Back to Journals » Nature and Science of Sleep » Volume 12
The Evaluation of Autonomic Arousals in Scoring Sleep Respiratory Disturbances with Polysomnography and Portable Monitor Devices: A Proof of Concept Study
Authors Mayer P, Herrero Babiloni A , Beetz G, Marshansky S , Kaddaha Z, Rompré PH, Jobin V, Lavigne GJ
Received 22 April 2020
Accepted for publication 30 June 2020
Published 16 July 2020 Volume 2020:12 Pages 443—451
DOI https://doi.org/10.2147/NSS.S258276
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Ahmed BaHammam
Pierre Mayer,1 Alberto Herrero Babiloni,1– 4 Gabrielle Beetz,2 Serguei Marshansky,1 Zeina Kaddaha,1 Pierre H Rompré,3 Vincent Jobin,1 Gilles J Lavigne1– 3
1Faculté de Médecine, Hôpital Hôtel-Dieu du Centre Hospitalier de l’Université de Montréal (CHUM), Université de Montréal, Montréal, Québec, Canada; 2Research Center, Hôpital du Sacré-Coeur de Montréal, CIUSSS du Nord-de-l’île-de-Montréal, Université de Montréal, Montréal, Québec, Canada; 3Department of Oral Health, Faculté de Médecine Dentaire, Université de Montréal, Montréal, Québec, Canada; 4Division of Experimental Medicine, McGill University, Montréal, Québec, Canada
Correspondence: Alberto Herrero Babiloni
Research Center, Hôpital du Sacré-Coeur de Montreal, CIUSSS du Nord-de-l’Île-de-Montréal, Université De Montréal, 5400 Boul Gouin O, Montréal, QC H4J 1C5, Canada
Tel +1 514-338-2222
Fax +1 514-238-2531
Email [email protected]
Background: Autonomic arousals can be considered as surrogates of electroencephalography (EEG) arousals when calculating respiratory disturbance index (RDI). The main objective of this proof of concept study was to evaluate the use of heart rate acceleration (HRa) arousals associated with sleep respiratory events in a population undergoing full polysomnography (type 1) and in another undergoing portable monitor study (type 3). Our hypothesis is that when compared to other commonly used indexes, RDI based on HRa will capture more events in both types of recording.
Materials and Methods: A retrospective analysis was performed in two different populations of patients with suspected OSA: a) 72 patients undergoing one night of type 1 recording and b) 79 patients undergoing one night of type 3 recording. Variables for type 1 were 4% oxygen desaturation index (ODI), apnea/hypopnea index (AHI), RDI based on EEG arousals (RDIe), and RDI based on HRa with threshold of 5bpm (RDIa5). For type 3, variables were 4% ODI, AHI, and RDIa5 (it is not possible to calculate RDIe due to the absence of EEG). Calculated data were 1) Mean values for each sleep disturbance index in type 1 and 3 recordings; 2) Frequency of migration from lower to higher OSA severity categories using RDIa5 in comparison to AHI (thresholds: ≥ 5/h mild, ≥ 15/h moderate, ≥ 30/h severe); and 3) Bland–Altman plots to assess agreement between AHI vs RDIe and RDIa5 in type 1 population, and AHI vs RDIa5 in type 3 populations.
Results: More respiratory disturbance events were captured with RDIa5 index in both type 1 and type 3 recordings when compared to the other indexes. In type 1 recording, when using RDIa5 37% of patients classified as not having OSA with AHI were now identified as having OSA, and a total of 59% migrated to higher severity categories. In type 3 recording, similar results were obtained, as 37% of patients classified as not having OSA with AHI were now identified as having OSA using RDIa5, and a total of 55% patients migrated to higher severity categories. Mean differences for RDIa5 and AHI in type 1 and 3 populations were similar.
Conclusion: The use of autonomic arousals such as HRa can help to detect more respiratory disturbance events when compared to other indexes, being a variable that may help to capture borderline mild cases. This becomes especially relevant in type 3 recordings. Future research is needed to determine its validity, optimization, and its clinical significance.
Keywords: sleep apnea, autonomic arousals, apnea-hypopnea index, respiratory disturbance index, polysomnography, portable monitor devices
Introduction
The overall prevalence of sleep disorder breathing was estimated to be 4% for males and 2% for females in the general population, according to earlier studies done in the United States.1 However, data from Europe (Swiss cohort) suggested that these numbers may be even higher, as the prevalence of moderate to severe sleep-disordered breathing was estimated to be 23.4% in women and 49.7% in men.2 Obstructive sleep apnea (OSA) is probably the most common sleep breathing disorder, and it has been identified as a risk factor for hypertension, heart disease, stroke, and obesity among others.2–4 The laboratory overnight polysomnography (PSG) also called type 1 study, is the standard diagnostic test for identifying OSA. Nevertheless, this test can be expensive and time-consuming.5 Moreover, different inconveniences such as limited access due to distance, familial obligation, and high cost can cause delays or absence of OSA diagnosis through PSG.6 To overcome these difficulties, home portable devices (also called type 3) have been developed as an alternative to PSG, which can be used in the appropriate clinical settings.7 Although portable monitor devices capture different variables of sleep including desaturation events, most of them do not record electroencephalography (EEG) parameters or respiratory effort-related arousals (RERA). Although for diagnosing OSA no EEG criteria are required, the assessment of RERAs provides a more comprehensive evaluation of respiratory disturbances. However, the lower number of events captured by type 3 recordings and the greater denominator in respiratory indexes (total recording time vs total sleep time in type 1 recordings) have led to the underestimation of OSA’s severity in type 3 studies,8 leading to the possible omission of its diagnosis in mild cases.
To score respiratory events associated with OSA, different respiratory outcome measures are used: 4% oxygen desaturation index (ODI), apnea-hypopnea index (AHI) with different hypopnea definitions including 3% or 4% oxygen desaturation, or respiratory disturbance index (RDI), which also considers RERA trough EEG arousals (ie, apnea + hypopnea + RERA). Nevertheless, AHI is considered the main index to diagnose and categorize OSA mainly considering apnea and hypopnea events, as RERA scoring is not performed homogeneously in all centers and that not all technologies are exactly comparable.9 Those previously mentioned events are characterized by repetitive episodes of total or partial upper airway closure and by increased respiratory efforts driven by chemoreceptor and mechanoreceptor stimuli, culminating in a micro-arousal from sleep.10 However, recent literature evaluating the association between AHI and mortality risks suggests that the assessment of OSA severity based on the current AHI thresholds is not optimal,11 highlighting the need of adjusting the current thresholds (accounting for age, sex, presence of comorbidities, etc.) or investigating the use of other indexes for its diagnosis. Indeed, although RDI is not the currently recommended index, it may provide more insight about sleep fragmentation, instability, and pathophysiology, as RERAs also disrupt sleep and have clinical consequences.12 RDI is usually calculated using EEG arousals (RDIe), defined by an increase of 16 Hz or more of the EEG frequency for more than 3 seconds (AASM 2007).13 Nevertheless, other parameters that are thought to be associated to respiratory arousals, such as autonomic ones, can be used to calculate RDI, especially in the absence of EEG. One recognized autonomic arousal is heat rate acceleration (HRa), defined as an increase of certain number of beats per minute after a respiratory event. The latter can be used to calculate an RDI index called RDI with autonomic arousals.14,15 These two types of micro-arousals (EEG and HRa) are thought to have different physiological origins,16 and it appears that autonomic arousals may reflect better physiologic subcortical responses linked to upper airway opening. Therefore, HRa may be used as another tool to score and investigate apnea and hypopnea respiratory events, perhaps being a more subtle method to assess the presence of upper airway obstructions or RERAs.17 Consequently, the use of HRa to calculate RDI may lead to a more representative evaluation of OSA’s severity not only in type 1 but especially in type 3 recordings, thus becoming very clinically relevant.
Therefore, the objective of this study was to evaluate as a proof of concept the use of heart rate acceleration (HRa) arousals associated with sleep respiratory events and compare it with other indexes in a population undergoing full polysomnography (type 1) and in another undergoing portable monitor study (type 3). Our hypothesis was that when compared with other indexes, RDI based on HRa will capture more events in both type of recordings. Moreover, we also aimed to assess the level of agreement between the autonomic index and AHI type 1 and 3 populations.
Materials and Methods
Sample
A retrospective analysis was conducted in data collected consecutively from June 2012 to October 2012 in two different populations of patients with suspected OSA: a) 72 (37 males, 35 females) undergoing one night of type 1 recording, and b) 79 patients (51 males, 28 females) undergoing one night of type 3 recording, for a total 151 patients studied. Patients identified as having high risk for OSA based on clinical evaluation, which included: male sex, body mass index (BMI) >27, report of snoring or observed apnea, high blood pressure, sleepiness (Epworth >10) were oriented for an ambulatory type 3 recording. Non-obese patients with a suspicion of another sleep disorder or with atypical symptoms were directed to a type 1 study or PSG. Urgent high-risk patients were also oriented to a type 1 study for a split-night protocol with CPAP titration. To be representative of the global population referred to the clinic, no restrictions were applied regarding medication or comorbidities. The study was done according to the rules of the Centre hospitalier de l’Université de Montréal (CHUM) Ethical Committee, the Records Department and the Professional Services Direction. The CHUM Ethical Committee approved the study and all data were maintained with confidentiality.
Recording Procedures
For the in-laboratory PSG procedures (type 1), standard sleep variables were monitored: EEG recordings of C4/A1, C3/A2, O1/A2, FZ/A1; electrooculogram (EOG) for eye movements (R-EOG/A1, L-EOG/A1), electromyogram (EMG) for chin and right and left anterior tibialis, and electrocardiogram (ECG) for heart rate (bipolar) using Alice (Philips Respironics, USA). A body position sensor (Braebon Medical Corp, Canada) attached to a thoracic belt was used to monitor body position. Oxygen saturation was measured by pulse oximetry (OxiMax, Nellcor Puritan Bennett (Melville) Ltd, Canada). Tidal airflow was monitored with a nasal or oronasal pressure cannula (Braebon Medical Corp, Canada). Respiratory efforts were measured by respiratory induced plethysmography belts placed around the thorax and abdomen. Patients were also monitored with a video camera and a microphone placed at the suprasternal notch to record snoring. For the home ambulatory recordings with portable monitor devices (type 3), all of the standard sleep variables monitored during polysomnography except EEG derivations and video camera recordings were recorded. The ambulatory recording system was Alice PDx (Philips Respironics, USA).
According to the American Academy of Sleep Medicine (AASM) scoring manual of 2007, sleep was visually scored by trained technicians blinded to experimental conditions and objectives.13 Apnea and hypopnea events were also scored according to the 2007 AASM scoring criteria.13 Hypopneas events were considered when a decrease of at least 30% of flow ≥10 seconds with a desaturation ≥4% occurred.13 EEG arousals included those following movement, breathing events or those that occur spontaneously, according to AASM 2007.9,13
Other sleep variables included total minutes scored as sleep, sleep latency (number of minutes from lights out until the first epoch scored as sleep), sleep efficiency (total sleep time as a percentage of time in bed), number of awakenings during sleep, and time spend in each sleep stage: non-rapid eye movement (NREM) stage N1, NREM stage N2, slow wave sleep stage N3, and REM sleep. Autonomic arousals were scored as an increase of 5 or more heartbeats per minute (bpm) on EKG less than 20 seconds post-respiratory event. This threshold was selected based on its sensitivity to capture more events.15 Increase in heart rate was measured manually using pulse rate from saturation signals.
Scoring Procedures
For the type 1 recording, the following respiratory outcomes were scored: i) ODI with 4% oxygen desaturation, automated analysis, ii) AHI with hypopnea definition of 4% oxygen desaturation, iii) respiratory disturbance index = RDIe, where “e” is for AHI + events (hypopnea and respiratory effort-related arousals associated with EEG arousals, and iv) respiratory disturbance index with acceleration in heart rate = RDIa5, RDIe + hypopnea and respiratory effort-related arousals associated with a heart rate increase of 5 beats per min (bpm) or more. For the type 3 recordings, ODI, AHI and RDIa5 = AHI + hypopnea and respiratory effort-related arousals associated with a heart rate increase of 5 beats per min (bpm) or more were obtained. RDIe was not scored due to the absence of EEG. Scoring was done by technicians without any previous automatic analysis.
Patients were classified into the usual AHI severity categories: no apnea (0; <5), mild (1; ≥5 to <15), moderate (2; ≥15 to <30), and severe (3; ≥30), according to the AASM scoring manual 2007 and 2012.12,17
Statistical Analyses
In this study, the following analyses were performed on all the respiratory outcomes in the two populations (type 1 and type 3):
- Mean values for each sleep disturbance index: ODI, AHI, RDIe, and RDIa5 in type 1; and ODI, AHI, and RDIa5 in type 3.
- The artificial migration (movement from one category to another) frequencies from lower to higher severity categories when comparing RDIa5 vs AHI and RDIe in type 1 and RDIa5 vs AHI in type 3, considering the current thresholds for AHI categorization as a gold standard.
- Bland-Altman (B-A) plots to assess agreement between AHI vs RDIe and RDIa5 in type 1 population, and AHI vs RDIa5 in type 3 populations. B-A are plotted as the mean difference between two variables against the average of the same two. The limits of agreement were defined as the interval of the mean difference ± 1.96 x standard deviation (SD).
Moreover, two-sample t-tests and Fisher’s exact tests were used to evaluate differences between the two populations. The analyses were done with SPSS statistical software.
Results
Demographic and Clinical Characteristics of both samples can be observed in Table 1. Except for age (type 1 population was slightly younger), there were no statistical significant differences between both studied populations in those characteristics.
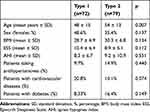 |
Table 1 Demographic and Clinical Characteristics of Both Samples |
Type 1 Scored Respiratory Disturbances Variables
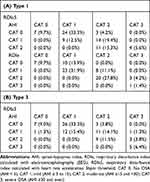
When patients were divided into three different categories according to their AHI (lower than 5, 5–15, and 15 to 30), higher number of events were observed with RDIa5 in all 3 categories in comparison to the other indexes (Figure 1). Additionally, when using RDIa5 vs AHI, 33% of patients moved to a mild OSA category (0 to 1) and 4% from no OSA to moderate (0 to 2), for a total of 37% of patients who were not considered as having OSA now having OSA with this index. Moreover, 19% of patients moved from mild to moderate category, and 6% from moderate to severe (Table 2). Overall, 58% of patients migrated to higher categories. Using RDIa5 vs RDIe, 14% of patients not detected with RDIe moved to a mild category (0 to 1), 11% from mild to moderate (1 to 2), and 4% from moderate to a severe category (2 to 3). Overall, 29% of the 72 patients migrated to a higher category (Table 2). B-A plots revealed a mean difference of 6.63 for RDIa5 vs AHI (limits of agreement −1.39 and 14.66) and 2.68 for RDIe vs AHI (limits of agreement −1.07 and 6.43) (Figure 2).
Type 3 Scored Respiratory Disturbances Variables
As in type 1 population, higher number of events were observed with RDIa5 when compared to ODI and AHI in all categories (Figure 3). Using RDIa5 vs AHI, 33% of patients not detected as OSA moved to a mild category (0 to 1), 4% from no AHI to moderate (0 to 2) for a total of 37% being detected as having OSA. Of the ones in the mild category, 14% moved to moderate (1 to 2), while 4% moved from the moderate to the severe category (2 to 3). Overall, 55% of the 79 patients recorded with Type 3 system migrated to a higher category (Table 2). B-A plots showed a mean difference of 6.91 between RDIa5 vs AHI (limits of agreement −2.90 and 16.73) (Figure 2).
Discussion
The results of this proof of concept study show that the use of HRa to calculate RDI leads to a higher number of captured events when compared with the reference standard (AHI) and other indexes in both type 1 and 3 recordings. The use of RDIa5 led to the migration of patients from lower to higher severity categories using the currently established thresholds in type 1 and 3 recordings, and notably, some patients with no diagnosis of OSA were categorized as having OSA with this index. Moreover, the similar difference observed between RDIa5 and AHI in type 1 and 3 populations suggests that RDIa5 may capture similar events in type 1 and 3 recordings, which is interesting given the absence of RDIe in the latter. Therefore, RDIa5 and other indexes based on autonomic arousals could have the potential to further characterize OSA, becoming of special interest in type 3 recordings where OSA’s severity is underestimated. However, the clinical implications of RDIa5 when compared to other indexes need to be evaluated in future studies.
There is certain controversy around the diagnosis and categorization of OSA, especially when AHI is used.18,19 AHI accounts for the number of apnea and hypopnea episodes per hour, but the definition of hypopnea may lead to considerable changes in the scoring, and therefore in the categorization of OSA according to its severity.18,20 In an older and perhaps more “conservative” definition, hypopneas were usually defined as a decrease in airflow of at least 30% accompanied by >4% oxyhemoglobin desaturation.21 However, with the claim that PSG scoring criteria have changed over time but not severity classification, a more “liberal” definition was proposed, where hypopnea is defined as a decrease in airflow of at least 30% accompanied by >3% oxyhemoglobin desaturation and/or an event-related arousal.22 Interestingly, despite being considered the gold standard index, it appears that AHI does not correlate well with clinical symptomatology18,19 and does not capture the diverse spectrum of the condition,23 which may question its use as it currently is. In fact, it appears that phenotyping OSA patients according to pathophysiologic risk factors or traits (eg, craniofacial morphology, arousal threshold, loop gain, muscle responsiveness) could help to capture OSA’s heterogeneity.23–26 RDI is another index that accounts for RERAs besides apnea/hypopnea episodes, consequently being more “inclusive” when compared with the classical AHI. While RERAs can impact sleep quality and have predictive value in cardiovascular complications according to some studies,27 evidence from a middle-aged population-based cohort study in Europe indicated a lack of association with negative clinical outcomes (eg, daytime sleepiness, hypertension, diabetes, or metabolic syndrome).28 However, despite the inclusion of RERAs in respiratory indexes is not currently mandatory, its oversight may lead to the underestimation of OSA’s severity and importantly to the possible lack of diagnosis in mild cases with type 3 recording.8
Autonomic arousals have been proposed as possible surrogates for EEG measurements in the calculation of RDI.14,15,29 HRa, pulse transit time (PTT), post-event hyperventilation, and pulse-wave amplitude (PWA) drops have been used as markers of autonomic arousals in different studies.14,15,29,30 Due to its accessibility in type 3 recordings, HRa has emerged as an interesting autonomic arousal marker, and its use as a surrogate has been explored already in other investigations.13,15,31 Although a previous study from our group showed that a cut-off value of 10 bpm correlated better with RDIe,14 to perform this proof of concept study we decided to use a cut-off value of 5 bpm based on previous literature and on the empirical impression that such a lower cut-off would help to capture more autonomic arousal events.31 However, the most accurate or appropriate cut-off is yet unknown and needs to be further investigated.
Our results indicate that the use of RDIa5 can capture more respiratory disturbance events when compared to other indexes in both type 1 and 3 recorded populations (Figure 1). Importantly, this finding is likely accentuated by using 5bpm as cut-off in our attempt to increase sensitivity, which could have increased the presence of false positives. Given the morbidity and mortality of OSA, it can be argued that its underestimation may be more clinically significant than its overestimation, as it may lead toward the absence of care or to a compromised care planning.32 Therefore, without trying to diminish the importance of a possible overestimation and presence of false positives, in this proof of concept study, we purposely selected 5bpm as a cut-off to be able to detect more events, especially in the type 3 population. Using 4% instead 3% as a threshold for ODI and hypopneas could have also increased the differences between indexes. Indeed, a 4% threshold was preferred to be capable of observing variances between indexes. Upcoming research should assess these outcomes considering different thresholds. Even though these results emphasize the possible value of using autonomic arousals to further understand and perhaps categorize better OSA patients, this study did not aim to assess the clinical relevance of these “new” detected events nor the optimal cut-off thresholds for improving accuracy. Future longitudinal studies are necessary to evaluate those in detail.
Additionally, as the magnitude between RDIa5 in type 1 and type 3 was found to be similar (Figure 2), it can be speculated that RDIa5 already includes RDIe events in type 3 studies, yet the relationship between these two indexes remains unclear. Indeed, EEG arousals may be present in the absence of autonomic arousals and vice versa. Similar results were found in other studies, where autonomic arousal was suggested to improve diagnostic accuracy of type 3 devices, thus being considered as surrogates of EEG cortical arousals (type 1 and type 3 performed in the same population).31 Nevertheless, our results must be interpreted cautiously, as the relevance of indexes based on autonomic arousals and their methodological refinement still has to be determined. More investigations evaluating HRa and other autonomic arousal measures in relation to OSA’s symptomatology, morbidity, and mortality are needed, in order to determine their clinical significance and optimize its potential use.
The possible significance of capturing the aforementioned events is illustrated by the artificial migration towards higher severity categories when using RDIa5, which was observed in both types of recordings (59% in type 1 and 55% in type 3). Thus, the use of RDIa5 could entail an important impact in the scoring and categorization of OSA, but particularly in detecting positive cases. Nevertheless, its possible association with clinical symptomatology (using different thresholds), which is fundamental for its management recommendations requires further investigation. Additionally, it is important to consider the potential influence that a “first night effect” and/or conditions associated with sleep fragmentation and autonomic dysfunction such as insomnia, chronic pain (eg, fibromyalgia, temporomandibular disorders), or mood disorders can have in the calculation these indexes.33–37 Future investigations are needed to assess the reproducibility of these indexes across different recorded nights and to evaluate how concomitant clinical disorders can affect their value.
Some limitations have to be considered in the present study. Patients with higher probabilities of having OSA are more likely to be studied with type 3 recordings, while patients with more borderline symptomatology are more likely to be studied with type 1. Hence, a selection bias is present in the studied sample. Although the patients were not preselected (besides orienting high-risk patients toward type 3 studies), the long waiting list for type 1 studies may have accentuated even more the selection bias for milder cases investigated with type 3, as they could have been redirected in order to have a first assessment more rapidly. Moreover, the retrospective nature of the data did not allow us to directly compare type 1 and 3 methods in the same sample of patients, which would have been optimal to reduce risk of bias and variability. Secondly, 9.7% of patients in the type 1 group and 14.9% in the type 3 group were taking beta-blockers, which may have affected autonomic function in those patients. However, some evidence suggests that beta-blockers have minimal to none influence in apnea related heart rate accelerations or decelerations,38 so the possible influence of these medications in our outcomes could be non-significant. Moreover, the intake of other medications or substances affecting sleep and/or autonomic function was not controlled. Finally, 4% of oxygen desaturation was used as a threshold for ODI and hypopneas instead of 3%. It is possible that this fact, along with the use of 5bpm for HRa, yielded to a higher difference between ODI/AHI and RDIa5.
Conclusion
This study serves as a proof of concept regarding the use of autonomic arousals such as HRa in aiding to the detection of more respiratory disturbance events when compared with other indexes, being of possible help capturing borderline mild cases. This becomes highly relevant in those type 3 recordings where RDIe cannot be calculated, as OSA is more likely to be underdiagnosed.
Acknowledgment
Funds from Canada Research Chair (GL) were used for the present study.
Disclosure
The authors declare that they have no conflict of interest.
References
1. Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328(17):1230–1235. doi:10.1056/NEJM199304293281704
2. Heinzer R, Vat S, Marques-Vidal P, et al. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015;3(4):310–318. doi:10.1016/S2213-2600(15)00043-0
3. Dredla BK, Castillo PR. Cardiovascular consequences of obstructive sleep apnea. Curr Cardiol Rep. 2019;21(11):137. doi:10.1007/s11886-019-1228-3
4. Lam JC, Mak JC, Ip MS. Obesity, obstructive sleep apnoea and metabolic syndrome. Respirology. 2012;17(2):223–236. doi:10.1111/j.1440-1843.2011.02081.x
5. Jobin V, Mayer P, Bellemare F. Predictive value of automated oxygen saturation analysis for the diagnosis and treatment of obstructive sleep apnoea in a home-based setting. Thorax. 2007;62(5):422–427. doi:10.1136/thx.2006.061234
6. Flemons WW, Douglas NJ, Kuna ST, Rodenstein DO, Wheatley J. Access to diagnosis and treatment of patients with suspected sleep apnea. Am J Respir Crit Care Med. 2004;169(6):668–672. doi:10.1164/rccm.200308-1124PP
7. Collop NA, Anderson WM, Boehlecke B, et al. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. Portable monitoring task force of the American academy of sleep medicine. J Clin Sleep Med. 2007;3(7):737–747.
8. Kapur VK, Auckley DH, Chowdhuri S, et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American academy of sleep medicine clinical practice guideline. J Clin Sleep Med. 2017;13(3):479–504. doi:10.5664/jcsm.6506
9. Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. Deliberations of the sleep apnea definitions task force of the American academy of sleep medicine. J Clin Sleep Med. 2012;8(5):597–619. doi:10.5664/jcsm.2172
10. Guilleminault C, Robinson A. Central sleep apnea. Neurol Clin. 1996;14(3):611–628. doi:10.1016/S0733-8619(05)70276-0
11. Korkalainen H, Toyras J, Nikkonen S, Leppanen T. Mortality-risk-based apnea-hypopnea index thresholds for diagnostics of obstructive sleep apnea. J Sleep Res. 2019;28(6):e12855. doi:10.1111/jsr.12855
12. Malhotra RK, Kirsch DB, Kristo DA, et al. Polysomnography for obstructive sleep apnea should include arousal-based scoring: an American academy of sleep medicine position statement. J Clin Sleep Med. 2018;14(7):1245–1247. doi:10.5664/jcsm.7234
13. Ruehland WR, O’Donoghue FJ, Pierce RJ, et al. The 2007 AASM recommendations for EEG electrode placement in polysomnography: impact on sleep and cortical arousal scoring. Sleep. 2011;34(1):73–81. doi:10.1093/sleep/34.1.73
14. Mayer P, Herrero Babiloni A, Aube JL, et al. Autonomic arousals as surrogates for cortical arousals caused by respiratory events: a methodological optimization study in the diagnosis of sleep breathing disorders. Nat Sci Sleep. 2019;11:423–431. doi:10.2147/NSS.S234703
15. Pitson DJ, Stradling JR. Autonomic markers of arousal during sleep in patients undergoing investigation for obstructive sleep apnoea, their relationship to EEG arousals, respiratory events and subjective sleepiness. J Sleep Res. 1998;7(1):53–59. doi:10.1046/j.1365-2869.1998.00092.x
16. Azarbarzin A, Ostrowski M, Moussavi Z, Hanly P, Younes M. Contribution of arousal from sleep to postevent tachycardia in patients with obstructive sleep apnea. Sleep. 2013;36(6):881–889. doi:10.5665/sleep.2716
17. Masa JF, Corral J, Martin MJ, et al. Assessment of thoracoabdominal bands to detect respiratory effort-related arousal. Eur Respir J. 2003;22(4):661–667. doi:10.1183/09031936.03.00010903
18. Punjabi NM. COUNTERPOINT: Is the apnea-hypopnea index the best way to quantify the severity of sleep-disordered breathing? No. Chest. 2016;149(1):16–19. doi:10.1378/chest.14-2261
19. Rapoport DM. POINT: Is the apnea-hypopnea index the best way to quantify the severity of sleep-disordered breathing? Yes. Chest. 2016;149(1):14–16. doi:10.1378/chest.15-1319
20. Macey PM, Woo MA, Kumar R, Cross RL, Harper RM. Relationship between obstructive sleep apnea severity and sleep, depression and anxiety symptoms in newly-diagnosed patients. PLoS One. 2010;5(4):e10211. doi:10.1371/journal.pone.0010211
21. Quan SF, Gillin JC, Littner MR, Shepard JW. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The report of an American Academy of sleep medicine task force. Sleep. 1999;22(5):667–689.
22. Hudgel DW. Sleep apnea severity classification - revisited. Sleep. 2016;39(5):1165–1166. doi:10.5665/sleep.5776
23. Zinchuk AV, Gentry MJ, Concato J, Yaggi HK. Phenotypes in obstructive sleep apnea: a definition, examples and evolution of approaches. Sleep Med Rev. 2017;35:113–123. doi:10.1016/j.smrv.2016.10.002
24. Lavigne GJ, Herrero Babiloni A, Beetz G, et al. Critical issues in dental and medical management of obstructive sleep apnea. J Dent Res. 2020;99(1):26–35. doi:10.1177/0022034519885644
25. Carberry JC, Amatoury J, Eckert DJ. Personalized management approach for OSA. Chest. 2018;153(3):744–755. doi:10.1016/j.chest.2017.06.011
26. Cistulli PA, Sutherland K. Phenotyping obstructive sleep apnoea-bringing precision to oral appliance therapy. J Oral Rehabil. 2019;46:1185–1191. doi:10.1111/joor.12857
27. Azarbarzin A, Ostrowski M, Hanly P, Younes M. Relationship between arousal intensity and heart rate response to arousal. Sleep. 2014;37(4):645–653. doi:10.5665/sleep.3560
28. Ogna A, Tobback N, Andries D, et al. Prevalence and clinical significance of respiratory effort-related arousals in the general population. J Clin Sleep Med. 2018;14(8):1339–1345. doi:10.5664/jcsm.7268
29. Pitson D, Chhina N, Knijn S, van Herwaaden M, Stradling J. Changes in pulse transit time and pulse rate as markers of arousal from sleep in normal subjects. Clin Sci. 1994;87(2):269–273. doi:10.1042/cs0870269
30. Masa JF, Corral J, Gomez de Terreros J, et al. Significance of including a surrogate arousal for sleep apnea-hypopnea syndrome diagnosis by respiratory polygraphy. Sleep. 2013;36(2):249–257. doi:10.5665/sleep.2384
31. Lachapelle P, Cascon J, Pamidi S, Kimoff RJ. Accuracy of portable devices in sleep apnea using oximetry-derived heart rate increases as a surrogate arousal marker. Sleep Breath. 2019;23(2):483–492. doi:10.1007/s11325-018-1708-5
32. Bianchi MT, Goparaju B. Potential underestimation of sleep apnea severity by at-home kits: rescoring in-laboratory polysomnography without sleep staging. J Clin Sleep Med. 2017;13(4):551–555. doi:10.5664/jcsm.6540
33. Rizzi M, Radovanovic D, Santus P, et al. Influence of autonomic nervous system dysfunction in the genesis of sleep disorders in fibromyalgia patients. Clin Exp Rheumatol. 2017;105(3):74–80.
34. Byun JH, Kim KT, Moon HJ, Motamedi GK, Cho YW. The first night effect during polysomnography, and patients’ estimates of sleep quality. Psychiatry Res. 2019;274:27–29. doi:10.1016/j.psychres.2019.02.011
35. Dubrovsky B, Janal MN, Lavigne GJ, et al. Depressive symptoms account for differences between self-reported versus polysomnographic assessment of sleep quality in women with myofascial TMD. J Oral Rehabil. 2017;44(12):925–933. doi:10.1111/joor.12552
36. Kalmbach DA, Buysse DJ, Cheng P, Roth T, Yang A, Drake CL. Nocturnal cognitive arousal is associated with objective sleep disturbance and indicators of physiologic hyperarousal in good sleepers and individuals with insomnia disorder. Sleep Med. 2019.
37. Kalmbach DA, Cuamatzi-Castelan AS, Tonnu CV, et al. Hyperarousal and sleep reactivity in insomnia: current insights. Nat Sci Sleep. 2018;10:193–201. doi:10.2147/NSS.S138823
38. Wolf J, Drozdowski J, Czechowicz K, et al. Effect of beta-blocker therapy on heart rate response in patients with hypertension and newly diagnosed untreated obstructive sleep apnea syndrome. Int J Cardiol. 2016;202:67–72. doi:10.1016/j.ijcard.2015.08.139
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.