Back to Journals » Clinical Interventions in Aging » Volume 15
The Impact of Advanced Age on Major Cardiovascular Events and Mortality in Patients with ST-Elevation Myocardial Infarction Undergoing a Pharmaco-Invasive Strategy
Authors Helber I , Alves CMR , Grespan SM, Veiga ECA, Moraes PIM , Souza JM, Barbosa AH , Gonçalves Jr I , Fonseca FAH , Carvalho ACC, Caixeta A
Received 9 June 2019
Accepted for publication 17 March 2020
Published 21 May 2020 Volume 2020:15 Pages 715—722
DOI https://doi.org/10.2147/CIA.S218827
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Izo Helber,1 Claudia Maria Rodrigues Alves,1 Stela Maris Grespan,1 Eduardo CA Veiga,2 Pedro IM Moraes,1 José Marconi Souza,1 Adriano H Barbosa,1 Iran Gonçalves Jr,1 Francisco AH Fonseca,1 Antônio Carlos C Carvalho1,†, Adriano Caixeta1,3
1Department of Medicine, Discipline of Cardiology, Escola Paulista de Medicina, Universidade Federal São Paulo, São Paulo, Brazil; 2Department of Gynecology and Obstetrics, Faculdade de Medicina da Universidade de São Paulo, São Paulo, Brazil; 3Department of Cardiology, Hospital Israelita Albert Einstein, São Paulo, Brazil
†Dr Antonio Carlos De Camargo Carvalho passed away on January 8, 2019
Correspondence: Adriano Caixeta
Escola Paulista de Medicina, Universidade Federal São Paulo, Av. Napoleão de Barros, 715, São Paulo, CEP: 04024002, Brazil
Tel +55 11- 5576-4000
Email [email protected] Twitter @adrianocaixeta
Background: There is little research in the efficacy and safety of a pharmaco-invasive strategy (PIS) in patients ≥ 75 years versus < 75 years of age. We aimed to evaluate and compare the influence of advanced age on the risk of death and major adverse cardiac events (MACE) in patients undergoing PIS.
Methods: Between January 2010 and November 2016, 14 municipal emergency rooms in São Paulo, Brazil, used full-dose tenecteplase to treat patients with STEMI as part of a pharmaco-invasive strategy for a local network implementation.
Results: A total of 1852 patients undergoing PIS were evaluated, of which 160 (9%) were ≥ 75 years of age. Compared to patients < 75 years, those ≥ 75 years were more often female, had lower body mass index, higher rates of hypertension; higher incidence of hypothyroidism, chronic renal failure, prior stroke, and diabetes. Compared to patients < 75 years of age, in-hospital MACE and mortality were higher in patients with ≥ 75 years (6.5% versus 19.4%; p< 0.001; and 4.0% versus 18.2%; p< 0.001, respectively). Patients ≥ 75 years had higher rates of in-hospital major bleeding (2.7% versus 5.6%; p=0.04) and higher incidence of cardiogenic shock (7.0% versus 19.6%; p< 0.001). By multivariable analysis, age ≥ 75 years was independent predictor of MACE (OR 3.57, 95% CI 1.72 to 7.42, p=0.001) and death (OR 2.07, 95% CI 1.12– 3.82, p=0.020).
Conclusion: In patients with ST-segment elevation myocardial infarction undergoing PIS, age ≥ 75 years was an independent factor that entailed a 3.5-fold higher MACE and 2-fold higher mortality rate compared to patients < 75 years of age.
Keywords: acute myocardial infarction, elderly, fibrinolysis, primary percutaneous coronary intervention, pharmaco-invasive strategy
Introduction
Primary percutaneous coronary intervention (pPCI) is the recommended method of reperfusion of patients with ST-segment elevation myocardial infarction (STEMI) when performed in a timely fashion by experienced operators (<120 min; class I, level of evidence A).1 In developing countries including Brazil, due to major logistical challenges, most patients do not have access to a hospital with pPCI facilities, leading to a delay in reperfusion therapy with an important impact on patient prognosis. It is estimated that <1% of the hospitals in Brazil can provide around-the-clock (24/7) pPCI care. Thus, a pharmaco-invasive treatment strategy (PIS), characterized by fibrinolysis followed by PCI within 24 h, represents an acceptable approach in situations in which pPCI is not available. PIS may reduce the differences between the pPCI and pharmacologic fibrinolysis results in patients with an early presentation.2
Although representing approximately 5% of the population, the elderly (≥75 years of age) are responsible for more than one-third of myocardial infarctions, with over half of them resulting in fatal outcomes.3 Despite decades of research and experience, the risk:benefit ratio of fibrinolytic therapy in this subgroup remains controversial. Elderly patients have higher ischemic events and mortality rate compared with non-elderly in the setting of STEMI undergoing pPCI;3 however, the impact of age on mortality and major adverse cardiac events (MACE) in a subgroup of very old patients undergoing a PIS strategy is unknown. We, therefore, sought to assess the impact of age ≥75 years on mortality and MACE in patients with STEMI undergoing PIS.
Methods
Between January 2010 and November 2016, 14 municipal emergency rooms at non-interventional Hospital and the Mobile Emergency Health Service (Serviço de Atendimento Móvel de Urgência; SAMU) in São Paulo, Brazil, used tenecteplase (TNK) to treat patients with STEMI as part of a local network implementation. Patients were subsequently transferred to a tertiary teaching hospital and underwent an early invasive management strategy of coronary angiography (˂24 h of TNK administration, designated as the PIS). The inclusion criteria included all patients with a clinical and electrocardiographic diagnosis of STEMI who received TNK in the past 24 h. The patients included in this retrospective study fulfilled the following criteria: Diagnosis of STEMI with electrocardiographic evidence showing ST-segment elevation in ≥2 contiguous leads or left bundle branch block (new or supposedly new), initial admission to a municipal emergency room facility or rescue by the SAMU, and transfer to the tertiary Hospital São Paulo of the Federal University of São Paulo. All patients in this analysis were treated with TNK (30–50 mg bolus according to patient weight) and underwent early cardiac catheterization or PCI when needed. After a bolus of TNK and loading dose of clopidogrel, enoxaparin was prescribed before transport to our PCI-capable hospital. Aspirin 100–200 mg chewed was given in the emergency department, followed by 100 mg orally daily during hospitalization and indefinitely thereafter. Antiplatelet therapy consisted of clopidogrel in a 300-mg loading dose (with no loading dose for patients ≥75 years of age), followed by 75 mg orally daily. Dual antiplatelet therapy was recommended for at least 1 year. PCI was performed according to current guidelines and standards. Only bare metal stents were available at the time of the study period and were used for PCI. Urgent coronary angiography was permitted at any time in the presence of hemodynamic or electrical instability, worsening ischemia, or progressive or sustained ST-segment elevation requiring immediate coronary intervention, according to the physician’s judgment. Whereas the term elderly has traditionally been applied to those aged ≥65 years, for the purposes of this discussion, we focus primarily on the group aged ≥75 years given the rapid rise in cardiovascular morbidity and mortality in this subset of patients. There are no dedicated adequately sized fibrinolytic trials in the elderly, and patients aged ≥75 years are under-represented in registries and landmark trials.
The study was approved by the local Hospital ethic committee (Comissão de Ética Médica do Hospital Universitário São Paulo da Universidade Federal de São Paulo) under protocol number 1838/11 (http://www.hospitalsaopaulo.org.br). The author declares that the procedures were followed complied with the Declaration of the Helsinki (WMA, 1964) – with respect to the principles of medical ethics, that forbid the disclosure of patient name, initials or Hospital record numbers. Yet the study was submitted to our ethics committee for permission to waive consent since it is a retrospective study. Our ethics committee is registered at the United States Department of Health and Human Services Office for Human Research Protections under protocol number FWA 00001891, IRB 00001889.
Variables and Clinical Outcomes
The primary endpoint of the present study is to compare the rates of in-hospital MACE and death in patients <75 years with those with aged ≥75 years. We collected data on participants’ demographics such as age, sex, cardiovascular risk factors, and comorbidities. Venous blood samples for laboratory tests were collected in all patients at hospital admission. In addition, data were also collected on clinical characteristics such as Killip class, intracranial hemorrhage, non-intracranial bleeding, cardiogenic shock, and all aspects of the interventional treatment and adjunctive pharmacology. MACE was defined as the composite of all-cause death, reinfarction, or stroke. Reinfarction was characterized as a new rise and fall of cardiac biomarkers along with symptoms of myocardial ischemia, new (or presumably new) significant ST-segment/T-wave change or new left bundle branch block, development of pathological Q-wave on ECG, and/or identification of new intracoronary thrombus by angiography. Stroke was classified as either hemorrhagic or ischemic. Major bleeding was defined according to the Thrombolysis in Myocardial Infarction (TIMI) bleeding criteria4 as the following: Any intracranial bleeding (excluding microhemorrhages <10 mm evident only on gradient-echo magnetic resonance imaging), clinically overt signs of hemorrhage associated with a drop in hemoglobin of ≥5 g/dL, or fatal bleeding (bleeding that directly results in death within 7 days). All the clinical outcomes including MACE and death were evaluated during hospitalization only.
Statistical Analysis
Categorical variables were described according to MACE and death with absolute and relative frequencies and association were tested by χ2 analysis or Fisher’s exact test. Continuous variables are expressed as means and were compared with the Student t test. Multivariate logistic regression was used to derive the independent predictors of MACE and death via backward stepwise selection (α=0.05). The risk factors of death from any cause or of MACE were analyzed during hospitalization. The following potential covariates were included in the model to identify the predictors of MACE and death: Age, female sex, glomerular filtration rate <60 mL/kg (using the Modification of Diet in Renal Disease [MDRD] study equation), troponin, HDL-cholesterol, aspartate aminotransferase, heart rate, smoking, Killip class, major bleeding, and fasting glucose.
To mitigate the multicollinearity phenomenon during logistic regression analysis from the variables that were closely related such as kidney function (MDRD) and risk scores (TIMI and Global Registry of Acute Coronary Events [GRACE], which include numerous clinical and laboratory variables), we selected those variables statistical significance via backward stepwise selection and clinical relevance (eg, MDRD to express kidney function) and excluded composite risk scores. All statistical tests were 2-tailed and were performed using SPSS version 20 (IBM, Armonk, NY, USA). A p value <0.05 was considered statistically significant.
Results
A total of 1852 patients undergoing PIS were evaluated, of which 160 (9%) were elderly (≥75 years). Table 1 shows the demographic and clinical characteristics in the two groups. Compared to patients <75 years of age, those ≥75 years of age were more often female, had a lower body mass index; higher rates of hypertension; higher incidence of hypothyroidism; more chronic renal failure (eGFR ˂60 mL/min evaluated by MDRD); more prior stroke; more diabetes; and more atypical chest pain. There were differences in the final TIMI flow 3 between the groups and in final blush grade 3, which were both lower in elderly patients.
 |
Table 1 Baseline Clinical Characteristics |
Clinical Outcomes
Table 2 shows in-hospital clinical outcomes comparing the two groups. Compared to patients <75 years, in-hospital MACE and mortality were higher in elderly patients (6.5% versus 19.4%, p<0.001; and 4.0% versus 18.2%, p<0.001; respectively). Elderly patients had higher rates of major bleeding (2.7% versus 5.6%, p=0.04) and a higher incidence of cardiogenic shock (7.0% versus 19.6%, p<0.001).
 |
Table 2 Procedural Characteristics Between Patients Age ˂75 Years and Age ≥75 Years |
By multivariable analysis, the independent predictors of MACE were: Age ≥75 years (odds ratio [OR] 3.57, 95% CI 1.72–7.42, p=0.001), female sex (OR 2.30, 95% CI 1.23–4.30, p=0.009), Killip class 3 or 4 (OR 14.75, 95% CI 7.77–28.03, p<0.001), fasting glucose (OR 1.06, 95% CI 1.03–1.09, p<0.001), and major bleeding (OR 4.83, 95% CI 2.00–11.64, p<0.001) (Table 3). The independent predictors of mortality were: Age ≥75 years (OR 2.07, 95% CI 1.12–3.82, p=0.02), female sex (OR 1.90, 95% CI 1.18–3.05, p=0.008), Killip class 3 or 4 (OR 6.82, 95% CI 4.11–11.33, p<0.001), fasting glucose (OR 1.04, 95% CI 1.01–1.06, p=0.003), and major bleeding (OR 3.46, 95% CI 1.60–7.51, p=0.002) (Table 4).
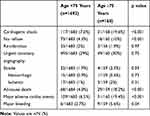 |
Table 3 In-Hospital Clinical Outcomes Between Patients Age ˂75 Years and Age ≥75 Years |
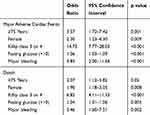 |
Table 4 Multivariable Logistic Regression Model for Major Adverse Cardiac Events and Death |
Discussion
The main findings of the present study are: (a) Compared to patients <75 years, those ≥75 years of age were more likely to be female, had a lower body mass index, and had more co-morbidities including hypertension, hypothyroidism, renal insufficiency, prior stroke, and diabetes; (b) the rate of cardiogenic shock and major bleeding was 2-fold higher in patients ≥75 years compared with those with <75 years; and (c) the rates of MACE and mortality in patients ≥75 years were 3 and 4 times higher than in patients <75 years, respectively, and after multivariable analysis, age ≥75 years was an independent predictor of MACE and death.
Increasing age not only leads to a greater number of comorbidities but also increases the time of exposure to them, impacting clinical outcomes mainly in patients ≥75 years. These patients are sicker by having more comorbidities than patients <75 years of age, which is consistent with the National Registry of Myocardial Infarction.3 Although randomized clinical trials have been the ultimate level of evidence for cardiovascular care guidelines, they have been conducted predominantly in populations <75 years, thereby resulting in inappropriate extrapolation of data to older patients. Efforts have been recently made to correct these biases. For instance, the recruitment of patients aged ≥75 years increased from 2% for studies published between 1966 and 1990 to 9% from 1991 to 2000; this remains well below their true representation (37%) among all patients with myocardial infarction.5 The present analysis is the first to evaluate the impact of PIS in individuals aged ≥75 years —representing 9% of the overall population—, adding more data in this neglected subset of patients.6,7
Although ischemic heart disease develops between 7 and 10 years later in women compared to men, acute myocardial infarction remains the main cause of death in both sexes. Acute coronary syndrome occurs 3 to 4 times more in men than in women aged <60 years old, but after 75 years, women represent the majority of the patients.8 These data are consistent with our study showing a 3-fold higher rate of STEMI in men up to 75 years and then decreasing to 50% in those ≥75 years.
Factors Associated with Delay
Notwithstanding their higher risk, elderly patients with STEMI less frequently receive evidence-based therapies than younger patients, even in the absence of contraindications.9 In the Register of Information and Knowledge About Swedish Heart Intensive Care Admissions (RISK-HIA), 43% of patients with STEMI with an average age of 75 years received no reperfusion therapy.10 In addition, prehospital delays are common in older adults, possibly related to diminished chest pain sensation, living alone, fear, cognitive impairment, comorbid illness, or social constraints.11 Along with older age additional factors associated with increased delay in seeking treatment for STEMI include female sex, diabetes, awareness of risk, passivity, barriers to self-care, lower education and socioeconomic levels, and inaccurate symptom attribution.12 Although in our analysis we have not found ischemic time as a predictor of MACE or death, pre-hospital delays plausibly contribute to poorer outcomes for the elderly. Atypical clinical presentation is a factor that affects reperfusion therapy time. Indeed, in our study, 15% of the patients ≥75 years exhibited atypical pain, twice the percentage of patients <75 years. This finding is similar to the Quebec Acute Coronary Care Working Group in which 20% of patients presented with atypical pain in acute coronary syndrome, progressively increasing with advancing age.9 In reperfusion therapy the higher absolute benefit is seen among patients with higher risk, including the elderly, especially when the treatment is offered up to 120 minutes after symptom onset.13,14 The GISSI15 trial showed little advantage in using fibrinolytic agents 6 hours after symptom onset. In the present study, the ischemic time was on median 220 minutes in patients <75 years and 240 minutes in those ≥75 years (60 min higher on those ≥75 years by mean), while it was only 100 minutes in the STREAM trial. Differences in symptom onset and fibrinolytic therapy delay between the two studies may explain the lower rate of angiographic reperfusion therapy success in our study, with a final TIMI 3 flow of 83% in patients <75 years of age and 67% of those ≥75 years. Conversely, 91.1% of the patients in the STREAM trial7 obtained a final TIMI 3 flow.
MACE and Bleeding
In older patients, the evidence concerning the risk: benefit ratio of fibrinolysis treatment is less well established because the risk of related complications increases with age16 and its efficacy may diminish.17 Some studies have shown a survival advantage associated with the use of fibrinolytic therapy in patients ≥75 years of age with STEMI,10 while others found an early mortality hazard.18 In the present study, patients ≥75 years of age had clearly more MACE, death, and major bleeding. Our rates of in-hospital MACE, cardiogenic shock, and death in patients ≥75 years (19.4%, 19.6%, and 18.4%, respectively) were much higher than in the PIS arm in the STREAM trial7 at 30 days (12.4%, 4.4%, and 3.3%, respectively), with a mean age of 59.7±12; however, they were comparable with our patients aged <75 years (6.5%, 7.0%, and 4%), with a mean age of 58.1±11.
Despite increasing risks with fibrinolytic therapy in the elderly, adverse outcomes for untreated STEMI remain high. Therefore, the risk of bleeding, especially intracranial hemorrhage, must be weighed against mortality risk.16 Hence, concerns persist in observational data that very elderly patients may experience short-term adverse effects from fibrinolytic therapy sufficient to counterbalance benefits, although the current STEMI guidelines do not exclude a PIS in the elderly.1 The reductions in age-specific doses were successfully performed in the adjuvant drugs to the fibrinolysis in the STREAM trial,6 but in our study no fibrinolytic dose adjustments were applied for elderly patients. The established benefit in lives saved in age-independent fibrinolysis needs to be balanced against the increased risk of bleeding, given that timely fibrinolytic therapy prevents 30 early deaths per 1000 patients treated within 6 hours after initiation of symptoms.18–20 Our higher (non-cerebral) bleeding rate occurred in 2.7% of patients <75 years and 5.6% of patients ≥75 years; in addition, there was a non-statistically but a numerically higher rate of hemorrhagic or ischemic stroke in patients ≥75 years compared with younger patients (1.9% and 2.5%, respectively). In the STREAM study,7 the highest bleeding rate was 6.5%, and the highest rate of hemorrhagic or ischemic stroke was 1.6%. In that study, prehospital fibrinolysis with timely coronary angiography resulted in effective reperfusion in patients with early STEMI who could not undergo primary PCI within 1 hour after the first medical contact. The impact of PIS versus pPCI in the older population with STEMI in unclear; the randomized STREAM-2 (NCT02777580) will address the safety and efficacy of PIS compared with pPCI in a population of elderly patients.
Limitations
This is a subgroup analysis from an observational study and does not intend to suggest that these results are better or equivalent to the systematic use of pPCI. Thus, whether which strategy (PIS versus pPCI) should be the preferred treatment in ≥75 years is still unresolved. The present analysis is limited to state that patients ≥75 years, once treated by PIS, have more no reflow phenomenon, major bleeding, cardiogenic shock, MACE and death than younger patients. It involved only municipal emergency department and ambulances that transfer cases identified as STEMI to our hospital. Nevertheless, no case was refused by a reference hospital and, therefore, these patients represent all-comers and consecutive series without any exclusion criteria regarding demographic characteristics, co-morbidity or clinical condition; representing a real wold population in which the results can be broadly generalized. In contrast, randomized trial has several inclusion criteria accounting for the uncertainty about the extrapolation of the results. The limited sample size of patients ≥75 years of age, representing around 9%, may explain the lower than expected rate of several complications including intracerebral bleeding even with full dose of TNK. Had specific data on neurological evaluation, imaging and procedural characteristics been available for this analysis, a more robust finding with regard to the lack of increased risk using TNK in the scenario of PIS would have been possible. The multivariable analyses performed in this study, although detailed, cannot exclude residual confounding from unmeasured variables. Furthermore, variability in the time from symptom onset to fibrinolysis measurement was not adequately accounted for in the multivariable analyses and therefore is an important potential confounder. MACE, death and other major complications were not evaluated by and independent clinical event committee (CEC); and thus, there may be disagreement between CEC and site investigator assessments in rates of clinical events. Finally, the results of this analysis apply only to STEMI patients when PIS was planned.
Conclusions
In patients with STEMI undergoing PIS, age ≥75 years was an independent factor that entailed a 3.5-fold higher rate of MACE and 2-fold higher rate of mortality compared to the patients aged <75 years. Future studies should address the risk:benefit ratio of PIS in this high-risk population.
Disclosure
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation. Francisco AH Fonseca reports grants from Astra Zeneca, personal fees from Amgen, Novo Nordisk, Takeda, Sanofi Aventis, and Novartis, outside the submitted work. The authors declare that they have no other conflicts of interest.
References
1. Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018;39(2):119–177.
2. Sharma V. Pharmaco-invasive strategy: an attractive alternative for management of ST-elevation myocardial infarction when timely primary percutaneous coronary intervention is not feasible. J Postgrad Med. 2018;64(2):73–74. doi:10.4103/jpgm.JPGM_353_17
3. Alexander KP, Newby LK, Armstrong PW, et al. Acute coronary care in the elderly, part II: ST-segment-elevation myocardial infarction: a scientific statement for healthcare professionals from the American Heart Association Council on Clinical Cardiology: in collaboration with the Society of Geriatric Cardiology. Circulation. 2007;115(19):2570–2589. doi:10.1161/CIRCULATIONAHA.107.182616
4. Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials. A consensus report from the Bleeding Academic Research Consortium. Circulation. 2011;123(23):2736–2747. doi:10.1161/CIRCULATIONAHA.110.009449
5. Lee PY, Alexander KP, Hammill BG, Pasquali SK, Peterson ED. Representation of elderly persons and women in published randomized trials of acute coronary syndromes. JAMA. 2001;286(6):708–713. doi:10.1001/jama.286.6.708
6. Sinnaeve PR, Danays T, Bogaerts K, Van de Werf F, Armstrong PW. Drug treatment of STEMI in the elderly: focus on fibrinolytic therapy and insights from the STREAM trial. Drugs Aging. 2016;33(2):109–118. doi:10.1007/s40266-016-0345-6
7. Armstrong PW, Gershlick AH, Goldstein P, et al. Fibrinolysis or primary PCI in ST-segment elevation myocardial infarction. N Engl J Med. 2013;368(15):1379–1387. doi:10.1056/NEJMoa1301092
8. Regitz-Zagrosek V, Oertelt-Prigione S, Prescott E, et al.; The EUGenMed Cardiovascular Clinical Study Group. Gender in cardiovascular diseases: impact on clinical manifestations, management, and outcomes. Eur Heart J. 2016;37(1):24–34.
9. Boucher JM, Racine N, Thanh TH, et al. Age-related differences in in-hospital mortality and the use of thrombolytic therapy for acute myocardial infarction. CMAJ. 2001;164(9):1285–1290.
10. Stenestrand U, Wallentin L. Register of information and knowledge about Swedish Heart Intensive Care Admissions (RISK-HIA). Fibrinolytic therapy in patients 75 years and older with ST-segment-elevation myocardial infarction: one-year follow-up of a large prospective cohort. Arch Intern Med. 2003;163(8):965–971. doi:10.1001/archinte.163.8.965
11. Avezum A, Makdisse M, Spencer F, et al. Impact of age on management and outcome of acute coronary syndrome: observations from the Global Registry of Acute Coronary Events (GRACE). Am Heart J. 2005;149:67–73. doi:10.1016/j.ahj.2004.06.003
12. Moser DK, Kimble LP, Alberts MJ, et al. Reducing delay in seeking treatment by patients with acute coronary syndrome and stroke: a scientific statement from the American Heart Association Council on Cardiovascular Nursing and Stroke Council. Circulation. 2006;114(2):168–182. doi:10.1161/CIRCULATIONAHA.106.176040
13. White HD. Thrombolytic therapy in the elderly. Lancet. 2000;356(9247):2028–2030. doi:10.1016/S0140-6736(00)03397-3
14. Boersma E, Maas ACP, Deckers JW, Simoons ML. Early thrombolytic treatment in acute myocardial infarction: reappraisal of the golden hour. Lancet. 1996;348(9030):771–775. doi:10.1016/S0140-6736(96)02514-7
15. Rovelli F, De Vita C, Feruglio GA, et al. Effectiveness of intravenous thrombolytic treatment in acute myocardial infarction. Lancet. 1986;327(8478):397–402.
16. Maggioni AP, Franzosi MG, Santoro E, White H, Van de Werf F, Tognoni G. The risk of stroke in patients with acute myocardial infarction after thrombolytic and antithrombotic treatment. N Engl J Med. 1992;327(1):1–6. doi:10.1056/NEJM199207023270101
17. Lesnefsky EJ, Lundergan CF, Hodgson JM, et al. Increased left ventricular dysfunction in elderly patients despite successful thrombolysis: the GUSTO-1 angiographic experience. J Am Coll Cardiol. 1996;28(2):331–337. doi:10.1016/0735-1097(96)00148-9
18. Thiemann DR, Coresh J, Schulman SP, Gerstenblith G, Oetgen WJ, Powe NR. Lack of benefit for intravenous thrombolysis in patients with myocardial infarction who are older than 75 years. Circulation. 2000;101(19):2239–2246. doi:10.1161/01.CIR.101.19.2239
19. White HD, Barbash GI, Califf RM, et al. Age and outcome with contemporary thrombolytic therapy. Results from the GUSTO-I trial. Global Utilization of Streptokinase and TPA for Occluded coronary arteries trial. Circulation. 1996;94(8):1826–1833. doi:10.1161/01.CIR.94.8.1826
20. Fibrinolytic Therapy Trialists’ (FTT) Collaborative Group. Indications for fibrinolytic therapy in suspected acute myocardial infarction: collaborative overview of early mortality and major morbidity results from all randomised trials of more than 1000 patients. Lancet. 1994;343(8893):311–322. doi:10.1016/S0140-6736(94)91161-4
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
