Back to Journals » Cancer Management and Research » Volume 10
The high pCR rate of sandwich neoadjuvant treatment in locally advanced rectal cancer may translate into a better long-term survival benefit: 5-year outcome of a Phase II clinical trial
Authors Hu YH, Wei JW, Chang H , Xiao WW, Lin JZ, Cai MY, Cai PQ, Kong LH, Chen G, Pan ZZ, Zeng ZF , Ding PR, Gao YH
Received 18 March 2018
Accepted for publication 3 July 2018
Published 10 October 2018 Volume 2018:10 Pages 4363—4369
DOI https://doi.org/10.2147/CMAR.S168573
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lu-Zhe Sun
Yong-Hong Hu,1,2,* Jia-Wang Wei,1–3,* Hui Chang,1,2,* Wei-Wei Xiao,1,2 Jun-Zhong Lin,1,4 Mu-Yan Cai,1,5 Pei-Qiang Cai,1,6 Ling-Heng Kong,1,4 Gong Chen,1,4 Zhi-Zhong Pan,1,4 Zhi-Fan Zeng,1,2 Pei-Rong Ding,1,4 Yuan-Hong Gao1,2
1State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, Guangdong, People’s Republic of China; 2Department of Radiation Oncology, Sun Yat-Sen University Cancer Center, Guangzhou, Guangdong, People’s Republic of China; 3Departments of Oncology, Ganzhou People’s Hospital, Ganzhou, Jiangxi, People’s Republic of China; 4Department of Colorectal Surgery, Sun Yat-sen University Cancer Center, Guangzhou, Guangdong, People’s Republic of China; 5Department of Pathology, Sun Yat-Sen University Cancer Center, Guangzhou, Guangdong, People’s Republic of China; 6Department of Medical Imaging and Interventional Radiology, Sun Yat-Sen University Cancer Center, Guangzhou, Guangdong, People’s Republic of China
*These authors contributed equally to this work
Background: In a Phase II clinical trial, we reported the effectiveness and safety of a sandwich neoadjuvant treatment based on a modified oxaliplatin plus capecitabine (XELOX) regimen for locally advanced rectal cancer (LARC). The pathologic complete response (pCR) rate was 42.2%, and no patient presented Grade 4 acute toxicities. This study was performed to evaluate whether the high pCR rate could translate into an improved long-term survival benefit by analyzing the 5-year follow-up results of the trial.
Methods: Fifty-one patients with LARC were initially enrolled in the trial. Of these, 2 cases were eliminated due to distant metastasis before treatment. In addition, 4 cases were eliminated for refusing surgery after neoadjuvant chemoradiotherapy (NACRT). Finally, a total of 45 patients were treated with the sandwich NACRT plus total mesorectal excision. We followed up these patients and calculated their overall survival (OS) and disease-free survival (DFS) through a Kaplan–Meier approach. A log-rank test and multivariate survival analysis based on a Cox proportional hazard model were performed to explore the risk factors influencing distant metastasis.
Results: The median follow-up time was 60.8 months, and among the 45 patients analyzed, 1 (2.2%) patient suffered local recurrence, and 9 (20.0%) suffered distant metastasis. The 3-year OS and DFS were 95.6% and 84.4%, respectively. In addition, the 5-year OS and DFS were 91.1% and 80.0%, respectively. In the multivariate analysis, postsurgical pathological N stage and carbohydrate antigen 19–9 before treatment maintained statistical significance on distant metastasis.
Conclusions: The sandwich NACRT with XELOX regimen might reduce distant metastasis and improve the survival of LARC patients. However, long-term benefits should be verified through further Phase III clinical trials.
Keywords: rectal neoplasms, neoadjuvant chemoradiotherapy, locally advanced, neoadjuvant therapy, prognosis
Introduction
Currently, neoadjuvant chemoradiotherapy (NACRT) plus total mesorectal excision (TME) is a standard mode of treating locally advanced rectal cancer (LARC).1 Because preoperative radiotherapy has been proven to reduce the local recurrence rate of LARC patients,2,3 the reducing effect becomes particularly more remarkable in combination with chemotherapy.4,5 However, unfortunately, the major cause of treatment failure is still distant metastasis, from which ~27.6% of LARC patients eventually die.6 To further improve survival, it is thus necessary to intensify systemic therapy that could eliminate distant metastasis. A number of clinical trials previously attempted to apply adjuvant chemotherapy after NACRT and TME. However, the value of adjuvant chemotherapy still remains uncertain.7,8 Moreover, adjuvant chemotherapy was proven to substantially increase severe toxic effects in LARC patients.7,9
Approaches have also sought to modify the NACRT regimen to maximize systemic control and minimize adverse effects. For example, after the German CAO/ARO/AIO-04 Phase III trial added oxaliplatin to fluorouracil-based NACRT and demonstrated a better pathologic complete response (pCR, 17% vs. 13%).10, oxaliplatin became one of the first-line treatments selected for LARC patients. Nevertheless, in some other Phase III trials, oxaliplatin combined with 5-fluorouracil (5-FU) or capecitabine appeared to result in more toxicities but not better treatment efficacy.11,12 In a Phase II trial, we attempted to modify the sequence of NACRT based on oxaliplatin. We developed a sandwich-type NACRT based on an optimized XELOX (oxaliplatin plus capecitabine) regimen administered prior to, concurrently with, and following radiation therapy13 and a satisfactory short-term curative effect was achieved. The rates of pCR and multivariate regression were 42.2% and 40.0%, respectively. In addition, the toxicities were tolerable, and the incidence of Grade 3/4 toxicities was 2.0%–10.2%. This study aimed to report the 5-year follow-up results of our Phase II trial and evaluate whether the high pCR and low toxicity rates obtained by the sandwich-type NACRT in LARC patients can translate to improve long-term survival benefits.
Patients and methods
Inclusion criteria
The trial design, inclusion criteria, treatment strategies, and outcome variables were previously published in detail.13 In brief, patients with pathologically diagnosed, non-metastatic, and resectable LARC were enrolled in the trial between January 1, 2012, and December 31, 2012. The inclusion criteria were as follows: 1) the inferior margin within 12 cm of the anal verge; 2) a clinical stage of Stage II or III, according to the seventh edition of the Union for International Cancer Control/American Joint Committee on Cancer TNM classification;14 3) age at diagnosis of 18–75 years old; and 4) Eastern Cooperative Oncology Group performance status ≤2. Patients were excluded for recurrent rectal cancer, prior radiation therapy to the pelvic region, or previous diagnosis of other malignancies. All patients were required to undergo a CT scan of the chest and abdomen, an MR scan of the pelvis, and measurement of serum tumor markers, such as carcinoembryonic antigen (CEA) and carbohydrate antigen 19–9 (CA19-9).
Before treatment, all patients received detailed oral and written information on the treatment protocol and possible adverse effects and then signed an informed consent form. The trial was approved by the Sun Yat-sen University Cancer Center and conducted in accordance with the Declaration of Helsinki.
Treatment
Radiotherapy
All patients were immobilized in a prone position using an AIO Bellyboard and Pelvic Solution System (AIO Solution; Orfit Industries, Wijnegem, Belgium). Volumetric-modulated arc therapy was the irradiation treatment modality used in this study. After a CT-based simulation, target volumes were delineated according to the guidelines of the International Commission on Radiation Units and Measurements reports 50 and 62. Gross tumor volume (GTV) included the macroscopic tumor and enlarged lymph nodes, as visualized on CT or MR images. The clinical target volume (CTV) covered the GTV with a radial margin of 2 cm and included high-risk regions of lymphatic drainage. Radiotherapy was conducted by conventional fractionation (2 Gy per fraction, 1 fraction per day, 5 days per week), in which the total dose of GTV and CTV were 50 Gy and 46 Gy, respectively (Figure 1).
  | Figure 1 Treatment schedules of the sandwich neoadjuvant treatment in LARC. Abbreviation: LARC, locally advanced rectal cancer. |
Neoadjuvant chemotherapy (NACT)
The XELOX chemotherapy regimen was used in sandwich NACRT and was repeated every 21 days. Before radiotherapy, all patients received a cycle of standard XELOX chemotherapy, in which 130 mg/m2 oxaliplatin was administered on day 1, and 1,000 mg/m2 capecitabine was administered twice daily from day 1–14.
Radiotherapy began 3 weeks after the first cycle of NACT. During radiotherapy, 2 cycles of modified XELOX chemotherapy were conducted concurrently. In the modified regimen, the dose of oxaliplatin on day 1 was reduced to 100 mg/m2, but the dose of capecitabine was the same as that on the first cycle.
After radiotherapy, the fourth cycle of NACT was supplemented with the standard XELOX regimen.
Radical resection and tumor response
All patients planned to receive a radical rectal resection 6–8 weeks after NACRT. The surgery was performed according to the principles of TME. Tumor response was evaluated in patients who underwent surgery. Specimens of each patient were examined by 2 pathologists who specialized in gastrointestinal cancers.
Adjuvant chemotherapy
Adjuvant chemotherapy was applied 3–8 weeks after surgery in patients who had an R0 resection. A total of 4 cycles of chemotherapy was administered with the standard XELOX regimen.
Follow-up and endpoints
Outpatient follow-up was conducted every 3–4 months in the first 2 years after treatment and, afterwards, semiannually until death or September 30, 2017, whichever occurred first. Follow-up evaluations mainly involved a complete physical examination, digital rectal examination, thoracoabdominal CT scan, endoscopic ultrasonography, pelvic MR scan, and tests of the levels of CEA and CA19-9.
The primary endpoint was overall survival (OS), which referred to the percentage of patients who were alive after a certain time period from diagnosis. Secondary endpoints were disease-free survival (DFS), local recurrence-free survival (RFS), and distant metastasis-free survival (MFS). DFS was defined as the percentage of living patients without local recurrence or distant metastasis after a certain time period from diagnosis. In addition, the RFS/MFS ratio was defined as the percentage of patients without local recurrence or distant metastasis after a certain time period.
Statistical analysis
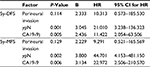
OS, DFS, RFS, and MFS were determined using a Kaplan–Meier method. A log-rank test was performed to evaluate whether the age, preoperative clinical T stage, preoperative clinical N stage, preoperative clinical stage, perineural invasion, postsurgical pathological T stage (ypT), postsurgical pathological N stage (ypN), and CEA and CA19-9 pretreatment levels were candidate risk factors of distant metastasis. Factors that achieved statistical significance were assessed by multivariate survival analysis based on a Cox proportional hazard model to confirm their independent predictive value.
Statistical analysis was conducted using SPSS Statistics 19.0 software (IBM Co., Armonk, NY, USA). A 2-sided P-value <0.05 was considered statistically significant.
Ethics approval and consent to participate
Before treatment, all patients received detailed oral and written information on the treatment protocol and possible adverse effects and signed an informed consent. The trial was approved by the institutional review board of our hospital (Sun Yat-sen University Cancer Center) and conducted in accordance with the Declaration of Helsinki.
Results
Patient profile
A total of 51 patients were involved in the trial since December 31, 2012. Of these, 2 patients were eliminated due to distant metastasis before treatment. The remaining 49 patients completed sandwich NACRT. However, 4 of these patients were excluded from this study because they had achieved a pCR after NACRT and refused the following surgery. Finally, 45 patients treated with both NACRT and TME were eligible for this study (Figure 2). Thirty-three patients received a sphincter-saving procedure. R0 resection of the primary tumor was obtained in all surgical patients, with negative distal and radial margins. The median number of retrieved lymph nodes was 5 (range 0–20 nodes) per specimen.
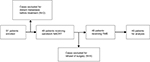  | Figure 2 Patients enrolled in this study. Abbreviations: NACRT, neoadjuvant chemoradiotherapy; TME, total mesorectal excision. |
Among the 45 included patients, there were 36 (80%) males and 9 (20%) females. Before surgery, 12 (26.7%) patients had clinical stage II disease, and 33 (73.3%) stage III disease. The numbers of patients with clinical T2, T3, and T4a diseases were 2 (4.4%), 24 (53.3%), and 19 (42.3%), respectively. In addition, the numbers of patients with clinical N0 and N1–2 diseases were 12 (26.7%) and 33 (73.3%), respectively. The distance between the inferior margin of the tumor and the anal verge was 2–12 cm (median: 6 cm). The baseline clinical characteristics of the 45 patients are summarized in Table 1.
Curative effect and toxicities
All 45 patients underwent an R0 resection. Tumor downstaging occurred in 37 (82.2%) patients, including 19 (42.2%) with ypT0N0 (pCR).
No patient presented Grade 4 acute toxicities. The incidence rates of Grade 3 leukopenia, thrombocytopenia, diarrhea, and palmar–plantar erythrodysesthesia were 2.0%, 10.2%, 8.2%, and 4.1%, respectively.
After follow-up, no patient presented Grade 4 late toxicities. Grade 3 late toxicities were mainly diarrhea and palmar–plantar erythrodysesthesia. In the first year of follow-up, 4 (8.9%) patients suffered Grade 3 diarrhea, and 1 (2.2%) patient suffered Grade 3 palmar–plantar erythrodysesthesia. From the second to fifth year, there was no new case with Grade 3 diarrhea or palmar–plantar erythrodysesthesia.
Survival analysis
The median follow-up time was 60.8 (21.6–64.7) months. There were 4 cancer deaths among the 45 patients treated with NACRT and TME. Of those, 2 cases died of pulmonary metastasis, 1 case died of hepatic metastases, and 1 died of multiple organ metastases (lungs, liver, and bones). The 3-year OS and DFS of the whole cohort analyzed were 95.6% and 84.4%, respectively. In addition, the 5-year OS and DFS were 91.1% and 80.0%, respectively. Survival curves of the whole cohort are shown in Figure 3A.
Considering the cause of treatment failure, there was 1 (2.2%) case of local recurrence and 9 (20%) cases of distant metastasis. Among the cases of distant metastasis, 4 (8.9%) suffered lung metastasis, 2 (4.4%) liver metastasis, 2 (4.4%) simultaneous metastasis in the lungs and bones, and 1 (2.2%) case suffered simultaneous metastases in the lungs, liver, and bones. Distant metastasis and local recurrence data of the whole cohort are summarized in Figure 3B.
Through the univariate analysis, perineural invasion, ypN+, and high serum level of CA19-9 pretreatment appeared to be candidate risk factors affecting distant metastasis (Table 1). Multivariate analysis by Cox regression showed that ypN+ and high serum levels of CA19-9 pretreatment were independent prognostic factors (Table 2).
Discussion
5-FU-based regimens have long been traditional NACRT regimens. However, the short-term curative effect of 5-FU-based NACRT is now unsatisfactory, such that the pCR rate is only 13%–17%, and the R0 resection rate is ~84%–95%.10,15 Moreover, the long-term clinical outcome of 5-FU-based NACRT is also very poor (5-year distant metastasis rate: 26%–36%; 5-year DFS: 68%–72%; and 5-year OS: 66%–76%).6,16
In a meta-analysis performed by An et al that included 4 trials (STAR-01, ACCORD12/0405, AIO-04, and NSABP R-04), there were 1,602 (83.2%) cases with clinical T3–4 and 1,158 (60.1%) with clinical N+ in the OX/FU group. The authors reported that the distant metastasis rate during the perioperative period was reduced (2.9% vs 5.4%, P=0.001) when 50–60 mg/m2/week oxaliplatin was added to 5-FU-based NACRT.17 However, this effect did not translate into a long-term benefit. Moreover, there was a simultaneous risk of increased toxicity. In the AIO-04 trial, a higher number of Grade 3–4 diarrheal and vomiting events were observed in patients treated with oxaliplatin plus 5-FU than in those treated with 5-FU only. In the STAR-01 trial, the oxaliplatin plus 5-FU group was also reported to have a higher incidence of Grade 3–4 adverse events than the 5-FU one (24%vs 8%, P<0.001).10,12,16,18 Therefore, modifying the NACRT sequence might be another option.
Indeed, before this Phase II trial, a number of studies attempted to implement chemotherapy before classical concurrent NACRT. Chua et al administered 4 cycles of chemotherapy with the standard XELOX regimen before NACRT. Although improved short-term curative effects were observed, compared with concurrent NACRT alone, the long-term clinical outcomes of XELOX plus NACRT were not obviously improved. The 3-year OS, DFS, and RFS were reported as 83%, 68%, and 74%, respectively, and the 3-year distant metastasis rate was 26%.19 Nevertheless, Gao et al reported the following acceptable incidences of acute toxicities: Grade three thrombocytopenia, 4.8%; Grade 3 diarrhea, 7.1%; Grade 3 proctitis, 4.8%; Grade 3 radiation dermatitis, 7.1%; and Grade 4 toxicities, 0.0%.20 In addition, some studies also attempted to administer chemotherapy between concurrent NACRT and surgery.21–23 Lee et al conducted 1 cycle of leucovorin plus 5-FU (LF) chemotherapy during the surgery waiting period and reported a 3-year OS and DFS of 86.9% and 79.5%, respectively.21 There was also no obvious improvement in the long-term outcomes. In addition, Lee et al and Habr-Gama et al reported that the Grade 3–4 toxicities of LF chemotherapy were 27.3%–93.0%.21,22
To further improve the long-term outcomes and reduce toxicities, we developed a distinct type of NACRT in a Phase II trial13 called the “sandwich type” NACRT, which involved concurrent NACRT “sandwiched” between 2 cycles of standard XELOX chemotherapy, with 1 before and 1 after NACRT. Additionally, when irradiation was applied, the dose of oxaliplatin was reduced by nearly 20%. We postulated that this sandwich NACRT regimen would bring long-term survival benefits to the LARC patients for the following reasons. First, it had a stronger dose intensity and time intensity than previous NACRT methods, which conferred it the ability to control potential subclinical distant metastasis before treatment. Second, it was inferred that the incidence of toxicities depended on the dose of the chemotherapy agents, especially the dose during radiotherapy. Lowering the dose of chemotherapy during radiotherapy might help reduce the toxicities related to concurrent NACRT.
Our Phase II trial indicated that the sandwich NACRT conferred an improved short-term curative effect compared with previous reports. Moreover, the pCR rate of the trial was 42.2%, and the toxicities were well tolerated. No patient presented Grade 4 toxicities, and Grade 3 toxicities corresponded to only 2.0%–10.2% of the patients. In addition, through this study, a satisfactory long-term outcome was found after a relatively long follow-up time. The 3-year distant metastasis rate, OS, and DFS (17.8%, 95.6%, and 84.4%, respectively) were both superior to those of all the prior clinical studies. Additionally, the 5-year OS and DFS were also ideal (91.1% and 80.0%, respectively). Some studies previously proved that the short-term DFS of LARC could serve as a surrogate endpoint of long-term OS.24,25 Therefore, we are confident that a survival benefit after a longer follow-up could be achieved.
Indeed, this study had several limitations. First, there was no control patient group because the trial was a single-arm study. Second, in multivariate analysis, CA19-9 maintained statistical significance on distant metastasis. However, because of the low number of events, this result could be a false positive. Third, the sample size of the trial was relatively small. A randomized controlled trial with larger sample sizes is needed to verify the results of this study. In fact, considering the satisfactory result achieved, we are now conducting a Phase III clinical trial.
Conclusion
In this study, preoperative sandwich NACRT based on the XELOX regimen may confer a good short-term curative effect that might reduce distant metastasis and improve survival in LARC patients. Moreover, although the results from this study require further verification by the Phase III clinical trial, this sandwich type of treatment could be well tolerated by LARC patients.
Data Sharing Statement
Please contact the author for data requests.
Disclosure
The authors report no conflicts of interest in this work.
References
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Rectal Cancer (Version 1. 2017). NCCN.org. Available from: https://www.nccn.org/store/login/login.aspx?ReturnURL=https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf. Accessed January 16, 2018. | ||
Peeters KC, Marijnen CA, Nagtegaal ID, et al. The TME trial after a median follow-up of 6 years: increased local control but no survival benefit in irradiated patients with resectable rectal carcinoma. Ann Surg. 2007;246(5):693–701. | ||
Chen C, Sun P, Rong J, Weng HW, Dai QS, Ye S. Short Course Radiation in the Treatment of Localized Rectal Cancer: A Systematic Review and Meta-Analysis. Sci Rep. 2015;5:10953. | ||
Bosset JF, Collette L, Calais G, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355(11):1114–1123. | ||
Cassidy RJ, Liu Y, Patel K, et al. Can we eliminate neoadjuvant chemoradiotherapy in favor of neoadjuvant multiagent chemotherapy for select stage II/III rectal adenocarcinomas: Analysis of the National Cancer Data base. Cancer. 2017;123(5):783–793. | ||
Sineshaw HM, Jemal A, Thomas CR, Mitin T. Changes in treatment patterns for patients with locally advanced rectal cancer in the United States over the past decade: An analysis from the National Cancer Data Base. Cancer. 2016;122(13):1996–2003. | ||
Fernández-Martos C, Pericay C, Aparicio J, et al. Phase II, randomized study of concomitant chemoradiotherapy followed by surgery and adjuvant capecitabine plus oxaliplatin (CAPOX) compared with induction CAPOX followed by concomitant chemoradiotherapy and surgery in magnetic resonance imaging-defined, locally advanced rectal cancer: Grupo cancer de recto 3 study. J Clin Oncol. 2010;28(5):859–865. | ||
Bosset JF, Calais G, Mineur L, et al. Fluorouracil-based adjuvant chemotherapy after preoperative chemoradiotherapy in rectal cancer: long-term results of the EORTC 22921 randomised study. Lancet Oncol. 2014;15(2):184–190. | ||
Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351(17):1731–1740. | ||
Rödel C, Liersch T, Becker H, Fietkau R, et al. Preoperative chemoradiotherapy and postoperative chemotherapy with fluorouracil and oxaliplatin versus fluorouracil alone in locally advanced rectal cancer: initial results of the German CAO/ARO/AIO-04 randomised phase 3 trial. Lancet Oncol. 2012;13(7):679–687. | ||
Gérard JP, Azria D, Gourgou-Bourgade S, et al. Comparison of two neoadjuvant chemoradiotherapy regimens for locally advanced rectal cancer: results of the phase III trial ACCORD 12/0405-Prodige 2. J Clin Oncol. 2010;28(10):1638–1644. | ||
Aschele C, Cionini L, Lonardi S, et al. Primary tumor response to preoperative chemoradiation with or without oxaliplatin in locally advanced rectal cancer: pathologic results of the STAR-01 randomized phase III trial. J Clin Oncol. 2011;29(20):2773–2780. | ||
Gao YH, Lin JZ, An X, et al. Neoadjuvant sandwich treatment with oxaliplatin and capecitabine administered prior to, concurrently with, and following radiation therapy in locally advanced rectal cancer: a prospective phase 2 trial. Int J Radiat Oncol Biol Phys. 2014;90(5):1153–1160. | ||
Edge S, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC Cancer Staging Manual. 7th ed. Berlin: Springer; 2010. Available from: https://cancerstaging.org/references-tools/deskreferences/Documents/AJCC%207th%20Ed%20Cancer%20Staging%20Manual.pdf. Accessed January 16, 2018. | ||
Petrelli F, Sgroi G, Sarti E, Barni S. Increasing the Interval Between Neoadjuvant Chemoradiotherapy and Surgery in Rectal Cancer: A Meta-analysis of Published Studies. Ann Surg. 2016;263(3):458–464. | ||
Rödel C, Graeven U, Fietkau R, et al. Oxaliplatin added to fluorouracil-based preoperative chemoradiotherapy and postoperative chemotherapy of locally advanced rectal cancer (the German CAO/ARO/AIO-04 study): final results of the multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2015;16(8):979–989. | ||
An X, Lin X, Wang FH, et al. Short term results of neoadjuvant chemoradiotherapy with fluoropyrimidine alone or in combination with oxaliplatin in locally advanced rectal cancer: a meta analysis. Eur J Cancer. 2013;49(4):843–851. | ||
Allegra CJ, Yothers G, O’Connell MJ, et al. Final results from NSABP protocol R-04: Neoadjuvant chemoradiation (RT) comparing continuous infusion (CIV) 5-fu with capecitabine (Cape) with or without oxaliplatin (ox) in patients with stage II and III rectal cancer. Climate of the Past. 2014;11(4):635–645. | ||
Chua YJ, Barbachano Y, Cunningham D, et al. Neoadjuvant capecitabine and oxaliplatin before chemoradiotherapy and total mesorectal excision in MRI-defined poor-risk rectal cancer: a phase 2 trial. Lancet Oncol. 2010;11(3):241–248. | ||
Gao YH, An X, Sun WJ, et al. Evaluation of capecitabine and oxaliplatin administered prior to and then concomitant to radiotherapy in high risk locally advanced rectal cancer. J Surg Oncol. 2014;109(5):478–482. | ||
Lee SJ, Kang BW, Chae YS, et al. Pilot Study of Neoadjuvant Chemoradiotherapy with Three Cycles of 5-Fluorouracil Plus Leucovorin for Treatment of Locally Advanced Rectal Cancer. Ann Surg Oncol. 2016;23(3):894–899. | ||
Habr-Gama A, Perez RO, Sabbaga J, Nadalin W, São Julião GP, Gama-Rodrigues J. Increasing the rates of complete response to neoadjuvant chemoradiotherapy for distal rectal cancer: results of a prospective study using additional chemotherapy during the resting period. Dis Colon Rectum. 2009;52(12):1927–1934. | ||
Gao YH, Zhang X, An X, et al. Oxaliplatin and capecitabine concomitant with neoadjuvant radiotherapy and extended to the resting period in high risk locally advanced rectal cancer. Strahlenther Onkol. 2014;190(2):158–164. | ||
Saad ED, Katz A, Hoff PM, Buyse M. Progression-free survival as surrogate and as true end-point: insights from the breast and colorectal cancer literature. Ann Oncol. 2009;21(1):7–12. | ||
Ciani O, Buyse M, Garside R, et al. Meta-analyses of randomized controlled trials show suboptimal validity of surrogate outcomes for overall survival in advanced colorectal cancer. J Clin Epidemiol. 2015;68(7):833–842. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.