Back to Journals » Biologics: Targets and Therapy » Volume 13
Systematic review of immunomodulatory therapies for hidradenitis suppurativa
Received 29 December 2018
Accepted for publication 22 March 2019
Published 13 May 2019 Volume 2019:13 Pages 53—78
DOI https://doi.org/10.2147/BTT.S199862
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Doris Benbrook
Shi Yu Derek Lim,1 Hazel H Oon2
1Internal Medicine Residency, National Healthcare Group, Singapore, Singapore; 2Department of Dermatology, National Skin Centre, Singapore, Singapore
Background: Greater understanding of the roles of tumor necrosis factor-α, IL-1β, IL-10, and the IL-23/T-helper (Th) 17 and IL-12/Th1 pathways in immune dysregulation in moderate/severe hidradenitis suppurativa (HS) has helped in developing new regimens. We aim to review the use of different immunomodulatory therapies used to manage HS.
Methods: A comprehensive literature search was conducted on the PubMed and Clinicaltrials.gov databases from 1 January 1947 to 31 December 2018. Only clinical trials, case reports, case series and retrospective analyses published in the English language were included.
Results: Our search yielded 107 articles and 35 clinical trials, of which 15 are still ongoing. The tumor necrosis factor-α inhibitors adalimumab and infliximab were the most comprehensively studied agents. Published data from clinical trials support the efficacy of adalimumab, infliximab, anakinra, ustekinumab, bermekimab and apremilast but not etanercept and MEDI8968. Clinical trials for CJM112 have been completed, with results awaiting publication. Trials are underway for secukinumab, IFX-1, INCB054707 and bimekizumab. Biologics used in smaller cohorts include canakinumab, golimumab and rituximab. Most agents are well tolerated and demonstrate a good safety profile, with the most commonly reported adverse event being infections.
Discussion and conclusions: To date, adalimumab is the only biologic which has been approved by the United States Food and Drug Administration for HS. However, other agents also show promise, with further trials underway to evaluate their efficacy, tolerability and safety profiles. Different clinical measurement scores and endpoints used to make direct comparison difficult. Longitudinal surveillance and pooled registry data are paramount to evaluate the long-term safety profile and efficacy of therapy.
Keywords: Hidradenitis suppurativa, biologics, tumor necrosis factor, adalimumab, infliximab, secukinumab
Introduction
Hidradenitis suppurativa (HS), which has an estimated worldwide prevalence of 1%, is a chronic inflammatory follicular occlusive disease predominantly involving the intertriginous areas.1 Clinically, its manifestations vary from inflammatory nodules and abscesses to the formation of sinus tracts and scarring.2 It has a profound adverse impact on patients’ quality of life, and has been closely linked with physical and psychiatric co-morbidities including obesity, hypertension, dyslipidemia, diabetes mellitus, thyroid disorders, polycystic ovarian syndrome, arthropathies and depression.3
Pathophysiology of HS
Histopathological examination of early lesions in HS demonstrates terminal follicular hyperkeratosis, hyperplasia of the follicular epithelium and perifolliculitis. The occlusion of the terminal hair follicle results in dilation and cyst formation, followed by rupture of the hair follicle. The introduction of follicular contents to the surrounding dermis induces an inflammatory response and subsequent formation of abscess, sinus tracts, fibrosis and scars. This is worsened by biofilm formation and secondary infection.4,5
The inflammatory response in HS has in recent years been better characterized, although there are many components that remain to be elucidated. In particular, tumor necrosis factor (TNF)-α, IL-1β, IL-10, and the IL-23/T-helper (Th) 17 and IL-12/Th1 pathways play key roles in immune dysregulation in HS.6,7 In studies of HS skin, significantly increased frequencies of CD4 T cells expressing Th17-associated cytokines and TNF were found infiltrating HS skin.7 Treatment with TNF inhibitors was also related with a significant decrease in IL-17 expressing CD4 T cells in HS skin.7
Staging of HS and implications on therapy
HS has traditionally been staged according to the Hurley staging system, first proposed in 1989 (Table 1).8
 | Table 1 Hurley staging of HS |
The Hurley staging system remains useful for determining the severity of disease in individual patients but is limited in monitoring the dynamic characteristics of disease in clinical trials.9 Hence, alternative scoring systems have been developed to better evaluate the efficacy of the intervention, as shown in Table 2.
 | Table 2 Scoring systems used in grading HS severity |
Conventional medical therapy, involving oral antibiotics and topical treatments, is suitable for treatment of mild to moderate HS. However, there are patients where HS remains resistant to conventional treatment. With the discovery of the key inflammatory mediators in HS, the role of biologics and other immunomodulatory therapies in the targeted treatment of moderate-to-severe HS has been closely studied. Of these, adalimumab remains the only Food and Drug Administration (FDA)-approved biologic for the treatment of HS.10
We present a review of all biologics and immunomodulatory therapies that have been reported in the treatment of HS.
Methods
A review of the literature was conducted by multiple PubMed searches using the keywords “hidradenitis suppurativa“ or ”acne inversa”; with publication date limits from 1 January 1947 to 31 December 2018. Retrieved references were critically appraised. The inclusion criteria were original articles, reports and letters in the English language reporting the treatment of HS with biologic or other immunomodulatory agents, either alone or in combination with conventional drugs or surgery. Articles which were judged to be irrelevant based on the title, abstract or full text, were excluded from the review.
A search of the website Clinicaltrials.gov for planned, in-progress, terminated and completed clinical trials with the terms “hidradenitis suppurativa“ and ”acne inversa” was also performed up to 31 December 2018.
Results
A total of 2,088 articles were retrieved by multiple PubMed searches conducted until 31 December 2018 using the keywords “hidradenitis suppurativa” or “acne inversa”. A total of 107 relevant articles were included in the analysis. A total of 47 case reports, 29 case series, 3 retrospective analyses, 4 cohort studies and 24 articles based on clinical trial data, were selected (Figure 1).
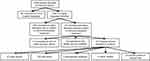 | Figure 1 Selection of articles identified by PubMed search. |
A total of 65 clinical trials were retrieved by a search of the Clinicaltrials.gov database conducted on 31 December 2018, of which 35 were related to immunomodulatory treatment. Twenty of these studies were completed or terminated. To access the results of these trials, the articles retrieved from the PubMed searches were reviewed and matched to their respective clinical trials, using the National Clinical Trial identifier, and the PubMed database was again searched using the terms (“hidradenitis suppurativa“ OR ”acne inversa”) and the medication name. Fourteen of the completed or terminated studies had published articles on PubMed (Figure 2). When trial results were not available in PubMed, results posted on Clinicaltrials.gov were used. In five cases, the trials were listed as “completed” on Clinicaltrials.gov, but neither PubMed indexed journal articles nor posted study results on Clinicaltrials.gov were found. A World Wide Web search was then performed to retrieve any study results available.
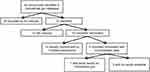 | Figure 2 Selection of articles identified by |
Biologic and other immunomodulatory therapies
A total of 19 biologic and other immunomodulatory agents reported in the treatment of HS were identified and categorized according to their mode of action (Table 3). Of these, efalizumab has been withdrawn and was thus excluded from this review. Information from individual published articles included in this review is available in Table 4.
 | Table 3 Biologics and other immunomodulatory therapies reported in the treatment of HS |
 | Table 4 Articles included in systematic review |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
 | Table 4 (Continued). |
TNF-α inhibitors
Adalimumab
Adalimumab is a recombinant human anti-TNF-α IgG1 monoclonal antibody. When used for HS, it is given subcutaneously as an initial dose of 160 mg, followed by a dose of 80 mg 2 weeks later, and a maintenance dose of 40 mg weekly thereafter.10 In 2015, it became the first and, to date, only FDA-approved biologic agent for the treatment of moderate/severe HS.10
A Phase II study by Kimball et al in 2012 first demonstrated that a significantly greater proportion receiving adalimumab weekly (17.6%) compared to placebo (3.9%) achieved the primary clinical endpoint of a HS-PGA score of clear, minimal or mild with at least a two-grade improvement relative to baseline scores at week 16. This effect was not significantly demonstrated in the group of patients receiving adalimumab every other week (9.6%) (weekly vs placebo difference 13.7%, p=0.025; every other week vs placebo difference 5.6%, p=0.25).11 After changing from weekly to every other week dosing, a decreased proportion of patients showed a clinical response.11
The subsequent PIONEER I and II Phase III trials involved a total of 633 patients with moderate-to-severe HS with an inadequate response to oral antibiotics. A significantly higher proportion of patients given adalimumab achieved HiSCR, compared to patients given placebo after 12 weeks of treatment (PIONEER I: 41.8% vs 26.0%, p=0.003; PIONEER II: 58.9% vs 27.6%, p<0.001).12 Most adverse events observed were mild or moderate in severity. Of note, in the group of patients treated with adalimumab, there were new psoriasiform eruptions and psoriasis in ten patients, one case of squamous cell carcinoma of the nose, and one death from cardiorespiratory arrest 42 days after the last dose of adalimumab in a 35-year-old man with a history of diabetes mellitus, smoking and a family history of ischemic heart disease.12 Secondary efficacy data also showed a greater proportion of subjects achieving a ≥30% reduction in the Patient’s Global Assessment of Skin Pain (PGA-SP) in both PIONEER I (adalimumab vs placebo [24.9%]; OR=2.03, p=0.004) and PIONEER II (adalimumab [61.2%] vs placebo [24.8%]; OR=4.78, p<0.001).13,14
An open-label extension trial of the PIONEER I and II trials also confirmed that patients who continued to receive weekly adalimumab maintained a long-term response with a HiSCR rate of 52.3% at week 168 and a decrease in the Dermatology Life Quality Index (DLQI) of 5.1–6.8 points at week 72, with no new safety risks identified.15
In a study by Ryan et al that analyzed the safety data of adalimumab in HS, there were no new safety concerns identified with the weekly dosing of adalimumab compared with every other week dosing.16
In case reports, adalimumab has also shown effectiveness in treating HS associated with pyoderma gangrenosum, acne and psoriatic arthritis.17–19 However, some case reports and series have drawn caution to the use of adalimumab, reporting adverse events such as erythroderma, melanoma, demyelinating disorders and drug-induced lupus.20–22
There are currently post-marketing surveillance trials of adalimumab underway, assessing quality of life, effectiveness of treatment and safety profile.23–27 In addition, the safety, efficacy and cost-effectiveness of adalimumab in conjunction with surgery are currently under investigation by two Phase IV trials.28,29
Infliximab
Infliximab is a chimeric mouse/human anti-TNF-α monoclonal antibody. It is currently FDA-approved for use in inflammatory bowel disease, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis and plaque psoriasis;30 and has been used as an off-label treatment in patients with HS resistant to adalimumab.9,31,32 It is currently dosed as an intravenous infusion 5 mg/kg body weight on week 0, 2, 6 and thereafter every 8 weeks.30 However, reports suggest that the dosing regimen for HS requires further refinement.31
In a descriptive single-center study involving 10 patients, no long-term curative effect was uniformly seen.33 In another study evaluating the efficacy of a single course (three infusions) of infliximab in 10 patients, three patients did not have a recurrence at 2 years, whereas the other seven had an average time of 8.5 months to recurrence of lesions (4.3–13.4 months).34
In a Phase II randomized study comprising a double-blind placebo-controlled treatment phase, an open-label crossover treatment phase and an observational phase, 38 patients with moderate-to-severe HS as defined by a HSSI score >8 were selected. More patients treated with regular infliximab responded with a 25% to <50% decrease in HSSI compared to placebo (60% vs 5.6%), whereas most patients treated with placebo had a <25% decrease in HSSI compared with infliximab (88.9% vs 13.3%, p<0.001).9 After 8 weeks of treatment, there were significant improvements in the infliximab group compared to the placebo group in terms of mean DLQI change (−10 vs −1.6, p=0.003) and mean PGA scores (1.8 vs 4.7, p<0.001).9 Most adverse events were mild and none were considered unexpected.9
In case reports, infliximab demonstrated efficacy in treating HS associated with pyoderma gangrenosum, acne, Crohn’s disease and systemic amyloidosis.35–42
However, infliximab has also in cases been associated with paradoxical worsening of facial acne vulgaris, demyelinating neuropathies, metastatic cutaneous squamous cell carcinoma and a case of Gemella morbillorum bacteremia complicated by brain abscesses.20,43–45
Etanercept
Etanercept is a dimeric TNF-α inhibitor. It is approved for use in rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, plaque psoriasis and psoriatic arthritis.46
After showing mixed results in open-label trials,47–50 it was examined under a double-blind, placebo-controlled study in 20 patients with moderate-to-severe HS. In patients given etanercept 50 mg twice weekly for 24 weeks, no significant improvement in HS was found.51
Golimumab
Golimumab is an anti-TNF-α human monoclonal antibody, approved for use in rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and ulcerative colitis.52
To date, it has been used in two case reports in the treatment of HS. In a case of a patient with concomitant Hurley Stage 3 HS and psoriatic arthritis, the use of subcutaneous golimumab 50 mg once weekly did not result in clinical improvement of HS (after adalimumab and anakinra had failed).53 However, in a later case report published in 2016 of a 42-year-old female with Hurley Stage 2 HS and pyostomatitis vegetans on a background of ulcerative colitis, golimumab subcutaneously 200 mg once followed by 100 mg every 4 weeks, together with amoxicillin-clavulanate, resulted in complete and sustained remission of HS, pyostomatitis vegetans and ulcerative colitis.54
There are no clinical trials underway to further assess golimumab in HS.
Certolizumab
Certolizumab is a PEGylated Fab fragment of a humanized TNF-α monoclonal antibody that is FDA-approved for the treatment of Crohn’s disease, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and plaque psoriasis.55 Its use in HS was described briefly in a case series, where it was used in two patients but found to be ineffective.56
IL-1 inhibitors
Anakinra
Anakinra is a recombinant IL-1 receptor inhibitor which is FDA-approved for use in rheumatoid arthritis and neonatal-onset multisystem inflammatory disease.57 It has been given as a 100 mg subcutaneous daily dose in HS.57
It has been studied in a double-blind, randomized, placebo-controlled Phase II clinical trial involving 20 patients. There were significantly more patients with a decreased disease activity score in the anakinra group compared to the placebo group after 12 weeks of treatment (78% vs 20%, p=0.02) and achieving HiSCR at the end of 12 weeks (78% vs 30%, p=0.04).58 However, at 24 weeks, the difference in patients achieving HiSCR was not statistically significant (10% vs 33%, p=0.28).58
In later case reports, there were also experiences of failure of anakinra therapy, or even worsening of HS related to anakinra use, suggesting the need for further clinical trials.59 Painful injection site reactions are also commonly reported with the use of anakinra, limiting its tolerability for some patients.60 It was also linked with drug-induced sarcoidosis in one case report.13
Canakinumab
Canakinumab is a human monoclonal anti-IL-1β antibody which is FDA-approved for use in cryopyrin-associated periodic syndromes and systemic juvenile idiopathic arthritis.61 It has been given up to 150 mg subcutaneous weekly dose in the treatment of HS. To date, it has shown mixed results in case reports and series.62–65
Bermekimab
Bermekimab (MABp1) is an anti-IL 1α human monoclonal antibody. In a recent Phase II trial involving 20 patients with moderate-to-severe HS either randomized to bermekimab or placebo for 12 weeks, 60% of patients on bermekimab achieved HiSCR at week 12 compared to 10% on placebo (P=0.035).66 Twelve weeks after cessation of treatment, 40% of patients on bermekimab had a positive HiSCR compared to 0% of patients on placebo.66 No adverse events related to bermekimab were reported.66
MEDI8968
MEDI8968 is a fully human immunoglobulin monoclonal antibody that selectively binds to the IL-1R1 receptor to inhibit activation by IL-1α and IL-1β. It has been studied for use in osteoarthritis, rheumatoid arthritis and chronic obstructive pulmonary disease.67–69 A Phase IIa study evaluating the safety, tolerability and efficacy of MEDI8968 for the treatment of subjects with moderate-to-severe HS was terminated early due to a lack of efficacy.70
IL-12/-23 inhibitors
Ustekinumab
Ustekinumab is a human monoclonal antibody that acts by binding to and inhibiting the p40 subunit on IL-12 and IL-23. It is FDA-approved for use in plaque psoriasis, psoriatic arthritis and Crohn’s disease.71 Patients weighing 100 kg and below receive 45 mg per dose, and those weighing above 100 kg receive 90 mg per dose.71
In a Phase II open-label study involving 17 patients on ustekinumab, the majority of patients showed moderate to marked improvement, as defined by a significant decrease in the mSS and modified HS Lesional Area Severity Index.72 Forty-seven percent of patients achieved HiSCR.72 Adverse events were mild and temporary, most commonly headache, fatigue and upper respiratory tract infections. The authors of the Phase II study suggested that the dosing regimen in HS may have to be further intensified.72
IL-17 inhibitors
Secukinumab
Secukinumab is a human IgG1k monoclonal antibody that acts as an IL-17A inhibitor. It is FDA-approved for moderate-to-severe plaque psoriasis, psoriatic arthritis and ankylosing spondylitis.73 Given at 300 mg subcutaneously weekly for 1 month followed by 4-weekly maintenance dosing, it has shown dramatic improvement in case reports of patients in whom other biologic therapies failed.74,75 An exploratory pilot study on the safety and feasibility of secukinumab in HS patients is currently underway,76 and there are two randomized double-blind multicenter trials to compare the efficacy, safety and tolerability of 2-weekly and 4-weekly secukinumab 300 mg in patients with moderate-to-severe HS.77,78
CJM112
CJM112 is a human monoclonal anti-IL-17A antibody. A Phase II study involving 66 patients with moderate-to-severe chronic -HS has been completed, but results are not available at present.79
Bimekizumab
Bimekizumab is a humanized anti-IL17A and IL-17F monoclonal antibody which has been studied and found to be effective in patients with psoriasis.80,81 A Phase II trial is currently underway to investigate its use in moderate-to-severe HS, with no results available at the time of writing.82
IL-23 inhibitors
Guselkumab
Guselkumab is an anti-IL-23 monoclonal antibody that has been FDA-approved for use in adults with moderate-to-severe plaque psoriasis. It is given subcutaneously 100 mg at week 0, week 4 and every 8 weeks thereafter.83 A case series involving three patients with severe HS, given guselkumab, found significant reductions in the IHS4, VAS for pain and (DLQI for all three patients.84 Another retrospective chart review of eight patients with moderate-to-severe HS given guselkumab found that 63% of patients reported improvements, with suggestions to further intensify the dosing regimen.85 No adverse events were documented in both articles. A Phase II multicenter randomized double-blind placebo-controlled trial has been initiated to evaluate its efficacy in the treatment of moderate-to-severe HS.86
Selective PDE-4 inhibitors
Apremilast
Apremilast is an orally administered PDE-4 inhibitor which is FDA-approved for use in patients with moderate-to-severe plaque psoriasis and active psoriatic arthritis. It is titrated to a target dose of 30 mg twice daily.87
In a reported case series of nine patients with Hurley stages II–III HS who had responded poorly to other treatments, five of six patients who persisted with treatment showed a good clinical response, with a significant improvement in the Sartorius score (73.17±67.76 to 56.17±44.89, p=0.028), VAS (7.17±0.98 to 2.00±2.10, p=0.026) and DLQI (21.33±8.91 to 9.33±5.85, p=0.027).88
In a double-blind, randomized, placebo-controlled trial involving 20 patients with moderate HS, 8 of 15 patients (53.3%) given apremilast achieved a positive HiSCR at week 16 compared to zero of five in the placebo group (p=0.055). Patients receiving apremilast also showed a significantly lower abscess and nodule count (mean difference −2.6; 95% CI −6.0 to −0.9; p=0.011), numerical rating scales for pain (mean difference −2.7; 95% CI −4.5 to −0.9; p=0.009), itch (mean difference −2.8; 95% CI −5.0 to −0.6; p=0.015) and disease burden (mean difference −1.8; 95% CI −3.7 to −0.01; p=0.049) compared to placebo. There were no major adverse events documented.89
Another Phase II open-label trial involving 20 patients has been completed, with no results available at present.90
Complement 5a inhibitors
IFX-1
IFX-1 is a human C5a-specific monoclonal antibody. Preliminary data from an open-label clinical study involving 12 patients, 75% of patients achieved HiSCR at day 50 (95% CI 0.43–0.95) and 83% at day 134 (95% CI 0.52–0.98).91,92
Another Phase II study is currently underway to determine its efficacy and safety.93
CD-20 inhibitors
Rituximab
Rituximab is a chimeric monoclonal antibody against the CD20 protein. It is FDA-approved for use in non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis and pemphigus vulgaris.94
In one case report in a kidney transplant recipient with idiopathic carpotarsal osteolysis who suffered chronic active antibody-mediated rejection and also developed HS, low-dose rituximab with two courses of 200 mg each were given with dramatic improvement of HS without remission of rejection.95
There are no further studies underway to evaluate the efficacy of rituximab.
JAK-1 inhibitors
INCB054707
INCB054707 is an orally administered inhibitor of the Janus kinase 1 pathway. There are currently two Phase II trials underway, with no other information available at the time of writing.96,97
Discussion
The increased understanding of the inflammatory pathways in HS provides many exciting therapeutic opportunities for patients with HS resistant to conventional methods of therapy. As more molecular targets are identified, immunomodulatory therapies can be developed, and their dosing regimens further refined. The efficacy, or lack thereof, of individual therapies also provides key insights into disease pathophysiology.
TNF-α inhibition in HS has been demonstrated to be useful. Adalimumab is presently the only FDA-approved biologic for use in HS and should thus be the drug of choice in moderate-to-severe HS where conventional treatment has proven ineffective. It is worth noting from our systematic review that where HS is associated with pyoderma gangrenosum, acne, Crohn’s disease or systemic amyloidosis, infliximab may also be considered as an effective off-label treatment.
Etanercept and MEDI8968 have already proven to be ineffective. Other therapies, involving smaller cohorts, have shown partial or mixed responses, with larger trials underway to further assess their efficacy in HS. The varying clinical measurement scores and endpoints used in these trials to determine treatment responsiveness potentially complicate direct comparison between different agents.
Most biologics and immunomodulatory therapies exhibit a generally well-tolerated safety profile. However, long-term safety concerns, including infection risks, especially latent tuberculosis reactivation, demyelinating disorders, and the development of malignancy from chronic immunosuppression will need to be evaluated through longitudinal surveillance and pooled registry data.98 These issues are especially pertinent in the treatment of HS, where the dosing regimen for biologics is typically more intensive compared to other inflammatory diseases, such as psoriasis. Patient selection remains important, as complete response is not the norm, not all patients with HS tolerate or respond well to immunomodulatory therapy, and may benefit from other modes of treatment, such as surgery.
In patients with severe HS that does not fully respond even to biologic treatment, we may consider biologics as an adjunct to surgery, where biologics are used to debulk disease to minimize the area required for surgical resection. Results from Phase IV trials to assess the combination of adalimumab with surgery will help refine the treatment approach for this category of severe HS.28,29
Ultimately, it is hoped that the use of immunomodulatory therapies will help overcome some of the challenges in treating severe HS, alleviating the impact on sufferers’ quality of life and morbidity associated with the disease. However, more quality data is required on their efficacy, safety and use in specific sub-populations before we can achieve truly targeted treatment of HS.
Disclosure
Hazel H Oon is a clinical investigator for Janssen, Novartis and Pfizer. She has also served as a speaker and advisory board member for AbbVie, Janssen, Novartis and Eli Lilly. Hazel H Oon reports grants and personal fees from AbbVie, Eli Lilly, Janssen, Novartis, and Pfizer outside the submitted work.The authors report no other conflicts of interest in this work.
References
1. Deckers IE, van der Zee HH, Prens EP. Epidemiology of hidradenitis suppurativa: prevalence, pathogenesis, and factors associated with the development of HS. Curr Dermatol Rep. 2014;3(1):54–60. doi:10.1007/s13671-013-0064-8
2. Sellheyer K, Krahl D. “Hidradenitis suppurativa” is acne inversa! An appeal to (finally) abandon a misnomer. Int J Dermatol. 2005;44(7):535–540. doi:10.1111/j.1365-4632.2004.02536.x
3. Shlyankevich J, Chen AJ, Kim GE, Kimball AB. Hidradenitis suppurativa is a systemic disease with substantial comorbidity burden: a chart-verified case-control analysis. J Am Acad Dermatol. 2014;71(6):1144–1150. doi:10.1016/j.jaad.2014.09.012
4. von Laffert M, Helmbold P, Wohlrab J, Fiedler E, Stadie V, Marsch WC. Hidradenitis suppurativa (acne inversa): early inflammatory events at terminal follicles and at interfollicular epidermis. Exp Dermatol. 2010;19(6):533–537. doi:10.1111/j.1600-0625.2009.00915.x
5. Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105–115. doi:10.2147/CCID.S111019
6. Kelly G, Hughes R, McGarry T, et al. Dysregulated cytokine expression in lesional and nonlesional skin in hidradenitis suppurativa. Br J Dermatol. 2015;173(6):1431–1439. doi:10.1111/bjd.14075
7. Moran B, Sweeney CM, Hughes R, et al. Hidradenitis suppurativa is characterized by dysregulation of the Th17: tregCell axis, which is corrected by anti-TNF therapy. J Invest Dermatol. 2017;137(11):2389–2395. doi:10.1016/j.jid.2017.05.033
8. Hurley H. Axillary hyperhidrosis, apocrine bromhidrosis, hidradenitis suppurativa, and familial benign pemphigus: surgical approach. Dermatologic Surgery New York: Marcel Dekker. 1989;729–739.
9. Grant A, Gonzalez T, Montgomery MO, Cardenas V, Kerdel FA. Infliximab therapy for patients with moderate to severe hidradenitis suppurativa: a randomized, double-blind, placebo-controlled crossover trial. J Am Acad Dermatol. 2010;62(2):205–217. doi:10.1016/j.jaad.2009.06.050
10.
11. Kimball AB, Kerdel F, Adams D, et al. Adalimumab for the treatment of moderate to severe Hidradenitis suppurativa: a parallel randomized trial. Ann Intern Med. 2012;157(12):846–855. doi:10.7326/0003-4819-157-12-201212180-00004
12. Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375(5):422–434. doi:10.1056/NEJMoa1504370
13. Friedman BE, English JC
14. Kimball AB, Sundaram M, Shields AL, et al. Adalimumab alleviates skin pain in patients with moderate to severe hidradenitis suppurativa: secondary efficacy results from the PIONEER I and PIONEER II randomized controlled trials. J Am Acad Dermatol. 2018;79:1141–1143. doi:10.1016/j.jaad.2018.05.015
15. Zouboulis CC, Okun MM, Prens EP, et al. Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open-label extension study. J Am Acad Dermatol. 2019;80(1):60–69.e2. doi: 10.1016/j.jaad.2018.05.040
16. Ryan C, Sobell JM, Leonardi CL, et al. Safety of Adalimumab Dosed Every Week and Every Other Week: focus on Patients with Hidradenitis Suppurativa or Psoriasis. Am J Clin Dermatol. 2018;19(3):437–447. doi:10.1007/s40257-017-0341-6
17. De Wet J, Jordaan HF, Kannenberg SM, Tod B, Glanzmann B, Visser WI. Pyoderma gangrenosum, acne, and suppurative hidradenitis syndrome in end-stage renal disease successfully treated with adalimumab. Dermatol Online J. 2017;23:12.
18. Saraceno R, Babino G, Chiricozzi A, Zangrilli A, Chimenti S. PsAPASH: a new syndrome associated with hidradenitis suppurativa with response to tumor necrosis factor inhibition. J Am Acad Dermatol. 2015;72(1):e42–44. doi:10.1016/j.jaad.2014.10.002
19. Murphy B, Morrison G, Podmore P. Successful use of adalimumab to treat pyoderma gangrenosum, acne and suppurative hidradenitis (PASH syndrome) following colectomy in ulcerative colitis. Int J Colorectal Dis. 2015;30(8):1139–1140. doi:10.1007/s00384-014-2110-9
20. Lozeron P, Denier C, Lacroix C, Adams D. Long-term course of demyelinating neuropathies occurring during tumor necrosis factor-alpha-blocker therapy. Arch Neurol. 2009;66(4):490–497. doi:10.1001/archneurol.2009.11
21. Zhao CY, Fernandez-Penas P. Is it worthy to treat hidradenitis suppurativa with adalimumab in patients with melanoma and other debilitating systemic diseases? A series of clinical dilemmas. Australas J Dermatol. 2018;59(4):e297–e298. doi:10.1111/ajd.12829
22. Benhadou F, Hellgren G, Willaert F, Del Marmol V. Acute erythroderma in a patient receiving TNF-alpha-blocking therapy for hidradenitis suppurativa. Case Rep Dermatol. 2018;10(1):7–12. doi:10.1159/000485911
23.
24.
25.
26.
27.
28.
29.
30.
31. Moriarty B, Jiyad Z, Creamer D. Four-weekly infliximab in the treatment of severe hidradenitis suppurativa. Br J Dermatol. 2014;170(4):986–987. doi:10.1111/bjd.12713
32. Delage M, Samimi M, Atlan M, Machet L, Lorette G, Maruani A. Efficacy of infliximab for hidradenitis suppurativa: assessment of clinical and biological inflammatory markers. Acta Derm Venereol. 2011;91(2):169–171. doi:10.2340/00015555-1025
33. Paradela S, Rodriguez-Lojo R, Fernandez-Torres R, Arevalo P, Fonseca E. Long-term efficacy of infliximab in hidradenitis suppurativa. J Dermatolog Treat. 2012;23(4):278–283. doi:10.3109/09546634.2012.683767
34. Mekkes JR, Bos JD. Long-term efficacy of a single course of infliximab in hidradenitis suppurativa. Br J Dermatol. 2008;158(2):370–374. doi:10.1111/j.1365-2133.2007.08332.x
35. Ozer I, Karacin C, Adisen E, Guz G, Ali Gurer M. Two diseases one remedy? Systemic amyloidosis secondary to hidradenitis suppurativa: treatment with infliximab. Dermatol Ther. 2017;30:2. doi:10.1111/dth.12445
36. Staub J, Pfannschmidt N, Strohal R, et al. Successful treatment of PASH syndrome with infliximab, cyclosporine and dapsone. J Eur Acad Dermatol Venereol. 2015;29(11):2243–2247. doi:10.1111/jdv.12765
37. Montes-Romero JA, Callejas-Rubio JL, Sanchez-Cano D, Gonzalez-Martinez FJ, Navas-Parejo A, Ortego-Centeno N. Amyloidosis secondary to hidradenitis suppurativa. Exceptional response to infliximab. Eur J Intern Med. 2008;19(6):e32–33. doi:10.1016/j.ejim.2007.11.014
38. Roussomoustakaki M, Dimoulios P, Chatzicostas C, et al. Hidradenitis suppurativa associated with Crohn‘s disease and spondyloarthropathy: response to anti-TNF therapy. J Gastroenterol. 2003;38(10):1000–1004. doi:10.1007/s00535-003-1185-9
39. Blazquez I, Gonzalez-Lama Y, Roustan G. Crohn‘s disease and hidradenitis suppurativa. An uncommon association that responds to infliximab. J Crohns Colitis. 2013;7(12):e717–718. doi:10.1016/j.crohns.2013.08.016
40. Groleau PF, Grossberg AL, Gaspari AA. Hidradenitis suppurativa and concomitant pyoderma gangrenosum treated with infliximab. Cutis. 2015;95(6):337–342.
41. Goertz RS, Konturek PC, Naegel A, et al. Experiences with a long-term treatment of a massive gluteal acne inversa with infliximab in Crohn‘s disease. Med Sci Monit. 2009;15(1):CS14–C18.
42. Martínez FNP, Benlloch S, Ponce J. Hidradenitis suppurativa and Crohn’s disease: response to treatment with infliximab. Inflamm Bowel Dis. 2001;7(4):323–326.
43. Vossen MG, Gattringer KB, Khalifeh N, et al. Gemella morbillorum bacteremia after anti-tumor necrosis factor alpha as acne inversa therapy. J Clin Microbiol. 2012;50(3):1109–1112. doi:10.1128/JCM.06161-11
44. Gori A, Rossari S, Bruscino N, Tripo L. Paradoxical effect of infliximab in a patient with hidradenitis suppurativa. Dermatol Ther. 2012;25(4):376–378. doi:10.1111/j.1529-8019.2012.01471.x
45. Scheinfeld N. A case of a patient with stage III familial hidradenitis suppurativa treated with 3 courses of infliximab and died of metastatic squamous cell carcinoma. Dermatol Online J. 2014;20:3.
46.
47. Giamarellos-Bourboulis EJ, Pelekanou E, Antonopoulou A, et al. An open-label phase II study of the safety and efficacy of etanercept for the therapy of hidradenitis suppurativa. Br J Dermatol. 2008;158(3):567–572. doi:10.1111/j.1365-2133.2007.08372.x
48. Pelekanou A, Kanni T, Savva A, et al. Long-term efficacy of etanercept in hidradenitis suppurativa: results from an open-label phase II prospective trial. Exp Dermatol. 2010;19(6):538–540. doi:10.1111/j.1600-0625.2009.00967.x
49. Lee RA, Dommasch E, Treat J, et al. A prospective clinical trial of open-label etanercept for the treatment of hidradenitis suppurativa. J Am Acad Dermatol. 2009;60(4):565–573. doi:10.1016/j.jaad.2008.11.898
50. Sotiriou E, Apalla Z, Ioannidos D. Etanercept for the treatment of hidradenitis suppurativa. Acta Derm Venereol. 2009;89(1):82–83. doi:10.2340/00015555-0545
51. Adams DR, Yankura JA, Fogelberg AC, Anderson BE. Treatment of hidradenitis suppurativa with etanercept injection. Arch Dermatol. 2010;146(5):501–504. doi:10.1001/archdermatol.2010.72
52.
53. van der Zee HH, Prens EP. Failure of anti-interleukin-1 therapy in severe hidradenitis suppurativa: a case report. Dermatology. 2013;226(2):97–100. doi:10.1159/000343221
54. Tursi A. Concomitant hidradenitis suppurativa and pyostomatitis vegetans in silent ulcerative colitis successfully treated with golimumab. Dig Liver Dis. 2016;48(12):1511–1512. doi:10.1016/j.dld.2016.09.010
55.
56. Sand FL, Thomsen SF. Off-label use of TNF-alpha inhibitors in a dermatological university department: retrospective evaluation of 118 patients. Dermatol Ther. 2015;28(3):158–165. doi:10.1111/dth.12222
57.
58. Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of Anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152(1):52–59. doi:10.1001/jamadermatol.2015.3903
59. Russo V, Alikhan A. Failure of Anakinra in a case of severe hidradenitis suppurativa. J Drugs Dermatol. 2016;15(6):772–774.
60. Leslie KS, Tripathi SV, Nguyen TV, Pauli M, Rosenblum MD. An open-label study of anakinra for the treatment of moderate to severe hidradenitis suppurativa. J Am Acad Dermatol. 2014;70(2):243–251. doi:10.1016/j.jaad.2013.09.044
61.
62. Sun NZ, Ro T, Jolly P, Sayed CJ. Non-response to interleukin-1 antagonist canakinumab in two patients with refractory pyoderma gangrenosum and hidradenitis suppurativa. J Clin Aesthet Dermatol. 2017;10(9):36–38.
63. Houriet C, Seyed Jafari SM, Thomi R, et al. Canakinumab for severe hidradenitis suppurativa: preliminary experience in 2 cases. JAMA Dermatol. 2017;153(11):1195–1197. doi:10.1001/jamadermatol.2017.2392
64. Jaeger T, Andres C, Grosber M, et al. Pyoderma gangrenosum and concomitant hidradenitis suppurativa–rapid response to canakinumab (anti-IL-1beta). Eur J Dermatol. 2013;23(3):408–410. doi:10.1684/ejd.2013.2018
65. Tekin B, Salman A, Ergun T. Hidradenitis suppurativa unresponsive to canakinumab treatment: a case report. Indian J Dermatol Venereol Leprol. 2017;83(5):615–617. doi:10.4103/ijdvl.IJDVL_147_16
66. Kanni T, Argyropoulou M, Spyridopoulos T, et al. MABp1 targeting IL-1alpha for moderate to severe hidradenitis suppurativa not eligible for adalimumab: a randomized study. J Invest Dermatol. 2018;138(4):795–801. doi:10.1016/j.jid.2017.10.030
67. Cohen SB, Proudman S, Kivitz AJ, et al. A randomized, double-blind study of AMG 108 (a fully human monoclonal antibody to IL-1R1) in patients with osteoarthritis of the knee. Arthritis Res Ther. 2011;13(4):R125. doi:10.1186/ar3430
68. Cardiel MH, Tak PP, Bensen W, et al. A phase 2 randomized, double-blind study of AMG 108, a fully human monoclonal antibody to IL-1R, in patients with rheumatoid arthritis. Arthritis Res Ther. 2010;12(5):R192. doi:10.1186/ar3163
69. Calverley PMA, Sethi S, Dawson M, et al. A randomised, placebo-controlled trial of anti-interleukin-1 receptor 1 monoclonal antibody MEDI8968 in chronic obstructive pulmonary disease. Respir Res. 2017;18(1):153. doi:10.1186/s12931-017-0633-7
70.
71.
72. Blok JL, Li K, Brodmerkel C, Horvatovich P, Jonkman MF, Horvath B. Ustekinumab in hidradenitis suppurativa: clinical results and a search for potential biomarkers in serum. Br J Dermatol. 2016;174(4):839–846. doi:10.1111/bjd.14338
73.
74. Thorlacius L, Theut Riis P, Jemec GBE. Severe hidradenitis suppurativa responding to treatment with secukinumab: a case report. Br J Dermatol. 2018;179(1):182–185. doi:10.1111/bjd.15769
75. Schuch A, Fischer T, Boehner A, Biedermann T, Volz T. Successful treatment of severe recalcitrant hidradenitis suppurativa with the interleukin-17A antibody secukinumab. Acta Derm Venereol. 2018;98(1):151–152. doi:10.2340/00015555-2794
76.
77.
78.
79.
80. Papp KA, Merola JF, Gottlieb AB, et al. Dual neutralization of both interleukin 17A and interleukin 17F with bimekizumab in patients with psoriasis: results from BE ABLE 1, a 12-week randomized, double-blinded, placebo-controlled phase 2b trial. J Am Acad Dermatol. 2018;79(2):277 e210–286 e210. doi:10.1016/j.jaad.2018.03.023
81. Glatt S, Helmer E, Haier B, et al. First-in-human randomized study of bimekizumab, a humanized monoclonal antibody and selective dual inhibitor of IL-17A and IL-17F, in mild psoriasis. Br J Clin Pharmacol. 2017;83(5):991–1001. doi:10.1111/bcp.13185
82.
83.
84. Kovacs M, Podda M. Guselkumab in the treatment of severe Hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2019;33(3):e140–e141. doi:10.1111/jdv.15368
85. Casseres RG, Kahn JS, Her MJ, Rosmarin D. Guselkumab in the treatment of hidradenitis suppurativa: a retrospective chart review. J Am Acad Dermatol. 2018. doi:10.1016/j.jaad.2018.12.017
86.
87.
88. Weber P, Seyed Jafari SM, Yawalkar N, Hunger RE. Apremilast in the treatment of moderate to severe hidradenitis suppurativa: a case series of 9 patients. J Am Acad Dermatol. 2017;76(6):1189–1191. doi:10.1016/j.jaad.2017.02.026
89. Vossen ARJVV, van der Zee HH, Prens EP. Apremilast for moderate hidradenitis suppurativa: results of a randomized controlled trial. J Am Acad Dermatol. 2019;80(1):80–88. doi:10.1016/j.jaad.2018.06.046
90.
91. Guo RHM, Zenker O, Giamarellos-Bourboulis EJ, Riedemann N IFX-1 blocking the anaphylatoxin 1 blocking the anaphylatoxin 1 blocking the anaphylatoxin C5a – an anti an anti-inflammatory effect in patients with hidradenitis suppurativa; 2017. Available from:
92.
93.
94.
95. Takahashi K, Yanagi T, Kitamura S, et al. Successful treatment of hidradenitis suppurativa with rituximab for a patient with idiopathic carpotarsal osteolysis and chronic active antibody-mediated rejection. J Dermatol. 2018;45(5):e116–e117. doi:10.1111/1346-8138.14144
96.
97.
98. Lee RA, Eisen DB. Treatment of hidradenitis suppurativa with biologic medications. J Am Acad Dermatol. 2015;73(5 Suppl 1):S82–88. doi:10.1016/j.jaad.2015.07.053
99. Kimball AB, Sobell JM, Zouboulis CC, et al. HiSCR (Hidradenitis Suppurativa Clinical Response): a novel clinical endpoint to evaluate therapeutic outcomes in patients with hidradenitis suppurativa from the placebo-controlled portion of a phase 2 adalimumab study. J Eur Acad Dermatol Venereol. 2016;30(6):989–994. doi:10.1111/jdv.13216
100. Sartorius K, Emtestam L, Jemec GB, Lapins J. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161(4):831–839. doi:10.1111/j.1365-2133.2009.09198.x
101. Amano M, Grant A, Kerdel FA. A prospective open-label clinical trial of adalimumab for the treatment of hidradenitis suppurativa. Int J Dermatol. 2010;49(8):950–955. doi:10.1111/j.1365-4632.2010.04545.x
102. Zouboulis CC, Tzellos T, Kyrgidis A, et al. Development and validation of the International Hidradenitis Suppurativa Severity Score System (IHS4), a novel dynamic scoring system to assess HS severity. Br J Dermatol. 2017;177(5):1401–1409. doi:10.1111/bjd.15748
103. Menis D, Maronas-Jimenez L, Delgado-Marquez AM, Postigo-Llorente C, Vanaclocha-Sebastian F. Two cases of severe hidradenitis suppurativa with failure of anakinra therapy. Br J Dermatol. 2015;172(3):810–811. doi:10.1111/bjd.13292
104.
105. Strober BE, Kim C, Siu K. Efalizumab for the treatment of refractory hidradenitis suppurativa. J Am Acad Dermatol. 2007;57(6):1090–1091. doi:10.1016/j.jaad.2007.07.032
106. Koilakou S, Karapiperis D, Tzathas C. A case of hidradenitis suppurativa refractory to anti-TNFalpha therapy in a patient with Crohn‘s disease. Am J Gastroenterol. 2010;105(1):231–232. doi:10.1038/ajg.2009.489
107. Moul DK, Korman NJ. The cutting edge. Severe hidradenitis suppurativa treated with adalimumab. Arch Dermatol. 2006;142(9):1110–1112. doi:10.1001/archderm.142.9.1110
108. Harde V, Mrowietz U. Treatment of severe recalcitrant hidradenitis suppurativa with adalimumab. J Dtsch Dermatol Ges. 2009;7(2):139–141. doi:10.1111/j.1610-0387.2008.06918.x
109. Samycia M, Brassard A. Adalimumab in treatment-resistant hidradenitis suppurativa following recurrence after extensive affected area excision: a review of biologics therapy. J Cutan Med Surg. 2013;17 Suppl 1:S23–S32. doi:10.2310/7750.2012.11144
110. Diamantova D, Lomickova I, Cetkovska P. Adalimumab treatment for hidradenitis suppurativa associated with Crohn‘s disease. Acta Dermatovenerol Croat. 2014;22(4):291–293.
111. Bosnic D, Zarkovic B, Baresic M, Zarkovic M, Anic B. Improvement of overlapping hidradenitis suppurativa and ankylosing spondylitis after the introduction of adalimumab. Reumatologia. 2016;54(6):321–325. doi:10.5114/reum.2016.64910
112. Shanmugam VK, Mulani S, McNish S, Harris S, Buescher T, Amdur R. Longitudinal observational study of hidradenitis suppurativa: impact of surgical intervention with adjunctive biologic therapy. Int J Dermatol. 2018;57(1):62–69. doi:10.1111/ijd.13798
113. Crowley EL, O‘Toole A, Gooderham MJ. Hidradenitis suppurativa with SAPHO syndrome maintained effectively with adalimumab, methotrexate, and intralesional corticosteroid injections. SAGE Open Med Case Rep. 2018;6:2050313X18778723.
114. Molina-Leyva A, Badiola J. Severe refractory hidradenitis suppurativa successfully treated with adalimumab in an HIV-positive/hepatitis C virus-positive patient. Aids. 2018;32(16):2436–2438. doi:10.1097/QAD.0000000000001998
115. Gorovoy I, Berghoff A, Ferris L. Successful treatment of recalcitrant hidradenitis suppurativa with adalimumab. Case Rep Dermatol. 2009;1(1):71–77. doi:10.1159/000251217
116. Zarchi K, Dufour DN, Jemec GB. Successful treatment of severe hidradenitis suppurativa with anakinra. JAMA Dermatol. 2013;149(10):1192–1194. doi:10.1001/jamadermatol.2013.5377
117. Zangrilli A, Esposito M, Mio G, Mazzotta A, Chimenti S. Long-term efficacy of etanercept in hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2008;22(10):1260–1262. doi:10.1111/j.1468-3083.2008.02617.x
118. Adams DR, Gordon KB, Devenyi AG, Ioffreda MD. Severe hidradenitis suppurativa treated with infliximab infusion. Arch Dermatol. 2003;139(12):1540–1542. doi:10.1001/archderm.139.12.1540
119. Rosi YL, Lowe L, Kang S. Treatment of hidradenitis suppurativa with infliximab in a patient with Crohn‘s disease. J Dermatolog Treat. 2005;16(1):58–61. doi:10.1080/09546630410024547
120. Husein-ElAhmed H, Fernandez-Pugnaire MA, Ruiz-Carrascosa JC. Severe hidradenitis suppurative in an HIV-positive male: use of multiple treatment modalities, including tumor necrosis factor blockade. AIDS Patient Care STDS. 2011;25(9):507–508. doi:10.1089/apc.2011.0158
121. von Preussen AC, Flux K, Hartschuh W, Hartmann M. Acne inversa successfully treated with infliximab. Int J Dermatol. 2012;51(8):1011–1013. doi:10.1111/j.1365-4632.2010.04670.x
122. Lebwohl B, Sapadin AN. Infliximab for the treatment of hidradenitis suppurativa. J Am Acad Dermatol. 2003;49(5 Suppl):S275–276. doi:10.1016/S0190
123. Thielen AM, Barde C, Saurat JH. Long-term infliximab for severe hidradenitis suppurativa. Br J Dermatol. 2006;155(5):1105–1107. doi:10.1111/j.1365-2133.2006.07528.x
124. Alecsandru D, Padilla B, Izquierdo JA, Fernandez-Cruz E, Sanchez-Ramon S. Severe refractory hidradenitis suppurativa in an HIV-positive patient successfully treated with infliximab. Arch Dermatol. 2010;146(12):1343–1345. doi:10.1001/archdermatol.2010.372
125. Poulin Y. Successful treatment of hidradenitis suppurativa with infliximab in a patient who failed to respond to etanercept. J Cutan Med Surg. 2009;13(4):221–225. doi:10.2310/7750.2008.08034
126. Kozub P, Simaljakova M. Hidradenitis suppurativa treated with combination of infliximab and dapsone. Bratisl Lek Listy. 2012;113(5):319–323.
127. Giuseppe P, Nicola P, Valentina C, et al. A case of moderate hidradenitis suppurativa and psoriasis treated with secukinumab. Ann Dermatol. 2018;30(4):462–464. doi:10.5021/ad.2018.30.4.462
128. Jorgensen AR, Yao Y, Thomsen SF. Therapeutic response to secukinumab in a 36-year-old woman with hidradenitis suppurativa. Case Rep Dermatol Med. 2018;2018:8685136.
129. Santos-Perez MI, Garcia-Rodicio S, Del Olmo-Revuelto MA, Pozo-Roman T. Ustekinumab for hidradenitis suppurativa: a case report. Actas Dermosifiliogr. 2014;105(7):720–722. doi:10.1016/j.ad.2013.09.011
130. Sharon VR, Garcia MS, Bagheri S, et al. Management of recalcitrant hidradenitis suppurativa with ustekinumab. Acta Derm Venereol. 2012;92(3):320–321. doi:10.2340/00015555-1229
131. Blanco R, Martinez-Taboada VM, Villa I, et al. Long-term successful adalimumab therapy in severe hidradenitis suppurativa. Arch Dermatol. 2009;145(5):580–584. doi:10.1001/archdermatol.2009.49
132. Chinniah N, Cains GD. Moderate to severe hidradenitis suppurativa treated with biological therapies. Australas J Dermatol. 2014;55(2):128–131. doi:10.1111/ajd.12136
133. Patil S. Low-dose adalimumab biosimilar (ZRC-3197) in the treatment of hidradenitis suppurativa. Indian J Dermatol Venereol Leprol. 2018;84(6):745–747. doi:10.4103/ijdvl.IJDVL_232_18
134. Cusack C, Buckley C. Etanercept: effective in the management of hidradenitis suppurativa. Br J Dermatol. 2006;154(4):726–729. doi:10.1111/j.1365-2133.2005.07067.x
135. Lasocki A, Sinclair R, Foley P, Saunders H. Hidradenitis suppurativa responding to treatment with infliximab. Australas J Dermatol. 2010;51(3):186–190. doi:10.1111/j.1440-0960.2010.00623.x
136. Elkjaer M, Dinesen L, Benazzato L, Rodriguez J, Logager V, Munkholm P. Efficacy of infliximab treatment in patients with severe fistulizing hidradenitis suppurativa. J Crohns Colitis. 2008;2(3):241–245. doi:10.1016/j.crohns.2008.02.002
137. Antonucci A, Negosanti M, Negosanti L, Iozzo I, Varotti C. Acne inversa treated with infliximab: different outcomes in 2 patients. Acta Derm Venereol. 2008;88(3):274–275. doi:10.2340/00015555-0397
138. Brunasso AM, Delfino C, Massone C. Hidradenitis suppurativa: are tumour necrosis factor-alpha blockers the ultimate alternative? Br J Dermatol. 2008;159(3):761–763. doi:10.1111/j.1365-2133.2008.08735.x
139. Moschella SL. Is there a role for infliximab in the current therapy of hidradenitis suppurativa? A report of three treated cases. Int J Dermatol. 2007;46(12):1287–1291. doi:10.1111/j.1365-4632.2007.03293.x
140. Sullivan TP, Welsh E, Kerdel FA, Burdick AE, Kirsner RS. Infliximab for hidradenitis suppurativa. Br J Dermatol. 2003;149(5):1046–1049.
141. Torres T, Selores M. Treatment of hidradenitis suppurativa with infliximab. An Bras Dermatol. 2010;85(4):576. doi:10.1590/S0365-05962010000400028
142. Usmani N, Clayton TH, Everett S, Goodfield MD. Variable response of hidradenitis suppurativa to infliximab in four patients. Clin Exp Dermatol. 2007;32(2):204–205.
143. Fernandez-Vozmediano JM, Armario-Hita JC. Infliximab for the treatment of hidradenitis suppurativa. Dermatology. 2007;215(1):41–44. doi:10.1159/000102032
144. Zhang J, Reeder VJ, Hamzavi IH. Use of biologics in the treatment of hidradenitis suppurativa: a review of the Henry Ford Hospital experience. Br J Dermatol. 2014;171(6):1600–1602. doi:10.1111/bjd.13186
145. DeFazio MV, Economides JM, King KS, et al. Outcomes after combined radical resection and targeted biologic therapy for the management of recalcitrant hidradenitis suppurativa. Ann Plast Surg. 2016;77(2):217–222. doi:10.1097/SAP.0000000000000584
146. Bahillo Monne C, Honorato Guerra S, Schoendorff Ortega C, Gargallo Quintero AB. Management of hidradenitis suppurativa with biological therapy: report of four cases and review of the literature. Dermatology. 2014;229(4):279–287. doi:10.1159/000365076
147. Gulliver WP, Jemec GB, Baker KA. Experience with ustekinumab for the treatment of moderate to severe hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2012;26(7):911–914. doi:10.1111/j.1468-3083.2011.04123.x
148. Van Rappard DC, Mekkes JR. Treatment of severe hidradenitis suppurativa with infliximab in combination with surgical interventions. Br J Dermatol. 2012;167(1):206–208. doi:10.1111/j.1365-2133.2012.10807.x
149. Martin-Ezquerra G, Masferrer E, Masferrer-Niubo M, et al. Use of biological treatments in patients with hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2015;29(1):56–60. doi:10.1111/jdv.12438
150. Bettoli V, Manfredini M, Calamo G. et al. Long-term adalimumab treatment of hidradenitis suppurativa: results and practical insights from a real-life experience. Dermatol Ther;2018:e12737. doi:10.1111/dth.12737
151. Kyriakou A, Trigoni A, Galanis N, Sotiriadis D, Patsatsi A. Efficacy of adalimumab in moderate to severe hidradenitis suppurativa: real life data. Dermatol Rep. 2018;10(2):7859. doi:10.4081/dr.2018.7859
152. van Rappard DC, Leenarts MF, Meijerink-van ‘T Oost L, Mekkes JR. Comparing treatment outcome of infliximab and adalimumab in patients with severe hidradenitis suppurativa. J Dermatolog Treat. 2012;23(4):284–289. doi:10.3109/09546634.2011.571657
153. Sbidian E, Hotz C, Seneschal J, et al. Antitumour necrosis factor-alpha therapy for hidradenitis suppurativa: results from a national cohort study between 2000 and 2013. Br J Dermatol. 2016;174(3):667–670. doi:10.1111/bjd.14199
154. Giamarellos-Bourboulis EJ, Sobell J, Ryan C, Wolkenstein PJ, Geng Z, Mulder GD. Infection-free clinical response among patients with hidradenitis suppurativa who were treated with adalimumab: results from two phase 3 studies. Wounds. 2017;29(11):E98–E102.
155. Kimball AB, Sundaram M, Shields AL, et al. Adalimumab alleviates skin pain in patients with moderate-to-severe hidradenitis suppurativa: secondary efficacy results from the PIONEER I and PIONEER II randomized controlled trials. J Am Acad Dermatol. 2018;79(6):1141–1143. doi:10.1016/j.jaad.2018.05.015
156. Jimenez-Gallo D, de la Varga-Martinez R, Ossorio-Garcia L, Collantes-Rodriguez C, Rodriguez C, Linares-Barrios M. Effects of adalimumab on T-helper-17 lymphocyte- and neutrophil-related inflammatory serum markers in patients with moderate-to-severe hidradenitis suppurativa. Cytokine. 2018;103:20–24. doi:10.1016/j.cyto.2017.12.020
157. Arenbergerova M, Gkalpakiotis S, Arenberger P. Effective long-term control of refractory hidradenitis suppurativa with adalimumab after failure of conventional therapy. Int J Dermatol. 2010;49(12):1445–1449. doi:10.1111/j.1365-4632.2010.04638.x
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
