Back to Journals » Infection and Drug Resistance » Volume 12
The efficacy and safety of nemonoxacin compared with levofloxacin in the treatment of community-acquired pneumonia: a systemic review and meta-analysis of randomized controlled trials
Authors Chang SP, Lee HZ , Lai CC , Tang HJ
Received 2 November 2018
Accepted for publication 19 December 2018
Published 14 February 2019 Volume 2019:12 Pages 433—438
DOI https://doi.org/10.2147/IDR.S193233
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Sahil Khanna
Shen-Peng Chang,1 Hong-Zin Lee,2 Chih-Cheng Lai,3 Hung-Jen Tang4
1Department of Pharmacy, Chi Mei Medical Center, Liouying, Taiwan; 2School of Pharmacy, China Medical University, Taichung, Taiwan; 3Department of Intensive Care Medicine, Chi Mei Medical Center, Liouying, Taiwan; 4Department of Medicine, Chi Mei Medical Center, Tainan, Taiwan
Objectives: This meta-analysis aims to assess the clinical efficacy and safety of nemonoxacin in comparison with levofloxacin in treating community-acquired pneumonia (CAP).
Materials and methods: The Pubmed, Embase, ClinicalTrials.gov., and the Cochrane databases were searched up to September 2018. Only randomized controlled trials (RCTs) evaluating nemonoxacin and levofloxacin in the treatment of CAP were included. The primary outcome was the clinical cure rate, and the secondary outcomes included the microbiologic response rate and the risk of adverse events.
Results: Three RCTs were included. Overall, nemonoxacin and levofloxacin had similar clinical cure rates in the treatment of CAP (OR =1.05, 95% CI =0.67–1.64, I2=0%). Nemonoxacin also had a microbiologic response rate similar to levofloxacin (OR =0.89, 95% CI =0.44–1.81, I2=0%). No significant differences were found in treatment-emergent adverse events between the two drugs (OR =1.08, 95% CI =0.81–1.43, I2=0%). In subgroup analysis, the similarities in the clinical cure rate, microbiologic response rate, and risk of adverse events of these two drugs remained unchanged with the dose of nemonoxacin (500 or 750 mg) and individual pathogens.
Conclusion: The clinical and microbiologic efficacy of nemonoxacin is comparable to that of levofloxacin in the treatment of CAP, and this agent is as well tolerated as levofloxacin.
Keywords: nemonoxacin, levofloxacin, community-acquired pneumonia
Introduction
Community-acquired pneumonia (CAP) is a common type of infection that can be caused by a variety of microorganisms, including typical pathogens such as Streptococcus pneumoniae and Haemophilus influenza and atypical pathogens such as Mycoplasma pneumoniae, Chlamydia pneumoniae, and Legionella species.1,2 In the face of the high morbidity and mortality of this disease, an appropriate antibiotic is the key to treatment.3 Respiratory quinolones, including levofloxacin and moxifloxacin, have good in vitro and in vivo activity against typical and atypical CAP pathogens and are recommended for treatment.3
Nemonoxacin is a recently developed novel quinolone. In contrast to other quinolones, nemonoxacin is a nonfluorinated C-8 methoxy quinolone which targets DNA gyrase and topoisomerase IV. Many in vitro studies have demonstrated its great antibacterial activity.4–8 Nemonoxacin also displays good in vitro activity against some antibiotic-resistant pathogens such as methicillin-resistant Staphylococcus aureus, penicillin-resistant Streptococcus pneumoniae, and ertapenem-non-susceptible Enterobacteriaceae.4,9–12 All of these findings suggest that nemonoxacin may play a role in the treatment of CAP.13 Nemonoxacin exhibits poor activity against Mycobacterium tuberculosis (tuberculosis [TB]), including both multidrug-resistant (MDR) TB and non-MDR-TB.14 Thus, unlike levofloxacin and moxifloxacin, which are active against TB, nemonoxacin may bring an additional benefit in the clinical setting of CAP as its use would not mask or delay the diagnosis of TB.13,15
Recently, several clinical trials16–18 have investigated the clinical efficacy and safety of nemonoxacin in the treatment of CAP. But there has been no systematic review or meta-analysis comparing the efficacy and safety of nemonoxacin and other quinolones in treating CAP. Therefore, we performed a comprehensive meta-analysis to provide better evidence on the efficacy and safety of nemonoxacin in treating CAP.
Materials and methods
Study search and selection
All clinical studies were identified by a systematic review of the literature in the PubMed, Embase, ClinicalTrials.gov., and Cochrane databases until September 2018 using the following search terms – nemonoxacin, TG-873870, and Taigexyn. Only the clinical studies that compared the clinical efficacy and adverse effects of nemonoxacin and other comparators were included. Two reviewers (Chang and Lee) searched and examined publications independently to avoid bias. When they disagreed, the third author (Lai) resolved the issue. The following data were extracted from every included study: year of publication, study design, sites and duration, antibiotic regimens of nemonoxacin and comparators, outcomes, and adverse events.
Definitions and outcomes
The primary outcome was overall clinical cure with resolution of clinical signs and symptoms of pneumonia, or recovery to the pretreatment state as the test of cure (TOC). Secondary outcomes included the microbiologic response rate and adverse events. A microbiologic response was defined as eradication (the baseline pathogen was absent) and presumed eradication (if an adequate source specimen was not available to culture, but the patient was assessed as clinically cured) at the TOC visit. Treatment-emergent adverse events were recorded, irrespective of causality. Finally, we used the results of intent-to-treat analysis for this meta-analysis.
Data analysis
This study used the Cochrane Risk of Bias Assessment tool to assess the quality of enrolled randomized controlled trials (RCTs) and the risk of bias.19 The software Review Manager, version 5.3 was used to conduct the statistical analyses. The degree of heterogeneity was evaluated with the Q statistic generated from the chi-squared test. The proportion of statistical heterogeneity was assessed by the I2 measure. Heterogeneity was considered significant when the P-value was <0.10 or the I2 was >50%. The random-effects model was used when the data were significantly heterogeneous, and the fixed-effect model was used when the data were homogenous. Pooled ORs and 95% CIs were calculated for outcome analyses.
Results
Study selection and characteristics
The search program yielded 189 references, including 47 from PubMed, 111 from Embase, 19 from the Cochrane database, and 12 from ClinicalTrials.gov. Finally, three studies16–18 fulfilling the inclusion criteria were included in this meta-analysis (Figure 1). All studies were randomized, double-blind, multicenter studies that were designed to compare the clinical efficacy and safety of nemonoxacin with levofloxacin for adult patients with CAP (Table 1). Two studies16,17 assigned CAP patients to one of three treatment groups (nemonoxacin 750 mg, nemonoxacin 500 mg, and levofloxacin 500 mg) in a 1:1:1 ratio. Another study18 assigned patients in a 2:1 ratio to receive nemonoxacin 500 mg or levofloxacin 500 mg for 7–10 days. Most of the domains were classified as having a low risk of bias, except for incomplete outcome data in one study17 (Figures 2 and 3).
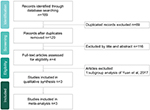  | Figure 1 Flow diagram of the study selection process. |
  | Table 1 Characteristics of included studies Abbreviation: CAP, community-acquired pneumonia |
  | Figure 2 Summary of risk of bias. |
  | Figure 3 Risk of bias per study and domain. |
Clinical efficacy
Overall, nemonoxacin had a clinical cure rate similar to levofloxacin in the treatment of CAP (OR =1.05, 95% CI =0.67–1.64, I2=0%; Figure 4). In addition, all the enrolled studies16–18 found no significant differences in the clinical cure rates of patients treated with nemonoxacin 500 mg and levofloxacin 500 mg (OR =1.01, 95% CI =0.62–1.65, I2=0%). Only two included studies16,17 compared the clinical cure rates of nemonoxacin 750 mg and levofloxacin 500 mg and no significant difference was found (OR =1.09, 95% CI =0.58–2.05, I2=0%). Nemonoxacin and levofloxacin exhibited similar clinical responses against Streptococcus pneumoniae (OR =1.20, 95% CI =0.21–6.75, I2=0%), Haemophilus spp. (OR =0.77, 95% CI =0.16–3.63, I2=0%), Staphylococcus aureus (OR =2.29, 95% CI =0.12–41.98, I2=0%), and atypical pathogens (OR =0.80, 95% CI =0.17–1.92, I2=0%).
  | Figure 4 The overall clinical cure rates of nemonoxacin and levofloxacin in the treatment of community-acquired pneumonia. |
Microbiologic response
Nemonoxacin had a microbiologic response rate similar to levofloxacin in the treatment of CAP (OR =0.89, 95% CI =0.44–1.81, I2=0%; Figure 5). Three enrolled studies16–18 compared the microbiologic response of nemonoxacin 500 mg with levofloxacin 500 mg and found no significant differences (OR =0.83, 95% CI =0.39–1.77, I2=0%). Two included studies16,17 compared the microbiologic response between nemonoxacin 750 mg and levofloxacin 500 mg and found no significant difference (OR =0.98, 95% CI =0.33–2.90, I2=0%).
  | Figure 5 The overall microbiologic response rates of nemonoxacin and levofloxacin in the treatment of community-acquired pneumonia. |
Adverse events
No significant differences were found for treatment-emergent adverse events in overall and subgroup comparisons (nemonoxacin at 500 or 750 mg vs levofloxacin 500 mg: OR =1.08, 95% CI =0.81–1.43, I2=0%, Figure 6; nemonoxacin at 500 mg vs levofloxacin 500 mg: OR =0.95, 95% CI =0.71–1.28, I2=0%; nemonoxacin at 750 mg vs levofloxacin 500 mg: OR =1.46, 95% CI =0.92–2.31, I2=0%).
  | Figure 6 The overall risks of treatment-emergent adverse events for nemonoxacin and levofloxacin in the treatment of community-acquired pneumonia. |
Comparison between nemonoxacin dosages of 750 and 500 mg
In subgroup analysis, there were no significant differences in the clinical cure rate (OR =0.99, 95% CI =0.33–2.99, I2=57%) and microbiologic response rate (OR =1.38, 95% CI =0.49–3.95, I2=0%) between different doses of nemonoxacin (750 and 500 mg). However, the 750 mg dosage of nemonoxacin had a higher risk of treatment-emergent adverse events than the 500 mg dose (OR =1.63, 95% CI =1.03–2.58, I2=0%).
Discussion
Several findings from this meta-analysis based on three RCTs showed that nemonoxacin has a clinical efficacy similar to levofloxacin in the treatment of adult patients with CAP. First, the clinical cure rate of nemonoxacin in treating CAP was as good as levofloxacin. Second, the microbiologic response rate of nemonoxacin was similar to levofloxacin. Third, subgroup analysis of different pathogens, including Streptococcus pneumoniae, Haemophilus spp., Staphylococcus aureus, and atypical pathogens, showed no significant differences in the clinical efficacy of these two drugs in the treatment of CAP. Finally, both the 500 and 750 mg dosages of nemonoxacin had clinical and microbiologic responses similar to levofloxacin. All of these findings are supported by in vitro and in vivo studies7,12,20 showing that the activity of nemonoxacin is comparable to levofloxacin. Therefore, based on the findings of these analyses, it is suggested that nemonoxacin can play an important role similar to levofloxacin in the treatment of CAP.
The risk of adverse events is another important concern in the treatment of CAP with this novel antimicrobial agent. Most of the treatment-emergent adverse events among nemonoxacin users were mild, and nausea, vomiting, leukopenia, and abnormal liver function were the most common adverse events. In this analysis, the pooled risks of treatment-emergent adverse effects were similar between nemonoxacin and levofloxacin. Even with a higher dose of nemonoxacin (750 mg), there was no significance difference in the safety between these two drugs. All of these findings suggest that nemonoxacin is as safe as levofloxacin in the treatment of CAP.
We also found that there were no significant differences in the clinical cure and microbiologic response rates between the nemonoxacin dosages of 500 and 750 mg. However, the 750 mg dosage had a significantly higher risk of adverse effects than the 500 mg dosage. Nemonoxacin 500 mg regimen may be adequate for the treatment of CAP.
This meta-analysis has one major strength. Only RCTs were included, so the risk of bias is minimized, and the level of evidence is strong. However, this meta-analysis also has several limitations. First, most cases of CAP in this meta-analysis were mild to moderate, and all patients in these three RCTs received only oral nemonoxacin. Therefore, further study is needed to investigate the use of nemonoxacin in severe CAP. Second, we did not evaluate the association between in vitro activity and the in vivo response of different organisms, especially for antibiotic-resistant pathogens. Finally, the numbers of studies and patients were limited in this meta-analysis, and therefore, the formal test for heterogeneity may be underestimating the degree of heterogeneity. In addition, only Asians and Africans were enrolled in these three RCTs. Therefore, these findings may not be generalized to other countries. Further large-scale study is warranted.
In conclusion, based on the findings of this meta-analysis of three RCTs, the clinical and microbiologic efficacy of nemonoxacin is as good as levofloxacin in the treatment of CAP, and this antibiotic is as well tolerated as levofloxacin. Thus, nemonoxacin can be recommended as an appropriate antibiotic therapy for CAP.
Disclosure
The authors report no conflicts of interest in this work.
References
Peto L, Nadjm B, Horby P, et al. The bacterial aetiology of adult community-acquired pneumonia in Asia: a systematic review. Trans R Soc Trop Med Hyg. 2014;108(6):326–337. | ||
Woodhead M. Community-acquired pneumonia in Europe: causative pathogens and resistance patterns. Eur Respir J Suppl. 2002;36:20S–27S. | ||
Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(Suppl 2):S27–S72. | ||
Li ZX, Liu YN, Wang R, Li AM. Nemonoxacin has potent activity against gram-positive, but not gram-negative clinical isolates. Clin Ter. 2015;166(6):e374–380. | ||
Lai CC, Liu WL, Ko WC, et al. Multicenter study in Taiwan of the in vitro activities of nemonoxacin, tigecycline, doripenem, and other antimicrobial agents against clinical isolates of various Nocardia species. Antimicrob Agents Chemother. 2011;55(5):2084–2091. | ||
Yang JC, Lee PI, Hsueh PR. In vitro activity of nemonoxacin, tigecycline, and other antimicrobial agents against Helicobacter pylori isolates in Taiwan, 1998–2007. Eur J Clin Microbiol Infect Dis. 2010;29(11):1369–1375. | ||
Lauderdale TL, Shiau YR, Lai JF, Chen HC, King CH. Comparative in vitro activities of nemonoxacin (TG-873870), a novel nonfluorinated quinolone, and other quinolones against clinical isolates. Antimicrob Agents Chemother. 2010;54(3):1338–1342. | ||
Chotikanatis K, Kohlhoff SA, Hammerschlag MR. In vitro activity of nemonoxacin, a novel nonfluorinated quinolone antibiotic, against Chlamydia trachomatis and Chlamydia pneumoniae. Antimicrob Agents Chemother. 2014;58(3):1800–1801. | ||
Chen YH, Liu CY, Ko WC, et al. Trends in the susceptibility of methicillin-resistant Staphylococcus aureus to nine antimicrobial agents, including ceftobiprole, nemonoxacin, and tyrothricin: results from the Tigecycline In Vitro Surveillance in Taiwan (TIST) study, 2006–2010. Eur J Clin Microbiol Infect Dis. 2014;33(2):233–239. | ||
Chen YH, Liu CY, Lu JJ, King CH, Hsueh PR. In vitro activity of nemonoxacin (TG-873870), a novel non-fluorinated quinolone, against clinical isolates of Staphylococcus aureus, enterococci and Streptococcus pneumoniae with various resistance phenotypes in Taiwan. J Antimicrob Chemother. 2009;64(6):1226–1229. | ||
Hsu MS, Liao CH, Liu CY, Yang CJ, Huang YT, Hsueh PR. In vitro susceptibilities of clinical isolates of ertapenem-non-susceptible Enterobacteriaceae to nemonoxacin, tigecycline, fosfomycin and other antimicrobial agents. Int J Antimicrob Agents. 2011;37(3):276–278. | ||
Li CR, Li Y, Li GQ, et al. In vivo antibacterial activity of nemonoxacin, a novel non-fluorinated quinolone. J Antimicrob Chemother. 2010;65(11):2411–2415. | ||
Huang CH, Lai CC, Chen YH, Hsueh PR. The potential role of nemonoxacin for treatment of common infections. Expert Opin Pharmacother. 2015;16(2):263–270. | ||
Tan CK, Lai CC, Liao CH, et al. Comparative in vitro activities of the new quinolone nemonoxacin (TG-873870), gemifloxacin and other quinolones against clinical isolates of Mycobacterium tuberculosis. J Antimicrob Chemother. 2009;64(2):428–429. | ||
Lai CC, Lee KY, Lin SW, et al. Nemonoxacin (TG-873870) for treatment of community-acquired pneumonia. Expert Rev Anti Infect Ther. 2014;12(4):401–417. | ||
Liu Y, Zhang Y, Wu J, et al. A randomized, double-blind, multicenter Phase II study comparing the efficacy and safety of oral nemonoxacin with oral levofloxacin in the treatment of community-acquired pneumonia. J Microbiol Immunol Infect. 2017;50(6):811–820. | ||
van Rensburg DJ, Perng RP, Mitha IH, et al. Efficacy and safety of nemonoxacin versus levofloxacin for community-acquired pneumonia. Antimicrob Agents Chemother. 2010;54(10):4098–4106. | ||
Yuan J, Mo B, Ma Z, et al. Safety and efficacy of oral nemonoxacin versus levofloxacin in treatment of community-acquired pneumonia: a Phase 3, multicenter, randomized, double-blind, double-dummy, active-controlled, non-inferiority trial. J Microbiol Immunol Infect. 2017;pii: S1684-1182(17):30151–30152. | ||
Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343(oct18 2):d5928. | ||
Li ZX, Liu YN, Wang R, Li AM. Nemonoxacin has potent activity against gram-positive, but not gram-negative clinical isolates. Clin Ter. 2015;166(6):e374–e380. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
