Back to Journals » Drug Design, Development and Therapy » Volume 13
Synthesis and antitumor evaluation of neolaxiflorin B inspired compounds
Authors Liu JP , Xiao YZ, Hu Y, Li XN , Wu MJ, Zhao Y, Zhao Y, Ma ZJ, Shen J
Received 15 March 2019
Accepted for publication 9 July 2019
Published 23 August 2019 Volume 2019:13 Pages 3021—3028
DOI https://doi.org/10.2147/DDDT.S202345
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sukesh Voruganti
Jing-Ping Liu,1 Ye-Zhi Xiao,1 Yu Hu,1 Xiao-Nian Li,2 Ming-Jiang Wu,3 Yong Zhao,1 Yan Zhao,1 Ze-Jin Ma,1 Jing Shen1
1Department of Chemistry, Yunnan Normal University, Kunming 650092, People’s Republic of China; 2State Key Laboratory of Phytochemistry and Plant Resources in West China, Kunming Institute of Botany, Kunming 650201, People’s Republic of China; 3Criminal Science and Technology Key Lab of Yunnan Police College, Kunming 650223, People’s Republic of China
Correspondence: Jing-Ping Liu; Jing Shen
Department of Chemistry, Yunnan Normal University, No. 298, 121st Street, Yunnan, Kunming 650092, People’s Republic of China
Tel/Fax +86 8 716 513 2144
Email [email protected]
[email protected]
Background: Neolaxiflorin B is derived from ent-kaurane like laxiflorin J and eriocalyxin B with a relatively low potency as an antitumor agent. During preliminary structure–activity relationship studies, the α,β-unsaturated ketone (enone) system is an important active group.
Methods: Seven neolaxiflorin B derivatives containing α,β-unsaturated ketone moieties were synthesized. In vitro, activity was evaluated against three human tumor cell lines and a rat myogenic cell line (HepG2, NSCLC-H292, SNU-1040, and L6, respectively) by MTT assay.
Results: Compound 15 appeared a promising antitumor lead due to its cytotoxic potency and relatively high selectivity, with an SI value of 13.14. Flow cytometry analysis was conducted to show that NSCLC-H292 cells were blocked in the G0/G1 phase in the presence of compound 15, thus inhibiting the proliferation of tumor cells.
Conclusion: This study has revealed that compound 15 is a promising antitumor lead due to the cytotoxic potencies and the high selectivity it displayed when compared to natural counterparts.
Keywords: neolaxiflorin B inspired compounds, unsaturated ketone, antitumor activity
Introduction
In the past decade, over 1000 ent-kaurane diterpenoids have been isolated and identified from the Isodon genus.1 According to recent reports, these diterpenoids possess a broad range of bioactivities, including anti-bacteria,2 cytotoxic, anti-HIV, anti-inflammatory, and anti-fungal activity.3 Laxiflorin, as one of the members of the diterpenoids, is assumed to share the same biogenic precursor, ent-kaurene (1), with eriocalyxin B and longikaurin A (Figure 1).4 Biosynthetically, there is a possibility of mutual transformation among these diterpenoids.
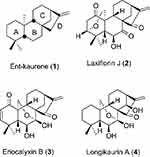 |
Figure 1 Structures of some Isodon diterpenoids and their biogenic precursor. |
According to the literature,5 neolaxiflorin B is derived from ent-kaurane like laxiflorin J and eriocalyxin B (Figure 2) with a relatively low potency as an antitumor agent. Although low in potency, its structure is novel, containing a unique scaffold, which we used as a basic template to synthesize novel natural product-like neolaxiflorin B derivatives to develop safe and effective antitumor agents.
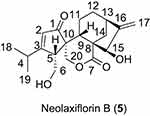 |
Figure 2 Structure of neolaxiflorin B. |
During preliminary structure–activity relationship studies, the removal of the α,β-unsaturated ketone (enone) system in the D-ring decreased its antitumor activity.6 Meanwhile, mounting evidence also demonstrates that dienone compounds with double α,β-unsaturated ketone functionalities, such as eriocalyxin B (3), can undergo two successive alkylations at the β-positions by reacting with cellular thiols during biological cascade reactions.7 We, therefore, aimed to synthesize a neolaxiflorin B-like scaffold with double α,β-unsaturated ketone.
Materials and methods
Materials and methods Information and methods
All reagents were used as purchased from Energy Chemical, Tansoole, Acros and Aldrich without further purification. Anhydrous CH2Cl2 was freshly distilled from CaH2, and THF was dried by Na. For those reactions that need to be performed in argon, at least three argon exchanges are required. The course of reaction was monitored by TLC. 1H-NMR and 13C-NMR spectra were recorded at ambient temperature with a Bruker Avance 300 (300 MHz for 1H and 75.5 MHz for 13C; Bruker, Germany) or a Bruker Avance 400 (400 MHz for 1H and 100 MHz for 13C; Bruker, Germany) instrument in which TMS was used as internal standard for all measurements. MS data were recorded by using a VG Auto spec-3000 spectrometer or a Finnigan MAT 90 instrument. IR spectra were measured as KBr pellets by using a Bio-Rad FTS-135 spectrometer. Characterization data for compounds are available free of charge via supporting information of associated content. All melting points are uncorrected. CC was performed by using silica gel (100–200 mesh; Qingdao Marine Chemical, China)
4-(hydroxymethyl)-3-isopropylcyclopent-2-enone (6)
To a solution of 5 (0.248 g, 2 mmol, 1eq) in n-hexane (5 mL) at room temperature under argon was added Co2(CO)8 (0.684 g, 2 mmol, 1eq). After 1 hr, the mixture was filtered through a short pad of neutral Al2O3 washed with n-hexane. The filtrate was added neutral Al2O3 (10 g) and concentrated under vacuum. To remove traces of oxygen, the residue is kept at 0.08 MPa for 15 mins, then the flask is flushed with argon. The procedure is repeated four times, further heated for 2 hrs at 50°C. The desired product 6 (70%) was achieved by silica gel column chromatography as a pale yellow liquid: 1H NMR (300 MHz, CDCl3) δ=5.83(s, 1H), 3.82–3.60 (m, 3H), 3.03 (s,1H), 2.58 (m,1H), 2.45–2.22 (m, 2H), 1.10 (dd, J=7.8 Hz, 6H) ppm; 13C NMR (75.5 MHz, CDCl3) δ=210.11, 189.49, 128.58, 62.56, 44.40, 39.32, 29.57, 21.37, 20.44ppm; IR (KBr): υmax =3388.32, 2964.75, 2874.24, 1718.07, 1676.32, 1603.50 cm−1; MS (ESI): m/z: 177; HRMS (ESI) Calcd for ([C9H14O2]+Na)+: 177.0891; found: 177.0888[M+Na]+.
Compound 7
To a solution of 6 (0.38 g, 2.47 mmol, 1eq) and imidazole (0.34 g, 4.94 mmol, 2eq) in CH2Cl2 (15 mL) at room temperature was added tert-Butyldimethylsilyl chloride (0.56 g, 3.71 mmol, 1.5eq). The solution was stirred overnight, and then the mixture was added water and extracted with CH2Cl2. The combined organic layers were dried over Na2SO4 and the solvent was removed under reduced pressure. The residue was purified by flash column chromatography to provide 7 (96%) as colorless liquid: 1H NMR (300 MHz, CDCl3) δ=5.93(s, 1H), 3.71(d, J=4.5 Hz,2H), 3.07(m, 1H), 2.66–2.62(m, 1H), 2.51–2.43(m, 1H), 2.30–2.23(m, 1H), 1.17(d, J=6.9 Hz, 3H), 1.14(d, J=7.2 Hz, 3H), 0.83(s, 9H), 0.01(s, 6H)ppm; 13C NMR (75.5 MHz, CDCl3) δ=209.19, 188.59, 128.81, 63.50, 44.42, 39.51, 29.74, 25.83, 21.76, 20.74, 18.24, 18.11, −3.46, −5.48ppm; IR (KBr): υmax =2959.33, 2928.81, 2874.91, 2857.09, 1717.84, 1681.76, 1605.36, 1258.86, 1100.42 cm−1; MS (ESI): m/z: 291; HRMS (ESI) Calcd for ([C15H28O2Si]+Na)+: 291.1756; found: 291.1758[M+Na]+.
Compound 8
Compound 8 is obtained as pale yellow liquid: 1H NMR (300 MHz, CDCl3) δ =7.43(d, J=9.9 Hz, 1H), 5.81(d, J=9.9 Hz, 1H), 5.26(s, 1H), 5.05(s, 1H), 4.19(q, J=7.2 Hz, 2H), 3.47(br., 1H), 2.86(d, J=95.4 Hz, 1H), 2.52(d, J=95.4 Hz, 1H), 2.25(m, 2H), 1.25(t, J=7.2 Hz, 3H)ppm; 13C NMR (75.5 MHz, CDCl3) δ=197.515, 172.761, 152.359, 143.136, 126.641, 113.126, 61.584, 57.960, 51.617, 43.207, 41.109, 14.120 ppm; IR (KBr): υmax =2979.98, 1729.06, 1685.40, 1280.90, 1222.04, 1193.09 cm−1; MS (ESI): m/z: 207; HRMS (ESI) Calcd for ([C12H14O3]+H)+: 207.1021; found: 207.1015 [M+H]+.
Compound 9
To a stirred solution of 7 (0.27 g, 1 mmol, 1eq) and 8 (0.23 g, 1.1 mmol, 1.1eq) in dry THF under Ar at −78°C was added LiHMDS (2 mL, 2eq, 1.0 M in THF) over a period of 5 mins and stirring at −78°C was continued for 45 mins. The resulting solution was warmed slowly to room temperature, quenched with saturated NH4Cl and extracted with Et2O. The combined organic layers were dried over Na2SO4 and the solvent was removed under reduced pressure. The residue was purified by flash column chromatography (EtOAc/petroleum ether, 1:10) to provide 9 (77%) as a crystal and its epimer 10 (9%) as a white powder (dr=9:1). 9 is obtained as crystal; m.p.:124–129°C; 1H NMR (400 MHz, CDCl3) δ =5.99(s, 1H), 5.07(s, 1H), 5.06(s, 1H), 4.18(q, J=7.2 Hz, 2H), 3.84(dd, J1=10 Hz, J2=3.2 Hz, 1H), 3.72(dd, J1=9.6 Hz, J2=4 Hz, 1H), 3.26(d, J=4.8 Hz, 1H), 3.07(d, J=9.2 Hz, 1H), 2.82–2.80(m, 2H), 2.71–2.46(m, 4H), 2.37–2.14(m, 2H), 1.67(d, J=16.8 Hz, 1H), 1.26(t, J=7.2 Hz, 3H), 1.17(d, J=6.8 Hz, 3H), 1.14(d, J=6.8 Hz, 3H), 0.78(s, 9H), 0.00(s, 3H), −0.01(s, 3H)ppm; 13C NMR (100 MHz, CDCl3) δ=209.37, 208.47, 190.18, 174.31, 145.78, 128.72, 109.93, 62.43, 61.12, 59.07, 53.17, 51.83, 47.30, 44.44, 42.67, 35.93, 29.67, 25.82, 25.74(3C), 21.80, 20.58, 18.14, 14.34, −5.62, −5.63ppm; IR (KBr): υmax =2957.43, 2929.36, 2885.29, 2857.41, 1733.92, 1713.56, 1689.57, 1655.97, 1615.37 cm−1; MS (ESI): m/z: 497; HRMS (ESI) Calcd for ([C27H42O5Si]+Na)+: 497.2699; found: 497.2698 [M+Na]+.
Compound 10
Compound 10 is obtained as white powder; m.p.:123–128°C; 1H NMR (400 MHz, CDCl3) δ=5.83(s, 1H), 5.07(s, 1H), 4.91(s, 1H), 4.13(q, J=7.2 Hz, 2H), 3.86(dd, J1=10 Hz, J2=2.8 Hz, 1H), 3.71(dd, J1=10 Hz, J2=3.6 Hz, 1H), 3.33(d, J=4.8 Hz, 1H), 3.20(d, J=12 Hz, 1H), 2.79–2.68(m, 6H), 2.33(s, 1H), 2.26–2.21(m, 1H), 1.80(d, J=16.8 Hz, 1H), 1.24(t, J=7.2 Hz, 3H), 1.12(d, J=6.8 Hz, 3H), 1.07(d, J=6.8 Hz, 3H), 0.78(s, 9H), 0.00(s, 6H) ppm; 13C NMR (100 MHz, CDCl3) δ=207.54, 206.18, 187.32, 174.88, 146.31, 128.91, 109.49, 60.96, 60.67, 59.82, 53.36, 52.97, 51.22, 44.20, 42.95, 36.33, 35.09, 29.28, 25.75, 21.76, 20.37, 18.14, 14.27, −5.47, −5.61 ppm; IR (KBr): υmax =2952.56, 2927.55, 2896.84, 2854.55, 1719.85, 1694.22, 1653.50, 1613.23, 1472.54 cm−1; MS (ESI): m/z: 497; HRMS (ESI) Calcd for ([C27H42O5Si]+Na)+: 497.2699; found: 497.2700 [M+Na]+.
Compound 11
To a stirred solution of 9 (0.47 g, 1 mmol, 1.0eq) in DCM, SeO2 (0.1 g, 0.9 mmol, 0.9eq) was added in one portion, and a solution of t-BuOOH (0.55 mL, 3 mmol, 5.5 M in decane, 3eq) was added dropwise. The solution was vigorously stirred at room temperature overnight, and then the mixture was filtered through a short pad of silica gel washed with EtOAc. The filtrate was concentrated under vacuum and purified by flash column chromatography to provide allylic alcohol 11 (71%) as colorless liquid: 1H NMR (400 MHz, CDCl3) δ=6.00(s, 1H), 5.42(s, 1H), 5.33(s, 1H), 4.61(s, 1H), 4.26(q, J=8 Hz, 2H), 3.85(m, 1H), 3.72m, 1H), 3.34(d, J=4.0 Hz, 1H), 3.09(d, J=8 Hz, 1H), 269–2.38(m, 4H), 2.15–2.02(m, 3H), 1.60(d, J=16 Hz, 1H), 1.28(t, J=8 Hz, 3H), 1.17(d, J=4 Hz, 3H), 1.13(d, J=8 Hz, 3H), 0.77(s, 9H), 0.00(s, 3H), −0.02(s, 3H)ppm; 13C NMR (100 MHz, CDCl3) δ=207.54, 206.18, 187.32, 174.88, 146.31, 128.91, 109.49, 60.96, 60.67, 59.82, 53.36, 52.97, 51.22, 44.20, 42.95, 36.33, 35.09, 29.28, 25.75, 21.76, 20.37, 18.14, 14.27, −5.47, −5.61ppm; IR (KBr): υmax =3435.15, 2958.67, 2929.86, 2900.27, 2857.55, 1732.39, 1719.69, 1704.16, 1613.47, 1471.93 cm−1; MS (ESI): m/z: 513; HRMS (ESI) Calcd for ([C27H42O6Si]+Na)+: 513.2648; found: 513.2655 [M+Na]+.
Compound 12
To a solution of 11 (0.49 g, 1 mmol, 1.0eq) in EtOAc (10 mL) was added IBX (0.56 g, 2 mmol, 2.0eq). The mixture was heated at 70°C for 4 hrs and filtered through a short pad of silica gel washed with EtOAc. The solvent was removed under reduced pressure and the residue was purified by flash column chromatography to provide 12 (50%) as colorless liquid: 1H NMR (400 MHz, CDCl3) δ =6.13(s, 1H), 5.95(s, 1H), 5.53(s, 1H), 4.18(q, J=8 Hz, 2H), 3.70(m, 3H), 3.04(m, 1H), 2.79–2.54(m, 5H), 2.29(t, J=4 Hz, 1H), 2.07(m, 1H), 1.26(t, J=8 Hz, 3H), 1.16(t, J=4 Hz, 6H), 0.8(s, 9H), 0.05(s, 6H)ppm; 13C NMR (100 MHz, CDCl3) δ=206.98, 204.92, 198.62, 186.14, 167.70, 141.98, 127.80, 118.79, 62.55, 60.67, 60.26, 53.62, 50.22, 48.93, 40.27, 37.66, 31.54, 28.67, 24.71, 20.51, 19.50, 17.13, 13.02, −6.69, −6.74 ppm; IR (KBr): υmax =2958.94, 2931.02, 2903.23, 2857.93, 1724.34, 1613.69, 1471.01 cm−1; MS (ESI): m/z: 511; HRMS (ESI) Calcd for ([C27H40O6Si]+Na)+: 511.2492; found: 511.2496 [M+Na]+.
General procedure for deprotection of silyl ether
HClO4-SiO2 (0.5 mmol/g, 50 mg) was added to a stirred solution of silyl ether substrate (1 mmol) in CH2Cl2 (5 mL) at room temperature. After complete conversion and filtration to remove the solid, saturated aqueous solution of NaHCO3 was added and separated. The aqueous solution was extracted with CH2Cl2 (5 mL×3). The organic layer was combined, washed with brine (5 mL), dried over MgSO4, and concentrated under reduced pressure. The residue was isolated through short column chromatography on silica gel, which was eluted with ethyl acetate-petroleum to give the target products.
Compound 13
Compound 13 is obtained as colorless liquid: 1H NMR (400 MHz, CDCl3) δ =6.04(s, 1H), 5.08(s, 1H), 5.07(s, 1H), 4.22(q, J=5.76 Hz, 2H), 3.90(dd, J1=2.96 Hz, J2=8.88 Hz, 1H), 3.68(dd, J1=4.64 Hz, J2=8.88 Hz, 1H), 3.28(d, J=4.12 Hz, 1H), 3.05–2.83(m, 4H), 2.66–2.50(m, 4H), 2.29–2.17(m, 3H), 1.29(t, J=5.76 Hz, 3H), 1.20(d, J=5.28 Hz, 3H), 1.18(d, J=5.64 Hz, 3H)ppm; 13C NMR (100 MHz, CDCl3) δ=208.94, 208.07, 189.65, 175.40, 145.51, 128.99, 109.95, 63.68, 61.56, 58.77, 53.37, 52.65, 47.71, 44.56, 42.26, 35.70, 35.35, 29.86, 21.60, 20.59, 14.18ppm; IR (KBr): υmax =3434.28, 3077.84, 2967.18, 2935.97, 2876.31, 1716.22, 1701.69, 1610.55, 1467.79 cm−1; MS (ESI): m/z: 383; HRMS (ESI) Calcd for ([C21H28O5i]+Na)+: 383.1834; found: 383.1837 [M+Na]+.
Compound 14
Compound 14 is obtained as colorless liquid: 1H NMR (400 MHz, CDCl3) δ=6.08(s, 1H), 5.46(s, 1H), 5.36(s,1H), 4.66(s, 1H), 4.30(q, J=5.72 Hz, 2H), 4.12(q, J=5.72 Hz, 2H), 3.91(dd, J1=2.96 Hz, J2=8.88 Hz, 1H), 3.76(dd, J1=4 Hz, J2=8.84 Hz, 1H), 3.37(d, J=4.2 Hz, 1H), 3.11(d, J=7.4 Hz, 1H), 2.83–2.36(m, 5H), 2.15(m, 2H), 1.34(t, J=5.68 Hz, 3H), 1.21(d, J=5.32 Hz, 3H), 1.18(d, J=5.64 Hz, 3H)ppm; 13C NMR (100 MHz, CDCl3) δ=208.42, 207.70, 189.86, 173.27, 149.67, 129.13, 114.97, 78.91, 63.00, 61.72, 60.42, 57.00, 52.22, 47.11, 40.61, 35.56, 32.16, 29.79, 21.63, 20.59, 14.20ppm; IR (KBr): υmax =3434.70, 3080.00, 2967.55, 2933.39, 2877.04, 1716.14, 1608.85, 1467.55 cm−1; MS (ESI): m/z: 399; HRMS (ESI) Calcd for ([C21H28O6i]+Na)+: 399.1784; found: 399.1788 [M+Na]+.
Compound 15
Compound 15 is obtained as colorless liquid: 1H NMR (400 MHz, CDCl3) δ=6.16(s, 1H), 5.98(s, 1H), 5.55(s, 1H), 4.20(q, J=5.72 Hz, 2H), 3.88(dd, J1=3.2 Hz, J2=8.52 Hz, 1H), 3.63(dd, J1=5.04 Hz, J2=8.52 Hz, 1H), 3.02–2.82(m, 3H), 2.63–2.38(m, 5H), 2.16(m, 2H), 1.30(t, J=5.72 Hz, 3H), 1.18(d, J=5.32 Hz, 3H), 1.16(d, J=5.64 Hz, 3H)ppm; 13C NMR (100 MHz, CDCl3) δ=207.98, 206.55, 199.67, 186.67, 169.01, 143.03, 129.05, 119.95, 63.77, 61.78, 61.07, 54.58, 51.33, 50.18, 41.52, 38.92, 32.79, 29.82, 21.47, 20.57, 14.04ppm; IR (KBr): υmax =3401.07, 2968.23, 2919.88, 2876.52, 2850.37, 1715.33, 1693.68, 1642.11, 1608.62, 1466.21 cm−1; MS (ESI): m/z: 397; HRMS (ESI) Calcd for ([C21H26O6i]+Na)+: 397.1627; found: 397.1629 [M+Na]+.
Cell culture and MTT test
Cells were purchased from the Wuxi Innovatbio Medicine Technology Co. Ltd (Wuxi, China) and were maintained at 37°C under the atmosphere of 5% CO2. The cells of human non-small lung cancer (H292) and colon cancer cell line (SNU-1040) cells were cultured in RPMI-medium and others (rat myoblasts and HepG2) were cultured in dulbecco’s modified eagle medium. Ten percent fetal bovine serum (Tianhang Biotechnology Co., Ltd., Zhejiang, China.) and 1% antibiotics (100 U/mL penicillin and 100 mg/mL streptomycin) was supplemented. Cells are inoculated in 96-well plate and cultured for 24 hrs. After attaching the culture bottle wall, the cells were divided into three groups, 1) control group; 2) positive control group; 3) experiment group. The administration concentrations of Cisplatin and compounds were 0.1 µM–10 μmol/L. After 24 hrs, 10% MTT was added and cells were cultured for 3.5 hrs. Then, the culture medium was discarded and 150 μL DMSO was added. Eventually, after incubation for 10 mins by shaking in the dark at room temperature, the absorbance was detected at 490 nm using a microplate reader, and the difference was analyzed by prism 7 software.
Flow cytometry
Cells were cultured in a six well plate and administrated with DTT and compound 15 for 48 hrs. After that, cells were digested and transferred into polyethylene pipe, followed by a centrifugation at 1000 for 5 mins. Then, 1 mL of precooled PBS was given to suspend the cells again. After another centrifugation, PBS was discarded and 70% ethanol was added to fix the cells overnight at 4°C. After rinsing with PBS, the cells were added to propidium iodide staining solution according to instructions. (Beyotime, China). The red fluorescence of the cells (1×105) was detected at 488 nm wavelength by flow cytometer (BD ACCURI C6 PLUS).
Results and discussion
We constructed dienone functionality in the A and D-ring and deleted the complex spiral B-ring moiety of neolaxiflorin B. Compound 6 was obtained through the Pauson-Khand reaction. Starting material 5 was treated with 10 mol% Chlorobis (ethylene) rhodium (I) dimer ([RhCl(COD)]2) under CO at 1atm in toluene at 100°C,8 but the desired product was not observed. Treatment of 5 with Molybdenum hexacarbonyl (Mo(CO)6) was also unsuccessful.9 We found that the conditions using classical dicobalt species Octacarbonyldicobalt (Co2(CO)8) for the Pauson-Khand reaction, however, were successful. We evaluated the Pauson-Khand reaction conditions for the synthesis of compound 6 (Table 1). The optimum conditions for 6 were achieved using the following procedure: Compound 5 was mixed with stoichiometric Co2(CO)8 in n-hexane under an inert argon atmosphere at rt. After 1 hr, neutral Al2O3 was added, and the solvent was removed using a rotary evaporator. The residue was incubated at 0.08 MPa for 15 mins at room temperature, then the flask was flushed with argon. The 0.08 MPa and argon flush steps were repeated four times and further heated at 50°C for 2 hrs. The desired product 6 was achieved by silica gel column chromatography.
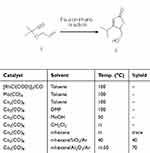 |
Table 1 Pauson–Khand reaction of compound 5 for the synthesis of compound 6 |
A silylation reaction of compound 6 was used to produce compound 7. Deprotonation of compound 7 with Lithium bis(trimethylsilyl)amide (LiHMDS), followed by reaction with compound 810 led to product 9 as the major product, along with the isomer 10 in 86% combined yield (dr =9:1) (Scheme 1). Compound 9 is a crystal that was confirmed by NMR spectral and X-ray crystallographic analysis (Figure 3), its epimer compound 10 is a powder.
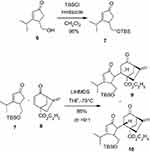 |
Scheme 1 Synthesis of compounds 7, 9, and 10. |
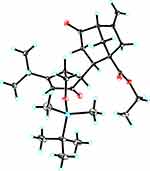 |
Figure 3 X-ray crystallographic analysis of compound 9. |
Compound 9 was subjected to allylic oxidation with selenium dioxide (SeO2), tert-butyl hydroperoxide (t-BuOOH) in CH2Cl2, and 2-Iodoxybenzoic acid (IBX) oxidation to form compounds 11 and 12, respectively (Scheme 2). Deprotection of the silyl ethers of compounds 9, 11, and 12 formed alcohol products in the presence of perchloric acid (HClO4) (Scheme 3).
 |
Scheme 2 Synthesis of compounds 11 and 12. |
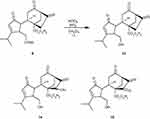 |
Scheme 3 Synthesis of some alcohols. |
The cytotoxic properties of all newly synthesized compounds were evaluated in vitro against three human tumor cell lines (HepG2, NSCLC-H292, SNU-1040) and one rat myotubes cell line (L6) by MTT assay. Eriocalycin B (5) was used as reference drug. The results are summarized in Table 2. (IC50 value is defined as the concentrations corresponding to 50% growth inhibition).
 |
Table 2 In vitro cytotoxic activities of compounds 9–15 (IC50, μM)a,b |
Results are displayed in Table 2. From the table, compounds 9 (IC50 =1.01 μM, NSCLC-H292), 10 (IC50 =3.67 μM, NSCLC-H292), 11 (IC50 =1.58 μM, HepG2), 13 (IC50 =1.23 μM, SNU-1040), and 14 (IC50 =1.08 μM, SNU-1040) showed potency when comparing to the reference eriocalyxin B with only an enone in the A-ring. Meanwhile, these results indicated that the presence of hydroxyl groups at the 6 and 15 position had no effect on the activity. Episomers 9 and 10 did not show significant differences in cytotoxicity and selectivity, so we chose compound 9 that was more readily available for further modification. Remarkably, compounds 12 and 15 with a dienone in the A and C ring, showed stronger activity against all tumor cell lines compared to those with one enone (compounds 9, 10, 11, 13, and 14). Deprotection of C-6 silyl ether of compound 12 produced compound 15, which greatly decreased the cytotoxic activity against rat myoblast cell line (L6). To obtain insight into the cytotoxic potential of these new compounds on normal cells, the effect of compound 15 and eriocalyxin B was evaluated in human liver carcinoma cells (HepG2) and rat myotubes (L6) cells. The potency and selectivity of compound 15 were higher than that of eriocalyxin B. The SI value (selective index, IC50 of normal cells/IC50 of tumor cells) of compound 15 was 123.14, and the Si value of eriocalyxin B was 54.33.
After cytotoxic assessment experiment, we conducted flow cytometry analysis to further understand potential underlying cellular mechanisms of actions of compound 15 (Table 3).
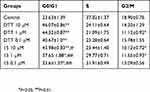 |
Table 3 Percentage of G0/G1, S, and G2/M phase |
We found that the percentage of NSCLC-H292 cells in G0/G1 phase increased significantly but those in G2/M decreased notably in the presence of DTT and compound 15, indicating that cells were blocked in G0/G1phase, thus inhibiting the proliferation of tumor cells.
Conclusion
In conclusion, we have designed and synthesized several compounds bearing an α, β-unsaturated ketone moiety. The in vitro activities were evaluated against HepG2, NSCLC-H292, and SNU-1040 tumor cell lines and an L6 myoblast cell line. This study has revealed that compound 15 is a promising antitumor lead due to the cytotoxic potencies and the high selectivity it displayed when compared to natural counterparts. Further biological investigations are currently undergoing to investigate how compound 15 inhibits tumor cell proliferation in our laboratory and the results will be reported in due course.
Acknowledgment
This work was supported by grants from the National Natural Science Foundation of China (21462053, 21564018), the Science Foundation of Yunnan Province Office of Education (2014Z044), Program of the Undergraduate Innovation (2018059).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Yan BC, Hu K, Sun HD, Puno PT. Recent advances in the synthesis of Isodon diterpenoids and schinortriterpenoids. Chin J Org Chem. 2018;38:2259. doi:10.6023/cjoc201806002
2. He ZA, Lu GZ, Zhu LL, Wang YX, Yan FL, Zhang JX. Antibacterial constituents from Isodon excisoides. J Chin Chem Soc. 2010;57:256–259. doi:10.1002/jccs.201000039
3. Liu M, Wang WG, Sun HD, Pu JX. Diterpenoids from Isodon species: an update. Nat Prod Rep. 2017;34:1090–1140. doi:10.1039/C7NP00027H
4. Lazarski KE, Moritz BJ, Thomson RJ. The total synthesis of Isodon diterpenes. Angew Chem Int Ed. 2014;53:10588–10599. doi:10.1002/anie.201404482
5. Wang WG, Du X, Li XN, et al. New bicyclo[3.1.0]hexane unit ent-kaurane diterpene and its seco-derivative from Isodon eriocalyx var. laxiflora. Org Lett. 2012;14:302–305. doi:10.1021/ol203061z
6. Zhao Y, Niu XM, Qian LP, Liu ZY, Zhao QS, Sun HD. Synthesis and cytotoxicity of some new eriocalyxin B derivatives. Eur J Med Chem. 2007;42:494–502. doi:10.1016/j.ejmech.2006.11.004
7. Niu XM, Li SH, Li ML, et al. Cytotoxic ent-kaurane diterpenoids from Isodon eriocalyx var. laxiflora. Planta Med. 2002;68:528–533. doi:10.1055/s-2002-32551
8. Lv C, Yan XH, Tu Q, et al. Isolation and asymmetric total synthesis of perforanoid A. Angew Chem Int Ed. 2016;55:7539. doi:10.1002/anie.201602783
9. Javier A, Marta RR, Juan CC. Mild and efficient molybdenum-mediated Pauson-Khand-type reaction. Org Lett. 2005;7:431–434. doi:10.1021/ol047678u
10. Hu Y, Li XN, Ma ZJ, et al. Synthesis of novel ent-kaurane-type diterpenoid derivatives effective for highly aggressive tumor cells. Molecules. 2018;23:3216. doi:10.3390/molecules23123216
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
