Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
Surgical treatment of an esophageal bronchogenic cyst with massive upper digestive tract hematoma without esophagectomy: a case report and the review of the literature
Authors Cheng Y, Chen D, Shi L, Yang W, Sang Y, Duan S, Chen Y
Received 3 October 2017
Accepted for publication 7 February 2018
Published 3 May 2018 Volume 2018:14 Pages 699—707
DOI https://doi.org/10.2147/TCRM.S153145
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Yuanjun Cheng,1,* Donglai Chen,2,* Li Shi,1 Wentao Yang,1 Yonghua Sang,1 Shanzhou Duan,1 Yongbing Chen1
1Department of Cardiothoracic Surgery, the Second Affiliated Hospital of Soochow University, Suzhou, China; 2Department of Thoracic Surgery, Shanghai Pulmonary Hospital, Tongji University School of Medicine, Shanghai, China
*These authors contributed equally to this work
Abstract: Esophageal bronchogenic cysts are extremely rare. Here we report a case of massive upper digestive tract hematoma and bronchogenic cyst mimicking aortic dissection that was safely removed without esophagectomy. A 30-year-old man was referred to our hospital for the treatment of a mediastinal cystic tumor located in the submucosa of the distal esophagus. His chief complaints were dysphagia > 1 week and severe persistent upper abdominal pain mimicking aortic dissection with constant vomiting for 1 day after gastroscopy examination. The serum level of carbohydrate antigen (CA)199 was > 1,000 U/mL and CA125 was 4,816 U/mL. Hemoglobin levels decreased from 122 g/L to 85 g/L in 5 days. Imaging examinations detected a huge hematoma of the gastric wall. Preoperative diagnosis was difficult. Although the pain indicated a possible aortic dissection, the abnormal levels of tumor biomarkers suggested malignancy. The patient underwent left thoracotomy. The cyst showed an exophytic lesion connected to the esophageal wall at the level of the gastroesophageal junction. Muddy brown contents were obtained by aspiration of the mass intraoperatively. Because enucleation could not be performed, esophageal myotomy in the distal esophagus and partial resection of the cyst were selected. Histopathological examination indicated a bronchogenic cyst of the esophagus. At a follow-up visit 3 months later, the patient had no signs of disease recurrence or any complaints. Postoperative tumor biomarkers returned to normal range. The present report summarizes the clinical details of the case and reviews the literature in order to improve the accuracy of diagnosis.
Keywords: esophagus, bronchogenic cyst, thoracotomy, tumor biomarkers, hematoma
Introduction
Bronchogenic cysts (BCs) are rare congenital malformations arising during the development of the ventral foregut, and they are most commonly found in the middle and superior mediastinum.1 They are mainly located near the tracheobronchial tree or lung parenchyma with or without a well-defined wall.2,3 Although these lesions are often benign, BCs can also cause symptoms such as chest pain, cough, dysphagia, and dyspnea due to compression of the surrounding structures.4 As the majority of cysts are within the mediastinum, paraesophageal BCs are frequently reported. Intramural esophageal BCs are reported occasionally.5 Typically, BCs are negative for carbohydrate antigen (CA)199 and CA125. Hematoma of the gastric wall has not been previously reported in any BC patients. We herein describe an extremely rare case of intramural esophageal BC and hematoma of the gastric wall with positive expressions of CA199 and CA125. Surgical resection is generally recommended in cases showing relevant complications and changes of the esophageal cyst. Complete excision of the entire cyst is necessary to avoid recurrence. This report describes the feasibility of thoracotomy for removing esophageal cysts and hematoma of the gastric wall associated with violent persistent abdominal pain mimicking aortic dissection. Although enucleation is a standard treatment for small-sized cysts, the technique has not been adopted for large cysts with severe inflammatory adhesions. To our best knowledge, no similar case has been previously reported. The patient signed an informed consent preoperatively.
Case report
A 30-year-old man was referred to our hospital for the treatment of a mediastinal cyst located in the submucosa of the distal esophagus in November 2016. His chief complaints were dysphagia > 1 week and severe persistent upper abdominal pain mimicking aortic dissection with constant vomiting for 1 day after gastroscopic examination. The patient was unable to take in solids or liquids. The patient denied any chest symptoms, such as coughing and chest pain. His past medical history was unremarkable.
Physical examination did not reveal any obvious abnormal signs, and preoperative laboratory data were within the normal range, with the exception of the tumor markers CA199 and CA125. The serum level of CA199 was > 1,000 U/mL (normal range: <37 U/mL), and CA125 was 4,816 U/mL (normal range: <37 U/mL), while carcinoembryonic antigen (normal range: <5 μg/L) and squamous cell carcinoma antigen (normal range: <1.5 μg/L) were within the normal range.
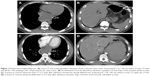
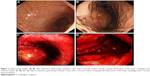
The thoracic computed tomography (CT) scan revealed a well-defined low-density mass originating from the distal esophageal wall, measuring 67 × 51 × 60 mm with a CT value of 28 HU. The day before admission, the gastroscopy examination showed a smooth surface causing partial obstruction of the lower esophagus and a normal stomach fundus. An abdominal CT revealed a normal shape of the stomach (Figure 1A and B). Five days later, transverse contrast-enhanced chest CT showed a sharply defined mass measuring 47 × 70 × 85 mm with a CT value of 31 HU. Transverse contrast-enhanced abdominal CT showed a huge lump in the stomach wall (Figure 1C and D). The distal esophageal lumen was almost closed. After admission, a mass with a smooth surface causing partial obstruction of the lower esophagus (labelled E on Figure 2) and stomach fundus (labelled G on Figure 2) was observed by gastroscopy (Figure 2A–D). Laboratory data showed that hemoglobin (HGB) was 85 g/L (normal range: 120–160 g/L) compared with 122 g/L 5 days before.
Given these findings and evaluation, an esophageal cyst with malignancy was suspected and the patient underwent surgery. Preoperatively, we chose among surgical procedures including enucleation, complete resection of the cyst, esophageal reconstruction, and radical resection of esophageal cancer according to the characteristics of the cyst and the hematoma of the gastric wall.
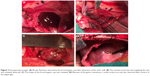
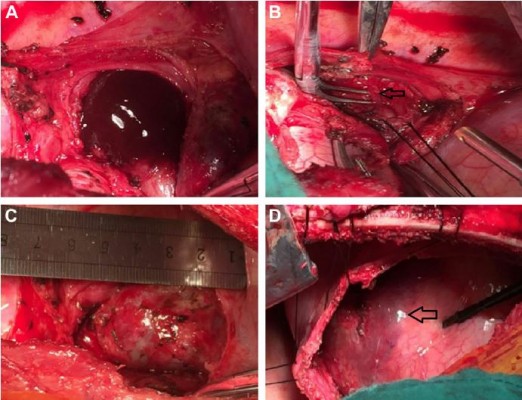
In the operation room, under general anesthesia, the left lung was collapsed using a double-lumen endotracheal tube while the patient was in a right lateral position. A left thoracotomy revealing a mass in the distal esophagus was performed. The mass was large and had severely adhered to the adjacent tissues, especially the diaphragm. The muddy brown contents were observed by aspiration of the mass. Because complete resection of the cyst was difficult, enucleation could not be performed. Esophageal myotomy in the distal esophagus was selected to preserve esophageal dynamic function and mucosal integrity. Partial resection of the cyst was performed simultaneously. A longitudinal 5-cm esophageal myotomy was performed, and a 40 × 70 × 80 mm unilocular cystic mass was identified. The cyst showed an exophytic swelling appearance and dense adhesions were observed between the cystic wall and the esophageal mucosa (Figure 3A–D). After separating the adhesions around the distal esophagus, the cyst was decompressed and intracystic fluid was suctioned to dissect the adjacent tissues easily and safely. The residual cyst wall was treated with electrocautery and iodine to prevent recurrence. The absence of mucosal perforation or esophagostenosis was confirmed intraoperatively by injecting water into the esophagus and stomach. The proper muscle layer of the esophagus was recovered approximately. Opening of the diaphragm and removal of the hematoma were achieved by making a small incision in the surface of the gastric muscle. A chest drainage tube was inserted through the eighth intercostal space.
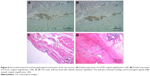
Histopathologically, the wall of the cyst was lined with ciliated columnar epithelium. The wall also contained cartilage and bronchogenic glands. We also detected positive expression of CA199 and CA125 in cyst sections by immunohistochemical staining (Figure 4A and B). A bronchial cyst of the esophagus was finally confirmed based on these findings (Figure 4C and D).
The postoperative process was favorable. A control esophagogram (barium meal) performed on postoperative day 30 showed neither stenosis nor leakage (Figure 5A–D), and oral fluid intake was resumed. The patient was discharged tolerating liquids and soft food uneventfully. The serum CA199 and CA125 levels decreased to normal levels after the operation. The patient is presently doing well at 3 months after surgery. No recurrence or dysphagia was noted during the follow-up period.
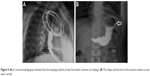  | Figure 5 (A) A control esophagogram showed that the esophagus (black arrow) had neither stenosis nor leakage. (B) The shape and function of the stomach (white arrow) were normal. |
Patient consent
Written informed consent was obtained from the patient for publication of clinical data including all images in this case report.
Discussion
BCs are congenital malformations arising from abnormal budding of the ventral foregut during the early stage of gestation. Considering the time of separation from the primary airways, BCs may present as mediastinal cysts approaching the tracheobronchial tree or as pulmonary cysts within the lung parenchyma.6–9 Over 20 cases have been reported in the literature since 2000 and are summarized in Table 1.2,4,10–34 The most common thoracic location of BCs is the middle mediastinum close to the carina, whereas the paratracheal, hilar, and intraparenchymal locations are rare. BCs have been reported in the posterior and anterior mediastinum, and occasionally in the diaphragm and intrapericardial or intracardiac regions.15,19 BCs are also located in extrathoracic regions such as cervical, skin, and retroperitoneum. Paraesophageal BCs are uncommon and difficult to differentiate from esophageal duplications. However, esophageal BCs are extremely rare, and their morbidity remains unknown because most patients are asymptomatic.35 Most often, these cysts are discovered at an early age, between 20 and 40 years, and they are rarely diagnosed in elderly patients or in the pediatric population.6–9 The present 30-year old patient is in his prime of life and his symptoms was consistent with his common age group of patients. Symptoms can be found incidentally or occasionally, and they vary according to the age of presentation, and the size of the cyst and its location. The symptoms are usually caused by compression of the surrounding structures or by complications related to the cyst.31 The most frequent symptoms are pain, cough, fever, and dyspnea; in rare cases, the cyst is complicated by a superior vena cava obstruction, pneumothorax, dysphagia, severe hemoptysis, or malignancies.6–9 Although BCs in the mediastinum are generally asymptomatic, the gastroscopy performed in our case irritated the cyst, resulting in intracapsular hemorrhage and hematoma of the gastric wall. The esophageal and gastric cavity almost disappeared, leading to dysphagia.
The correct preoperative diagnosis is very difficult due to the rarity of the disease and the lack of specific imaging methods. Different laboratory tests and imaging techniques can be used for preoperative diagnosis, and some differential diagnoses need to be considered, such as aortic aneurysm, recurrent tuberculosis, tuberculoma, aspergilloma, hydatid disease, fungal disease, benign or malignant tumor, paraesophageal cyst, cystic nervous tumor, pulmonary sequestration, congenital cystic adenomatoid malformation, esophageal leiomyoma, pleural fibroma, lymphadenopathy, and intracystic hemorrhage.24 However, the characteristics of the mass need to be defined by histopathological examination. Intracystic hemorrhage can be detected by decreased HGB levels, as in the present case.
The characteristics of the mass can be assessed by measuring tumor marker levels. CA199 is the primary marker of choice for pancreatic, colorectal, and hepatocellular cancers, and it is also elevated in pancreatitis, cirrhosis, and obstructive disease of the bile duct.36 CA125 is the marker of choice for ovarian cancer.37 High levels of tumor markers such as CA199 and CA125 are rarely observed in patients with esophageal cysts. Reports of high levels of both CA199 and CA125 are extremely rare.16,33,38 In the present case, abnormal levels of CA199 and CA125 were also detected in the serum and the cyst sections, and the elevated CA199 and CA125 levels decreased to a normal range postoperatively. These findings suggested that the serum levels of CA199 and CA125 were partly correlated with epithelial cell secretion from the esophageal BC in our patient. Although all these cases have remarkably elevated CA199 and CA125 levels, only the present case has developed massive upper digestive tract hematoma, leading to severe, tearing pain in the abdomen. To our best knowledge, no similar cases have been previously reported.
Despite the use of several imaging techniques such as radiography, barium meal, CT scan, magnetic resonance imaging (MRI), endoscopic ultrasound (EUS), EUS-fine needle aspiration (EUS-FNA), and gastroscopy for the diagnosis of esophageal cysts, a definitive diagnosis remains difficult preoperatively. Mediastinal BCs usually present as non-specific middle or posterior mediastinal masses on chest radiographs. Barium meal can show a local exogenous mass in the esophagus, although it is difficult to discriminate esophageal cysts from other diseases. CT can reveal the location of a cyst and its relationship with surrounding tissues and it is superior to radiography. BCs typically manifest as spherical masses of either water or soft tissue attenuation. If BCs manifest as water attenuation masses in CT scans, they can be differentiated from other mediastinal masses such as lymphadenopathy or neoplasia. However, BCs are detected as soft tissue attenuation masses in CT scans; therefore, differentiation from solid lesions is difficult. If lesions located in the esophagus compress the esophageal lumen, precise definition of the outer wall of the esophagus remains difficult. Differences in CT values enable the characterization of different tissues. CT values vary from water density (0–20 HU) to high density (80–90 HU) according to the contents of the cyst, such as protein, calcium, and the presence of infection.39 However, cysts cannot be distinguished from tumor parenchyma based only on CT values. MRI is more sensitive and more accurate than CT regarding location and diagnosis. St-Georges et al reported that the diagnostic accuracy rate of preoperative CT is 69.2%, whereas the diagnostic accuracy of MRI is 100%.40 However, when a cyst is infected or contains protein or calcium, its density may be the same as that of a solid mass, which increases the diagnostic difficulty.14,41 EUS uses the esophagus as an acoustic medium to image the mediastinum layers. EUS has been proposed as a modality for the accurate diagnosis of esophageal cystic lesions. This technique identifies lesions of the esophagus and can distinguish between cystic and solid lesions by differentiating anechoic tissue from that containing dense hyperechoic debris in the intracystic space. EUS-FNA can provide a definitive diagnosis of mediastinal tumors; however, fluid cytology cannot be used to determine the origin of esophageal cysts. Furthermore, direct puncture of the cyst can induce infection, making the operation more difficult. Therefore, the universal application of EUS-FNA is not recommended for all BCs. In particular, the present case should not be recommended for EUS-FNA because of the probability of hemorrhage of the tumor. Gastroscopy is used to distinguish cysts from esophageal cancer by observing the integrity of the esophageal mucosa.
Surgical procedures such as video-assisted thoracoscopy (VATS), thoracotomy, and mediastinoscopy remain the standard treatment for esophageal BCs. A precise diagnosis is based on the surgical specimen, and even if patients are asymptomatic, precautions need to be taken against future complications such as infection, rupture, intracystic hemorrhage, and cancerization.27,42 Thoracoscopy is a mini-traumatic operation that is effective and as safe as open surgery. It can provide a good field of vision, allowing identification of both the vagal nerve and the phrenic nerve during dissection. In addition, the advantages of thoracoscopy include reducing postoperative pain, promoting patient recovery, and minimizing incisions. Thoracotomy is the conventional approach to remove an esophageal cyst, and it allows maintenance of the integrity of the esophageal mucosa. Despite certain disadvantages associated with postoperative recovery, thoracotomy would be preferred over thoracoscopy as the surgical approach for the present patient in case of potential intraoperative emergency because of the uncertainty of preoperative diagnosis.
Regardless of the preferred surgical approach, complete resection is necessary in cases of recurrence. However, if a cyst is severely adhered to vital tissues (such as the thoracic aorta or vena cava), complete resection is not possible and thoracoscopy can increase the surgical risk. Thoracotomy and total resection with de-epithelization may be a good option in these cases. The residual epithelial layer should be destroyed using toxic agents.43 In the present case, the adhesion to the distal esophagus and a huge hematoma of the gastric wall were resolved using thoracotomy. This technique enabled not only a reduction of the risk of mucosal damage, but also the simultaneous removal of the hematoma. The integrity of the mucosa was preserved, which was confirmed by injecting water into esophagus and stomach. Close observation and timely postoperative care provided further confirmation of the lack of perforation or putrescence of the esophageal and gastric mucosa.
Conclusion
Esophageal BC mimicking aortic dissection is extremely rare. The preoperative diagnosis is complicated and the definite diagnosis is based on pathological examination. Surgical excision is the principal treatment for this disease. The rare case of massive upper digestive tract hematoma and BC mimicking aortic dissection was safely removed without esophagectomy. The procedure described had a favorable outcome and is worthy of reference. To the best of our knowledge, no similar case has ever been reported previously in the currently available literature.
Disclosure
The authors report no conflicts of interest in this work.
References
Thaller P, Blanchet C, Badr M, Mesnage R, Leboucq N, Mondain M, Cambonie G. Neonatal respiratory distress syndrome revealing a cervical bronchogenic cyst: A case report. BMC Pediatr. 2015;15:72. | ||
Wang W, Ni Y, Zhang LW, et al. A case report of para-esophageal bronchogenic cyst with esophageal communication. J Cardiothorac Surg. 2012;7:94. | ||
Cioffi U, de Simone M. Should video-assisted surgery be the first-line approach for bronchogenic cysts? Asian Cardiovasc Thorac Ann. 2011;19(3/4):289. | ||
Vannucci J, Pecoriello R, Tassi V, Ceccarelli S, Puma F. Giant thoracoabdominal esophageal bronchogenic cyst. Dis Esophagus. 2013;26(3):340. | ||
Suda K, Sueyoshi R, Okawada M, Koga H, Lane GJ, Yamataka A, Doi T. Completely intramural bronchogenic cyst of the cervical esophagus in a neonate. Pediatr Surg Int. 2015;31:683–687. | ||
Maier HC. Bronchogenic cysts of the mediastinum. Ann Surg. 1948;127(3):476–502. | ||
Limaïem F, Ayadi-Kaddour A, Djilani H, Kilani T, El Mezni F. Pulmonary and mediastinal bronchogenic cysts: A clinicopathologic study of 33 cases. Lung. 2008;186(1):55–61. | ||
Ribet ME, Copin MC, Gosselin BH. Bronchogenic cysts of the lung. Ann Thorac Surg. 1996;61(6):1636–1640. | ||
Ribet ME, Copin MC, Gosselin BH. Bronchogenic cysts of the mediastinum. J Thorac Cardiovasc Surg. 1995;109(5):1003–1010. | ||
Sashiyama H, Miyazaki S, Okazaki Y, et al. Esophageal bronchogenic cyst successfully excised by endoscopic mucosal resection. Gastrointest Endosc. 2002;56(1):141–145. | ||
Hallani H, Eslick GD, Cox M, Wyatt JM, Lee CH. Chest pain? Cause. Lancet. 2004;363(9407):452. | ||
Westerterp M, van den Berg JG, van Lanschot JJ, Fockens P. Intramural bronchogenic cysts mimicking solid tumors. Endoscopy. 2004;36(12):1119–1122. | ||
Melo N, Pitman MB, Rattner DW. Bronchogenic cyst of the gastric fundus presenting as a gastrointestinal stromal tumor. J Laparoendosc Adv Surg Tech A. 2005;15(2):163–165. | ||
Pages ON, Rubin S, Baehrel B. Intra-esophageal rupture of a bronchogenic cyst. Interact Cardiovasc Thorac Surg. 2005;4(4):287–288. | ||
Ko SF, Hsieh MJ, Lin JW, Huang CC, Li CC, Cheung YC, Ng SH. Bronchogenic cyst of the esophagus: Clinical and imaging features of seven cases. Clin Imaging. 2006;30(5):309–314. | ||
Akutsu Y, Matsubara H, Hayashi H, Okazumi S, Aoki T, Kozu T, Ochiai T. Endoscope-assisted thoracoscopic technique for esophageal bronchogenic cyst which presented elevated CA 125. Dig Surg. 2006;23(4):209–214. | ||
Chuang KH, Huang TW, Cheng YL, et al. Esophageal bronchogenic cyst: A rare entity. Z Gastroenterol. 2007;45(9):958–960. | ||
Grover M, Gupta A, Wagner DP, Orringer MB. Hard to swallow. Am J Med. 2007;120(12):1023–1025. | ||
Turkyilmaz A, Eroglu A, Subasi M, Findik G. Intramural esophageal bronchogenic cysts: A review of the literature. Dis Esophagus. 2007;20(6):461–465. | ||
Eom DW, Kang GH, Kim HW, Ryu DS. Unusual bronchopulmonary foregut malformation associated with pericardial defect: bronchogenic cyst communicating with tubular esophageal duplication. J Korean Med Sci. 2007;22(3):564–567. | ||
Kiral H, Tezel CS, Kosar A, Keles M. Clinicopathologic demonstration of complex bronchopulmonary foregut malformation. Ann Thorac Surg. 2008;85(6):2114–2116. | ||
Rubin S, Sandu S, Durand E, Baehrel B. Diaphragmatic rupture during labour, two years after an intra-oesophageal rupture of a bronchogenic cyst treated by an omental wrapping. Interact Cardiovasc Thorac Surg. 2009;9(2):374–376. | ||
Diaz Nieto R, Naranjo Torres A, Gómez Alvarez M, et al. Intraabdominal bronchogenic cyst. J Gastrointest Surg. 2010;14(4):756–758. | ||
Chafik A, Benjelloun A, Qassif H, et al. Intramural esophageal bronchogenic cysts. Asian Cardiovasc Thorac Ann. 2011;19(1):69–71. | ||
Fernandez JL, Bauza G, McAneny DB. Minimally invasive management of lesser sac bronchogenic cyst. JSLS. 2011;15(4):571–574. | ||
Barbetakis N, Asteriou C, Kleontas A, Papadopoulou F, Tsilikas C. Video-assisted thoracoscopic resection of a bronchogenic esophageal cyst. J Minim Access Surg. 2011;7(4):249–252. | ||
Ghobakhlou M, Fatemi SR, Dezfouli AA, Tirgary F, Zali MR. Long-term dysphagia due to bronchogenic cyst of the esophagus. Endoscopy. 2012;44(Suppl.2):E129–E130. | ||
Ballehaninna UK, Shaw JP, Brichkov I. Subdiaphragmatic bronchogenic cyst at the gastroesophageal junction presenting with dysphagia: A case report. Surg Laparosc Endosc Percutan Tech. 2013;23(4):e170–e172. | ||
Tang X, Jiang B, Gong W. Endoscopic submucosal tunnel dissection of a bronchogenic esophageal cyst. Endoscopy. 2014;46(Suppl.1)E626–E627. | ||
Gou Y, Wang Y, Fang H, Xu X, Yu W, Zhang K, Yu Y. Bronchogenic cyst in the hepatogastric ligament masquerading as an esophageal mesenchymal tumor: A case report. Int J Clin Exp Pathol. 2015;8(11):15307–15311. | ||
Altieri MS, Zheng R, Pryor AD, Heimann A, Ahn S, Telem DA. Esophageal bronchogenic cyst and review of the literature. Surg Endosc. 2015;29(10):3010–3015. | ||
Trehan M, Singla S, Singh J, Garg N, Mahajan A. A rare case of intra-abdominal bronchogenic cyst – a case report. J Clin Diagn Res. 2015;9(11):PD03–PD04. | ||
Han C, Lin R, Yu J, et al. A case report of esophageal bronchogenic cyst and review of the literature with an emphasis on endoscopic ultrasonography appearance. Medicine (Baltimore). 2016;95(11):e3111. | ||
Lin JS, Yu YR, Chiou E, Chumpitazi BP, Schady D, Brandt ML. Intramural esophageal bronchogenic cyst mimicking achalasia in a toddler. Pediatr Surg Int. 2017;33(1):119–123. | ||
Liang JS, Yin GL, Zhang XM, Zhu SB, Dong YQ. Intramural esophageal bronchogenic cyst with wall hemorrhage results in acute esophageal obstruction: Report of two cases. Am Surg. 2014;80(1):E27–E29. | ||
Sun LH, Lu L, Fu WH, Li WD, Liu T. Gastric bronchogenic cyst presenting as a gastrointestinal stromal tumor. Int J Clin Exp Pathol. 2015;8(10):13606–13612. | ||
Randolph LK, Hopkins MK, Hopkins MP, Wasdahl DA. Hepatoid carcinoma of the ovary: A case report and review of the literature. Gynecologic Oncol Rep. 2015;13:64–67. | ||
Goto T, Maeshima A, Oyamada Y, Kato R. Esophageal cyst producing CA19-9 and CA125. Interact Cardiovasc Thorac Surg. 2010;10(3):448–450. | ||
Chang YC, Chen JS, Chang YL, et al. Video-assisted thoracoscopic excision of intradiaphragmatic bronchogenic cysts: Two cases. J Laparoendosc Adv Surg Tech A. 2006;16(5):489–492. | ||
St-Georges R, Deslauriers J, Duranceau A, et al. Clinical spectrum of bronchogenic cysts of the mediastinum and lung in the adult. Ann Thorac Surg. 1991;52(1):6–13. | ||
Lim LL, Ho KY, Goh PM. Preoperative diagnosis of a paraesophageal bronchogenic cyst using endosonography. Ann Thorac Surg. 2002;73(2):633–635. | ||
Hasegawa T, Murayama F, Endo S, Sohara Y. Recurrent bronchogenic cyst 15 years after incomplete excision. Interact Cardiovasc Thorac Surg. 2003;2(4):685–687. | ||
Kosar A, Tezel C, Orki A, Kiral H. Arman B. Bronchogenic cysts of the lung: Report of 29 cases. Heart Lung Circ. 2009;18(3):214–218. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.