Back to Journals » International Medical Case Reports Journal » Volume 8
Successful radiofrequency catheter ablation of atrioventricular nodal reentrant tachycardia in a patient with dextrocardia due to unilateral pulmonary agenesis: a case report
Authors Aksu T, Guler T, Golcuk E, Erden I, Ozcan K
Received 25 November 2014
Accepted for publication 16 December 2014
Published 2 February 2015 Volume 2015:8 Pages 41—45
DOI https://doi.org/10.2147/IMCRJ.S78095
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ronald Prineas
Tolga Aksu, Tumer Erdem Guler, Ebru Golcuk, Ismail Erden, Kazim Serhan Ozcan
Department of Cardiology, Kocaeli Derince Education and Research Hospital, Derince, Kocaeli, Turkey
Abstract: Radiofrequency catheter ablation of the slow pathway is considered to be the treatment of choice for patients with atrioventricular nodal reentrant tachycardia. We report a 34-year-old female with mirror image dextrocardia due to unilateral pulmonary agenesis who underwent successful slow pathway ablation for typical atrioventricular nodal reentrant tachycardia. Using contrast injection, cardiac anatomy was identified in a short time and successfully ablated.
Keywords: dextrocardia, AVNRT, ablation, pulmonary agenesis
Introduction
Radiofrequency ablation of the slow pathway is considered to be the gold standard treatment for patients with atrioventricular nodal reentrant tachycardia (AVNRT). Pulmonary artery agenesis is a rare anomaly that may occur during embryological development of the heart.1 This agenesis may be accompanied by a complete or partial absence of the lung. In the great majority of the cases, the diagnosis is usually made at or soon after birth and it can be associated with multiple anomalies. However, extremely rare cases which have unilateral agenesis may survive until adulthood. An otherwise normal heart may be displaced into the right hemithorax by unilateral pulmonary agenesis.2
Catheter ablation of AVNRT in the setting of dextrocardia is potentially challenging. There have been a few reports of successful slow pathway ablation in patients with dextrocardia, but usually at the expense of prolonged procedure and fluoroscopy times.3–7 To our knowledge, slow pathway ablation of a patient with dextrocardia due to pulmonary agenesis has not been described to date.
Case report
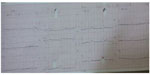
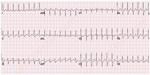
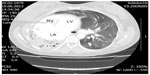
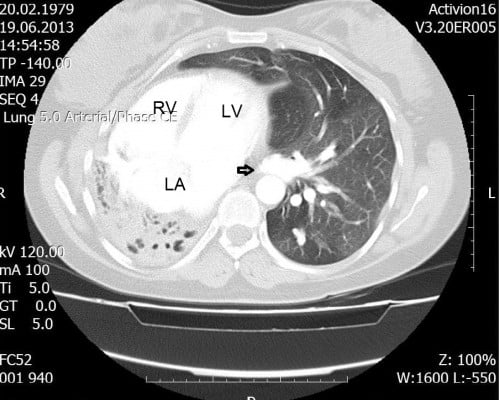
A 35-year-old woman was admitted to our cardiology clinic with complaints of palpitation. She had a 7-year history of paroxysmal palpitations. The tachycardia, at a rate of 170–200 bpm, was associated with dyspnea and chest tightness and lasted for up to 60 minutes at a time. Her physical examination revealed no breathing sound on the right and a normal breathing sound on the left hemithorax, while the heart’s sound was heard from the right hemithorax. The chest radiogram incidentally showed a rudimentary, opacified right hemithorax with mediastinal shift and herniation of the contralateral lung. Except for dextroposition of the heart, two-dimensional echocardiogram showed no underlying heart disease. A 12-lead resting electrocardiogram (ECG) showed sinus rhythm at a rate of 64 bpm, a positive QRS complex in aVR, a positive P wave in aVR and a biphasic P wave in aVL, and a prominent R wave in V1 with undetermined horizontal axis (Figure 1). The clinical tachycardia was a regular, narrow QRS tachycardia at a rate of 170 bpm, during which the P wave was indiscernible (Figure 2). Computed tomography showed a complete absence of the right lung with dextroposition of the heart (Figure 3). Electrophysiological study was performed after obtaining written informed consent and discontinuation of all drugs for five half-lives.
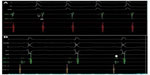
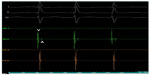
The right atrium and the right ventricle were imaged on left lateral and anteroposterior views by contrast injection through inferior vena cava to reveal the cardiac anatomy (Figure 4). Then, a 4 mm-tipped ablation catheter (Cordis Webster, Baldwin Park, CA, USA) was advanced through the 6F sheath into the right atrium and a four-pole fixed-curve diagnostic catheter was placed on the bundle of His. During the electrophysiological study, a narrow QRS tachycardia with a cycle length of 270 ms was reproducibly induced by atrial premature stimulation with an extrastimulus (Figure 5A). The earliest atrial activation was recorded from the His bundle region. The ventriculoatrial interval, measured from the onset of ventricular activation on the surface ECG to the earliest deflection of the atrial activation in the His bundle electrogram, was <60 ms, and the diagnosis of typical slow–fast AVNRT was confirmed. The tachycardia could be terminated by atrial burst pacing. The atrioventricular node effective refractory time before ablation was 240 ms.
  | Figure 4 Fluoroscopic view in the 15° anteroposterior projection after contrast injection. |
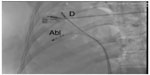
His bundle records were taken with an electroanatomical approach. Then, slow pathway potentials were recorded (Figure 5B). Fluoroscopic views of the catheters during the ablation and mapping of the coronary sinus ostium are shown in Figures 6 and 7. During ablation, the radiofrequency energy was adjusted to obtain a catheter tip temperature between 50°C and 60°C. The current was applied for 60 seconds after junctional tachycardia was observed during ablation (Figure 8). ventriculoatrial block was not revealed during radiofrequency application.
After the ablation, there was no residual slow pathway conduction and the clinical arrhythmia was not inducible without or with infusion of isoproterenol. The total procedure time was 30 minutes with a total radiofrequency delivery time of 8 minutes and fluoroscopy time of only 6 minutes. No complication occurred after the procedure and the patient was free of symptoms at 1-year follow-up.
Discussion
Dextrocardia or complex cardiac anatomy of the heart may be challenging to electrophysiologists during catheter ablation procedures. There have been only a few case reports of catheter ablation of supraventricular tachyarrhythmias in patients with dextrocardia.3–7
Pulmonary agenesis can be localized to a single lobe and it can affect an entire lung or, in rare cases, both lungs.1 Although a majority of patients with unilateral agenesis die soon after birth or in early childhood, in some extreme cases, patients can survive up to adulthood, like our patient. Three types of dextrocardia, are described in the literature.2 These are as follows. 1) Dextrocardia with situs inversus. The most common and familiar form is the “mirror image of normal” or mirror-image dextrocardia, in which the migration of the apex of the L-bulboventricular loop is into the right hemithorax, as expected. The anterior–posterior relationship of the various parts of the heart is normal, but their right-to-left orientation is reversed. 2) Dextrocardia with situs solitus. The second most common type is dextroversion, in which the heart appears to be rotated into the right hemithorax relative to its normal position. 3) The third type of dextrocardia is dextroposition, in which an otherwise normal heart is displaced or shifted into the right hemithorax by other extracardiac causes such as agenesis or fibrosis of the right lung. Our case was a type 3 dextrocardia.
To recognize the ECG findings of dextrocardia necessitates a clear understanding of the electrical axis. Global negativity in lead I, a positively deflected QRS complex in aVR, and right axis deviation and reverse R wave progression in precordial leads are the mean ECG findings in dextrocardia. The finding of a positively deflected QRS complex in aVR and a negatively deflected ORS complex in lead I may lead to misdiagnosis. The most common cause for this finding is reversed electrode placement of arm leads (left and right).
During a standard catheter ablation procedure, the catheter is targeted to the ablation region based on typical local electrogram characteristics (slow-pathway potentials) and anatomical landmarks. In contrast to patients with normal anatomy, localization of the slow pathway and stabilization of the catheter are more difficult in patients with dextrocardia. Therefore, the time spent in the catheterization laboratory and the fluoroscopy time are markedly longer than in cases with normal hearts.
Our case illustrates the role of imaging by contrast injection in a patient with dextrocardia. The identification of the accurate anatomy using imaging modalities such as computerized tomography and magnetic resonance imaging and three-dimensional image reconstruction using mapping systems may be useful not only to help the electrophysiologist, enabling a safe and successful catheter ablation procedure, but also to understand the complex anatomical structures and to guide optimal catheter access. However, these modalities are quite expensive and are not easily accessible in every unit. Before the ablation procedure, we easily localized the coronary sinus ostium and His bundle position. As such, we did not need an additional catheter in this ablation procedure, but an additional catheter might further facilitate the procedure.
We are presenting this pulmonary agenesis case in view of its atypical presentation in adulthood, which is exceptionally rare. As demonstrated in this case, revealing cardiac anatomy by contrast injection or other imaging modalities may shorten the procedure time and decrease the complication rates.
Disclosure
The authors report no conflicts of interest in this work.
References
Kravitz RM. Congenital malformations of the lung. Pediatr Clin North Am. 1994;41(3):453–472. | |
Maldjian PD, Saric M. Approach to dextrocardia in adults: review. AJR Am J Roentgenol. 2007;188(6 Suppl):S39–S49; quiz S35–S38. | |
Abe H, Araki M, Nagatomo T, Miura Y, Nakashima Y. Radiofrequency catheter ablation of an accessory pathway in dextrocardia. Pacing Clin Electrophysiol. 1997;20(9 Pt 1):2284–2285. | |
Wu TJ, Chen SA, Chiang CE, et al. Radiofrequency catheter ablation of sustained intraatrial reentrant tachycardia in a patient with mirror-image dextrocardia. J Cardiovasc Electrophysiol. 1994;5(9):790–794. | |
Hatala R, Weiss C, Koschyk DH, Siebels J, Cappato R, Kuck KH. Radiofrequency catheter ablation of left atrial tachycardia originating within the pulmonary vein in a patient with dextrocardia. Pacing Clin Electrophysiol. 1996;19(6):999–1002. | |
Reithmann C, Hoffmann E, Dorwarth U, Remp T, Steinbeck G. Slow pathway ablation in a patient with common AV nodal reentrant tachycardia and complete situs inversus. Europace. 1999;1(4):283–285. | |
Aizawa Y, Takatsuki S, Kimura T, et al. Successful radiofrequency catheter ablation of atrioventricular nodal reentrant tachycardia in a patient with dextrocardia and situs inversus. Herz. 2013;38(1):102–104. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.