Back to Journals » Cancer Management and Research » Volume 10
Survival and recurrence patterns of multifocal glioblastoma after radiation therapy
Authors Syed M, Liermann J, Verma V, Bernhardt D , Bougatf N , Paul A, Rieken S, Debus J , Adeberg S
Received 20 February 2018
Accepted for publication 25 June 2018
Published 4 October 2018 Volume 2018:10 Pages 4229—4235
DOI https://doi.org/10.2147/CMAR.S165956
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Kenan Onel
Mustafa Syed,1–3 Jakob Liermann,1–3 Vivek Verma,4 Denise Bernhardt,1,2 Nina Bougatf,1,3 Angela Paul,1,2 Stefan Rieken,1–3 Jürgen Debus,1–3,5 Sebastian Adeberg1,2,5
1Heidelberg Institute of Radiation Oncology, Im Neuenheimer Feld 400, 69120 Heidelberg, Germany; 2Department of Radiation Oncology, University Hospital Heidelberg, Im Neuenheimer Feld 400, 69120 Heidelberg, Germany; 3Heidelberg Ion-Beam Therapy Center (HIT), Im Neuenheimer Feld 450, 69120 Heidelberg, Germany; 4Department of Radiation Oncology, University of Nebraska Medical Center, Omaha, NE 68198, USA; 5Clinical Cooperation Unit Radiation Oncology, German Cancer Research Center (DKFZ), Im Neuenheimer Feld 280, 69120 Heidelberg, Germany
Purpose: It is hypothesized that multifocal glioblastoma (mGBM) is associated with worse prognosis compared to unifocal disease (uGBM). This study aims to investigate the differences in survival rates and progression patterns of patients between these two groups after radiation therapy.
Patients and methods: We retrospectively analyzed 265 patients with primary GBM undergoing radiation therapy at the Department of Radiation Oncology, University Hospital Heidelberg, Germany, between 2004 and 2013. Of these, 202 (76%) were uGBMs and 63 (24%) were mGBMs. First, progression-free survival (PFS) and overall survival (OS) between groups were compared using the Kaplan–Meier method. Second, univariate and multivariate Cox proportional hazards regression was applied to discern prognostic and predictive factors with PFS and OS in the cohorts. Third, recurrence patterns of uGBMs and mGBMs were assessed on follow-up MRIs and compared using the chi-squared test.
Results: As compared to patients with uGBM, patients with mGBM experienced significantly worse median OS (11.5 vs 14.8 months, P=0.032). Overall, 195 (73.0%) patients experienced tumor progression: 153 (75.7%) patients with uGBM and 46 (73.0%) patients with mGBM. There were no significant differences in PFS between the respective groups (6.5 vs 6.6 months, P=0.750). Of note, concomitant temozolomide treatment was associated with an OS benefit in both uGBM and mGBM by about five months (P=0.006 and P<0.001). Furthermore, there were no significant differences in progression patterns of uGBM and mGBM. Both recurred as unifocal and multifocal disease (P=0.51), and local vs distant brain recurrences occurred similarly in both groups (OR=1.33, P=0.53).
Conclusion: Multifocality is an independent predictor of survival in GBM. Concomitant temozolomide treatment improved OS of patients with mGBM and uGBM. Both disease types showed similar patterns of progression. Current target volume concepts seem to be adequate in both unifocal and multifocal GBMs. GBM, the most common primary brain tumor in adults, is associated with poor survival. We show herein that multifocality is an independent prognostic factor for survival. We also illustrate that the progression patterns of both unifocal and multifocal GBM are similar.
Keywords: temozolomide, chemotherapy, progression, multifocal, glioblastoma, gliomatosis, high grade glioma, target volume
Introduction
Glioblastoma (GBM) is the most common primary malignant tumor of the central nervous system in adults, with survival rates less than 15 months after trimodality therapy.1 However, individual heterogeneity in the survival rates is undoubtedly observed in light of several prognostic factors that have been established in the recent years. These include age, Karnofsky performance status (KPS), tumor locality, and others. Additionally, molecular factors such as O6-methylguanine-DNA-methyltransferase (MGMT) promoter methylation or isocitrate dehydrogenase (IDH) mutation are increasingly playing an important role concerning therapy response and individual survival.2–7
Multifocal disease at presentation has several definitions and varies from study to study, but demonstrates decreased overall survival (OS) as compared to unifocal disease.8,9 However, although progression patterns of recurrent GBM have been described,4 those of the multifocal GBM (mGBM) subset have been understudied.10,11
In this study, we compare unifocal GBM (uGBM) and mGBM regarding progression free survival (PFS) and OS while also evaluating other factors associated with outcome. Furthermore, we systematically review progression patterns in each disease type; in other words, whether they progress in similar unifocal or multifocal patterns and the anatomical location of the recurrent lesion in comparison to the original lesion were evaluated.
Implications of our data include differentiating between GBMs regarding variations in growth and recurrence patterns with the potential necessity of altering radiation therapy (RT) target volumes. Furthermore, any differences in recurrence and progression patterns may require revisions in target volume contouring in the re-irradiation setting.
Patients and methods
We retrospectively analyzed 265 patients with primary GBM undergoing RT at our institution (Department of Radiation Oncology, University Hospital Heidelberg, Heidelberg, Germany) between 2004 and 2013. Patient characteristics are listed in Table 1.
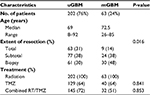  | Table 1 Patient characteristics Abbreviations: mGBM, multifocal glioblastoma; RT, radiotherapy; TMZ, temozolomide; uGBM, unifocal glioblastoma. |
Though there is no consensus in the definition of multifocal disease, and various definitions are utilized, for the purposes of this study, mGBM was characterized as at least two non-connected foci of disease at least 1 cm apart from each other on magnetic resonance imaging (MRI).12 Edema and/or T2/FLAIR signal abnormality was allowed to connect the gross tumor as per other studies.8
All patients underwent RT for the primary disease. In brief, patients were immobilized using custom head masks and simulated with computed tomography and MRI scanning. Target definition and treatment planning were carried out via several methods, techniques, and doses depending on the particular case and time period (Figure 1). Follow-up was at a regular interval of 3 months including repeat MRI.
Progression-free survival (PFS, months) and OS (months) were calculated from the date of the initiation of RT to the date of radiologic progression and to the day of death or last follow-up, respectively. Survival curves for PFS and OS were made using the Kaplan–Meier method and the mGBM/uGBM groups compared with the log-rank test. Univariate and multivariate Cox proportional hazards models were used to evaluate the influence of cofactors on survival.
For progression analysis, we evaluated pre-therapeutic and post-therapeutic contrast enhanced MRI according to the Response Assessment in Neuro-Oncology criteria.13 Progression was defined as the appearance of a new lesion, a significant increase in non-enhancing T2/FLAIR lesions, or as a ≥25% increase in T1 enhancing lesions.13 If pseudoprogression was clinically suspected, repeat imaging was obtained at a subsequent interval to confirm; if corroborated, the initial MRI was utilized as the date of progression.
Progression patterns were studied by reviewing the appearance of the pertinent area in comparison to the site of the initial lesion on MRI. We studied whether the progression occurred locally, in the same hemisphere, in corpus callosum, or in the contralateral hemisphere. An appearance inside the initial planning target volume (PTV) or ≤1 cm was regarded as a local progression, whereas an appearance ≥1 cm of PTV was considered as a distant progression. We also investigated if the progression arose as unifocal or multifocal disease (defined as above), regardless of its initial disease occurrence.
Statistical analyses were performed using SigmaPlot™ (Systat Software GmbH, Erkrath, Germany) software. A P-value<0.05 was considered statistically significant. Comparisons between the mGBM and uGBM groups were made using the Wilcoxon rank-sum test for means, and Fisher’s exact or chi-squared tests for proportions.
This study was approved by the university review board and ethics committee and in accordance to the declaration of Helsinki of 1975 in its most recent version. Ethics approval for the study was obtained from the local ethics committee, University Hospital Heidelberg (Nr. S-056/2015). Patients provided written informed consent for their data to be used in the study.
Results
Patient characteristics
In our analysis, 202 (76%) out of 265 patients had a unifocal disease, whereas 63 (24%) of them had a multifocal disease (Table 1) and were followed for a median follow-up time of 13.29 months (range 1.6–55.9 months). The patients in uGBM group had a median age of 69 years (range 8–92 years), whereas the multifocal group consisted of patients with a median age of 72.5 years (range 26–85 years).
Twenty-seven percent of the patients underwent a total resection and 38% of patients underwent a subtotal resection, whereas 35% only had a biopsy done. All of the patients were treated with radiotherapy to a median dose of 60.0 Gy (range 39.5–68.0 Gy) in 2.0 Gy (range 1.8–3.0 Gy) fractions. The dose on the lower range was due to earlier termination of RT. Sixty-three percent of the patients were treated with temozolomide (TMZ) overall, and 66% received concomitant TMZ therapy (Table 1).
A total of 199 (75.1%) patients experienced tumor progression on follow-up MRI scans, which corresponded to 153 (75.7%) patients with uGBM and 46 (73.0%) patients with mGBM.
Survival analysis
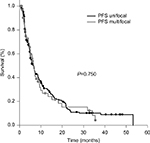
There was no significant difference in PFS between the unifocal (median 6.6 months, range 0.5–53.2 months) and the multifocal groups (median 6.5 months, range 1.1–24.2 months) (P=0.75, Figure 2).
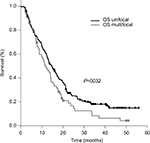
However, patients with mGBM experienced significantly worse median OS of 11.5 months (range 1.6–25 months) as compared to patients with uGBM (median 14.8 months, range 1–55.9 months, P=0.032) (Figure 3). Similarly, the two-year survival was 1.8% in mGBM and significantly lower than in uGBM patients (18.5%) (P=0.004).
Univariate analysis showed that patients >60 years experienced significantly worse PFS (P=0.02), whereas those treated with concomitant TMZ (P<0.001) and with a KPS >60% (P=0.05) experienced a significantly higher PFS (Table 2). The univariate analysis for OS did not show a significant effect of all of the abovementioned factors except for multifocal disease (P<0.001) (Table 2).
Multivariate analysis, taking the most common confounders (age, KPS, resection status, TMZ therapy, MGMT promoter methylation status, multifocal disease) into account, showed that concomitant TMZ therapy and KPS >60% years remained prognostic factors for significantly better PFS (P=0.002 and P=0.032, respectively), while age >60 years was a negative prognostic factor (P=0.015) (Table 3). Regarding multivariate analysis for OS, only multifocal disease showed a significantly worse prognostic impact (P<0.001, Table 3).
Further evaluation showed that concomitant TMZ therapy was associated with significantly better OS in both mGBM (8.3 vs 14.2 months, P=0.006) and uGBM (11.7 vs 17.0 months, P<0.001). Thus, the OS benefit with TMZ was about 5 months in both groups.
Univariate and multivariate analyses for OS revealed a positive prognostic effect for concomitant TMZ treatment in both mGBM (P=0.008) and uGBM (P<0.001).
Progression analysis
Lastly, when evaluating disease recurrence patterns of uGBM and mGBM, both appeared to recur either unifocally or multifocally. Of the uGBM group, 67.1% of recurrences were unifocal and 32.9% were multifocal. These numbers in the mGBM group were 60.5% and 32.9%, respectively. There were no differences between both cohorts in terms of this parameter (P=0.51).
Additionally, we also analyzed the localization of the new lesions compared to the original site. The recurrence pattern of new lesions was also similar in both groups; “local” and “distant” recurrences occurred in 67% and 33% of the uGBM cohort and 60% and 40% of the mGBM cohort (P=0.53), respectively.
Discussion
MGBMs make up a substantial percentage of all GBMs. The goal of this work was to evaluate the nature of recurrences in this cohort, as juxtaposed with uGBMs. In addition to corroborating inferior OS in the mGBM cohort, we found no differences in PFS. Multifocality was an independent correlate of OS as well. Of note, TMZ treatment was associated with an OS benefit in both uGBM and mGBM. Lastly, there were no dissimilarities between mGBMs and uGBMs in terms of unifocal vs multifocal recurrence, along with the location of the recurrence.
Our data are in agreement with existing findings in that uGBMs and mGBMs tend to most frequently recur in-field,14 although that publication found a PFS but not an OS difference, contrary to our findings. Moreover, we could also confirm the two most established therapy – independent prognostic factors age and KPS score – in the univariate and multivariate analyses.5,15 To some extent, our data contradict those of Thomas et al,16 who found no independent correlation between multifocality and outcomes. However, as multifocality was associated with lower KPS in that study, it is possible that an independent effect was unable to be ascertained therein.
TMZ therapy has become a standard convention and is the most commonly used chemotherapeutic agent in patients with newly diagnosed GBM.1 Concomitant TMZ treatment improved OS of patients with mGBM and uGBM by approximately 5 months. We propose that radiotherapy with concomitant TMZ treatment should also be considered as the first-line treatment of patients with mGBM. Data regarding TMZ maintenance therapy were not available for our cohort as this treatment is usually not prescribed by our radiation oncology department.
The worse prognosis of mGBM may be explained by recent pathogenesis-related discoveries. Sahm et al17 described IDH1-mutant GBM cells and cell protrusions in the contralateral brain hemisphere. Osswald et al18 demonstrated that GBM cells have long protrusions with which the cells invade, interconnect, and communicate with each other using intercellular calcium waves. The result is a dynamic functional intercellular network, which is resistant to standard treatment including RT. The cells are able to distribute high levels of calcium through their protrusions to other cells in the network. This bypassing of high intracellular calcium concentrations is typically induced by radiotherapy and leads to apoptosis.19
Although the lesions in mGBM might appear as individual lesion with no macroscopic-radiological connection, our results and the abovementioned studies support the idea that the lesion are interconnected microscopically and are a part of all-brain systemic syncytium.
These findings are underlined by the results of our progression analysis. To our knowledge, this study is the only one to date comparing the recurrence patterns of uGBM with mGBM. If multifocal disease were more likely to recur in a multifocal pattern, re-irradiation may involve larger targets margins. Our data, however, show that there are no substantial correlations between proclivity to recur multifocally and the type of the original tumor. Hence, re-irradiation margins appear to be adequate regardless of focality of initial disease.
For our study, we characterized multifocal disease as at least two non-connected, contrast enhancing foci at least 1 cm apart from each other on T1 MRI. The lesions were typically connected by T2/FLAIR signal abnormality. Several other studies have used different, inconsistent definitions and also the term multicentric GBM. In contrast to mGBM, this GBM subtype demonstrates multiple foci without interconnecting T2/FLAIR signal abnormality. These tumors were thought to be separate tumors, occurring synchronously.9–11 However, with improved MRI imaging, the frequency of the diagnosis of multicentric GBM has been shrinking as connections between lesions could be seen.8 With the abovementioned histological findings, multicentric GBMs appear rather to be mGBMs.
Lastly, a major focus of risk stratification of GBMs is occurring with the advent of molecular and genetic signatures of cerebral neoplasms. These results for mGBMs vs uGBMs have shown rather conflicting data. Two studies have found no differences in terms of gene expression signatures9,14 but a third has postulated distinct epigenetic modifications in mGBMs.20,21 Further translational studies are clearly indicated to address this issue, including analysis of microRNA expression.
The strength of this study is a comparably large and homogenous patient collective with a long follow-up time. Furthermore, as TMZ has become a standard chemotherapy now, our patient collective received uniform treatment. All MRIs were re-analyzed by a senior radiologist to minimize intraobserver variability and provide consistency. Nevertheless, our findings have several limitations aside from the retrospective nature. First, molecular characteristics were incompletely assessed in the cohorts. Second, because the definition of mGBM and parameters related to progression vary between studies, generalization and direct comparison with other data are problematic.
Conclusion
Multifocality is an independent predictor for survival in GBM. Both uGBM and mGBM benefit from TMZ therapy, and progression patterns of both types do not differ from each other.
Acknowledgments
The abstract of this paper was presented at the German Society of Radiation Oncology (DEGRO) Annual Meeting 2016 as a poster presentation with interim findings. The poster’s abstract was published in “Abstracts DEGRO 2016” in Strahlentherapie und Onkologie: https://link.springer.com/article/10.1007%2Fs00066-016-0974-z.
Author contributions
SA, AP, SR, and JD treated the patients. SA and MS collected the data. MS and SA evaluated the dataset and performed statistical analysis. MS, SA, JL, VV, and DB wrote and edited the manuscript. All the authors read and approved the manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Stupp R, Hegi ME, Mason WP, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10(5):459–466. | ||
Carrillo JA, Lai A, Nghiemphu PL, et al. Relationship between tumor enhancement, edema, IDH1 mutational status, MGMT promoter methylation, and survival in glioblastoma. AJNR Am J Neuroradiol. 2012;33(7):1349–1355. | ||
Verma V, Mehta MP. Clinical ramifications of “genomic staging” of low-grade gliomas. J Neurooncol. 2016;129(2):195–199. | ||
Gebhardt BJ, Dobelbower MC, Ennis WH, Bag AK, Markert JM, Fiveash JB. Patterns of failure for glioblastoma multiforme following limited-margin radiation and concurrent temozolomide. Radiat Oncol. 2014;9:130. | ||
Weller M, Felsberg J, Hartmann C, et al. Molecular predictors of progression-free and overall survival in patients with newly diagnosed glioblastoma: a prospective translational study of the German Glioma Network. J Clin Oncol. 2009;27(34):5743–5750. | ||
Chen JR, Yao Y, Hz X, Qin ZY. Isocitrate Dehydrogenase (IDH)1/2 Mutations as Prognostic Markers in Patients With Glioblastomas. Medicine (Baltimore). 2016;95(9):e2583. | ||
Hegi ME, Liu L, Herman JG, et al. Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMT activity. J Clin Oncol. 2008;26(25):4189–4199. | ||
Lasocki A, Gaillard F, Tacey M, Drummond K, Stuckey S. Multifocal and multicentric glioblastoma: Improved characterisation with FLAIR imaging and prognostic implications. J Clin Neurosci. 2016;31:92–98. | ||
Patil CG, Yi A, Elramsisy A, et al. Prognosis of patients with multifocal glioblastoma: a case-control study. J Neurosurg. 2012;117(4):705–711. | ||
Hassaneen W, Levine NB, Suki D, et al. Multiple craniotomies in the management of multifocal and multicentric glioblastoma. Clinical article. J Neurosurg. 2011;114(3):576–584. | ||
Zamponi N, Rychlicki F, Ducati A, Regnicolo L, Salvolini U, Ricciuti RA. Multicentric glioma with unusual clinical presentation. Childs Nerv Syst. 2001;17(1-2):101–105. | ||
Singh G, Mehrotra A, Sardhara J, et al. Multiple glioblastomas: Are they different from their solitary counterparts? Asian J Neurosurg. 2015;10(4):266–271. | ||
Wen PY, Macdonald DR, Reardon DA, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol. 2010;28(11):1963–1972. | ||
Paulsson AK, Holmes JA, Peiffer AM, et al. Comparison of clinical outcomes and genomic characteristics of single focus and multifocal glioblastoma. J Neurooncol. 2014;119(2):429–435. | ||
Showalter TN, Andrel J, Andrews DW, Curran WJ Jr, Daskalakis C, Werner-Wasik M. Multifocal glioblastoma multiforme: prognostic factors and patterns of progression. Int J Radiat Oncol Biol Phys. 2007;69(3):820–824. | ||
Thomas RP, Xu LW, Lober RM, Li G, Nagpal S. The incidence and significance of multiple lesions in glioblastoma. J Neurooncol. 2013;112(1):91–97. | ||
Sahm F, Capper D, Jeibmann A, et al. Addressing diffuse glioma as a systemic brain disease with single-cell analysis. Arch Neurol. 2012;69(4):523–526. | ||
Osswald M, Jung E, Sahm F, et al. Brain tumour cells interconnect to a functional and resistant network. Nature. 2015;528(7580):93-8. | ||
Tombal B, Denmeade SR, Gillis JM, Isaacs JT. A supramicromolar elevation of intracellular free calcium ([Ca(2+)](i)) is consistently required to induce the execution phase of apoptosis. Cell Death Differ. 2002;9(5):561–573. | ||
Liu Q, Liu Y, Li W, et al. Genetic, epigenetic, and molecular landscapes of multifocal and multicentric glioblastoma. Acta Neuropathol. 2015;130(4):587–597. | ||
Abou-El-Ardat K, Seifert M, Becker K, et al. Comprehensive molecular characterization of multifocal glioblastoma proves its monoclonal origin and reveals novel insights into clonal evolution and heterogeneity of glioblastomas. Neuro Oncol. 2017;19(4):546–557. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.