Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 15
The mRNA Expression Profile of Psoriatic Lesion Distinct from Non-Lesion
Authors Li X, Xing J, Wang F, Li J, Li J, Hou R, Zhang K
Received 17 August 2022
Accepted for publication 13 September 2022
Published 27 September 2022 Volume 2022:15 Pages 2035—2043
DOI https://doi.org/10.2147/CCID.S385894
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jeffrey Weinberg
Xinhua Li,1 Jianxiao Xing,1 Fangdi Wang,1 Juan Li,1 Junqin Li,1 Ruixia Hou,1 Kaiming Zhang2
1Shanxi Key Laboratory of Stem Cell for Immunological Dermatosis, Taiyuan Central Hospital of Shanxi Medical University, Taiyuan, Shanxi Province, 030009, People’s Republic of China; 2Shanxi Key Laboratory of Stem Cell for Immunological Dermatosis, Taiyuan Central Hospital, Taiyuan, Shanxi Province, 030009, People’s Republic of China
Correspondence: Kaiming Zhang, Taiyuan Central Hospital, No, 5 Dong San Dao Xiang, Jiefang Road, Taiyuan, Shanxi Province, People’s Republic of China, Tel +86-351-5656080, Email [email protected]
Purpose: Psoriasis is a chronic recurring autoimmune skin disease with a complex etiology and chronic progression; however, its molecular mechanisms remain unclear.
Patients and Methods: We performed transcriptomic analysis to profile the mRNA expression of psoriatic lesions (PL) and non-lesion (NL) tissues from psoriasis patients along with normal skin from healthy donors. RT-qPCR was used to validate the mRNA expression profiles.
Results: A total of 237 differentially expressed genes were screened and identified by RNA sequencing. GO and KEGG analysis indicated that these DEGs were enriched in the PPAR signaling pathway and intermediate filament cytoskeleton. For PPAR signaling pathway, the expression of five genes, including ADIPOQ, AQP7, PLIN1, FABP4 and LPL, were all significantly decreased in psoriatic lesions compared to normal skin by RT-qPCR. There is a clear difference between psoriatic lesions and non-lesion in the expression of ADIPOQ, AQP7 and LPL. For intermediate filament cytoskeleton, including KRT27, KRT25, KRT71, KRT86 and KRT85 were significantly decreased in the psoriasis lesions, showing agreement with the RNA-seq data.
Conclusion: This study revealed a significant difference between the mRNA expression profiles of PL, NL tissue and normal skin.
Keywords: psoriasis, RNA-sequencing, PPAR signaling pathway, keratin
Introduction
Psoriasis is an autoimmune disease characterized by1 erythema and scale lesions that2,3 affects approximately 2–4% of the population. Although the exact pathogenesis of this disease has not yet been fully determined, it is thought to be a systemic disease as its pathogenesis and development are determined by4,5 cellular immune disorders, genetic, environmental, and6 infectious factors, and interaction between metabolic, environmental, and7 genetic factors.8 Psoriasis is a cytokine-driven skin disease in which lesions are caused by abnormal thickening of the epidermis and hyperproliferation of keratinocytes (KCs).9,10 Transcriptome studies of psoriasis have been performed using large patient cohorts to understand how gene expression is altered at lesions compared to macroscopic normal skin.
Studies have found differences in the mRNA expression of many genes between psoriasis and normal individuals.11 C-myc, c-fos, and c-jun transcripts were significantly induced over in vivo levels 2–4 h after organ culture of normal or psoriatic keratome biopsies, demonstrating that these genes can be highly expressed in the context of tissue injury.12 Haptoglobin protein expression was also markedly increased in the 138 psoriasis group compared with in the normal groups. There are also many genes expression is down-regulated, such as13 DUSP1,14 SMAD2, TGFbeta receptor I.
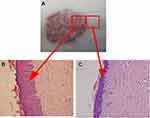
However, the characteristic lesions of psoriasis only appear locally and are distinctly different from the adjacent skin both in appearance and histopathology, as shown in Figure 1. Therefore, there may be other factors that contribute toward the manifestation of erythema and scales in psoriatic lesions that are absent in adjacent skin.15 Although Jabbari et al examined the coding psoriatic transcriptome of lesional and normal skin samples from psoriatic individuals, the study did not examine normal skin samples collected adjacent to the psoriatic lesions. To elucidate the factors that are specifically present in psoriatic lesions, our study provides a comprehensive mRNA profile of patients with psoriasis and directions for further study of the molecular mechanisms of psoriasis.
Materials and Methods
Samples
We collected 10 psoriatic lesions (PL) and adjacent normal skin (non-lesion, NL) samples from patients with psoriasis (five males and five females; 23–39 years-old) and 10 normal tissue samples from healthy volunteers (six males and four females; 25–40 years-old) from Taiyuan Central Hospital. All tissue specimens were stored at −80°C. All participants provided written informed consent and the study protocol was approved by the ethics committee of Taiyuan Central Hospital.
RNA Sequencing
Total RNA was extracted from tissues using TRIzol (Invitrogen, Carlsbad, CA, USA) and rRNA was depleted using a Ribo-Zero™ Magnetic Kit. RNA purity was checked using a KaiaoK5500® spectrophotometer (Kaiao, Beijing, China) and sequenced using an Illumina HiSeq 4000 platform on a 150 bp paired end run. Raw data was filtered to remove rRNA, low quality samples, linker contamination, and unknown base N content. The rRNA sequences were identified by mapping reads to the ribosome database with SOAP. Bowtie 2 was used to align the clean reads to the reference sequence and RSEM was used to calculate the number of reads mapping to genes.
DEG Analysis and Functional Analysis
Differential gene expression analysis was performed using the Limma R/Bioconductor software package in R (v. 3.22.7), which provides an integrated solution for both the differential expression and differential splicing analysis of RNA-seq data. The Benjamini–Hochberg method was used as an FDR adjustment for multiple testing correction. An FDR threshold of < 0.05 was used to define statistical significance. Metascape (http://metascape.org) was used to perform gene enrichment and functional annotation analyses.
Real-Time Quantitative PCR (RT-qPCR)
RNA was extracted from the samples using Trizol (Invitrogen, Carlsbad, CA, USA) and RT-qPCR was performed on ten preselected psoriasis-related genes using the qPCR primers shown in Supplementary Table 1. Each RT-qPCR reaction was performed in duplicate and the mean threshold cycle (Ct) value for each sample was used for data analysis. The 2−ΔΔCt method was used to determine the fold-change in expression level normalized to β-actin. Paired t-test analyses were performed on the 2−ΔΔCt values to compare the two groups of samples.
Statistical Analysis
Statistical analyses were carried out using the R software package (http://cran.r-project.org/). Differences between the mRNA expression levels of patients with psoriasis and normal individuals were evaluated by paired t-tests. Statistical significance was set at P < 0.05.
Results
Genome-Wide mRNA Analysis of Psoriatic Lesion (PL) and Non-Lesion (NL)
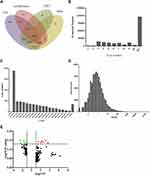
To comprehensively determine the mRNA landscape of psoriasis, we profiled the mRNA expression of psoriatic lesion (PL) and non-lesion (NL) samples from patients with psoriasis by RNA sequencing. A total of 1.5–2.0 billion clean reads were obtained for each sample using hierarchical indexing for spliced alignment of transcripts (HISAT). Furthermore, we used CPC, txCdsPredict, pfam, and CNCI to assess transcription coding ability (Figure 2A), obtaining a total set of 187,731 transcripts (including 29,750 novel mRNAs) that were defined as candidate mRNAs (Figure 2B and C). We also quantitatively analyzed the mRNA expression levels for read counts and Fragments per Kilobase Million (FPKM), finding that most of the mRNAs contained over 10 exons with a transcript length of over 500 bp (Figure 2D). Moreover, we evaluated the transcription levels of the mRNAs. To investigate whether the differentially expressed mRNAs regulate genes and signaling pathways associated with psoriasis, we used target prediction programs to predict potential targets of the dysregulated mRNAs. Two hundred and thirty-seven differentially expressed mRNAs were identified through RNA-seq analysis. Among them, 164 were significantly upregulated, and 73 were significantly downregulated (Fold Change ≥2.0, P-value ≤ 0.05) (Figure 2E).
Signaling Pathway Enrichment Analysis
GO analysis revealed considerable functional overlap among the 237 predicted target genes and found that they were enriched in cellular components including lipid metabolic processes, small molecule metabolic processes, chemical homeostasis, and response to oxygen-containing compounds (P < 0.05), and in biological processes including extracellular region, extracellular space, and anchored membrane components (Figure 3A and B). KEGG-based pathway enrichment analysis was performed using the DAVID database and signaling pathways with P < 0.05 were selected. There was considerable overlap among the signaling transduction pathways of the target genes, with KEGG pathway analysis highlighting the peroxisome proliferator-activated receptor (PPAR) signaling pathway and arachidonic acid metabolism (P < 0.05; Figure 3C).
 |
Figure 3 Gene Ontology (GO) and KEGG analysis of target genes. (A) GO biological process analysis. (B) GO cellular component analysis. (C) KEGG pathway analysis. |
RT-qPCR Analysis Expression of Gene in Psoriatic Lesion (PL), Non-Lesion (NL) and Normal Skin(Nor)
According to RNA-seq analysis, we retrieved ten genes related to the peroxisome proliferator activated receptor (PPAR) signaling pathway and intermediate filament cytoskeleton (Figure 4A). These genes are down-regulated in psoriatic lesions compared with non-psoriatic lesions. To verify the RNA-seq results, we selected ten dysregulated mRNAs for RT-qPCR analysis. For PPAR signaling pathway, we selected five genes, including adipoq (ADIPOQ), aquaglyceroporin 7 (AQP7), fatty acid-binding protein (FABP4), lipoprotein lipase (LPL), and perilipin 1 (PLIN1), which were all significantly decreased in psoriatic lesions compared to normal skin by RT-qPCR (Figure 4C). There is a clear difference between psoriatic lesions and non-lesion in the expression of ADIPOQ, AQP7 and LPL. For intermediate filament cytoskeleton, including KRT27, KRT25, KRT71, KRT86 and KRT85 were significantly decreased in the psoriasis lesions, showing agreement with the RNA-seq data (Figure 4B).
Discussion
Psoriasis is a clinically heterogeneous lifelong skin disease that presents in multiple forms such as16,17 plaque, flexural, guttate, pustular or erythrodermic.18 Psoriatic lesions result from complex interactions between dermal or epidermal cells, resident and infiltrating immune cells, and a variety of cytokines.19 Psoriasis has differences in gene expression between lesions and non-lesions, such as atopic dermatitis and cutaneous lupus.20,21 Previous studies have performed transcriptomic analyses to investigate molecular abnormities in patients with psoriasis. However, these studies have focused on skin lesions and therefore the transcriptomic signature of the peripheral circulation in psoriasis, a systematic autoinflammatory disease, remains largely unknown. In this study, we conducted an RNA-seq-based transcriptomic analysis of psoriatic lesions and adjacent normal skin, which may provide insights into the pathogenesis of psoriasis and potential biomarkers for its diagnosis and treatment.
Currently, many psoriasis-associated mRNAs have been investigated that may play an important role in susceptibility to psoriasis. To the best of our knowledge, this study determined the mRNA expression profiles of psoriatic lesions and adjacent normal skin. We identified 237 psoriasis-associated genes via multiple methods to provide a convincing set of genes for further enrichment and pathogenicity analyses. GO analysis was performed to investigate the biological functions that were enriched among the dysregulated mRNAs, revealing that differentially expressed mRNAs in psoriatic lesions and adjacent normal skin may be involved in the intermediate filament cytoskeleton, extracellular region, and fatty acid metabolic processes.
Interestingly, the intermediate filament cytoskeleton contained the differentially expressed genes KRT27, KRT25, KRT71, KRT86, and KRT85, which are known to be involved in the development of psoriasis. RT-qPCR revealed that KRT27, KRT25, KRT71, KRT86 and KRT85 expression were markedly higher in the normal control group compared with the psoriasis and NL groups, consistent with the RNA seq-analysis results.22,23 Keratins (KRT) are the major components of the epithelial cytoskeleton and are responsible for maintaining the structural stability and integrity of keratinocytes.24 Keratins also regulate keratinocyte mobility via cytoplasmic viscosity during stratification or wound healing, while25 keratinocyte hyperproliferation is considered a hallmark of psoriasis.26 K17 exerts both pro-proliferative and pro-inflammatory effects on keratinocytes. Moreover, K17 peptides trigger autoreactive T cells and promote psoriasis-related cytokine production. In this study, KRT27, KRT25, KRT71, KRT86 and KRT85 mRNA levels were observed in patients with psoriasis and may promote excessive keratinocyte proliferation,27,28 consistent with psoriasis characterized by cornified layer-hyperkeratosis expansion.
Moreover, KEGG pathway analysis revealed considerable overlap between the signal transduction pathways of the target gene set, particularly the PPAR signaling pathway and arachidonic acid metabolism, suggesting that they may be closely related to the biosynthesis and metabolism of psoriasis and include ADIPOQ, AQP7, PLIN1, FABP4, and the expression of those genes were significantly decreased in the psoriasis group. Furthermore,29–31 these genes are known participate in the PPAR signaling pathway.32 Reducing the level of the PPARγ gene expression may be a result of the overregulation of several cascades. Pattern recognition receptors (TLRs, NOD1, NOD2, and CLEC7A) highly expressed during the infection may be one of such cascades.33 Transcription factors including NF-kBs, JUN-FOS, AHR, GATA3, HIF1A, FOXO1, and FOSL1 can directly inhibit PPARγ expression. All these transcription factors are overstimulated in the inflammatory and immune response. Recently,34 PPARs and their ligands have been identified in skin, where they control important cellular functions, such as inflammation, proliferation, and differentiation.35,36 PLIN1 is significantly enriched in the fat metabolism-related PPAR signaling pathway via the DNA demethylation of the PPAR-response elements of the PLIN1 gene promoter upon differentiation. Two other target genes, ADIPOQ and AQP7, were also down-regulated in the psoriasis group compared with the normal control and NL groups.37 ADIPOQ has been reported to increase AQP3 expression via PPARα-mediated signaling in hepatic stellate cells, while38 ADIPOQ has been shown to activate PPARα and affect the AMPK pathway,39 which is involved in the occurrence and development of psoriasis.40 Furthermore, the PPAR signaling pathway has been suggested as a potential therapeutic target for the treatment of hyperproliferative skin diseases.
Limitations
Our research has several limitations. The number of tested samples was relatively small. More works are needed to verify our findings and illustrate the detailed mechanism of these gene based on larger sample size in the future.
Conclusion
In summary, we screened psoriasis-related mRNA expression profiles by using bioinformatics approaches. The experimentally-validated targets genes of intermediate filament cytoskeleton and PPAR signaling pathway are key regulators of psoriasis lesions. Our results provide a new experimental basis and directions for further research on the effects of mRNA on the occurrence and development of psoriasis.
Ethics Statement
The study was approved by the institutional ethics committee of Taiyuan City Central Hospital (2016005). The study was carried out with the informed consent of the patients themselves and in accordance with the Declaration of Helsinki Principles.
Acknowledgments
The authors are grateful for funding from the National Natural Science Foundation of China (nos. 81602768). We would like to thank AJE (https//www.aje.cn/) for English language editing.
Disclosure
The authors declare no conflicts of interest for this work.
References
1. Nickoloff BJ, Nestle FO. Recent insights into the immunopathogenesis of psoriasis provide new therapeutic opportunities. J Clin Invest. 2004;113(12):1664–1675. doi:10.1172/JCI200422147
2. Leonardi CL, Powers JL, Matheson RT, et al. Psoriasis Study Group Etanercept as monotherapy in patients with psoriasis. N Engl J Med. 2003;349(21):2014–2022. doi:10.1056/NEJMoa030409
3. Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61(3):451–485. doi:10.1016/j.jaad.2009.03.027
4. Batalla A, Coto E, González-Lara L, et al. Association between single nucleotide polymorphisms IL17RA rs4819554 and IL17E rs79877597 and psoriasis in a Spanish cohort. J Dermatol Sci. 2015;80(2):111–115. doi:10.1016/j.jdermsci.2015.06.011
5. Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41(5):514–521. doi:10.1111/ced.12841
6. Honma M, Hayashi K. Psoriasis: recent progress in molecular-targeted therapies. J Dermatol. 2021;48(6):761–777. doi:10.1111/1346-8138.15727
7. Coto E, Santos-Juanes J, Coto-Segura P, et al. New psoriasis susceptibility genes: momentum for skin-barrier disruption. J Invest Dermatol. 2011;131(5):1003–1005. doi:10.1038/jid.2011.14
8. Li B, Tsoi LC, Swindell WR, et al. Transcriptome analysis of psoriasis in a large case-control sample: RNA-seq provides insights into disease mechanisms. J Invest Dermatol. 2014;134:1828–1838. doi:10.1038/jid.2014.28
9. Swindell WR, Sarkar MK, Stuart PE, et al. Psoriasis drug development and GWAS interpretation through in silico analysis of transcription factor binding sites. Clin Transl Med. 2015;4:13. doi:10.1186/s40169-015-0054-5
10. Swindell WR, Sarkar MK, Liang Y, et al. Cross-disease transcriptomics: unique IL-17A signaling in psoriasis lesions and an autoimmune PBMC signature. J Invest Dermatol. 2016;136(9):1820–1830. doi:10.1016/j.jid.2016.04.035
11. Elder JT, Tavakkol A, Klein SB, et al. Protooncogene expression in normal and psoriatic skin. J Invest Dermatol. 1990;94(1):19–25. doi:10.1111/1523-1747.ep12873313
12. Tian FJ, Zhang YY, Liu LQ, et al. Haptoglobin protein and mRNA expression in psoriasis and its clinical significance. Mol Med Rep. 2016;14(4):3735–3742. doi:10.3892/mmr.2016.5672
13. Kjellerup RB, Johansen C, Kragballe K, et al. The expression of dual-specificity phosphatase 1 mRNA is downregulated in lesional psoriatic skin. Br J Dermatol. 2013;168(2):339–345. doi:10.1111/bjd.12020
14. Yu H, Mrowietz U, Seifert O. Downregulation of SMAD2, 4 and 6 mRNA and TGFbeta receptor I mRNA in lesional and non-lesional psoriatic skin. Acta Derm Venereol. 2009;89(4):351–356. doi:10.2340/00015555-0634
15. Jabbari A, Suarez-Farinas M, Dewell S, et al. Transcriptional profiling of psoriasis using RNA-seq reveals previously unidentified differentially expressed genes. J Invest Dermatol. 2012;132(1):246–249. doi:10.1038/jid.2011.267
16. Zheng Q-Y, Liang S-J, Xu F. C5a/C5aR1 pathway is critical for the pathogenesis of psoriasis. Front Immunol. 2019;10:1866. doi:10.3389/fimmu.2019.01866
17. Raharja A, Mahil SK, Barker JN. Psoriasis: a brief overview. Clin Med. 2021;21(3):170–173. doi:10.7861/clinmed.2021-0257
18. Wang L, Yu X, Wu C, et al. RNA sequencing-based longitudinal transcriptomic profiling gives novel insights into the disease mechanism of generalized pustular psoriasis. BMC Med Genomics. 2018;11(1):52. doi:10.1186/s12920-018-0369-3
19. Wei XY, Fricker K, Enk AH, et al. Altered expression of keratin 14 in lesional epidermis of autoimmune skin diseases. Int J Dermatol. 2016;55(6):620–628. doi:10.1111/ijd.13011
20. Choy DF, Hsu DK, Seshasayee D, et al. Comparative transcriptomic analyses of atopicdermatitis and psoriasis reveal shared neutrophilic inflammation. J Allergy Clin Immunol. 2012;130(6):1335–1343. doi:10.1016/j.jaci.2012.06.044
21. Gao Y, Yi X, Ding Y, et al. Combined transcriptomic analysis revealed AKR1B10 played an important role in psoriasis through the dysregulated lipid pathway and overproliferation of keratinocyte. Biomed Res Int. 2017;2017:8717369. doi:10.1155/2017/8717369
22. Coulombe PA. The molecular revolution in cutaneous biology: keratin genes and their associated disease: diversity, opportunities, and challenges. J Invest Dermatol. 2017;137(5):e67–e71. doi:10.1016/j.jid.2016.04.039
23. Zhang X, Yin M, Zhang LJ. Keratin 6, 16 and 17 critical barrier alarmin molecules in skin wounds and psoriasis. Cells. 2019;8(8):807. doi:10.3390/cells8080807
24. Gu LH, Coulombe PA. Keratin function in skin epithelia: a broadening palette with surprising shades. Curr Opin Cell Biol. 2007;19(1):13–23. doi:10.1016/j.ceb.2006.12.007
25. Zhang J, Li X, Wei J. Gallic acid inhibits the expression of keratin 16 and keratin 17 through Nrf2 in psoriasis-like skin disease. Int Immunopharmacol. 2018;65:84–95. doi:10.1016/j.intimp.2018.09.048
26. Lin Y, Zhang W, Li B, et al. Keratin 17: a critical player in the pathogenesis of psoriasis. Semin Cell Dev Biol. 2022;128:112–119. doi:10.1016/j.semcdb.2021.06.018
27. Wang W, Yu X, Wu C, et al. IL-36γ inhibits differentiation and induces inflammation of keratinocyte via Wnt signaling pathway in psoriasis. Int J Med Sci. 2017;14(10):1002–1007. doi:10.7150/ijms.20809
28. Zhang J, Fang H, Wang R, et al. Effect of calcipotriol on IFN-γ-induced Keratin 17 expression in immortalized human epidermal keratinocyte cells. Med Sci Monit. 2017;23:6049–6056. doi:10.12659/MSM.904850
29. Walker CG, Holness MJ, Gibbons GF, et al. Fasting-induced increases in aquaporin 7 and adipose triglyceride lipase mRNA expression in adipose tissue are attenuated by peroxisome proliferator-activatedreceptor alpha deficiency. Int J Obes. 2007;31(7):1165–1171. doi:10.1038/sj.ijo.0803555
30. Li B, Weng Q, Dong C, et al. A key gene, PLIN1, can affect porcine intramuscular fat content based on transcriptome. AnalysisGenes. 2018;9:194. doi:10.3390/genes9040194
31. Smetnev S, Klimushina K, Kutsenko K, et al. Associations of SNPs of the ADIPOQ gene with serum adiponectin levels, unstable angina, and coronary artery disease. Biomolecules. 2019;9(10):537. doi:10.3390/biom9100537
32. Sobolev V, Nesterova A, Soboleva A, et al. The model of PPARγ-downregulated signaling in psoriasis. PPAR Res. 2020;2020:6529057. doi:10.1155/2020/6529057
33. Sobolev V, Nesterova A, Soboleva A, et al. Analysis of PPARγ signaling activity in psoriasis. Int J Mol Sci. 2021;22(16):8603. doi:10.3390/ijms22168603
34. Ostadhadi S, Nikoui V, Haj-Mirzaian A, et al. The role of PPAR-gamma receptor in pruritus. Eur J Pharmacol. 2015;762:322–325. doi:10.1016/j.ejphar.2015.06.009
35. Lyu Y, Su X, Deng J, et al. Defective differentiation of adipose precursor cells from lipodystrophic mice lacking perilipin 1. PLoS One. 2015;10(2):e0117536. doi:10.1371/journal.pone.0117536
36. Fujiki K, Shinoda A, Kano F, et al. PPARγ-induced PARylation promotes local DNA demethylation by production of 5-hydroxymethylcytosine. Nat Commun. 2013;4:2262. doi:10.1038/ncomms3262
37. Sertznig P, Seifert M, Tilgen W, et al. Peroxisome proliferator-activated receptors (PPARs) and the human skin. Am J Clin Dermatol. 2008;9(1):15–31. doi:10.2165/00128071-200809010-00002
38. Tardelli M, Claudel T, Bruschi FV, et al. Adiponectin regulates AQP3 via PPARα in human hepatic stellate cells. Biochem Biophys Res Commun. 2017;490(1):51–54. doi:10.1016/j.bbrc.2017.06.009
39. Kishida K, Shimomura I, Nishizawa H, et al. Enhancement of the aquaporin adipose gene expression by a peroxisome proliferator-activated receptor gamma. J Biol Chem. 2001;276(51):48572–48579. doi:10.1074/jbc.M108213200
40. Garcin G, Guiraud I, Lacroix M, et al. AMPK/HuR-driven IL-20 post-transcriptionalregulation in psoriatic skin. J Invest Dermatol. 2015;135(11):2732–2741. doi:10.1038/jid.2015.282
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.