Back to Journals » Neuropsychiatric Disease and Treatment » Volume 14
Structural equation modeling approach between salience network dysfunction, depressed mood, and subjective quality of life in schizophrenia: an ICA resting-state fMRI study
Authors Ohta M , Nakataki M , Takeda T , Numata S, Tominaga T, Kameoka N, Kubo H, Kinoshita M, Matsuura K, Otomo M , Takeichi N, Harada M, Ohmori T
Received 19 January 2018
Accepted for publication 17 April 2018
Published 15 June 2018 Volume 2018:14 Pages 1585—1597
DOI https://doi.org/10.2147/NDT.S163132
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Taro Kishi
Masashi Ohta,1 Masahito Nakataki,1 Tomoya Takeda,1 Shusuke Numata,1 Takeo Tominaga,1 Naomi Kameoka,2 Hiroko Kubo,1 Makoto Kinoshita,1 Kanae Matsuura,2 Maki Otomo,3 Naoya Takeichi,4 Masafumi Harada,3 Tetsuro Ohmori1
1Department of Psychiatry, Graduate School of Biomedical Sciences, Tokushima University, Tokushima, Japan; 2Department of Psychiatry, Tokushima University Hospital, Tokushima, Japan; 3Department of Radiology and Radiation Oncology, Graduate School of Biomedical Sciences, Tokushima University, Tokushima, Japan; 4Department of Radiology, Tokushima University Hospital, Tokushima, Japan
Purpose: Quality of life (QOL) is an important clinical outcome for patients with schizophrenia, and recent studies have focused on subjective QOL. We evaluated the causal relationship between psychosocial aspect of subjective QOL, symptoms, cognitive functions, and salience network (SN) dysfunction in schizophrenia using structural equation modeling (SEM).
Patients and methods: We performed a cross-sectional study of 21 patients with symptomatically stabilized schizophrenia and 21 age-, sex-, and education level-matched healthy controls who underwent resting-state functional magnetic resonance imaging. We evaluated SN dysfunction in schizophrenia using independent component analysis (ICA). We rated participant psychopathology using the Positive and Negative Syndrome Scale (PANSS), the Brief Assessment of Cognition in Schizophrenia (BACS), and the Calgary Depression Scale for Schizophrenia (CDSS). We rated psychosocial aspect of subjective QOL using the Schizophrenia Quality of Life Scale (SQLS) psychosocial subscale. We applied SEM to examine the relationships between SN dysfunction, PANSS positive and negative scores, CDSS total scores, BACS composite scores, and SQLS psychosocial subscale scores.
Results: In second-level analysis after group ICA, patient group had significant lower right pallidum functional connectivity (FC) within the SN than the controls did (Montreal Neurological Institute [MNI] [x y z] = [22 -2 -6]) (p = 0.027, family-wise error [FWE] corrected). In SEM, we obtained a good fit for an SEM model in which SN dysfunction causes depressed mood, which in turn determines psychosocial aspect of subjective QOL (chi-squared p = 0.9, root mean square error of approximation (RMSEA) < 0.001, comparative fit index [CFI] = 1.00, and standardized root mean square residual [SRMR]= 0.020).
Conclusion: We found a continuous process by which SN dysfunction causes depressed moods that determine psychosocial aspect of subjective QOL in schizophrenia. This is the first report that offers a unified explanation of functional neuroimaging, symptoms, and outcomes. Future studies combining neuroimaging techniques and clinical assessments would elucidate schizophrenia’s pathogenesis.
Keywords: depressed mood, salience network, schizophrenia, structural equation modeling, subjective quality of life, resting-state fMRI
Introduction
Living in the community is thought to contribute to quality of life (QOL).1 Most patients commonly have the deficit of functional outcome,2,3 which is defined as a wide range of real-world functions including independent living, financial management, employment, and leisure/social activities,4 persistently. QOL is therefore an important clinical outcome for patients with schizophrenia.5
QOL in patients with schizophrenia has been measured subjectively and objectively. Objective QOL includes indicators of health and living conditions, sociodemographic items, and a functioning role in society, whereas subjective QOL includes indicators of general and domain-specific life satisfaction.6 Because patients exhibit cognitive dysfunction,7 their subjective QOL has historically been considered unreliable,8 but it is currently contended that symptomatically stable patients can assess their own QOL.9 However, researchers should note that objective and subjective QOLs can differ due their potentially different influencing factors.10,11
Subjective QOL for patients with schizophrenia is associated with multiple factors including depressed mood,12 positive symptoms,13 negative symptoms,14,15 neuroleptic doses,10 and cognitive function,16 but studies have reported conflicting results, such as QOL being positively correlated with cognitive function17,18 or unrelated to it.13,19,20 This inconsistency suggests spurious correlations or asymmetric relationships between factors associated with subjective QOL,21 such that one factor affects others, or several factors reciprocally affect each other. A causal interpretation of cross-sectional data is needed prior to any randomized experiments mechanistically examining subjective QOL. Understanding the neural basis of patients’ behavior is particularly important for optimizing future experiments.
In recent years, magnetic resonance imaging (MRI) has been used to elucidate the pathology of schizophrenia. Resting-state functional MRI (rsfMRI) studies have reported associations between resting-state network (RSN) abnormalities and psychiatric symptoms.22–25 Previous studies have focused on dysfunctions in three important RSNs: the default mode network (DMN), the central executive network (CEN), and the salience network (SN).26 Specifically, the SN, which includes the frontoinsular cortex and anterior cingulate cortex,27 plays a central role among them. The SN identifies salient stimuli, regulates the DMN and CEN related to subsequent processing, and forms emotions.28,29 Therefore, SN dysfunction has been proposed as a crucial mechanism for several schizophrenia symptoms30 and is related to depressed mood, positive and negative symptoms, and cognitive function in psychiatric diseases.31 This suggests that it affects subjective QOL, but the exact interrelationships between SN dysfunction, subjective QOL, depressed mood, positive symptoms, negative symptoms, and cognitive function remain unclear, as do the relative contributions of each of these determinants.
We conducted a cross-sectional study to evaluate the relationship between subjective QOL, symptoms, cognitive functions, and SN connectivity of rsfMRI in symptomatically stabilized patients with schizophrenia. In evaluating subjective QOL, we focused on psychosocial aspect of that because it includes various emotional problems such as solitude, depressed mood, and despair that markedly influence overall subjective QOL.32 And we evaluated SN dysfunction in schizophrenia using resting-state fMRI with combined group independent component analysis (ICA) for inclusion of SN connectivity in the following path analyses. Our aim was to determine the causal interpretation between psychosocial aspect of subjective QOL, symptoms, cognitive functions, and SN dysfunction in schizophrenia. To understand psychosocial aspect of subjective QOL clearly, we tried to find the biological markers which affect the symptoms and the outcomes in schizophrenia. Here, we focused on brain function as a biological marker. In patients with schizophrenia, we expected a continuous process that SN, which plays a central role in RSNs, has decreased functional connectivity (FC), causes symptoms, and concludes psychosocial aspect of subjective QOL. We used structural equation modeling (SEM) to test a theory involving non-straightforward relationships between them. The SEM is a useful statistical method for the specification of directional influences among variables in cross-sectional studies,33 and be widely used in test of causal assumption based on cross-sectional data of psychiatric disorders.34–36 In particular, we modeled several causal assumptions based on the existing evidence, tested our data-set using SEM, and determined the best model.
Patients and methods
Participants
This study was conducted through the Department of Psychiatry at the Tokushima University Graduate School between June 2016 and December 2016. All subjects were recruited form Tokushima University Hospital, Japan, and gave written informed consent, and the study was approved by the Tokushima University ethics committee and performed in accordance with the principles of the Declaration of Helsinki.
We enrolled 21 patients and 21 age-, sex-, and education level-matched, healthy control subjects. The patients had been diagnosed with schizophrenia based on the criteria in the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. At the time enrolment, all patients were clinically stable, as judged by a therapeutic psychiatrist. The criteria for determining clinical stability were: no schedule to change treatment contents; essentially no psychopathological changes; judgment of clinically stable by a therapeutic psychiatrist and patients themselves. We performed psychopathological assessments on each patient before and after the study, and we observed no major changes. Each patient’s clinical stability was assessed from medical records, self-reports, and the observations of psychiatric staff and relatives. The exclusion criteria were a past history or presence of any serious disorders affecting the brain or cognitive functioning, such as epilepsy, serious head injury, or brain tumor; alcohol abuse; active drug use in the past year; or pregnancy or intention to become pregnant during the study period.
The healthy control subjects were evaluated with structured clinical interviews to confirm the absence of schizophrenia and had no history of neurological or psychiatric disorders or any first-degree relatives with psychotic episodes. The demographic and clinical data of two groups are shown in Table 1. Significant difference between two groups was found in IQ (Student’s t-test, p < 0.001) rated by the Wechsler Adult Intelligence Scale-III short form.37,38
  | Table 1 Demographic and clinical parameters of the healthy control and patient groups |
Clinical assessments
Psychopathology was rated using the Positive and Negative Syndrome Scale (PANSS).39 Cognitive function was rated using the Brief Assessment of Cognition in Schizophrenia (BACS).40,41 Depressed mood was rated using the Calgary Depression Scale for Schizophrenia (CDSS).42,43 Psychosocial aspect of subjective QOL was rated using the Schizophrenia Quality of Life Scale (SQLS) psychosocial subscale32,44 (Table 1). The SQLS is a self-reported, 30-item questionnaire for measuring QOL specific to patients with schizophrenia with good reliability and validity. It is composed of three scales: psychosocial, motivation/energy, and symptoms/side effects. Lower scores indicate higher levels of subjective QOL.32,44 As motivation/energy subscale and symptoms/side-effects subscale did not differ significantly between the two groups, we did not consider them for following analyses.
Image acquisition and pre-processing
All subjects underwent rsfMRI scanning while lying down with their eyes closed during resting wakefulness. We placed foam pads to prevent head movement during scanning. No stimuli were presented during scanning. For each subject, the rsfMRI scan lasted for 10 minutes.
We obtained a series of MRI scans on a 3-T Discovery MR750 device (GE Medical Systems, Chicago, IL, USA) with a 24-channel phased-array head coil at the Tokushima University Hospital Department of Radiology. Structural images were obtained using a three-dimensional axial T1-weighted fast spoiled gradient-recalled-echo (GRE) sequence (echo time = 3.03 ms, retention time = 6.93 ms, flip angle = 11°, matrix = 512 × 512, voxel size = 0.46 × 0.46 mm2, slice thickness = 1.2 mm, inter-slice gap = 0 mm, number of slices = 392). At the same locations as those of the anatomical slices, functional images were collected in runs using a T2*-weighted GRE planar imaging sequence (echo time = 30 ms, retention time = 2,500 ms, flip angle = 80°, matrix = 64 × 64, slice thickness = 3.2 mm, number of slices = 40) sensitive to blood oxygenation level-dependent contrast. We acquired one structural T1-weighted volume and 240 functional T2*-weighted volumes.
After a quality check, both structural and functional images were processed using SPM8 (Wellcome Trust Centre for Neuroimaging, London, UK; http://www.fil.ion.ucl.ac.uk/spm/). The slice time-corrected functional data were realigned and registered to their mean by correcting for head motion using a six-parameter rigid-body transformation. All subjects did not have excessive head motion (>2.0 mm translation and/or > 2.0° rotation) during scan. Furthermore, we also compared the differences of four head motion metrics (Mean Motion, Maximum Motion, Number of Movements, and Rotation),45 which were calculated from the translation and rotation parameters from the rigid body correction, between groups. There were no significant differences in these head motion metrics between patients and controls. Therefore, the imaging data of all subjects were taking into consideration in further analyses. The realigned images were indirectly normalized to Montreal Neurological Institute (MNI) space. The resulting data were smoothed with an isotropic 8 mm full-width at half-maximum Gaussian kernel.
Group ICA
Pre-processed images were analyzed with group ICA using the Group ICA fMRI Toolbox (GIFT; http://mialab.mrn.org/software/gift/index.html)46,47 implemented in Matlab (MathWorks, Natick, MA, USA). For computational feasibility, the previously concatenated data from all subjects were reduced using principal component analysis, after which ICA was performed with the infomax algorithm.48 This identified a spatial map of 20 consistent spatially independent components for each subject that was reconstructed using group ICA and converted to z-values. To identify the component that represented the SN, the components were sorted in GIFT using cross-voxel correlation with an SN template from Shirer et al (http://findlab.stanford.edu/functional_ROIs.htm).49 The component with the highest spatial correlation with the SN template was selected as the SN. Subject-specific SN components were then entered into second-level analysis in SPM8.
Model specification
In order to apply SEM, we developed the hypotheses based on previous studies as follows. SN dysfunction has been reported to be related to depressed mood, positive and negative symptoms, and cognitive function in psychiatric disease.31 Subjective QOL has been reported to be most related to depressed mood in schizophrenia.50 It has also been reported that subjective QOL is related to positive and negative symptoms in schizophrenia.15 It has been reported that subjective QOL is related to negative symptoms rather than positive symptoms.14 In contrast, it has been reported that subjective QOL is related to positive symptoms rather than negative symptoms.13 Furthermore, subjective QOL has been reported to be related to cognitive function in schizophrenia.16
By considering these previous studies, we proposed that symptoms (positive, negative, or depressed mood) or cognitive function mediate the relationship between SN dysfunction and psychosocial aspect of subjective QOL. Based on our hypotheses, we made the following models (Figure S1). To examine the mediation effect of symptoms and cognitive function between SN dysfunction and psychosocial aspect of subjective QOL, we tested the coefficient for direct and indirect relationship between SN dysfunction and psychosocial aspect of subjective QOL in models 5–8.
Model 1: SN dysfunction influences psychosocial aspect of subjective QOL indirectly via positive symptoms.13,15,31
Model 2: SN dysfunction influences psychosocial aspect of subjective QOL indirectly via negative symptoms.14,15,31
Model 3: SN dysfunction influences psychosocial aspect of subjective QOL indirectly via depressed mood.31,50
Model 4: SN dysfunction influences psychosocial aspect of subjective QOL indirectly via cognitive function.16,31
Model 5: SN dysfunction influences psychosocial aspect of subjective QOL, both directly and indirectly via positive symptoms.
Model 6: SN dysfunction influences psychosocial aspect of subjective QOL, both directly and indirectly via negative symptoms.
Model 7: SN dysfunction influences psychosocial aspect of subjective QOL, both directly and indirectly via depressed mood.
Model 8: SN dysfunction influences psychosocial aspect of subjective QOL, both directly and indirectly via cognitive function.
Statistical analyses
We compared subject-specific SN component maps in the patient and control groups using two-sample t-tests. Z-values of these maps were used to calculate FC strength. We used voxel- and cluster-level family-wise error (FWE) corrections to guard against type I errors. And then, mean FC z-values were extracted from the region with significant differences between two groups for purposes of following path analyses.
Depending on whether the Shapiro–Wilk test indicated normal or non-normal distributions, we used Pearson’s or Spearman’s correlation coefficients to analyze correlations between SN dysfunction, positive symptoms, negative symptoms, depressed mood, cognitive function, and psychosocial aspect of subjective QOL. In addition, correlation analyses between chlorpromazine-equivalent antipsychotic doses and experimental variables, Drug Induced Extra-Pyramidal Symptoms Scale (DIEPSS) and experimental variables were conducted to assess the effects of medications and side effects. These analyses were performed in SPSS version 22 (IBM, Armonk, NY, USA). All tests were two-sided, and Bonferroni corrections were applied to maintain an overall type I error rate of 0.05.
SEM was then conducted using lavaan51 and semTools52 packages for R (R Project for Statistical Computing, Vienna, Austria). The model parameters were determined through re-sampling method by bootstrapping (n = 1,000). We evaluated the model fit using the chi-squared statistic (p ≥ 0.05), the root mean square error of approximation (RMSEA < 0.05), the comparative fit index (CFI ≥ 0.95), and the standardized root mean square residuals (SRMR ≤ 0.08).53 The path coefficient’s strength was defined as weak (r < 0.2), moderate (r = 0.2–0.5), or strong (r > 0.5), and significance was defined as p < 0.05.54 Correlation analyses and SEM were performed with ICA results, PANSS positive and negative scores, CDSS total scores, BACS composite scores, and SQLS psychosocial subscale scores. A composite score was calculated by averaging all of the six standardized primary measures from the BACS.41
Results
Group ICA
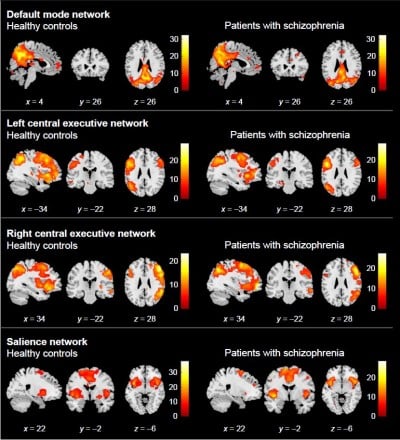
Using a group ICA approach, we identified a consistent SN component in all subjects. Figure 1 shows the two groups’ SN spatial patterns (FWE-corrected one-sample t-test: p < 0.05). The groups had similar SN expressions, but the patients had significant lower right pallidum FCs within the SN than the controls did (MNI [x y z] = [22 −2 −6]) (p = 0.027, FWE-corrected) (Table 2, Figure 2A and B). For purposes of SEM, mean FC z-values were extracted from the right pallidum cluster.
  | Figure 1 The ICA analysis results. |
Correlations between variables
The correlation analysis results are shown in Table 3. We found significant correlations between SQLS psychosocial subscale scores and CDSS total scores (Spearman’s r = 0.785, p < 0.001), and between PANSS positive and negative scores (Pearson’s r = 0.746, p < 0.005). There were no significant correlations between antipsychotic doses and any experimental variables nor DIEPSS and any experimental variables.
SEM
We conducted the SEM on eight models (Figure S1). Table 4 presents their fit measures. Model 3 provided a good fit (chi-squared p = 0.9, RMSEA < 0.001, CFI = 1.00, and SRMR = 0.020) that was better than that of the other models, as indicated by its smaller Akaike’s and Bayesian information criteria (Table 4). We did not find direct effect from right pallidum FC to SQLS psychosocial subscale scores because direct path between them had no significant association in Models 5–8. Only Models 3 and 7 had significant and moderate-to-strong associations between right pallidum FC and CDSS total scores (path coefficient = −0.43, p = 0.02; path coefficient = −0.43, p = 0.02) and between CDSS total scores and SQLS psychosocial subscale scores (path coefficient = 0.76, p < 0.001; path coefficient = 0.79, p < 0.001), which are common in both models. Direct path between right pallidum FC and SQLS psychosocial subscale scores may be removable. Model 1 was the second best model (chi-squared p = 0.49, RMSEA = 0.017, CFI = 1.00, and SRMR = 0.082); however, all of their paths were not statistically significant. From abovementioned findings, we concluded that Model 3 is the best model (Figure 3).
Discussion
Our goal was to explore the relationship between SN dysfunction, symptoms, and QOL in patients with schizophrenia. We hypothesized that symptoms mediate the relationship between SN dysfunction and QOL. Through SEM, we identified a causal relationship confirming this hypothesis.
The SN is thought to be a central RSN system that enables transitions between various brain states. It is therefore described as the core region in emotional regulation and cognitive processing for various stimuli.28,29 SN dysfunction is related to depressed mood, positive and negative symptoms, and cognitive function in psychiatric diseases.31 Furthermore, it has been proposed as a mechanism underlying several core symptoms of schizophrenia.30 Our study is the first rsfMRI study to find an association between SN dysfunction and depressed mood in schizophrenia. This finding is important for understanding the SN’s role in schizophrenia-associated psychosocial dysfunction and suggests that treating SN dysfunction could improve psychosocial functioning.
The patients exhibited reduced SN connectivity in the pallidum, an SN-associated basal ganglion located deep beneath the cerebral cortex.55–57 Previous studies have reported that pallidum activity is related to depressive symptoms in schizophrenia58,59 and that pallidum structural abnormalities occur in schizophrenia.60,61 The pallidum is thought to be related to motor sensation, motivation, and desire.62 Our patients exhibited a cluster with significantly decreased FC that was mainly located in the ventral pallidum, which plays a particularly important role in processing and executing motivated behavior.63,64 This motivational system is related to depressive symptoms.65 In schizophrenia, motivational deficits are associated with depressed mood that is difficult to distinguish from negative symptoms66 or medication-induced extrapyramidal symptoms (EPS).67 However, the CDSS evaluates depressed mood in schizophrenia independently of EPS or negative symptoms.68,69 Consistently, we found no significant correlations between CDSS and DIEPSS and PANSS negative scores. Our finding provides further evidence that SN dysfunction in schizophrenia causes depressive symptoms independent of EPS or negative symptoms.
We also found a strong association between psychosocial aspect of subjective QOL and depressed mood. Previous studies suggested that subjective QOL is more relevant to depressed mood than are positive symptoms, negative symptoms, or cognitive function.11,50,70 Given that we detected no significant causal roles for positive symptoms, negative symptoms, or cognitive function, our findings are consistent with these previous studies. However, the association of positive symptoms, negative symptoms, and cognitive function with SN dysfunction and subjective QOL have been reported in previous studies.13–16,31 Our patients were symptomatically stabilized, so they did not exhibit conspicuous positive symptoms, negative symptoms, or cognitive dysfunction, which may account for the discrepancy with the previous study. An association of depressed mood with psychosocial aspect of subjective QOL may only be seen in symptomatically stabilized patients. Furthermore, such an inconsistency might be due to the measurement of several different aspects of subjective QOL in these previous studies. SQLS is composed of three subscales: psychosocial, motivation/energy, and symptoms/side effects. The motivation/energy subscale measures various problems of motivation and activity.32 The relationship between neurocognition and motivation has been reported in previous study.71 Therefore, cognitive function may affect the motivation/energy subscale. The symptoms/side-effects subscale measures such issues as sleep disturbance, blurred vision, dizziness, muscle twitches, and dry mouth.32 Patients with severe positive and/or negative symptoms may suffer from sleep disturbance and may use large amount of medication, which can cause more side-effects. Further studies involving various aspects of subjective QOL are needed in symptomatically diverse patients.
This is the first report to present a unified explanation of functional neuroimaging, symptoms, and outcomes in schizophrenia. Combining neuroimaging findings and clinical assessments provided better explanations for the mechanisms underlying psychosocial aspect of subjective QOL than were possible with clinical assessments alone. Through SEM, we found that decreased pallidum FC causes depressed moods that worsen psychosocial aspect of subjective QOL in schizophrenia. Our findings suggest that QOL may be improved by targeting depressed moods72,73 and the SN reward system30 through approaches such as cognitive behavioral therapy or pharmacotherapy.
This study has several limitations. First, all the patients were taking medications, so we could not exclude medication effects. However, we found no significant correlations between antipsychotic doses and any experimental variable, so we doubt that medications had an effect.
Second, we could not exclude the possibility that other networks, such as DMN and CEN, affected depressed mood and psychosocial aspect of subjective QOL. However, on additional group comparison between controls and patients, we could not find any significant group differences in DMN nor CEN (Figure S2). This suggests that SN may affect depressed mood more than DMN or CEN in symptomatically stabilized patients with schizophrenia.
Third, this was a cross-sectional study, not a prospective one. These findings derived from cross-sectional data should be confirmed by further large-scale and longitudinal research.
Fourth, psychosocial aspect of subjective QOL can be affected by socioeconomic factors such as support services, living environment, and frequency of contact with friends and family.74 These aspects could not be controlled in our study. Further studies are necessary to explore the influence of such factors.
Finally, the relatively small sample size may limit the interpretation of our findings. Studies with larger sample sizes are needed to replicate our findings.
Conclusion
We indicated a continuous process by which SN dysfunction causes depressed moods that determine psychosocial aspect of subjective QOL in schizophrenia. Depressed mood is a common symptom of schizophrenia, and we suggested that focusing on depressed mood is important for improving patients’ QOL. We think that our findings will be one of the scientific evidences leading to future studies. Combining neuroimaging techniques with clinical assessments would improve future studies into the pathogenesis of schizophrenia.
Acknowledgments
This work was supported in part by Grants-in-Aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science and Technology (TO). The funding source had no role in study design, data collection, data analysis, data interpretation, or writing of the report and the decision to submit it for publication.
We would like to thank the staff at Tokushima University Hospital for support with fMRI data acquisition. We would like to thank Editage (www.editage.jp) for English language editing. We also thank the participants for their involvement and dedication to this study.
Author contributions
M Ohta and M Nakataki designed the study. M Nakataki and T Ohmori managed the research project. M Ohta, M Nakataki, S Numata, T Tominaga, N Kameoka, H Kubo, and M Kinoshita recruited patients and discussed the results. T Takeda and K Matsuura conducted psychological examinations. M Otomo, N Takeichi, and M Harada performed fMRI. M Ohta and T Takeda organized the data. M Ohta, M Nakataki, and T Takeda searched the literature. M Ohta analyzed the data and wrote the first draft of this manuscript. M Nakataki and T Ohmori supervised data analysis and revised the manuscript. All authors read and approved the final manuscript. All authors contributed toward data analysis, drafting, and revising the paper and agreed to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
The World Health Organization Quality of Life assessment (WHOQOL): position paper from the World Health Organization. Soc Sci Med. 1995;41(10):1403–1409. | ||
Wiersma D, Wanderling J, Dragomirecka E, et al. Social disability in schizophrenia: its development and prediction over 15 years in incidence cohorts in six European centres. Psychol Med. 2000;30(5):1155–1167. | ||
Hegarty JD, Baldessarini RJ, Tohen M, Waternaux C, Oepen G. One hundred years of schizophrenia: a meta-analysis of the outcome literature. Am J Psychiatry. 1994;151(10):1409–1416. | ||
Harvey PD, Bellack AS. Toward a terminology for functional recovery in schizophrenia: is functional remission a viable concept? Schizophr Bull. 2009;35(2):300–306. | ||
Hofer A, Baumgartner S, Edlinger M, et al. Patient outcomes in schizophrenia I: correlates with sociodemographic variables, psychopathology, and side effects. Eur Psychiatry J Assoc Eur Psychiatr. 2005;20(5–6):386–394. | ||
Fitzgerald PB, Williams CL, Corteling N, et al. Subject and observer-rated quality of life in schizophrenia. Acta Psychiatr Scand. 2001;103(5):387–392. | ||
Doyle M, Flanagan S, Browne S, et al. Subjective and external assessments of quality of life in schizophrenia: relationship to insight. Acta Psychiatr Scand. 1999;99(6):466–472. | ||
Awad AG, Voruganti LP. Cost-utility analysis in schizophrenia. J Clin Psychiatry. 1999;60(Suppl 3):22–26; discussion 27–28. | ||
Voruganti L, Heslegrave R, Awad AG, Seeman MV. Quality of life measurement in schizophrenia: reconciling the quest for subjectivity with the question of reliability. Psychol Med. 1998;28(1):165–172. | ||
Tomotake M, Kaneda Y, Iga J-I, et al. Subjective and objective measures of quality of life have different predictors for people with schizophrenia. Psychol Rep. 2006;99(2):477–487. | ||
Hayhurst KP, Massie JA, Dunn G, Lewis SW, Drake RJ. Validity of subjective versus objective quality of life assessment in people with schizophrenia. BMC Psychiatry. 2014;14(1):4. | ||
Aki H, Tomotake M, Kaneda Y, et al. Subjective and objective quality of life, levels of life skills, and their clinical determinants in outpatients with schizophrenia. Psychiatry Res. 2008;158(1):19–25. | ||
Chino B, Nemoto T, Fujii C, Mizuno M. Subjective assessments of the quality of life, well-being and self-efficacy in patients with schizophrenia. Psychiatry Clin Neurosci. 2009;63(4):521–528. | ||
Packer S, Husted J, Cohen S, Tomlinson G. Psychopathology and quality of life in schizophrenia. J Psychiatry Neurosci. 1997;22(4):231–234. | ||
Yamauchi K, Aki H, Tomotake M, et al. Predictors of subjective and objective quality of life in outpatients with schizophrenia. Psychiatry Clin Neurosci. 2008;62(4):404–411. | ||
Woon PS, Chia MY, Chan WY, Sim K. Neurocognitive, clinical and functional correlates of subjective quality of life in Asian outpatients with schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(3):463–468. | ||
Alptekin K, Akvardar Y, Kivircik Akdede BB, et al. Is quality of life associated with cognitive impairment in schizophrenia? Prog Neuropsychopharmacol Biol Psychiatry. 2005;29(2):239–244. | ||
Herman M. Neurocognitive functioning and quality of life among dually diagnosed and non-substance abusing schizophrenia inpatients. Int J Ment Health Nurs. 2004;13(4):282–291. | ||
Agid O, McDonald K, Siu C, et al. Happiness in first-episode schizophrenia. Schizophr Res. 2012;141(1):98–103. | ||
Smith TE, Hull JW, Goodman M, et al. The relative influences of symptoms, insight, and neurocognition on social adjustment in schizophrenia and schizoaffective disorder. J Nerv Ment Dis. 1999;187(2):102–108. | ||
Simon HA. Spurious correlation: a causal interpretation. J Am Stat Assoc. 1954;267(49):467–479. | ||
Alonso-Solís A, Vives-Gilabert Y, Grasa E, et al. Resting-state functional connectivity alterations in the default network of schizophrenia patients with persistent auditory verbal hallucinations. Schizophr Res. 2015;161(2–3):261–268. | ||
Rotarska-Jagiela A, van de Ven V, Oertel-Knöchel V, Uhlhaas PJ, Vogeley K, Linden DEJ. Resting-state functional network correlates of psychotic symptoms in schizophrenia. Schizophr Res. 2010;117(1):21–30. | ||
Sommer IE, Clos M, Meijering AL, Diederen KMJ, Eickhoff SB. Resting state functional connectivity in patients with chronic hallucinations. PLoS One. 2012;7(9):e43516. | ||
Woodward ND, Rogers B, Heckers S. Functional resting-state networks are differentially affected in schizophrenia. Schizophr Res. 2011;130(1–3):86–93. | ||
Menon V. Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn Sci. 2011;15(10):483–506. | ||
Seeley WW, Menon V, Schatzberg AF, et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci Off J Soc Neurosci. 2007;27(9):2349–2356. | ||
Craig ADB. How do you feel now? The anterior insula and human awareness. Nat Rev Neurosci. 2009;10(1):59–70. | ||
Menon V, Uddin LQ. Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct. 2010;214(5–6):655–667. | ||
Palaniyappan L, White TP, Liddle PF. The concept of salience network dysfunction in schizophrenia: from neuroimaging observations to therapeutic opportunities. Curr Top Med Chem. 2012;12(21):2324–2338. | ||
Peters SK, Dunlop K, Downar J. Cortico-striatal-thalamic loop circuits of the salience network: a central pathway in psychiatric disease and treatment. Front Syst Neurosci. 2016;10:104. | ||
Wilkinson G, Hesdon B, Wild D, et al. Self-report quality of life measure for people with schizophrenia: the SQLS. Br J Psychiatry J Ment Sci. 2000;177:42–46. | ||
MacCallum RC, Austin JT. Applications of structural equation modeling in psychological research. Annu Rev Psychol. 2000;51:201–226. | ||
Kochunov P, Coyle TR, Rowland LM, et al. Association of white matter with core cognitive deficits in patients with schizophrenia. JAMA Psychiatry. 2017;74(9):958–966. | ||
Thomas ML, Green MF, Hellemann G, et al. Modeling deficits from early auditory information processing to psychosocial functioning in schizophrenia. JAMA Psychiatry. 2017;74(1):37–46. | ||
Boyer L, Cermolacce M, Dassa D, et al. Neurocognition, insight and medication nonadherence in schizophrenia: a structural equation modeling approach. PLoS One. 2012;7(10):e47655. | ||
Japanese WAIS-III Publication Committee. Nihonban WAIS-III chinou kensa [Japanese Wechsler Adult Intelligence Scale]. 3rd ed. Tokyo: Nihon bunka kagakusha; 2006. Japanese. | ||
Fujita K, Maekawa H, Dairoku K, Yamanaka K. WAIS-III no kaisyaku jirei to rinshou kenkyu. Tokyo: Nihon bunka kagakusha; 2011. Japanese. | ||
Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261–276. | ||
Kaneda Y, Sumiyoshi T, Keefe R, Ishimoto Y, Numata S, Ohmori T. Brief Assessment of Cognition in Schizophrenia: validation of the Japanese version. Psychiatry Clin Neurosci. 2007;61(6):602–609. | ||
Keefe RSE, Goldberg TE, Harvey PD, Gold JM, Poe MP, Coughenour L. The Brief Assessment of Cognition in Schizophrenia: reliability, sensitivity, and comparison with a standard neurocognitive battery. Schizophr Res. 2004;68(2–3):283–297. | ||
Addington D, Addington J, Maticka-Tyndale E. Assessing depression in schizophrenia: the Calgary Depression Scale. Br J Psychiatry Suppl. 1993;(22):39–44. | ||
Kaneda Y, Fujii A, Ohmori T. Psychometric properties of the Japanese version of the Calgary Depression Scale for Schizophrenics. J Nerv Ment Dis. 2000;188(4):237–239. | ||
Kaneda Y, Imakura A, Fujii A, Ohmori T. Schizophrenia Quality of Life Scale: validation of the Japanese version. Psychiatry Res. 2002;113(1–2):107–113. | ||
Van Dijk KRA, Sabuncu MR, Buckner RL. The influence of head motion on intrinsic functional connectivity MRI. NeuroImage. 2012;59(1):431–438. | ||
Calhoun VD, Adali T, Pearlson GD, Pekar JJ. A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp. 2001;14(3):140–151. | ||
Esposito F, Scarabino T, Hyvarinen A, et al. Independent component analysis of fMRI group studies by self-organizing clustering. NeuroImage. 2005;25(1):193–205. | ||
Bell AJ, Sejnowski TJ. An information-maximization approach to blind separation and blind deconvolution. Neural Comput. 1995;7(6):1129–1159. | ||
Shirer WR, Ryali S, Rykhlevskaia E, Menon V, Greicius MD. Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb Cortex N Y N. 2012;22(1):158–165. | ||
Tomotake M. Quality of life and its predictors in people with schizophrenia. J Med Investig JMI. 2011;58(3–4):167–174. | ||
Rosseel Y. Lavaan: an R package for structural equation modeling. J Stat Softw. 2012;48(2):1–36. | ||
semTools Contributors [webpage on the Internet]. semTools: useful tools for structural equation modeling. R package version 0.4–14. Available from: http://cran.r-project.org/web/packages/semTools/index.html. Accessed March 24, 2018. | ||
Kline RB. Principles and Practice of Structural Equation Modeling. 2nd ed. New York, NY: The Guilford Press; 2005. | ||
Cohen J. Statistical Power Analysis for Behavioural Sciences. Hillsdale, NJ: Lawrence Erlbaum Associates; 1982. | ||
García-García I, Jurado MÁ, Garolera M, et al. Alterations of the salience network in obesity: a resting-state fMRI study. Hum Brain Mapp. 2013;34(11):2786–2797. | ||
Lei D, Li K, Li L, et al. Disrupted functional brain connectome in patients with posttraumatic stress disorder. Radiology. 2015;276(3):818–827. | ||
Orliac F, Naveau M, Joliot M, et al. Links among resting-state default-mode network, salience network, and symptomatology in schizophrenia. Schizophr Res. 2013;148(1–3):74–80. | ||
Baumann B, Bogerts B. The pathomorphology of schizophrenia and mood disorders: similarities and differences. Schizophr Res. 1999;39(2):141–148; discussion 162. | ||
Kumari V, Peters E, Guinn A, et al. Mapping depression in schizophrenia: a functional magnetic resonance imaging study. Schizophr Bull. 2016;42(3):802–813. | ||
Okada N, Fukunaga M, Yamashita F, et al. Abnormal asymmetries in subcortical brain volume in schizophrenia. Mol Psychiatry. 2016;21(10):1460–1466. | ||
van Erp TGM, Hibar DP, Rasmussen JM, et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol Psychiatry. 2016;21(4):585. | ||
Smith KS, Tindell AJ, Aldridge JW, Berridge KC. Ventral pallidum roles in reward and motivation. Behav Brain Res. 2009;196(2):155–167. | ||
Haber SN, Knutson B. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology. 2010;35(1):4–26. | ||
Root DH, Melendez RI, Zaborszky L, Napier TC. The ventral pallidum: subregion-specific functional anatomy and roles in motivated behaviors. Prog Neurobiol. 2015;130:29–70. | ||
Vergara C, Roberts JE. Motivation and goal orientation in vulnerability to depression. Cogn Emot. 2011;25(7):1281–1290. | ||
Prosser ES, Csernansky JG, Kaplan J, Thiemann S, Becker TJ, Hollister LE. Depression, parkinsonian symptoms, and negative symptoms in schizophrenics treated with neuroleptics. J Nerv Ment Dis. 1987;175(2):100–105. | ||
Van Putten T, May RP. “Akinetic depression” in schizophrenia. Arch Gen Psychiatry. 1978;35(9):1101–1107. | ||
Addington D, Addington J, Maticka-Tyndale E, Joyce J. Reliability and validity of a depression rating scale for schizophrenics. Schizophr Res. 1992;6(3):201–208. | ||
Addington D, Addington J, Schissel B. A depression rating scale for schizophrenics. Schizophr Res. 1990;3(4):247–251. | ||
Narvaez JM, Twamley EW, McKibbin CL, Heaton RK, Patterson TL. Subjective and objective quality of life in schizophrenia. Schizophr Res. 2008;98(1–3):201–208. | ||
Nakagami E, Xie B, Hoe M, Brekke JS. Intrinsic motivation, neurocognition and psychosocial functioning in schizophrenia: testing mediator and moderator effects. Schizophr Res. 2008;105(1–3):95–104. | ||
Levinson DF, Umapathy C, Musthaq M. Treatment of schizoaffective disorder and schizophrenia with mood symptoms. Am J Psychiatry. 1999;156(8):1138–1148. | ||
Sensky T, Turkington D, Kingdon D, et al. A randomized controlled trial of cognitive-behavioral therapy for persistent symptoms in schizophrenia resistant to medication. Arch Gen Psychiatry. 2000;57(2):165–172. | ||
Gaite L, Vázquez-Barquero JL, Borra C, et al. Quality of life in patients with schizophrenia in five European countries: the EPSILON study. Acta Psychiatr Scand. 2002;105(4):283–292. | ||
Inagaki A, Inada T. Dose equivalence of novel antipsychotics. Part 5. Jpn J Clin Psychopharm. 2008;11:887–890. Japanese. |
Supplementary materials
  | Figure S1 Model specification. |
  | Figure S2 Comparison of connectivity maps for patients and controls across three RSNs (DMN, CEN, and SN). |
References
Chino B, Nemoto T, Fujii C, Mizuno M. Subjective assessments of the quality of life, well-being and self-efficacy in patients with schizophrenia. Psychiatry Clin Neurosci. 2009;63(4):521–528. | ||
Packer S, Husted J, Cohen S, Tomlinson G. Psychopathology and quality of life in schizophrenia. J Psychiatry Neurosci. 1997;22(4):231–234. | ||
Yamauchi K, Aki H, Tomotake M, et al. Predictors of subjective and objective quality of life in outpatients with schizophrenia. Psychiatry Clin Neurosci. 2008;62(4):404–411. | ||
Woon PS, Chia MY, Chan WY, Sim K. Neurocognitive, clinical and functional correlates of subjective quality of life in Asian outpatients with schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(3):463–468. | ||
Peters SK, Dunlop K, Downar J. Cortico-striatal-thalamic loop circuits of the salience network: a central pathway in psychiatric disease and treatment. Front Syst Neurosci. 2016;10:104. | ||
Tomotake M. Quality of life and its predictors in people with schizophrenia. J Med Investig JMI. 2011;58(3–4):167–174. | ||
Shirer WR, Ryali S, Rykhlevskaia E, Menon V, Greicius MD. Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb Cortex N Y N. 2012;22(1):158–165. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





