting tissue repair. Studies have progressed over time from in vitro to in vivo studies, first in animals and now in humans, with several Phase I/II trials completed and Phase III trials underway. As therapeutic hypothermia has become the standard of care in treating term infants with hypoxic-ischemic encephalopathy, studies must now evaluate other neuroprotective agents, including erythropoietin, used in concert with therapeutic hypothermia. Erythropoietin has shown promise as a neuroprotective agent in animal and human models, both alone and together with hypothermia.
Keywords: neonate, brain injury
Perinatal asphyxia
Lack of oxygen and tissue perfusion in the perinatal period can lead to neonatal hypoxic-ischemic encephalopathy (HIE), which occurs in one to three/1,000 live births in developed countries.1 In 2008, it was estimated that birth asphyxia caused between 563,000 and 997,000 deaths worldwide, 9% of all deaths in children younger than 5 years of age.2 Recently, therapeutic hypothermia has proven to be effective at improving mortality and neurodevelopmental outcomes in infants with moderate-to-severe HIE.3,4 However, even with therapeutic hypothermia, HIE still causes significant morbidity and mortality, with approximately 48% of infants dying or having major neurodevelopmental disability at 18 months of age.4 Additional interventions are clearly needed to further improve outcomes, and these must be tested in the context of therapeutic hypothermia.
Mechanisms of brain injury
Perinatal asphyxia results from disruption in cerebral perfusion and oxygenation, often caused by an interruption in blood flow and gas exchange across the placenta. The resulting brain injury is characterized by an evolving process, which spans the period of initial interruption of blood flow through the period of recovery after reperfusion. The first phase occurs during the period of decreased oxygen delivery to the infant. The body must switch to anaerobic metabolism, resulting in significantly less adenosine triphosphate (ATP) being generated for each molecule of glucose metabolized. The decreased availability of ATP causes failure of the ATP-dependent Na+/K+ pump, leading to a sodium influx into cells. The sodium influx is followed by chloride and water influx, leading to cellular swelling and, eventually, lysis with cell death by necrosis.5 The failure of the ATP-dependent Na+/K+ pump also causes membrane depolarization, leading to increased glutamate release and decreased glutamate uptake. The increased concentration of extracellular glutamate, along with activation of ion-gated calcium channels and failure of energy-dependent processes of calcium removal from the cell, causes accumulation of calcium in the cytosol, which has significant negative effects including membrane injury, generation of free radicals and nitric oxide, and further decreases in ATP production.5,6 The number of cells that die during this initial phase is related to the severity of the insult, with a higher number of cells dying in the initial phase after a more severe insult.6 The next phase consists of secondary energy failure that occurs 6 to 48+ hours after the original injury and involves inflammation, cytotoxic edema, nitric oxide synthesis, mitochondrial dysfunction, and further accumulation of excitotoxins.6,7 This phase correlates best with neurodevelopmental outcomes and has the potential to be affected by neuroprotective interventions.3,6,8 Hypoxia and ischemia can cause injury to both white and gray matter regions, depending on the type, duration, timing, and other circumstances of the injury. In term infants, the most common patterns of injury include watershed injury (plus cortical gray matter injury when severe), deep gray matter injury (involving deep grey nuclei, hippocampi, and perirolandic cortex, with additional cortical damage when severe), and multicystic encephalopathy in infants who experience an acute event superimposed on more chronic mild-to-moderate hypoxia.9
Mechanisms of cell death
Three mechanisms of cell death can occur in response to hypoxic-ischemic injury: necrosis, apoptosis, and autophagy. These cell death programs are complex, interrelated, and involve signaling pathways which can potentially be interrupted or modified, allowing for targeted neuroprotective strategies.10 Necrotic cell death tends to occur early following HIE. It is characterized by profound cellular swelling leading to cell rupture, membrane disintegration, and release of intracellular contents.5 Necrotic cell death requires less energy than apoptosis or autophagy, but still involves activation of specific signaling pathways.11,12 Neuronal necrosis that occurs in the context of excitotoxicity and hypoxic-ischemic injury is mediated by membrane depolarization caused by glutamate-triggered influx of calcium into the cell. Necrosis occurs predominantly in sites of profound energy deprivation, such as the core of an ischemic region, and is responsible for much of the immediate cell death during the first phase after injury, but there is a continuum between necrotic and apoptotic cell death.13,14 In contrast to necrosis, apoptosis is a form of programmed cell death characterized by immunologically silent cell shrinkage with nuclear pyknosis and intact plasma membranes.5 It can be activated by intrinsic or extrinsic pathways. The intrinsic, or mitochondrial, pathway depends on the balance of antiapoptotic proteins (such as Bcl-2 and Bcl-xL) and proapoptotic proteins (such as BAX and BAD).15,16 Apoptosis can also be triggered by external signals such as Fas ligand and tumor necrosis factor-alpha (TNF-α) activation of proapoptotic receptors on the cell surface, which is known as the extrinsic pathway. Proapoptotic proteins cause permeabilization of the mitochondrial membrane, allowing factors including cytochrome c to be released into the cytosol, leading to apoptosis.17 Neuroapoptosis following hypoxic-ischemic injury typically occurs in the ischemic penumbra during the secondary phase of brain injury, making this pathway an excellent target for neuroprotection.10,18
Autophagy is a homeostatic process by which unwanted proteins and damaged organelles are eliminated from cells. It is a catabolic process involving intracellular degradation of cytosolic proteins and organelles by autophagosomes, which fuse with lysosomes to form autolysosomes.10 Autophagy is now recognized as a distinct mechanism of cell death that is interrelated to both necrosis and apoptosis.19 There are several proposed mechanisms for the role of autophagy in cell death following hypoxic-ischemic injury, including as an independent mechanism and as a trigger for apoptotic cell death.10
The mechanism of cell death that predominates in hypoxic-ischemic injury is influenced by characteristics of the individual including age (neonates are more sensitive to apoptosis than adults, and the location of calcium permeable membrane receptors switches from white matter to gray matter over time), sex (different specific pathways predominate in males compared to females), and other factors, such as energy availability.20–22 Many cells die from hybrids of multiple pathways (such as apoptosis and necrosis or apoptosis and autophagy), as there are significant interconnections between pathways.23,24 The overlap in cell death pathways makes identifying targets for neuroprotective agents complex, because cell death can proceed down an alternative pathway if one pathway is inhibited.10 Each pathway is important for normal development, thus blocking all pathways completely can have negative effects.25–28 As understanding of the complex interactions between mechanisms of cell death and survival improves, neuroprotective strategies may include use of multiple complimentary agents, either to target different pathways or to use one drug to extend the therapeutic window for another.
Therapeutic hypothermia
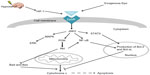
Therapeutic hypothermia has become the standard of care treatment for HIE.3,29 It has multiple neuroprotective effects, including decreased energy depletion, inhibition of glutamate release and decreased impairment in glutamate reuptake, deceased free radical generation and inflammation, and blockade of pathways leading to apoptosis (Figure 1).5 It has been shown in trials to reduce the risk of death or major neurodevelopmental disability by approximately 50% with a number needed to treat of 7–9.3,30 As hypothermia has become the standard of care, research into other neuroprotective agents, particularly in humans but also in animal models, has shifted from study of a neuroprotective agent alone to investigation of the combined effects of the agent along with hypothermia.
Erythropoietin
Erythropoietin (Epo) is a 30.4 kDa hematopoietic cytokine that was originally recognized for its role in erythropoiesis. It is produced primarily in the kidney of adults and the liver of fetuses, although Epo production also occurs in the brain, testis, and placenta.31,32 Endogenous Epo is required for normal brain development, function, and repair. Epo is primarily produced by astrocytes but can also be detected in oligodendrocytes, neurons, endothelial cells, and microglia.33–38 In the setting of hypoxia-ischemia, Epo receptors (EpoRs) in neurons, astrocytes, and microglia are massively upregulated.39,40 Increased Epo expression follows, via hypoxia-mediated stabilization of neuronal transcription factor hypoxia-inducible factor 1α, if the insult is of sufficient duration.41,42 Hypoxia-inducible factor-2 (HIF-2) has also been found to regulate the production of Epo in response to hypoxia in many tissues, though its precise role is less clear.43–46 In the absence of Epo–EpoR binding, cells are predisposed to apoptosis, while, in the presence of Epo, cells are preserved.47,48 This creates an important rationale for exogenous Epo administration, given that brain injury can occur after brief but catastrophic insults, such as placental abruption or cord accidents, which are insufficient to stimulate an increase in endogenous Epo synthesis.49
Mechanisms of action of Epo
Epo binds to the EpoR homodimer, which activates Jak2 kinase to phosphorylate Jak2 and EpoR (Figure 2).50–52 This activates multiple signaling cascades, including MAPK/ERK, PI3K/Akt, Stat5, and NF-κB.53,54 NF-κB and Stat5 move into the nucleus and act as transcription factors in the production of Bcl-2 and Bcl-xL, which are antiapoptotic proteins.40,55 Epo also inhibits the function of Bax and Bad, which are proapoptotic, via AKT. The balance of these proteins determines whether a cell undergoes apoptosis.40 In addition to the EpoR homodimer, other receptor complexes have been implicated in the neuroprotective effects of Epo. In particular, the common beta receptor (CβR)–EpoR heterodimer has been found in some studies to be essential in Epo neuroprotective abilities, but other studies have not confirmed these findings.56,57 These signaling pathways have multiple downstream neuroprotective and neurotrophic effects.
Epo appears to have both acute and long-term effects following brain injury (Figure 1). Through multiple mechanisms, Epo decreases cell death acutely and also promotes cell and tissue repair, affecting many components of the neurovascular unit. Many studies have demonstrated Epo’s antiapoptotic effects.50,53,58–69 Additionally, Epo has also been shown to have anti-inflammatory, neurotrophic, and antioxidant properties, along with having a role in promoting angiogenesis, neurogenesis, and oligodendrogenesis.47,70–80 Epo may protect the brain from edema by upregulation of aquaporin channels.81 Epo also increases reticulocytosis in preterm infants, which in turn increases iron utilization. When iron is unbound, it can produce free radicals that cause oxidative injury, so, by increasing iron utilization, Epo may secondarily decrease injury.82,83
Animal and human studies of Epo
The effects of Epo on neonatal brain injury have been studied in multiple animal models (Table 1), most commonly in the rat model of unilateral carotid ligation followed by hypoxia (Vannucci model) and the middle cerebral artery occlusion model.84,85 These models are commonly used to produce gray matter injury similar to that seen in term infants who experience hypoxic-ischemic injury or perinatal stroke, respectively. Animal models have been used both to demonstrate Epo’s neuroprotective effects on gross and histological brain injury and neurobehavioral outcomes and to elucidate the mechanism of neuroprotection. There is some variability in the results of these studies, likely related to variability in methodology, including duration of hypoxia/ischemia; timing, dose, and frequency of Epo administration; and timing of the outcome studied. Overall, however, Epo has been shown to be protective over a wide range of doses, in multiple animal models, by multiple investigators (see Table 1).
As hypothermia became the standard of care for neonates with HIE, Epo began to be incorporated in animal studies of HIE.49,86,87 Fan et al published a study in 2012 comparing no treatment, hypothermia alone, Epo alone, and hypothermia + Epo on brain injury and behavior in rats subject to a hypoxic-ischemic insult using the Vannucci model.87 Hypothermia in this study consisted of 3 hours at 32.5°C–33°C immediately after the hypoxic period was complete. Epo was dosed at 5,000 U/kg given intraperitoneally immediately after hypothermia and repeated 24 and 48 hours later. The investigators found that administration of Epo alone modestly improved behavioral outcomes at 2 and 5 weeks post-injury (measured using the cylindrical rearing test) but had no effect on histologic brain injury. Similarly, Epo in addition to hypothermia had a mildly additive effect on hypothermia alone in improving behavioral outcomes but no additive effect for histological injury. Interestingly, the authors found that the neuroprotective effects of hypothermia were more pronounced in female animals, while Epo did not have sex-specific effects. In the same journal, Fang et al published a similar study evaluating the effects of Epo and hypothermia on neonatal rats subjected to hypoxic-ischemic injury using the Vannucci model.86 In this study, therapeutic hypothermia consisted of 8 hours at 32°C and Epo was dosed at 1,000 U/kg, given immediately after injury but prior to hypothermia and repeated at 24 hours and 7 days post-injury. The authors found no differences between untreated animals and animals treated with hypothermia, Epo, or both in either histopathological or behavioral outcomes other than improved histopathological outcomes in male animals treated with Epo. Traudt et al completed a study in 2013 comparing hypothermia alone to hypothermia + Epo in a nonhuman primate (pigtail macaque) model of perinatal asphyxia.49 The macaques were exposed to 15 or 18 minutes of umbilical cord occlusion and were then treated with 72 hours of therapeutic hypothermia at 33.5°C with or without Epo or were untreated. Epo was initially dosed intravenously at 3,500 U/kg for one dose, followed by three doses of 2,500 U/kg given at 24 and 72 hours and 7 days post-injury, but was then switched to 1,000 U/kg for all four doses based on pharmacokinetic data. The authors found that, among macaques exposed to umbilical cord occlusion, there was a 44% incidence of death or moderate-to-severe cerebral palsy in the untreated animals and a 43% incidence in the animals treated with hypothermia alone compared to a 0% incidence in the hypothermia + Epo group. Animals treated with hypothermia + Epo also showed improvement in motor and cognitive outcomes, cerebellar growth, and diffusion tensor imaging (DTI) measures.
These three studies clearly have disparate results regarding the efficacy of both hypothermia and Epo. This variability could be related to many factors, including differences in the models used, Epo dosing, and the duration and degree of hypothermia.88 Traudt’s study is the first study of Epo and hypothermia in a nonhuman primate model of HIE. Large-animal models of brain injury offer both advantages and disadvantages compared to small animal models. Large animals (sheep, piglets, nonhuman primates) are expensive and require more resources for their care, thus it is not possible to include the same number of subjects as are typically included in small-animal studies. However, in many ways, their brains are more similar to those of humans. Large animals possess a gyrencephalic brain, a white to gray matter ratio that better approximates that of humans, and similar vascular patterns to humans and they can be monitored and cared for in a manner similar to neonates in an intensive care unit.89
Human studies of Epo
In the past 5 years, several studies on the neuroprotective effects of Epo on human infants with HIE have been published (Table 2). The initial study by Zhu et al in 2009 compared Epo to supportive care in infants with moderate-to-severe encephalopathy.90 Epo was dosed at either 300 or 500 U/kg and given subcutaneously immediately following injury and repeated every other day for 2 weeks intravenously. The authors demonstrated decreased incidence of moderate-to-severe disability or death at 18 months of age in infants given either of the two doses of Epo, particularly in infants with moderate compared to severe HIE, without adverse hematopoietic side effects. The second study, by Elmahdy et al in 2010, compared Epo to supportive care in infants with mild-to-moderate HIE.91 Epo was dosed at 2,500 U/kg subcutaneously, started within 4–6 hours of injury, and repeated daily for five total doses. These authors also demonstrated improved outcomes in infants treated with Epo, including decreased seizure activity, decreased endogenous nitric oxide production, and improved neurodevelopmental outcomes up to 6 months. The third study, by Wu et al in 2012, was a Phase I/II study of the safety and pharmacokinetics of Epo at escalating doses in infants with HIE being treated with therapeutic hypothermia.92 Doses ranged from 250 to 2,500 U/kg and were administered intravenously, starting at less than 24 hours of age and continuing every 48 hours for up to six total doses. The authors showed that dosing at 1,000 U/kg produced plasma concentrations similar to those found to be neuroprotective in animals and was well tolerated. At mean age 22 months, infants who received Epo exhibited a relatively low rate of moderate-to-severe disability, even in the setting of significant brain injury.93 The most recent study, by El Shimi et al in 2014, examined whether a single dose of Epo was as safe and effective as hypothermia in treating HIE, given that hypothermia was not available in many lower-resource nations, despite being the standard of care for HIE in developed nations.94 Epo was dosed at 1,500 U/kg given subcutaneously on day 1 of life. Hypothermia was accomplished using cold packs to maintain a rectal temperature between 33°C and 34°C for 72 hours. This small study showed improved survival in the infants treated with hypothermia compared to single-dose Epo and supportive care, particularly in infants with moderate or Sarnat stage II encephalopathy. There was a trend toward improved MRI brain injury score and functional outcomes in infants treated with hypothermia. Larger Phase III studies to test efficacy are planned or under way in France (Neurepo, NCT01732146), Australia (PAEAN), and the US (NEAT O, NCT01913340).
  | Table 2 Human infant studies of Epo |
Epo dosing and adverse effects
The optimal dosing regimen in human neonates is unknown. Epo is ineffective at promoting neuroprotection at very low doses and may cause harm at very high doses.82,95 Kellert et al’s dose comparison study in rats demonstrated that three doses of 5,000 U/kg resulted in the most consistent neuroprotection with the lowest total dose exposure.62 Traudt et al’s study, which showed strong benefits of Epo when used in conjunction with hypothermia, found that dosing hypothermic macaques at 1,000 U/kg produced similar pharmacokinetic parameters to rats dosed at 5,000 U/kg.49 Interestingly, these authors noted that a dosing regimen in hypothermic macaques produced a 25% higher peak Epo concentration than expected based on pharmacokinetic data obtained in normothermic human neonates, suggesting that hypothermia alters Epo’s pharmacokinetics. Wu et al’s pharmacokinetic study of Epo in human neonates undergoing therapeutic hypothermia for HIE also found that 1,000 U/kg of Epo produced similar pharmacokinetic parameters as doses that have been found to be neuroprotective in animal models.92
The significance and severity of adverse effects related to Epo administration also remain controversial. As Epo is used primarily as an erythropoietic agent, it certainly has effects on red blood cell formation. Polycythemia has not been seen in trials of neuroprotection in term infants, and two trials did not find a significant difference in hematocrit and number of red blood cell transfusions between treated and untreated infants, although Zhu et al’s trial did show that use of Epo prevented the decrease in hematocrit over time seen in control and hypothermia-only infants.90–94 In adults, Epo has been associated with an increased risk for hypertension, but this has not been the case in neonates.83,96–99 In premature infants, Epo has been linked to several possible adverse effects. Early studies demonstrated a risk of neutropenia after treatment with Epo, but this has not been confirmed in later studies of erythropoietic or neuroprotective dosing of preterm infants.83,97–100 There has also been concern about increased risk of retinopathy of prematurity in preterm infants treated with Epo, particularly early in life, but the data are conflicting and this is not relevant to term infants.98,99,101–104 In one retrospective study, an increased risk of cutaneous hemangiomas was reported with Epo exposure in preterm infants, but, as cutaneous hemangiomas are common in preterm infants, a causal relationship has not been established.83,105–108
There are theoretical concerns regarding clotting abnormalities in infants treated with both Epo and therapeutic hypothermia. Hypothermia has negative effects on hemostasis and leads to increased risk of disseminated intravascular coagulation.109,110 Epo may also affect clot formation, as use of Epo has been associated with increased risk for thromboembolic events in adults with strokes.111 At this time, no studies of normothermic or hypothermic neonates treated with Epo have demonstrated increased risk of clotting abnormalities, but we must continue to be vigilant about this potential adverse effect. Overall, significant adverse effects have not been seen in term or preterm infants treated with Epo.
Limitations of studies to date
Animal models
The animal models described previously all attempt to reproduce hypoxic-ischemic injury in term infants.112 While these models can simulate the human neonatal experience, they are not exact reproductions. Infants that meet clinical criteria for HIE can have had very different antenatal and perinatal experiences. HIE may be caused by a single acute event, such as a placental abruption or umbilical cord prolapse; a more chronic process like intrauterine growth restriction or infection; or a combination of events, such as a difficult delivery in a stressed infant who does not tolerate delivery well due to maternal chorioamnionitis or longstanding placental insufficiency. Thus, infants with HIE who meet entry criteria for a study may in fact have disparate mechanisms of injury, which likely helps to explain variability in response to treatments and, therefore, outcomes. Additionally, it is likely that infants with mild, moderate, and severe HIE will respond differently to neuroprotective therapies.3,90 Many of the studies on the neuroprotective effects of Epo following HIE use the Vannucci model, which models acute, severe hypoxic ischemic brain injury, but does not reflect more chronic or combined inflammatory and hypoxic injuries. Thus, it is important to glean information from a wide variety of animal models and, as clinical trials are planned, to target neuroprotective therapies toward specific mechanisms of injury that show improvement in preclinical trials.
Human studies
There is currently a paucity of human trials available from which to assess the efficacy of Epo neuroprotection for HIE. Phase I/II trials have been undertaken to establish rudimentary safety, pharmacokinetics, and feasibility of larger studies and have provided limited long-term outcome data. Because of the nature of these studies, there is also significant heterogeneity in study design, with Epo doses ranging from 250–2,500 U/kg, dosing intervals ranging from 24–48 hours, and dose numbers ranging from one to seven. Despite these limitations, these studies have laid the groundwork for future Phase III studies in which the safety and efficacy of Epo neuroprotection can be rigorously tested.
Conclusion
In the past 5 years, there has been a significant advancement in treatment of HIE. Therapeutic hypothermia has been proven to improve outcomes, with a number needed to treat of 7. Despite this, infants with moderate-to-severe HIE who receive hypothermia still experience high rates of death (26%) and, among survivors, developmental delay (23%), cerebral palsy (19%), deafness (4%), and blindness (6%). The overall rate of death or major disability despite hypothermia thus remains unacceptably high (48%).4 Adjuvant therapies are therefore needed to further improve outcomes.
As we further our understanding of how cells die after neonatal hypoxic-ischemic brain injury, we can develop new neuroprotective strategies that promote or inhibit specific pathways. The complex and interconnecting pathways of cell death illustrate the need to approach neuroprotection from multiple angles. Several agents, including Epo, have shown promise as neuroprotective agents and are being studied further. If these therapies interact at different points in tissue response and healing pathways following injury, it is possible that, ultimately, a “cocktail” of therapeutic agents will be used to promote optimal healing.
Disclosure
The authors report no conflicts of interest in this work.
References
Graham EM, Ruis KA, Hartman AL, Northington FJ, Fox HE. A systematic review of the role of intrapartum hypoxia-ischemia in the causation of neonatal encephalopathy. Am J Obstet Gynecol. 2008;199(6):587–595. | |
Black RE, Cousens S, Johnson HL, et al; Child Health Epidemiology Reference Group of WHO and UNICEF. Global, regional, and national causes of child mortality in 2008: a systematic analysis. Lancet. 2010; 375(9730):1969–1987. | |
Jacobs SE, Berg M, Hunt R, Tarnow-Mordi WO, Inder TE, Davis PG. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev. 2012;1:CD003311. | |
Tagin MA, Woolcott CG, Vincer MJ, Whyte RK, Stinson DA. Hypothermia for neonatal hypoxic ischemic encephalopathy: an updated systematic review and meta-analysis. Arch Pediatr Adolesc Med. 2012;166(6):558–566. | |
Volpe JJ. Neurology of the Newborn. Elsevier Health Sciences; 2008. | |
Inder TE, Volpe JJ. Mechanisms of perinatal brain injury. Semin Neonatol. 2000;5(1):3–16. | |
Perlman JM. Intervention strategies for neonatal hypoxic-ischemic cerebral injury. Clin Ther. 2006;28(9):1353–1365. | |
Roth SC, Baudin J, Cady E, et al. Relation of deranged neonatal cerebral oxidative metabolism with neurodevelopmental outcome and head circumference at 4 years. Dev Med Child Neurol. 1997;39(11):718–725. | |
Sie LT, van der Knaap MS, Oosting J, de Vries LS, Lafeber HN, Valk J. MR patterns of hypoxic-ischemic brain damage after prenatal, perinatal or postnatal asphyxia. Neuropediatrics. 2000;31(3):128–136. | |
Puyal J, Ginet V, Clarke PG. Multiple interacting cell death mechanisms in the mediation of excitotoxicity and ischemic brain damage: a challenge for neuroprotection. Prog Neurobiol. 2013;105:24–48. | |
Galluzzi L, Berghe TV, Vanlangenakker N, et al. Programmed Necrosis: From Molecules to Health and Disease. 1st ed. Elsevier Inc.; 2011:1–35. | |
Chavez-Valdez R, Martin LJ, Northington FJ. Programmed necrosis: a prominent mechanism of cell death following neonatal brain injury. Neurol Res Int. 2012;2012:257563. | |
Northington FJ, Ferriero DM, Graham EM, Traystman RJ, Martin LJ. Early neurodegeneration after hypoxia-ischemia in neonatal rat is necrosis while delayed neuronal death is apoptosis. Neurobiol Dis. 2001;8(2):207–219. | |
Northington FJ, Chavez-Valdez R, Martin LJ. Neuronal cell death in neonatal hypoxia-ischemia. Ann Neurol. 2011;69(5):743–758. | |
Llambi F, Moldoveanu T, Tait SW, et al. A unified model of mammalian BCL-2 protein family interactions at the mitochondria. Mol Cell. 2011;44(4):517–531. | |
Antonsson B, Martinou JC. The Bcl-2 protein family. Exp Cell Res. 2000;256(1):50–57. | |
Culmsee C, Mattson MP. p53 in neuronal apoptosis. Biochem Biophys Res Commun. 2005;331(3):761–777. | |
Broughton BR, Reutens DC, Sobey CG. Apoptotic mechanisms after cerebral ischemia. Stroke. 2009;40(5):e331–e339. | |
Chaabane W, User SD, El-Gazzah M, et al. Autophagy, apoptosis, mitoptosis and necrosis: interdependence between those pathways and effects on cancer. Arch Immunol Ther Exp (Warsz). 2013;61(1):43–58. | |
Talos DM, Fishman RE, Park H, et al. Developmental regulation of alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptor subunit expression in forebrain and relationship to regional susceptibility to hypoxic/ischemic injury. I. Rodent cerebral white matter and cortex. J Comp Neurol. 2006;497(1):42–60. | |
Renolleau S, Fau S, Charriaut-Marlangue C. Gender-related differences in apoptotic pathways after neonatal cerebral ischemia. Neuroscientist. 2008;14(1):46–52. | |
Zhu C, Wang X, Xu F, et al. The influence of age on apoptotic and other mechanisms of cell death after cerebral hypoxia-ischemia. Cell Death Differ. 2005;12(2):162–176. | |
Nakajima W, Ishida A, Lange MS, et al. Apoptosis has a prolonged role in the neurodegeneration after hypoxic ischemia in the newborn rat. J Neurosci. 2000;20(21):7994–8004. | |
Eisenberg-Lerner A, Bialik S, Simon HU, Kimchi A. Life and death partners: apoptosis, autophagy and the cross-talk between them. Cell Death Differ. 2009;16(7):966–975. | |
Haberny KA, Paule MG, Scallet AC, et al. Ontogeny of the N-methyl-D-aspartate (NMDA) receptor system and susceptibility to neurotoxicity. Toxicol Sci. 2002;68(1):9–17. | |
Hardingham GE, Bading H. The Yin and Yang of NMDA receptor signalling. Trends Neurosci. 2003;26(2):81–89. | |
Ikonomidou C, Bosch F, Miksa M, et al. Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science. 1999;283(5398):70–74. | |
Ikonomidou C, Turski L. Why did NMDA receptor antagonists fail clinical trials for stroke and traumatic brain injury? Lancet Neurol. 2002;1(6):383–386. | |
Committee on Fetus and Newborn, Papile LA, Baley JE, et al. Hypothermia and neonatal encephalopathy. Pediatrics. 2014;133(6):1146–1150. | |
Edwards AD, Brocklehurst P, Gunn AJ, et al. Neurological outcomes at 18 months of age after moderate hypothermia for perinatal hypoxic ischaemic encephalopathy: synthesis and meta-analysis of trial data. BMJ. 2010;340:c363. | |
Dame C, Fahnenstich H, Freitag P, et al. Erythropoietin mRNA expression in human fetal and neonatal tissue. Blood. 1998;92(9):3218–3225. | |
Zanjani ED, Poster J, Burlington H, Mann LI, Wasserman LR. Liver as the primary site of erythropoietin formation in the fetus. J Lab Clin Med. 1977;89(3):640–644. | |
Chen ZY, Asavaritikrai P, Prchal JT, Noguchi CT. Endogenous erythropoietin signaling is required for normal neural progenitor cell proliferation. J Biol Chem. 2007;282(35):25875–25883. | |
Sakanaka M, Wen TC, Matsuda S, et al. In vivo evidence that erythropoietin protects neurons from ischemic damage. Proc Natl Acad Sci U S A. 1998;95(8):4635–4640. | |
Juul SE, Anderson DK, Li Y, Christensen RD. Erythropoietin and erythropoietin receptor in the developing human central nervous system. Pediatr Res. 1998;43(1):40–49. | |
Bernaudin M, Marti HH, Roussel S, et al. A potential role for erythropoietin in focal permanent cerebral ischemia in mice. J Cereb Blood Flow Metab. 1999;19(6):643–651. | |
Masuda S, Okano M, Yamagishi K, Nagao M, Ueda M, Sasaki R. A novel site of erythropoietin production. Oxygen-dependent production in cultured rat astrocytes. J Biol Chem. 1994;269(30):19488–19493. | |
Sugawa M, Sakurai Y, Ishikawa-Ieda Y, Suzuki H, Asou H. Effects of erythropoietin on glial cell development; oligodendrocyte maturation and astrocyte proliferation. Neurosci Res. 2002;44(4):391–403. | |
Wen TC, Rogido M, Genetta T, Sola A. Permanent focal cerebral ischemia activates erythropoietin receptor in the neonatal rat brain. Neurosci Lett. 2004;355(3):165–168. | |
van der Kooij MA, Groenendaal F, Kavelaars A, Heijnen CJ, van Bel F. Neuroprotective properties and mechanisms of erythropoietin in in vitro and in vivo experimental models for hypoxia/ischemia. Brain Res Rev. 2008;59(1):22–33. | |
Tan CC, Eckardt KU, Firth JD, Ratcliffe PJ. Feedback modulation of renal and hepatic erythropoietin mRNA in response to graded anemia and hypoxia. Am J Physiol. 1992;263(3 Pt 2):F474–F481. | |
Jones NM, Bergeron M. Hypoxic preconditioning induces changes in HIF-1 target genes in neonatal rat brain. J Cereb Blood Flow Metab. 2001;21(9):1105–1114. | |
Kumar P. The transcriptional activator hypoxia inducible factor 2 (HIF-2/EPAS-1) regulates the oxygen-dependent expression of erythropoietin in cortical astrocytes. J Neurosci. 2006;26(37):9471–9481. | |
Rankin EB, Biju MP, Liu Q, et al. Hypoxia-inducible factor-2 (HIF-2) regulates hepatic erythropoietin in vivo. J Clin Invest. 2007;117(4):1068–1077. | |
Wiesener MS, Jürgensen JS, Rosenberger C, et al. Widespread hypoxia-inducible expression of HIF-2alpha in distinct cell populations of different organs. FASEB J. 2003;17(2):271–273. | |
Rosenberger C, Mandriota S, Jürgensen JS, et al. Expression of hypoxia-inducible factor-1alpha and -2alpha in hypoxic and ischemic rat kidneys. J Am Soc Nephrol. 2002;13(7):1721–1732. | |
Jantzie LL, Miller RH, Robinson S. Erythropoietin signaling promotes oligodendrocyte development following prenatal systemic hypoxic-ischemic brain injury. Pediatr Res. 2013;74(6):658–667. | |
Mazur M, Miller RH, Robinson S. Postnatal erythropoietin treatment mitigates neural cell loss after systemic prenatal hypoxic-ischemic injury. J Neurosurg Pediatr. 2010;6(3):206–221. | |
Traudt CM, McPherson RJ, Bauer LA, et al. Concurrent erythropoietin and hypothermia treatment improve outcomes in a term nonhuman primate model of perinatal asphyxia. Dev Neurosci. 2013;35(6):491–503. | |
Digicaylioglu M, Lipton SA. Erythropoietin-mediated neuroprotection involves cross-talk between Jak2 and NF-kappaB signalling cascades. Nature. 2001;412(6847):641–647. | |
Kawakami M, Iwasaki S, Sato K, Takahashi M. Erythropoietin inhibits calcium-induced neurotransmitter release from clonal neuronal cells. Biochem Biophys Res Commun. 2000;279(1):293–297. | |
Sola A, Rogido M, Lee BH, Genetta T, Wen TC. Erythropoietin after focal cerebral ischemia activates the Janus kinase-signal transducer and activator of transcription signaling pathway and improves brain injury in postnatal day 7 rats. Pediatr Res. 2005;57(4):481–487. | |
Um M, Lodish HF. Antiapoptotic effects of erythropoietin in differentiated neuroblastoma SH-SY5Y cells require activation of both the STAT5 and AKT signaling pathways. J Biol Chem. 2006;281(9):5648–5656. | |
Shang Y, Wu Y, Yao S, Wang X, Feng D, Yang W. Protective effect of erythropoietin against ketamine-induced apoptosis in cultured rat cortical neurons: involvement of PI3K/Akt and GSK-3 beta pathway. Apoptosis. 2007;12(12):2187–2195. | |
Kumral A, Tüzün F, Oner MG, Genç S, Duman N, Ozkan H. Erythropoietin in neonatal brain protection: The past, the present and the future. Brain Dev. 2011;33(8):632–643. | |
Sanchez PE, Navarro FP, Fares RP, et al. Erythropoietin receptor expression is concordant with erythropoietin but not with common beta chain expression in the rat brain throughout the life span. J Comp Neurol. 2009;514(4):403–414. | |
Brines M, Cerami A. Emerging biological roles for erythropoietin in the nervous system. Nat Rev Neurosci. 2005;6(6):484–494. | |
Ruscher K, Freyer D, Karsch M, et al. Erythropoietin is a paracrine mediator of ischemic tolerance in the brain: evidence from an in vitro model. J Neurosci. 2002;22(23):10291–10301. | |
Wang CH, Liang CL, Huang LT, et al. Single intravenous injection of naked plasmid DNA encoding erythropoietin provides neuroprotection in hypoxia-ischemia rats. Biochem Biophys Res Commun. 2004;314(4):1064–1071. | |
Sun Y, Zhou C, Polk P, Nanda A, Zhang JH. Mechanisms of erythropoietin-induced brain protection in neonatal hypoxia-ischemia rat model. J Cereb Blood Flow Metab. 2004;24(2):259–270. | |
Villa P, Bigini P, Mennini T, et al. Erythropoietin selectively attenuates cytokine production and inflammation in cerebral ischemia by targeting neuronal apoptosis. J Exp Med. 2003;198(6):971–975. | |
Kellert BA, McPherson RJ, Juul SE. A comparison of high-dose recombinant erythropoietin treatment regimens in brain-injured neonatal rats. Pediatr Res. 2007;61(4):451–455. | |
Kaindl AM, Sifringer M, Koppelstaetter A, et al. Erythropoietin protects the developing brain from hyperoxia-induced cell death and proteome changes. Ann Neurol. 2008;64(5):523–534. | |
Wu Y, Shang Y, Sun S, Liang H, Liu R. Erythropoietin prevents PC12 cells from 1-methyl-4-phenylpyridinium ion-induced apoptosis via the Akt/GSK-3beta/caspase-3 mediated signaling pathway. Apoptosis. 2007;12(8):1365–1375. | |
Lee ST, Chu K, Sinn DI, et al. Erythropoietin reduces perihematomal inflammation and cell death with eNOS and STAT3 activations in experimental intracerebral hemorrhage. J Neurochem. 2006;96(6):1728–1739. | |
Yamada M, Burke C, Colditz P, Johnson DW, Gobe GC. Erythropoietin protects against apoptosis and increases expression of non-neuronal cell markers in the hypoxia-injured developing brain. J Pathol. 2011;224(1):101–109. | |
Olgun Y, Kirkim G, Kolatan E, et al. Otoprotective effect of recombinant erythropoietin in a model of newborn hypoxic-ischemic encephalopathy. Int J Pediatr Otorhinolaryngol. 2013;77(5):739–746. | |
Yis U, Kurul SH, Kumral A, et al. Effect of erythropoietin on oxygen-induced brain injury in the newborn rat. Neurosci Lett. 2008;448(3):245–249. | |
Kim MS, Seo YK, Park HJ, et al. The neuroprotective effect of recombinant human erythropoietin via an antiapoptotic mechanism on hypoxic-ischemic brain injury in neonatal rats. Korean J Pediatr. 2010;53(10):898–908. | |
Shingo T, Sorokan ST, Shimazaki T, Weiss S. Erythropoietin regulates the in vitro and in vivo production of neuronal progenitors by mammalian forebrain neural stem cells. J Neurosci. 2001;21(24):9733–9743. | |
Campana WM, Misasi R, O’Brien JS. Identification of a neurotrophic sequence in erythropoietin. Int J Mol Med. 1998;1(1):235–241. | |
Byts N, Samoylenko A, Fasshauer T, et al. Essential role for Stat5 in the neurotrophic but not in the neuroprotective effect of erythropoietin. Cell Death Differ. 2008;15(4):783–792. | |
Viviani B, Bartesaghi S, Corsini E, et al. Erythropoietin protects primary hippocampal neurons increasing the expression of brain-derived neurotrophic factor. J Neurochem. 2005;93(2):412–421. | |
Solaroglu I, Solaroglu A, Kaptanoglu E, et al. Erythropoietin prevents ischemia-reperfusion from inducing oxidative damage in fetal rat brain. Childs Nerv Syst. 2003;19(1):19–22. | |
Kumral A, Gonenc S, Acikgoz O, et al. Erythropoietin increases glutathione peroxidase enzyme activity and decreases lipid peroxidation levels in hypoxic-ischemic brain injury in neonatal rats. Biol Neonate. 2005;87(1):15–18. | |
Tsai PT, Ohab JJ, Kertesz N, et al. A critical role of erythropoietin receptor in neurogenesis and post-stroke recovery. J Neurosci. 2006;26(4):1269–1274. | |
Wang L, Zhang Z, Wang Y, Zhang R, Chopp M. Treatment of stroke with erythropoietin enhances neurogenesis and angiogenesis and improves neurological function in rats. Stroke. 2004;35(7):1732–1737. | |
Iwai M, Cao G, Yin W, Stetler RA, Liu J, Chen J. Erythropoietin promotes neuronal replacement through revascularization and neurogenesis after neonatal hypoxia/ischemia in rats. Stroke. 2007;38(10):2795–2803. | |
Sun Y, Calvert JW, Zhang JH. Neonatal hypoxia/ischemia is associated with decreased inflammatory mediators after erythropoietin administration. Stroke. 2005;36(8):1672–1678. | |
Juul SE, Beyer RP, Bammler TK, McPherson RJ, Wilkerson J, Farin FM. Microarray analysis of high-dose recombinant erythropoietin treatment of unilateral brain injury in neonatal mouse hippocampus. Pediatr Res. 2009;65(5):485–492. | |
Brissaud O, Villega F, Pieter Konsman J, et al. Short-term effect of erythropoietin on brain lesions and aquaporin-4 expression in a hypoxic-ischemic neonatal rat model assessed by magnetic resonance diffusion weighted imaging and immunohistochemistry. Pediatr Res. 2010;68(2):123–127. | |
Juul S. Neuroprotective role of erythropoietin in neonates. J Matern Fetal Neonatal Med. 2012;25 Suppl 4:105–107. | |
Juul SE, McPherson RJ, Bauer LA, Ledbetter KJ, Gleason CA, Mayock DE. A phase I/II trial of high-dose erythropoietin in extremely low birth weight infants: pharmacokinetics and safety. Pediatrics. 2008;122(2):383–391. | |
Rice JE 3rd, Vannucci RC, Brierley JB. The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol. 1981;9(2):131–141. | |
Levine S. Anoxic-ischemic encephalopathy in rats. Am J Pathol. 1960;36:1–17. | |
Fang AY, Gonzalez FF, Sheldon RA, Ferriero DM. Effects of combination therapy using hypothermia and erythropoietin in a rat model of neonatal hypoxia-ischemia. Pediatr Res. 2012;73(1):12–17. | |
Fan X, van Bel F, van der Kooij MA, Heijnen CJ, Groenendaal F. Hypothermia and erythropoietin for neuroprotection after neonatal brain damage. Pediatr Res. 2012;73(1):18–23. | |
Juul SE. Hypothermia plus erythropoietin for neonatal neuroprotection? Commentary on Fan et al. and Fang et al. Pediatr Res. 2013;73(1):10–11. | |
Silbereis JC, Huang EJ, Back SA, Rowitch DH. Towards improved animal models of neonatal white matter injury associated with cerebral palsy. Dis Model Mech. 2010;3(11–12):678–688. | |
Zhu C, Kang W, Xu F, et al. Erythropoietin improved neurologic outcomes in newborns with hypoxic-ischemic encephalopathy. Pediatrics. 2009;124(2):e218–e226. | |
Elmahdy H, El-Mashad AR, El-Bahrawy H, El-Gohary T, El-Barbary A, Aly H. Human recombinant erythropoietin in asphyxia neonatorum: pilot trial. Pediatrics. 2010;125(5):e1135–e1142. | |
Wu YW, Bauer LA, Ballard RA, et al. Erythropoietin for neuroprotection in neonatal encephalopathy: safety and pharmacokinetics. Pediatrics. 2012;130(4):683–691. | |
Rogers EE, Bonifacio SL, Glass HC, et al. Erythropoietin and Hypothermia for Hypoxic-Ischemic Encephalopathy. Pediatr Neurol. 2014;51:657–662. | |
El Shimi MS, Awad HA, Hassanein SM, et al. Single dose recombinant erythropoietin versus moderate hypothermia for neonatal hypoxic ischemic encephalopathy in low resource settings. J Matern Fetal Neonatal Med. 2014;27(13):1295–1300. | |
Weber A, Dzietko M, Berns M, et al. Neuronal damage after moderate hypoxia and erythropoietin. Neurobiol Dis. 2005;20(2):594–600. | |
Tonia T, Mettler A, Robert N. Erythropoietin or darbepoetin for patients with cancer. … Database Syst Rev. 2012. | |
Fauchère JC, Dame C, Vonthein R, et al. An approach to using recombinant erythropoietin for neuroprotection in very preterm infants. Pediatrics. 2008;122(2):375–382. | |
Ohlsson A AS. Early erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants (Review). 2012:1–96. | |
Aher SM OA. Late erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants (Review). 2012:1–92. | |
Ohls RK, Christensen RD. Recombinant erythropoietin compared with erythrocyte transfusion in the treatment of anemia of prematurity. J Pediatr. 1991;119(5):781–788. | |
Xu XJ, Huang HY, Chen HL. Erythropoietin and retinopathy of prematurity: a meta-analysis. Eur J Pediatr. 2014;173(10):1355–1364. | |
Romagnoli C, Zecca E, Gallini F, Girlando P, Zuppa AA. Do recombinant human erythropoietin and iron supplementation increase the risk of retinopathy of prematurity? Eur J Pediatr. 2000;159(8):627–628. | |
Mehmet S, Fusun A, Sebnem C, et al. One-year experience in the retinopathy of prematurity: frequency and risk factors, short-term results and follow-up. Int J Ophthalmol. 2011;4(6):634–640. | |
Slusarski JD, McPherson RJ, Wallace GN, Juul SE. Pediatric research – abstract of article: high-dose erythropoietin does not exacerbate retinopathy of prematurity in rats. Pediatr Res. 2009. | |
Leung SP. Multiple strawberry haemangiomas-side effect of rhuEpo? Acta Paediatr. 2000;89(7):890. | |
Zaffanello M, Franchini M, Rugolotto S. Recombinant human erythropoietin might induce strawberry haemangiomas in very-low-birthweight preterm infants. Acta Paediatr. 2003;92(11):1353–1354. | |
Xiong T, Qu Y, Mu D, Ferriero D. International Journal of Developmental Neuroscience. Int J Dev Neurosci. 2011;29(6):583–591. | |
Doege C, Pritsch M, Frühwald MC, Bauer J. An association between infantile haemangiomas and erythropoietin treatment in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2011;97(1):F45–F49. | |
Eicher DJ, Wagner CL, Katikaneni LP, et al. Moderate hypothermia in neonatal encephalopathy: safety outcomes. Pediatr Neurol. 2005;32(1):18–24. | |
McPherson RJ, Juul SE. Erythropoietin for infants with hypoxic-ischemic encephalopathy. Curr Opin Pediatr. 2010;22(2):139–145. | |
Ehrenreich H, Weissenborn K, Prange H, et al; EPO Stroke Trial Group. Recombinant human erythropoietin in the treatment of acute ischemic stroke. Stroke. 2009;40(12):e647–e656. | |
Yager JY, Ashwal S. Animal models of perinatal hypoxic-ischemic brain damage. Pediatr Neurol. 2009;40(3):156–167. | |
Matsushita H, Johnston MV, Lange MS, Wilson MA. Protective effect of erythropoietin in neonatal hypoxic ischemia in mice. Neuroreport. 2003;14(13):1757–1761. | |
Kumral A, Ozer E, Yilmaz O, et al. Neuroprotective effect of erythropoietin on hypoxic-ischemic brain injury in neonatal rats. Biol Neonate. 2003;83(3):224–228. | |
Spandou E, Soubasi V, Papoutsopoulou S, et al. Erythropoietin prevents hypoxia/ischemia-induced DNA fragmentation in an experimental model of perinatal asphyxia. Neurosci Lett. 2004;366(1):24–28. | |
Kumral A, Uysal N, Tugyan K, et al. Erythropoietin improves long-term spatial memory deficits and brain injury following neonatal hypoxia-ischemia in rats. Behav Brain Res. 2004;153(1):77–86. | |
Wang X, Zhu C, Wang X, et al. The nonerythropoietic asialoerythropoietin protects against neonatal hypoxia-ischemia as potently as erythropoietin. J Neurochem. 2004;91(4):900–910. | |
Demers EJ, McPherson RJ, Juul SE. Erythropoietin protects dopaminergic neurons and improves neurobehavioral outcomes in juvenile rats after neonatal hypoxia-ischemia. Pediatr Res. 2005;58(2):297–301. | |
Spandou E, Papadopoulou Z, Soubasi V, et al. Erythropoietin prevents long-term sensorimotor deficits and brain injury following neonatal hypoxia-ischemia in rats. Brain Res. 2005;1045(1–2):22–30. | |
Kumral A, Genc S, Ozer E, et al. Erythropoietin downregulates bax and DP5 proapoptotic gene expression in neonatal hypoxic-ischemic brain injury. Biol Neonate. 2006;89(3):205–210. | |
McClure MM, Threlkeld SW, Fitch RH. The effects of erythropoietin on auditory processing following neonatal hypoxic-ischemic injury. Brain Res. 2006;1087(1):190–195. | |
McClure MM, Threlkeld SW, Fitch RH. Auditory processing and learning/memory following erythropoietin administration in neonatally hypoxic-ischemic injured rats. Brain Res. 2007;1132:203–209. | |
McPherson RJ, Demers EJ, Juul SE. Safety of high-dose recombinant erythropoietin in a neonatal rat model. Neonatology. 2007;91(1):36–43. | |
Statler PA, McPherson RJ, Bauer LA, Kellert BA, Juul SE. Pharmacokinetics of high-dose recombinant erythropoietin in plasma and brain of neonatal rats. Pediatr Res. 2007;61(6):671–675. | |
Kim SS, Lee KH, Sung DK, et al. Erythropoietin attenuates brain injury, subventricular zone expansion, and sensorimotor deficits in hypoxic-ischemic neonatal rats. J Korean Med Sci. 2008;23(3):484–491. | |
van der Kooij MA, Groenendaal F, Kavelaars A, Heijnen CJ, van Bel F. Combination of deferoxamine and erythropoietin: therapy for hypoxia-ischemia-induced brain injury in the neonatal rat? Neurosci Lett. 2009;451(2):109–113. | |
Iwai M, Stetler RA, Xing J, et al. Enhanced oligodendrogenesis and recovery of neurological function by erythropoietin after neonatal hypoxic/ischemic brain injury. Stroke. 2010;41(5):1032–1037. | |
Fan X, Heijnen CJ, van der Kooij MA, Groenendaal F, van Bel F. Beneficial effect of erythropoietin on sensorimotor function and white matter after hypoxia-ischemia in neonatal mice. Pediatr Res. 2011;69(1):56–61. | |
Alexander ML, Hill CA, Rosenkrantz TS, Fitch RH. Evaluation of the therapeutic benefit of delayed administration of erythropoietin following early hypoxic-ischemic injury in rodents. Dev Neurosci. 2012;34(6):515–524. | |
Chen H, Spagnoli F, Burris M, et al. Nanoerythropoietin is 10-times more effective than regular erythropoietin in neuroprotection in a neonatal rat model of hypoxia and ischemia. Stroke. 2012;43(3):884–887. | |
Chang YS, Mu D, Wendland M, et al. Erythropoietin improves functional and histological outcome in neonatal stroke. Pediatr Res. 2005;58(1):106–111. | |
Wen TC, Rogido M, Peng H, Genetta T, Moore J, Sola A. Gender differences in long-term beneficial effects of erythropoietin given after neonatal stroke in postnatal day-7 rats. Neuroscience. 2006;139(3):803–811. | |
Gonzalez FF, McQuillen P, Mu D, et al. Erythropoietin enhances long-term neuroprotection and neurogenesis in neonatal stroke. Dev Neurosci. 2007;29(4–5):321–330. | |
Gonzalez FF, Abel R, Almli CR, Mu D, Wendland M, Ferriero DM. Erythropoietin sustains cognitive function and brain volume after neonatal stroke. Dev Neurosci. 2009;31(5):403–411. | |
Gonzalez FF, Larpthaveesarp A, McQuillen P, et al. Erythropoietin increases neurogenesis and oligodendrogliosis of subventricular zone precursor cells after neonatal stroke. Stroke. 2013;44(3):753–758. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.