Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
The expressions of NGF and VEGF in the fracture tissues are closely associated with accelerated clavicle fracture healing in patients with traumatic brain injury
Authors Zhang R, Liang Y, Wei S
Received 2 August 2018
Accepted for publication 11 October 2018
Published 21 November 2018 Volume 2018:14 Pages 2315—2322
DOI https://doi.org/10.2147/TCRM.S182325
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Ran Zhang,1,2 Yi Liang,1 Shuxiang Wei1
1Department of Orthopedics, Liuzhou General Hospital, Liuzhou 545006, Guangxi, China; 2The Department of Orthopedics, The Affiliated Liuzhou General Hospital of Guangxi University of Technology, Liuzhou 545006, Guangxi, China
Background: Angiogenesis and bone formation are vital for fracture healing. Nerve growth factor (NGF) not only promotes neuronal survival but also enhances the proliferation and differentiation of osteoblasts. Vascular endothelial growth factor (VEGF) plays an important role in angiogenesis. However, the potential correlation of NGF and VEGF levels with fracture healing in patients with traumatic brain injury (TBI) remains unclear.
Methods: This study enrolled 22 patients with clavicle fracture and concomitant TBI (CFT group) and 25 patients with clavicle fracture alone (CF group). Serum NGF levels were measured with ELISA. The expressions of NGF, VEGF, and CD31 in callus tissues were measured with immunohistochemistry.
Results: The fracture healing time in CFT group (82.22±13.61 days) was significantly shorter than that in CF group (127±25.05 days; P<0.001). The expression of CD31, marker of blood vessels, in callus tissues of CFT group was higher compared with that of CF group. Serum NGF levels and the expression of NGF in callus tissues of CFT group were higher than those in CF group (P<0.01). The expressions of CD31, NGF, and VEGF are correlated with shorter fracture healing time.
Conclusion: The formation of blood vessels was increased in CFT group compared with CF group. NGF and VEGF levels were higher in CFT group than in CF group and correlated with shorter fracture healing time. Accelerated fracture healing in patients with TBI may be due to NGF- and VEGF-mediated angiogenesis at the fracture site.
Keywords: traumatic brain injury, fracture healing, clavicle fracture, NGF, VEGF, angiogenesis
Introduction
Traumatic brain injury (TBI) is defined as damage to the brain caused by external mechanical force.1 Previous studies have shown an association between TBI and accelerated fracture healing.2–4 Angiogenesis and bone formation are vital for fracture healing. During fracture healing process, immune cells are rapidly recruited to the injury sites for initiating inflammatory response.5,6 Thus, mesenchymal and hematopoietic cells are recruited and activated to promote angiogenesis and bone formation.7–10 However, the mechanism of the angiogenesis and bone formation in patients with TBI remains elusive.
Fracture healing process may require a variety of cytokines, such as bone morphogenetic protein, transforming growth factor-β, fibroblast growth factor-2, and platelet-derived growth factors. Nerve growth factor (NGF) is a well-studied protein that is involved in the regulation of growth, proliferation, maintenance, and survival of certain target neurons.11,12 NGF has been shown to promote peripheral nerve regeneration in rats model.13 NGF has also been shown to accelerate wound healing.14 NGF expression has been detected in bone marrow mesenchymal stem cells of both human and rodents.15,16 The expression of NGF and its receptor in the regional callus tissue were found to be increased.17 NGF has been shown to play a vital role in osteogenesis process. NGF-TrkA signaling mediates the communication between osteoblasts and sensory nerves, which is essential for bone formation in mice.18 NGF treatment promotes osteogenesis and bone formation.19,20
Vascular endothelial growth factor (VEGF) is a signal protein that is critical for the formation of new blood vessels during embryonic development and post injury.21,22 VEGF and its corresponding receptors levels are rapidly upregulated after traumatic injury of the central nervous system,23 suggesting that VEGF could promote angiogenesis after TBI. In addition, VEGF could be released in rheumatoid arthritis in response to TNF-α, thus enhancing endothelial permeability and swelling, thereby stimulating angiogenesis.24
However, there has been no study about the association of NGF and VEGF with accelerated fracture healing in TBI patients so far. Therefore, the present study aims to address the potential role of both NGF and VEGF in the process of fracture healing in clavicle fracture patients concomitant with TBI compared to patients with clavicle fracture alone.
Materials and methods
Study subjects
This study was approved by the Ethical Review Committee of Liuzhou General Hospital and conducted under the guidance of the Declaration of Helsinki. From January 2009 to July 2017, there were 47 patients with clavicle fracture enrolled in Liuzhou General Hospital. All subjects were fully informed, and they provided written consent prior to participation. Among these 47 patients, there were 25 patients with clavicle fracture alone (CF group) and 22 patients with clavicle fracture and concomitant TBI (CFT group). Patients in CF group underwent surgery within 1 or 2 weeks post injury. Patients in CFT group include 12 patients with TBI treated with open reduction and internal fixation of the clavicle within 1 week post injury and 10 patients underwent surgery between 8 days and 14 days after brain injury. On admission, both CF and CFT groups were imaged by conventional X-rays at the clavicle fracture and diagnosed with the Glasgow Coma Scale (GCS) system. The GCS score of CFT group was <8 points, while the GCS score of CF group was >13 points. Callus volume was preoperatively measured with length, width, and height obtained from CT scan images. The following formula was used to calculate the callus volume: volume=length × width × height × π /6. The width and height in the formula were subtracted from the width and height of clavicle bone, respectively. The brain injuries were clinically defined as loss of consciousness, post-traumatic amnesia, disorientation, confusion and/or neurological deficit after injuries including cerebral concussion, cerebral contusion, laceration, subarachnoid hemorrhage, subdural hemorrhage, and intracerebral hemorrhage. Participants with any forms of prior nervous system or bone-related diseases, malignant disease, multiple fractures, diabetes, autoimmune disease, rheumatoid arthritis, other chronic inflammation diseases as well as nonsteroidal antiinflammatory drugs, immunosuppressant, history of long-time steroid, survived <1 year after surgery or bisphosphonate therapy were excluded.
Follow-up and assessment of fracture healing
Both CF and CFT groups were followed up for at least 6 months post surgery. The follow-up assessment was based on examinations at 1, 2, 4, 8, 12, 16, 20, and 24 week post injury. The fracture was recorded as clinical heal when the clinical indications as follow were achieved: 1) the patient had no tenderness and no pain in the surgical site; 2) the patient could lift a 1 kg weight for 1 minute with the affected side; and 3) the fracture line was blurred and continuous epiphyseal based on the X-ray images. The healing time was independently evaluated by two radiologists who were blinded to the presence or absence of TBI.
Immunohistochemistry
The anti-NGF/P75 (Boster Biological Technology, cat#: M01187), anti-VEGF (Boster Biological Technology, cat#: P802), and anti-CD31 (Boster Biological Technology, cat#: BM4213) were used for immunohistochemistry for measuring corresponded proteins expressions at the callus tissue between fractures. To block the endogenous peroxidase, H2O2 was added to the section after antigen repair and incubated for 10 minutes at room temperature and then washed with PBS buffer for 3 minutes three times and incubated with 100 μL of the blocking normal calf serum working solution for 15 minutes at room temperature. Then, it was incubated with 100 μL of primary antibody overnight at 4°C after removing the serum. On the second day, it was washed with PBS buffer for 3 minutes three times after being incubated at room temperature for 15 minutes. Then, it was incubated with100 μL biotin-labeled goat anti-mouse/rabbit IgG polymer at room temperature for 13 minutes and washed with PBS buffer for 3 minutes three times. It was then incubated with 100 μL of horseradish peroxidase-labeled streptolysin working solution at room temperature for10 minutes and washed with PBS buffer for 3 minutes three times. Appropriate amount of 3,3′-Diaminobenzidine (DAB) was freshly prepared and incubated for 5–8 minutes. Hematoxylin and eosin stain was incubated for 20 seconds and tap water was used for counterstaining.
ELISA
The procedure of ELISA for detecting serum NGF levels was followed with manufacturer’s instruction (EK0471, Boster Biological Technology). Briefly, the serum and standard solution were added to each well. The reactions were incubated for 2.5 hours at room temperature. After that, the wells were washed with washing buffer and then incubated with anti-NGF antibody for 1 hour at room temperature. After washing, streptavidin solution was added and incubated for 45 minutes and then substrate reagent was incubated for 30 minutes at room temperature. Finally, stop solution was added to stop the reaction. SPECTROstar Nano plate reader (BMG Labtech) was used to measure the value of OD 450 nm. NGF concentrations were calculated by comparing the value of the serum to the standard.
Image analysis
Scanning was performed on each slice, and the top left, top right, bottom right, bottom left, and the middle region were selected for five fields of view, which were imaged with the Minmei microscope digital imaging system (V9.5.2). Image-pro plus 6.0 (Media Cybernetics, Rockville, MD, USA) was used to analyze the images. In order to train the software, positive cells were manually selected in several images. After that, the sensitivity was set to level 4, and all positive cells were automatically counted and processed by the software. The percentage of blood vessels in each visual field (CD31 positive area) was calculated as a percentage of the total visual field area with the Image pro software. The following formula was used to calculate vessel density: vessel density =vessel area/total area*100%.
Statistical analysis
Statistical Package for Social Sciences software (SPSS Inc., Chicago, IL, USA) version 16.0 for Windows was used for statistical analysis. The mean SD or median (interquartile range) was used for presenting the data. Data normality was analyzed by Kolmogorov–Smirnov test. The significance in clinical characteristics between CF and CFT groups was assessed by Student’s t-test, Mann–Whitney U test, or chi-squared test. The significance of healing time difference between CF and CFT group was assessed by clinical variables Pearson correlation. Statistical significance and correlations were defined as P<0.05.
Results
Baseline characteristics of the study groups
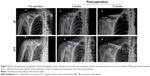
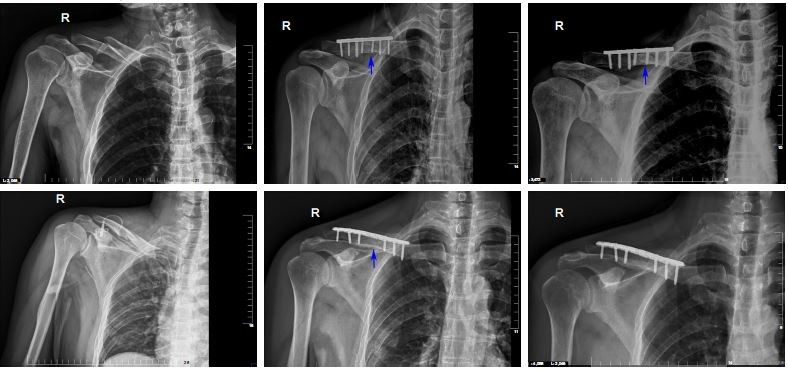
This study includes 22 patients with fracture and concomitant TBI (CFT group) and 25 patients with fracture alone (CF group). In Table 1, the demographics and clinical profile of patients in CF and CFT groups, including age, gender, fracture time from injury to surgery, are not significantly different between CF and CFT groups (for all, P>0.05). CFT group has a significantly lower GCS scores (11.5 vs 15; Table 1), shorter healing time (82.2 vs 127.0 days; Figure 1A), and larger fracture callus volume (21.9 vs 8.5 cm3; Figure 1B) compared with CF group (for all, P<0.001). Callus volume is a very important indicator of bone fracture healing. Based on our clinical experience, the larger the callus volume, the shorter will be the fracture healing time. The representative, longitudinal chest radiographs from the same patients showed fractures before surgery and the fixation at around 4 and 12 weeks after surgery (Figure 2). Healed clavicle fracture was observed at around 12 weeks after surgery in the patients of CFT group, rather than the CF group (Figure 2). In CFT group, there were eleven patients with a combined cerebral contusion, laceration, and subdural hemorrhage, seven patients with cerebral contusion and laceration only, one patient with cerebral contusion and subdural hematoma, one patient with subdural hematoma only, and two patients with cerebral concussion.
The serum NGF levels increased during fracture healing process
To characterize the relationship between NGF levels and fracture healing, we measured the serum NGF levels of each patient with ELISA at 1, 7, 14, and 21 days after surgery, respectively. The concentration of serum NGF in both CF and CFT groups increases with time and reaches the peak at 14 days after surgery (Figure 3).
The NGF and VEGF levels are higher in CFT group compared with CF group
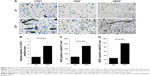
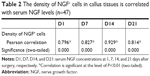
The concentration of serum NGF in CFT group is significantly higher than that in CF group at 1, 7, 14, and 21 days after surgery (P<0.001; Figure 3). CD31 is specifically expressed in endothelial cells, and its expression level represents the intensity of angiogenesis in callus tissues. Interestingly, the representative immunohistochemistry images showed that the expression of CD31 in callus tissues in CFT group is significantly higher compared with CF group (Figure 4A). Furthermore, we measured the NGF and VEGF levels in callus tissues in both CFT and CF groups with immunohistochemistry. The representative immunohistochemistry images showed that NGF and VEGF levels in CFT group are significantly higher compared with CF group (Figure 4A). To solidify this observation, we analyzed the images with Image-pro plus 6.0 and calculated the density of CD31, NGF, and VEGF positive cells, which are higher in CFT group compared with CF group (for all, P<0.001; Figure 4B–D). In addition, the density of NGF positive cells in callus tissues is correlated with serum NGF levels at 1, 7, 14, and 21 days after surgery (P<0.01; Table 2).
Higher NGF and VEGF levels predict shorter fracture healing time
To analyze the correlation among healing time and clinical variables, we performed partial correlation analysis by using surgery time post injury as control variable, because the expressions of NGF, VEGF, and CD31 might be affected by the surgery time post injury. Consistent with previous results, there is a significant negative correlation between fractures healing time and injury type (r=−0.747, P<0.001) or callus volume (r=−0.632, P<0.001). Furthermore, the fracture healing time is also negatively correlated with the density of NGF (r=−0.837, P<0.001), VEGF (r=−0.831, P<0.001), and CD31 (r=−0.847, P<0.001) positive cells, but not with age and gender (for both, P>0.05; Table 3).
Discussion
Bone fracture patients with contaminant TBI including subarachnoid hemorrhage, subdural hemorrhage, and intracerebral hemorrhage present accelerated fracture healing process compared with the patients with bone fracture alone,3,4 and the types of brain injury did not show significant difference of effect on fracture healing.25 During fracture healing process, NGF promotes regeneration of injured nerves after bone fracture,26 while VEGF stimulates the formation of blood vessels.21 However, the relative importance of NGF and VEGF levels during the fracture healing process in patients with TBI remains poorly understood.
In this study, we find that the expression of CD31 in callus tissues in CFT group is significantly higher compared with CF group, suggesting that the angiogenesis is enhanced in CFT group. Furthermore, we find that serum NGF levels increased with time and reached the peak at 14 days after surgery in both CF and CFT groups. This finding is consistent with previous observation that the peak of NGF mRNA expression occurred 10 days after distraction, and therefore, NGF may promote the development of nerves around bone tissue.27 Beside its expression in bone marrow mesenchymal stromal cells (MSCs), NGF expression was also detected in osteogenic cells, periosteal mesenchymal osteoprogenitor cells, superficial osteocytes, and osteoblastic progenitor cells and upregulated during the proliferation phase.28 VEGF expression is not limited in endothelial cells and is also detected in various nonvascular cells, such as type 2 pneumocytes, monocytes, megakaryocytes, dendritic cells, hematopoietic stem cells, lens epithelial cells, and a variety of neural cells.29 Hypoxia is one of the most important stimuli of VEGF expression and beneficial for fracture healing.30,31 After bone injury, peripheral blood enters the fracture site and forms a hematoma, which involves cells from both peripheral and intramedullary blood, as well as bone marrow cells. At this early stage, M1 macrophages, neutrophil granulocytes, and CD8+ cytotoxic T cells release proinflammatory cytokines to attract further immune cells and CD34+ endothelial progenitors. Subsequently, the expression of proangiogenic factors, such as VEGF, drive angiogenic processes and a first wave of revascularization. Furthermore, fibroblasts and MSCs migrate to the fracture site from peripheral blood and surrounding tissue for facilitating callus progression.6 In the CFT group, the accelerated healing is probably caused by enhanced angiogenesis process, as expression of VEGF increased in callus tissue. Since the NGF and VEGF levels are higher in CFT group compared with CF group, it will be of future interest to investigate the regulatory mechanism of the elevated levels of NGF and VEGF in patients with clavicle fracture and concomitant TBI. Interestingly, we showed that higher NGF and VEGF levels predict shorter fracture healing time. It could be supported by the results that NGF and VEGF promote the healing of fractures in animal models.32,33 During the inflammatory phase of bone fracture healing, NGF upregulates the expression of adhesion molecules and stimulates the proliferation of endothelial cells.34,35 Combining the correlation between NGF and VEGF levels, there is a possibility that NGF induces VEGF expression and thus enhances the proliferation of endothelial cells. NT-3, another member of NGF protein family, has been shown to induce VEGF expression at the injured site and promote angiogenesis in vitro.36 The upregulated NGF and VEGF levels may promote fracture healing through enhancing angiogenesis at the fracture site.
Although this study is the first report that NGF and VEGF levels are correlated with accelerated fracture healing in patients with TBI, however, there are several limitations. Firstly, the power to elucidate association between the NGF and VEGF levels and clavicle fracture and concomitant TBI was limited by small number subjects (n=47). Secondly, fracture and concomitant TBI animal model is needed to validate the role of NGF and VEGF in fracture process. It will be of future interest to perform prospective studies based on the results from large samples.
Conclusion
Serum NGF levels increased during repairing stage in both CF and CFT groups. NGF and VEGF levels are higher in CFT group compared with CF group correlated with a shorter fracture healing time. To our knowledge, this is the first report that NGF and VEGF levels are associated with accelerated fracture healing in patients with TBI. However, the role of NGF and VEGF levels in fracture and concomitant TBI must be carefully validated in animal models. Although NGF and VEGF levels predict shorter fracture healing time in 47 samples, prospective studies are required based on the results from larger samples.
Acknowledgment
The study was supported by the Science and Technology Program of Liuzhou (No 2015 J030524).
Disclosure
The authors report no conflicts of interest in this work.
References
Maas AI, Stocchetti N, Bullock R. Moderate and severe traumatic brain injury in adults. Lancet Neurol. 2008;7(8):728–741. | ||
Locher RJ, Lünnemann T, Garbe A, et al. Traumatic brain injury and bone healing: radiographic and biomechanical analyses of bone formation and stability in a combined murine trauma model. J Musculoskelet Neuronal Interact. 2015;15(4):309–315. | ||
Bajwa NM, Kesavan C, Mohan S. Long-term consequences of traumatic brain injury in bone metabolism. Front Neurol. 2018;9:115. | ||
Seemann R, Graef F, Garbe A, et al. Leptin-deficiency eradicates the positive effect of traumatic brain injury on bone healing: histological analyses in a combined trauma mouse model. J Musculoskelet Neuronal Interact. 2018;18(1):32–41. | ||
Baht GS, Vi L, Alman BA. The role of the immune cells in fracture healing. Curr Osteoporos Rep. 2018;16(2):138–145. | ||
Schlundt C, El Khassawna T, Serra A, et al. Macrophages in bone fracture healing: their essential role in endochondral ossification. Bone. 2018;106:78–89. | ||
Lienau J, Schmidt-Bleek K, Peters A, et al. Differential regulation of blood vessel formation between standard and delayed bone healing. J Orthop Res. 2009;27(9):1133–1140. | ||
Pajarinen J, Lin T, Gibon E, et al. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials. pii: S0142-9612(17)30834-7. | ||
Zheng Y, Huang C, Liu F, et al. Reactivation of denervated Schwann cells by neurons induced from bone marrow-derived mesenchymal stem cells. Brain Res Bull. 2018;139:211–223. | ||
Peng S, Cao L, He S, et al. An overview of long noncoding RNAs involved in bone regeneration from mesenchymal stem cells. Stem Cells Int. 2018;2018:8273648. | ||
Minnone G, de Benedetti F, Bracci-Laudiero L. NGF and its receptors in the regulation of inflammatory response. Int J Mol Sci. 2017;18(5):E1028. | ||
Bracci-Laudiero L, de Stefano ME. NGF in early embryogenesis, differentiation, and pathology in the nervous and immune systems. Curr Top Behav Neurosci. 2016;29:125–152. | ||
Sun W, Sun C, Lin H, et al. The effect of collagen-binding NGF-beta on the promotion of sciatic nerve regeneration in a rat sciatic nerve crush injury model. Biomaterials. 2009;30(27):4649–4656. | ||
Kawamoto K, Matsuda H. Nerve growth factor and wound healing. Prog Brain Res. 2004;146:369–384. | ||
Brohlin M, Kingham PJ, Novikova LN, Novikov LN, Wiberg M. Aging effect on neurotrophic activity of human mesenchymal stem cells. PLoS One. 2012;7(9):e45052. | ||
Taghi GM, Ghasem Kashani Maryam H, Taghi L, Leili H, Leyla M. Characterization of in vitro cultured bone marrow and adipose tissue-derived mesenchymal stem cells and their ability to express neurotrophic factors. Cell Biol Int. 2012;36(12):1239–1249. | ||
Majuta LA, Longo G, Fealk MN, McCaffrey G, Mantyh PW. Orthopedic surgery and bone fracture pain are both significantly attenuated by sustained blockade of nerve growth factor. Pain. 2015;156(1):157–165. | ||
Tomlinson RE, Li Z, Li Z, Minichiello L, Riddle RC, Venkatesan A, et al. NGF-TrkA signaling in sensory nerves is required for skeletal adaptation to mechanical loads in mice. Proc Natl Acad Sci U S A. 2017;114(18):E3632–E3641. | ||
Jin P, Yin F, Huang L, Zheng L, Zhao J, Zhang X. Guangxi cobra venom-derived NGF promotes the osteogenic and therapeutic effects of porous BCP ceramic. Exp Mol Med. 2017;49(4):e312. | ||
Matsumoto K, Ema M. Roles of VEGF-A signalling in development, regeneration, and tumours. J Biochem. 2014;156(1):1–10. | ||
Cao J, Wang L, Lei DL, Liu YP, Du ZJ, Cui FZ. Local injection of nerve growth factor via a hydrogel enhances bone formation during mandibular distraction osteogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(1):48–53. | ||
Wang H, Qiu L, Ma Y, et al. Naoxintong inhibits myocardial infarction injury by VEGF/eNOS signaling-mediated neovascularization. J Ethnopharmacol. 2017;209:13–23. | ||
Mecollari V, Nieuwenhuis B, Verhaagen J. A perspective on the role of class III semaphorin signaling in central nervous system trauma. Front Cell Neurosci. 2014;8:328. | ||
Zhang J, Li C, Zheng Y, Lin Z, Zhang Y, Zhang Z. Inhibition of angiogenesis by arsenic trioxide. Oncotarget. 2017;8(43):73529–73546. | ||
Yang TY, Wang TC, Tsai YH, Huang KC. The effects of an injury to the brain on bone healing and callus formation in young adults with fractures of the femoral shaft. J Bone Joint Surg Br. 2012;94(2):227–230. | ||
Sang Q, Sun D, Chen Z, Zhao W. NGF and PI3K/Akt signaling participate in the ventral motor neuronal protection of curcumin in sciatic nerve injury rat models. Biomed Pharmacother. 2018;103:1146–1153. | ||
Aiga A, Asaumi K, Lee YJ, et al. Expression of neurotrophins and their receptors tropomyosin-related kinases (Trk) under tension-stress during distraction osteogenesis. Acta Med Okayama. 2006;60(5):267–277. | ||
Su YW, Zhou XF, Foster BK, Grills BL, Xu J, Xian CJ. Roles of neurotrophins in skeletal tissue formation and healing. J Cell Physiol. 2018;233(3):2133–2145. | ||
D’Amore PA. Vascular endothelial cell growth factor-a: not just for endothelial cells anymore. Am J Pathol. 2007;171(1):14–18. | ||
Clarkin CE, Gerstenfeld LC. VEGF and bone cell signalling: an essential vessel for communication? Cell Biochem Funct. 2013;31(1):1–11. | ||
Wilson SS, Wong A, Toupadakis CA, Yellowley CE. Expression of angiopoietin-like protein 4 at the fracture site: regulation by hypoxia and osteoblastic differentiation. J Orthop Res. 2015;33(9):1364–1373. | ||
Bei C, Lin Z, Yang Z, et al. Study on effect of NGF on fracture healing. Zhongguo Xiu Fu Chong Jian WaiKe ZaZhi. 2009;23(5):570–576. | ||
Frey SP, Jansen H, Raschke MJ, Meffert RH, Ochman S. VEGF improves skeletal muscle regeneration after acute trauma and reconstruction of the limb in a rabbit model. Clin Orthop Relat Res. 2012;470(12):3607–3614. | ||
Kolostova K, Taltynov O, Pinterova D, et al. Tissue repair driven by two different mechanisms of growth factor plasmids VEGF and NGF in mice auricular cartilage: regeneration mediated by administering growth factor plasmids. Eur Arch Otorhinolaryngol. 2012;269(7):1763–1770. | ||
Raychaudhuri SK, Raychaudhuri SP, Weltman H, Farber EM. Effect of nerve growth factor on endothelial cell biology: proliferation and adherence molecule expression on human dermal microvascular endothelial cells. Arch Dermatol Res. 2001;293(6):291–295. | ||
Su YW, Chung R, Ruan CS, et al. Neurotrophin-3 induces BMP-2 and VEGF activities and promotes the bony repair of injured growth plate cartilage and bone in rats. J Bone Miner Res. 2016;31(6):1258–1274. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.