Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
The analyses of risk factors for COPD in the Li ethnic group in Hainan, People’s Republic of China
Authors Ding Y, Xu J, Yao J, Chen Y, He P, Ouyang Y, Niu H, Tian Z, Sun P
Received 10 April 2015
Accepted for publication 17 July 2015
Published 30 November 2015 Volume 2015:10(1) Pages 2593—2600
DOI https://doi.org/10.2147/COPD.S86402
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Yipeng Ding,1,* Junxu Xu,2,* Jinjian Yao,1 Yu Chen,2 Ping He,1 Yanhong Ouyang,1 Huan Niu,1 Zhongjie Tian,1 Pei Sun1
1Department of Emergency, People’s Hospital of Hainan Province, 2Department of Respiratory, The Third People’s Hospital of Haikou, Haikou, Hainan, People’s Republic of China
*These authors contributed equally to this work
Objective: To study the risk factors for chronic obstructive pulmonary disease (COPD) in Li population in Hainan province, People’s Republic of China.
Methods: Li people above 40 years of age from Hainan were chosen by stratified random cluster sampling between 2012 and 2014. All participants were interviewed with a home-visiting questionnaire, and spirometry was performed on all eligible participants. Patients with airflow limitation (forced expiratory volume in 1 second [FEV1]/forced vital capacity [FVC] <0.70) were further examined by postbronchodilator spirometry, and those with a postbronchodilator FEV1/FVC <0.70 was diagnosed with COPD. The information of physical condition and history, smoking intensity, smoking duration, second-hand smoking, education, job category, monthly household income, working years, residential environment, primary fuel for cooking and heating (biomass fuel including wood, crop residues, dung, and charcoal, or modern fuel such as natural gas, liquefied petroleum gas, electricity, and solar energy), ventilated kitchen, heating methods, air pollution, recurrent respiratory infections, family history of respiratory diseases, cough incentives, and allergies of COPD and non-COPD subjects was analyzed by univariate and multivariate logistic regression models to identify correlated risk factors for COPD.
Results: Out of the 5,463 Li participants, a total of 277 COPD cases were identified by spirometry, and 307 healthy subjects were randomly selected as controls. Univariate logistic regression analyses showed that older people (65 years and above), low body mass index (BMI), biomass smoke, 11–20 and >20 cigarettes/day, smoking for 40 years or more, second-hand smoking, recurrent respiratory infections, and induced cough were risk factors for COPD, whereas high BMI, high education level, and presence of ventilated kitchen were protective factors. Subsequent multivariate logistic regression model further demonstrated that aging, low BMI, biomass smoke, >20 cigarettes/day, and recurrent respiratory tract infections were high-risk factors for COPD in the Li population.
Conclusion: The incidence of COPD has a strong correlation with age, BMI, biomass smoke, >20 cigarettes/day, and recurrent respiratory infections, suggesting they were high-risk factors for COPD in Li population.
Keywords: chronic obstructive pulmonary disease, epidemiology, survey, Li population
Introduction
Chronic obstructive pulmonary disease (COPD) is a common chronic respiratory disease that is characterized by a reduction in lung function due to airflow obstruction that is not fully reversible.1 The disease is normally progressive and seriously affects the work ability and quality of life of patients. COPD has been reported as the fourth leading cause of morbidity and mortality in the USA, with gross medical costs up to $24 billion in 1993.2 Although COPD was previously regarded as a disease of high-income countries, its prevalence has now been recognized in low- and middle-income countries as well.3,4 According to the World Health Organization (WHO), COPD is currently the fourth leading cause of death worldwide,5 and is estimated to become the third by 2020.6–8 As the most common respiratory disease in the People’s Republic of China, COPD is the third and fourth leading cause of death in rural and urban areas,9 respectively, and ranks first among the burdens of diseases in the People’s Republic of China.10 Recently, the incidence and mortality of COPD is increasing with the population aging and increasing environmental pollution in the People’s Republic of China.11
The major risk factor of COPD has different patterns according to the geographic areas. Tobacco smoke (including second-hand or passive exposure) causes the COPD burden in high- and middle-income countries, whereas exposure to indoor air pollution, such as the use of biomass fuels (wood, crop residues, and charcoal) for cooking and heating, is the primary cause of COPD in low-income countries.12–15 It has been reported that almost 3 billion people worldwide use biomass as their main source of energy for cooking, heating, and other household needs, and therefore are exposed to high levels of indoor air pollution that is responsible for higher COPD risk in these communities.15,16 Other risk factors for COPD include occupational dusts and chemicals (such as vapors, irritants, and fumes), socioeconomic status (such as malnutrition and poverty), recurrent respiratory infections, a history of tuberculosis, untreated asthma, and heredity.17
Hainan, an island province at the southernmost coast of People’s Republic of China, is a multiethnic mix province, where Han and Li are the two main ethnic populations. The Li population has lived in the tropical regions for many generations with little intermarriage with the outside world. Although the prevalence of COPD in the People’s Republic of China has been well studied,9,18 in our previous study on the incidence of COPD in Hainan, it was revealed that the incidence of COPD in the Han population in Hainan was higher compared with other regions in the People’s Republic of China probably due to unique climate-related effects in Hainan province (Ding et al., unpublished data, 2011–2013). Nevertheless, little has been known about the incidence of COPD and related risk factors in the Li population. In this study, we investigated the prevalence of COPD and analyzed its association with a variety of risk factors in the Li population. To the best of our knowledge, this is the first population-based, observational study with comprehensive statistical analysis on COPD in the Li population in Hainan province in the People’s Republic of China.
Methods
Study participants
This observational study was conducted between January 2012 and December 2014 in the Qicha district in the rural area of Hainan in the People’s Republic of China. We chose Qicha district because it had a stable Li population. Participants aged above 40 years in 32 villages were chosen by stratified random cluster sampling in Qicha district. All patients and controls were members of the Li ethnic group. Healthy individuals without any chronic or severe endocrinological, metabolic, or nutritional diseases were selected as controls. Each participant signed an informed consent form. The study was approved by the Research Ethics Committee at the Hainan Provincial People’s Hospital and the Council for National Science Foundation.
Data collection
Weight and height of the participants were measured. A screening questionnaire developed from different validated questionnaires was completed during a face-to-face interview to collect personal data, including age, sex, occupation, education, respiratory symptoms, smoking intensity, smoking duration, second-hand smoking, education, job category, monthly household income, working years, residential environment, primary fuel for cooking and heating (biomass fuel including wood, crop residues, dung, and charcoal, or modern fuel such as natural gas, liquefied petroleum gas, electricity, and solar energy), ventilated kitchen, air pollution, recurrent respiratory infections, family history of respiratory diseases, cough incentives, and allergies. All interviewers were licensed respiratory physicians. After initial eligibility evaluation for spirometry, the indicated participants underwent spirometry examination according to the procedure for spirometry recommended by American Thoracic Society using portable spirometers (PowerCube-Body, Ganshorn, Germany). At least three reproducible blows with the largest and second-largest values for both forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1) within 150 mL or no more than 5% difference were recorded for each subject. The largest values for FVC and FEV1 were used for further analysis. The FEV1: FVC ratio (FEV1%) was also determined for each subject. A prebronchodilator FEV1:FVC ratio, 0.7 was used to determine whether a subsequent postbronchodilator assessment was needed to be performed. Subjects with asthma, bronchiectasis, past history of tuberculosis, or resent infection within 4–6 weeks were also excluded. The selected participants took a short-acting bronchodilator (200 μg of salbutamol), and spirometry was repeated 15 minutes thereafter. According to the criteria established by the National Heart, Lung, and Blood Institute/WHO Global Initiative for Chronic Obstructive Lung Disease,1 subjects with postbronchodilator FEV1/FVC <0.07 were defined to have COPD. Spirometers were calibrated daily with a 1 L syringe. The total number of participants in our study were 5,463 Li people, including 277 COPD patients and 5,186 healthy people. All COPD patients were included in the subsequent statistic analyses. Out of the healthy population, 307 people were randomly selected as controls in the statistic analyses.
Statistical analysis
All statistical tests were performed with SPSS version 11.5 (SPSS Inc., Chicago, IL, USA), and a P-value of 0.05 was considered statistically significant. The COPD prevalence was calculated as the ratio of the number of COPD patients to the number of all participants. Age group, body mass index (BMI), smoking intensity (the number of cigarettes/day), education, smoking duration, recurrent respiratory infections, and second-hand smoking were treated as categorical variables (Table 1). The associations between COPD and possible risk factors were evaluated by univariate logistic regression analysis. Risk factors that had a P-value <0.1 in the univariate analyses was included in subsequent multivariate regression analysis. Odd ratios (ORs) and 95% confidence interval (CI) values for COPD in relation to potential risk factors were calculated with multivariate logistic regression models. The variables of age groups, BMI, and recurrent respiratory infections were forced into the final multivariate logistic regression model.
  | Table 1 The categorical variables in logistic regression analyses |
Results
Basic characteristics of study participants
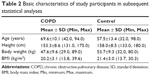
A total of 5,463 Li people (2,727 males and 2,736 females) were invited to participate in the survey, including 2,393 (43.8%) people aged 40–49 years, 1,175 (21.5%) aged 50–59 years, 1,062 (19.4%) aged 60–69 years, 567 (10.3%) aged 70–79 years, and 266 (4.9%) aged above 80 years. A total of 277 COPD cases (140 males and 137 females) were identified by spirometry tests and subjected to subsequent statistical analyses. Out of the 5,186 healthy participants, 307 (141 males and 166 females) were randomly selected as controls in the statistical analyses. Basic characteristics of both COPD and healthy control groups including age, height, body weight, and BMI are listed, respectively, in Table 2. Males represented 50.5% of the cases and 45.9% of the controls. Sex was equally distributed among COPD cases and control subjects. Cases were, on average, older than control subjects (aged 69.6 years versus 57.5 years) and showed a slightly lower BMI (20.2 kg/m2 versus 21.4 kg/m2). The number of cases in the elderly group was significantly higher than the number of control subjects (71.8% versus 32.2%). The number of underweight cases was much higher compared with control subjects. FVC, FEV1, and FEV1% of study participants were summarized in Table 3.
Univariate logistic regression analysis of risk factors for COPD
As listed in Table 4, older people (65 years and above) (OR =5.36, 95% CI, 3.76–7.64), low BMI (OR =2.62, 95% CI, 1.73–3.97), biomass smoke (OR =12.61, 95% CI, 7.55–18.28), smoking intensity (11–20 cigarettes/day, OR =1.87, 95% CI, 1.06–3.32; >20 cigarettes/day, OR =5.14, 95% CI, 2.60–10.06), smoking duration (40 years or more, OR =4.93, 95% CI, 2.48–9.82), second-hand smoking (OR =1.64, 95% CI, 1.04–2.57), recurrent respiratory infections (1–3 times/year, OR =22.47, 95% CI, 11.30–44.68; 4–5 times/year, OR =67.55, 95% CI, 29.43–155.06; and >5 times/year, OR =89.68, 95% CI, 37.52–214.32), and induced cough (OR =10.61, 95% CI, 6.52–17.25) were risk factors for COPD, whereas high BMI (OR =0.61, 95% CI, 0.37–0.99), high education level (OR =0.50, 95% CI, 0.31–0.82), and having a ventilated kitchen (OR =2.24, 95% CI, 0.56–9.06) were protective factors as indicated by univariate logistic regression analysis. The correlation between these variables and COPD was also analyzed by χ2 test (data not shown), and the results were consistent with those in the univariate logistic regression analysis. In addition, it was demonstrated that smoking history including the duration of smoking, quitting, and age of quitting smoking did not significantly affect the incidence of COPD in Li population (P>0.05). No correlation was observed between other categorical variables such as sex, family gross income, occupation, living environment, use of biomass fuels for cooking and heating, family history of respiratory diseases, cough symptoms, allergies, etc and COPD (P>0.05).
Multivariate logistic regression analysis of risk factors for COPD
Variables that had a P-value <0.1 in the univariate analyses including age, BMI, education, smoking intensity, smoke duration, second-hand smoking, recurrent respiratory infections, and induced cough were forced into subsequent multivariate logistic regression model. As listed in Table 5, older age (65 years and above), low BMI, biomass smoke, >20 cigarettes/day, and recurrent respiratory infections were risk factors for COPD. Results demonstrated that the risk for COPD in elderly group was significantly higher compared with nonelderly group (P<0.05). The risk in underweight BMI group was three times higher than that in the normal BMI group. The risk in biomass smoke group was eight times higher compared with the group using modern fuels. The risk in the group consuming >20 cigarettes/day was four times higher than that in other groups with less smoking. The risk in groups with recurrent respiratory infections (1–3, 4–5, and >5 times/year) was approximately 48, 95, and 174 times higher, respectively, compared with control group without any respiratory infection. Furthermore, no significant correlation between education, poorly ventilated kitchen, smoke duration, second-hand smoking, and induced cough was identified by multivariate logistic regression analyses (P>0.05), suggesting that they were not high-risk factors for COPD.
Discussion
Currently, COPD has been ranked the fourth leading cause of death in urban areas and the third in rural areas in the People’s Republic of China.9 Therefore, it is crucial to investigate the high-risk factors and high-risk groups for COPD in order for effective prevention and treatment. The Li population is a unique minority of Hainan province in the People’s Republic of China, and data on the prevalence of COPD and related risk factors were extremely scarce. This study was one of the first population-based, randomized surveys focused on the prevalence and risk factors for COPD in the Li population. The prevalence of spirometry-defined COPD in Li population older than 40 years was 5.07%, which was slightly lower compared with that of the general population in the People’s Republic of China (~8%) probably due to excellent air quality with little pollution in Hainan province. Results in univariate logistic regression model of correlated risk factors for COPD showed that age (65 years and above), low BMI, biomass smoke, smoking intensity (11–20 and >20 cigarettes/day), smoking duration (40 years or more), second-hand smoking, recurrent respiratory infections, and induced cough were risk factors of COPD, whereas high BMI, high education level, and presence of ventilated kitchen are protective factors. However, a strong correlation between older age (65 years and above), low BMI, biomass smoke, >20 cigarettes/day, and recurrent respiratory infections and COPD was detected in subsequent multivariate logistic regression analyses, suggesting that they were high-risk factors for COPD in the Li population.
The correlation between age and COPD has been well studied. Aging has been known as a risk factor for COPD. The incidence of COPD gradually increases with increasing age, and significant differences were detected among the incidence of COPD in different age groups, especially in rural areas surrounding cities worldwide.19–22 In 2005, Xiao et al23 found that the incident of COPD was positively correlated with age in Jining, People’s Republic of China. They further reported significant differences among the incidence of COPD in different age groups of both males and females. Jin et al also identified older age as a risk factor for COPD in Jilin, People’s Republic of China. The incidence of COPD in the age group (≥70 years) was approximately three times higher compared with that of 40–49 age group.24 Similarly, in a study conducted in Japan, the incidence of COPD in the age group ≥70 years was approximately four times higher than that in the 40–49 age group. The positive correlation between age and the incidence of COPD was confirmed in population in other countries such as the Netherlands and Uganda.7,12 Consistent with previous studies, this work demonstrated a significantly higher risk for COPD in elderly group (≥65 years) compared with nonelderly group (<65 years), indicating that older age was a risk factor for COPD in the Li population in Hainan, People’s Republic of China.
There are only few researches on the correlation between BMI and COPD, which remain controversial. In a case-control study of risk factors for COPD by Wu,25 no correlation between BMI and COPD was observed (P>0.05). Nevertheless, BMI was found to be closely associated with the incidence of COPD in other researches, and low BMI was one of the risk factors for COPD.23,26 The incidence of COPD in underweight group was significantly higher compared with normal group probably due to malnutrition and poor immune response to infections.26 Consistent with these studies, we identified BMI as one of the high-risk factors for COPD in the Li population. The risk of COPD in low BMI group was substantially increased compared with normal group (OR =3.01, 95% CI, 1.61–5.63). Being underweight might be related to malnutrition and/or low socioeconomic status, which are regarded as environmental risk factors for COPD. On the other hand, being underweight might be one of the symptoms in patients with advanced COPD since these patients often experience weight loss, loss of appetite, as well as depression and anxiety.27
It has been well known that smoking is a major risk factor for COPD.28–30 Of those who smoke, approximately 20% will get COPD,31 and of those who are lifelong smokers approximately 50% will develop COPD.32 In the US and UK, 80%–95% of those who have got COPD are either current smokers or were former smokers.31,33 The incidence rate of COPD increases with higher total smoking intensity (the product of daily smoking and smoking duration).29 Additionally, the likelihood of developing COPD is found to be associated with the age at which the person starts smoking.30 In the People’s Republic of China, it has been shown that the occurrence of COPD is closely related to smoking, in particular, smoking intensity.34 The incidence of COPD in Chinese males is found to be significantly higher than that in Chinese females, which may probably be attributed to the markedly higher smoking rate in Chinese males.19,24 Consistent with previous studies, results of univariate analysis identified smoking intensity (10–20 and >20 cigarettes/day) and smoking duration (smoking for 40 years or more) as risk factors for COPD. Moreover, the multivariate analysis showed that >20 cigarettes/day was a high-risk factor for COPD (OR =4.11).
Biomass smoke has been previously reported as a primary risk factor for the development of COPD, especially in low- and middle-income countries.12 Biomass fuels, including wood, crop residues, dung, and charcoal, are often burned inefficiently in open fires, causing high levels of indoor air pollution, and therefore lead to high incidence of COPD.24 In this study, results of univariate analysis showed that the incidence rate of COPD associated with biomass smoke was significantly higher than that of modern energy. Multivariate logistic regression analysis confirmed that the incidence rate of COPD in biomass fuel group was 8.39 times higher compared with that in modern energy group, indicating that biomass smoke was a high-risk factor for COPD.
The occurrence of COPD has been previously found to be associated with frequent childhood cough and a family history of respiratory disease (before the age of 14), indicating that recurrent respiratory infections play an important role in COPD.26,35 Respiratory infection refers to a number of infectious diseases involving the respiratory tract, and it is generally classified as an upper or a lower respiratory tract infection. Xiao et al23 found that respiratory infections were a risk factor for COPD in a multivariate logistic regression model (OR =1.389).23 In this study, recurrent respiratory infections were identified as a high-risk factor for COPD in the Li population. The risk of COPD was significantly increased with increasing number of infections. Specifically, the risk of COPD in group with more than five respiratory infections was 174 times higher compared with control subjects without any respiratory infection. Nevertheless, recurrent infection might represent exacerbation of COPD, and pulmonary function test (PFT) data before and after infection are needed to further confirm the findings in this study. It has been known that infection, especially respiratory infection, is one of the environmental risk factors for COPD and plays an important role in the pathogenesis and progression of COPD. Streptococcus pneumoniae and Haemophilus influenzae might be major pathogens for acute exacerbations of COPD.27 In addition, it was suggested that viruses such as chlamydia and Mycoplasma pneumonia might be crucial for the occurrence and development of COPD.36 However, further research will be needed to elucidate the close association between these microbes and the pathogenesis of COPD.
In conclusion, our epidemiological study has shown that older age (65 years and above), low BMI, biomass smoke, >20 cigarettes/day, and recurrent respiratory infections are high-risk factors for COPD in the Li population in Hainan, People’s Republic of China. COPD could be a major health threat to the Li population living in the rural areas of Hainan. However, the occurrence and development of COPD is a complex process involving a wide range of factors including genetic polymorphisms such as those in hypoxia-inducible factor 1α, cholinergic receptor, neuronal nicotinic, alpha polypeptide-5, and IRE-BP 2,37 and environmental factors such as air pollution.27 Further in-depth researches are needed to investigate the short- and long-term effects of these risk factors, their roles in the early development of COPD, and to develop affordable and effective intervention strategies for COPD in the Li population.
Acknowledgments
We would thank all patients and individuals in this study. We are also grateful to all clinicians who helped us to complete the interviews and spirometry in the study. This work was supported by the National Natural Science Foundation of China (number 81160008).
Disclosure
The authors report no conflicts of interest in this work.
References
Rabe KF, Hurd S, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2007;176(6):532–555. | ||
Sullivan SD, Ramsey SD, Lee TA. The economic burden of COPD. Chest. 2000;117:5S–9S. | ||
WHO. Global Surveillance, Prevention and Control of Chronic Respiratory Diseases: A Comprehensive Approach. Geneva, Switzerland: World Health Organization; 2007. | ||
Mannino M, Buist AS. Global burden of COPD: risk factors, prevalence, and future trends. Lancet. 2007;370:765–773. | ||
WHO. World Health Statistics. Geneva, Switzerland: World Health Organization; 2008. | ||
Mannino DM, Gagnon RC, Petty TL, Lydick E. Obstructive lung disease and low lung function in adults in the United States: data from the National Health and Nutrition Examination Survey, 1988–1994. Arch Intern Med. 2000;160(11):1683–1689. | ||
Rutten van-Molken MP, Feenstra TL. The burden of asthma and chronic obstructive pulmonary disease: data from The Netherlands. Pharmacoeconomics. 2001;19(Suppl 2):1–6. | ||
Murray CJL, Lopez AD. Evidence-based health policy-lessons from the global burden of disease study. Science. 1996;274:740–743. | ||
Zhong N, Wang C, Yao W, et al. Prevalence of chronic obstructive pulmonary disease in China: a large, population-based survey. Am J Respir Crit Care Med. 2007;176(8):753–760. | ||
Lopez AD, Murray CC. The global burden of disease, 1990–2020. Nat Med. 1998;4:1241–1243. | ||
Afonso AS, Verhamme KM, Sturkenboom MC, Brusselle GG. COPD in the general population: prevalence, incidence and survival. Respir Med. 2011;105(12):1872–1884. doi:10.1016/j.rmed.2011.06.012. | ||
Van Gemert F, Kirenga B, Chavannes N, et al. Prevalence of chronic obstructive pulmonary disease and associated risk factors in Uganda (FRESH AIR Uganda): a prospective cross-sectional observational study. Lancet Glob Health. 2015;3(1):e44–e51. doi:10.1016/S2214-109X(14)70337-7. | ||
Kurmi OP, Lam KB, Ayres JG. Indoor air pollution and the lung in low- and medium-income countries. Eur Respir J. 2012;40:239–254. | ||
Perez-Padilla R, Schilmann A, Riojas-Rodriguez H. Respiratory health effects of indoor air pollution. Int J Tuberc Lung Dis. 2010;14:1079–1086. | ||
Salvi S, Barnes PJ. Is exposure to biomass smoke the biggest risk factor for COPD globally? Chest. 2010;138:3–6. | ||
Van Gemert F, van der Molen T, Jones R, Chavannes N. The impact of asthma and COPD in sub-Saharan Africa. Prim Care Respir J. 2011;20:240–248. | ||
Forum of International Respiratory Societies. Respiratory diseases in the world: reality of today – opportunities for tomorrow. Available from: http://www.ersnet.org/images/firs-world-report.pdf. Accessed November 25, 2014. | ||
Lou P, Zhu Y, Chen P, et al. Prevalence and correlations with depression, anxiety, and other features in outpatients with chronic obstructive pulmonary disease in China: a cross-sectional case control study. BMC Pulm Med. 2012;12:53. doi:10.1186/1471-2466-12-53. | ||
Wang LP. Analysis of risk factors for chronic obstructive pulmonary disease in 422 cases in rural area. China Healthcare Nutr. 2012;7:2241–2242. Chinese. | ||
Wei X, Wei LQ. Chronic obstructive pulmonary disease: 2011 annual review. J Logistics Univ CPAPF. 2012;21(6):397–400. Chinese. | ||
Van Schayck CP, Loozen JMC. Detecting patients at a high risk of developing chronic obstructive pulmonary disease in general practice: cross sectional case finding study. Br Med J. 2002;324:1370. | ||
Ling M, Rong Y, Gou AS, et al. Risk factors for chronic obstructive pulmonary disease in rural areas of Xinjiang. Chin J Tuberculosis Respir Dis. 2011;34(9):666–668. Chinese. | ||
Xiao YL, Li HY, Lin Y, et al. Prevalence of chronic obstructive pulmonary disease and associated risk factors. Chin J Behav Med Sci. 2005;14(9):786–787. Chinese. | ||
Jin SG, Zhang JP, Shi L, Wang HT, Li XJ. Analysis of risk factors for chronic obstructive pulmonary disease in rural areas of Jilin. Chin Rural Health Service Admin. 2009;29(7):547–549. Chinese. | ||
Wu CJ. Risk factors for chronic obstructive pulmonary disease and health management. Theory Pract Chin Med. 2013;26(11):1523–1524. Chinese. | ||
Jiang RG, Luo DS, Huang CP, Li WM. The prevalence of chronic obstructive pulmonary disease and correlated risk factors in the rural areas in Hubei province. Chin J Epidemiol. 2007;28(10):976–979. Chinese. | ||
COPD Study Group of Chinese Society of Respiratory Disease. Treatment guidelines for chronic obstructive pulmonary disease. Chin J Tuberculosis Respir Dis. 2002;25(8):453–460. | ||
Vestbo J. Definition and overview (PDF). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Global Initiative for Chronic Obstructive Lung Disease. 2013:1–7. Accessed March 18, 2014. | ||
Tzanakis N, Anagnostopoulou U, Filaditaki V, et a1. Prevalence of COPD in Greece. Chest. 2004;125:892–900. | ||
Jyrki-Tapani K, Sovijarvi A, Lundback B. Chronic obstructive pulmonary disease in Finland: prevalence and risk factors. COPD. 2005;2:331–339. | ||
Ward H. Oxford Handbook of Epidemiology for Clinicians. Oxford, UK: Oxford University Press; 2012:289–290. | ||
Laniado-Laborín R. Smoking and chronic obstructive pulmonary disease (COPD). Parallel epidemics of the 21st century. Int J Environ Res Public Health. 2009;6(1):209–224. doi:10.3390/ijerph6010209. | ||
Rennard S. Clinical Management of Chronic Obstructive Pulmonary Disease. 2nd ed. New York, NY: Informa Healthcare; 2013:23. | ||
Liu SM, Wang XP, Wang DL, et al. Investigation of chronic obstructive pulmonary disease in Guangdong province, China. Natl Med J China. 2005;85(11):747–752. | ||
Shaheen SO, Sterne JAC, Tucker JS, et al. Birth weight, childhood lower respiratory tract infection and adult lung function. Thorax. 1998;53:549–553. | ||
Park SJ, Lee YC, Rhee YK, Lee HB. Seroprevalence of Mycoplasma pneumoniae and Chlamydia pneumoniae in stable asthma and chronic obstructive pulmonary disease. J Korean Med Sci. 2005;20(2):225–228. | ||
Ding Y, Yang D, Xun X, et al. Association of genetic polymorphisms with chronic obstructive pulmonary disease in the Hainan population: a case-control study. Int J Chron Obstruct Pulmon Dis. 2014;10:7–13. doi:10.2147/COPD.S73042. eCollection 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.