Back to Journals » International Journal of General Medicine » Volume 13
Status Quo of Diagnostic Procedures and Treatment of Inpatients with Infective Endocarditis at the Department of General Medicine at a University Hospital in a Suburban City in Japan: A Single-Hospital-Based Retrospective Study
Authors Yamashita S , Tago M , Tokushima M, Nakashima T, Katsuki NE , Anzai K , Yamashita S
Received 25 May 2020
Accepted for publication 14 July 2020
Published 26 August 2020 Volume 2020:13 Pages 547—557
DOI https://doi.org/10.2147/IJGM.S264497
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Shun Yamashita,1 Masaki Tago,1 Midori Tokushima,1 Tomotaro Nakashima,1 Naoko E Katsuki,1 Keizo Anzai,2 Shu-ichi Yamashita1
1Department of General Medicine, Saga University Hospital, Saga, Japan; 2Division of Metabolism and Endocrinology, Faculty of Medicine, Saga University, Saga, Japan
Correspondence: Masaki Tago
Department of General Medicine, Saga University Hospital, 5-1-1 Nabeshima, Saga 849-8501, Japan
Tel +81952343238
Fax +81952342029
Email [email protected]
Background: Our hospital’s department of general medicine is often involved in the diagnosis and treatment of diseases that are considered by other hospitals or other departments in our hospital to be difficult to diagnose correctly.
Objective: The aim of this study was to clarify how patients with infective endocarditis (IE) being admitted to our hospital’s department of general medicine were examined and treated and to elucidate their prognosis compared with patients admitted to other departments.
Materials and Methods: Inpatients of Saga University Hospital with definite IE from September 2007 to August 2017 were divided into 2 groups: those admitted to the general medicine department (the GM group) and those admitted to other departments (the non-GM group).
Results: Seventy-four patients were included; 17 (23%) were admitted to the general medicine department. In the GM group, the percentage of patients diagnosed with definite or suspected IE was lower (0% vs 32%, p=0.008), as was the rate of patients with echocardiographic findings that fulfilled the major modified Duke’s criteria (71% vs 98%, p≤ 0.001), preadmission. The GM group had higher percentages of patients with back or joint pain (41% vs 9%, p=0.001) and complications, including pyogenic spondylitis (35% vs 2%, p≤ 0.001), deep-seated abscesses (24% vs 5%, p=0.024), pyogenic arthritis (18% vs 0%, p=0.001), and glomerulonephritis (77% vs 37%, p=0.004) than did the non-GM group. Mortality within 30 days of admission to our hospital (12% vs 14%, p=0.753) and overall in-hospital mortality (12% vs 18%, p=0.570) did not significantly differ.
Conclusion: The general medicine department could have accurately diagnosed IE, given appropriate treatments, and obtained similar prognoses to those of IE patients treated by other departments, including cardiology and cardiovascular surgery, even in patients for whom diagnosing IE was more difficult or who had less typical echocardiographic findings preadmission.
Keywords: infective endocarditis, echocardiographic findings of infective endocarditis, general medicine, cardiology, cardiovascular surgery
Introduction
Infective endocarditis (IE) reportedly has an annual incidence that reaches 32.4 per 1,000,000 people.1 Many clinical departments in addition to those specializing in cardiology or cardiovascular surgery can be involved in diagnosing and treating patients with IE owing to its various nonspecific manifestations and findings on physical and laboratory examinations.2 Regardless of which department the patient is admitted to, modified Duke’s criteria are usually used to make the diagnosis.3 Among these departments, the general medicine departments, especially at university hospitals in Japan, which are often in charge of diagnosing diagnostically challenging diseases, including fevers of unknown origin, are likely to see patients with IE.4–7 Our previous research showed that the general medicine department of Saga University Hospital in Japan diagnosed and treated the third-highest number of inpatients with IE after the departments of cardiology and cardiovascular surgery.4
The department of cardiovascular surgery plays a clearly delineated role in treating IE in terms of when and how to operate and how to manage patients after surgery.8 The roles of cardiology departments in Japan were depicted in a report showing the clinical features of IE in Japanese patients treated and registered only by cardiologists at multiple centers through the web-based registration system, Cardiac Disease Registration.9 To our knowledge, however, the clinical characteristics of patients with IE who have been admitted to and treated by general medicine departments, especially in university hospitals in Japan, are unreported. The aim of this study was to clarify how patients with IE admitted to our hospital’s department of general medicine were examined and treated and to elucidate their prognosis.
We herein report how the general medicine department of Saga University Hospital, located in a suburban city in Japan, is involved in diagnosing and treating IE by comparing the characteristics of patients treated by the general medicine department to those of patients treated by other departments, including cardiology and cardiovascular surgery.
Materials and Methods
Study Design and Patients
The present study was a single-hospital-based retrospective study, performed as a secondary analysis of our previous report of the clinical status quo of IE at a university hospital in Japan, especially regarding clinical manifestations, by investigating all patients diagnosed with IE admitted to any departments of Saga University Hospital, Japan, from 2007 to 2017.4 As in our previous study, inpatients diagnosed with IE at Saga University Hospital were identified as per the International Statistical Classification of Diseases and Related Health Problems-10 (ICD-10, code number I-330). Patients who were subsequently diagnosed with “definite IE” according to modified Duke’s clinical criteria or pathological criteria were enrolled in this retrospective study. Subjects were divided into 2 groups: patients who were admitted to the department of general medicine, including those only transiently before moving to another department (the GM group) and those admitted to any other departments without having been admitted to the general medicine department during this hospitalization (the non-GM group). The primary outcome of this study was to clarify the clinical manifestations of patients with IE in the GM group, by comparing the characteristics of patients in the non-GM group.
Setting
Saga University Hospital is in Saga Prefecture, which has a population of 800,000 and is located on the southern island of Kyushu, Japan. It is a university hospital with fully equipped outpatient clinics and 604 inpatient beds in 29 clinical departments, including the departments of general medicine, cardiology, and cardiovascular surgery. The hospital provides high-level medical services mainly for patients in acute disease phases. The Division of Infectious Disease/Prevention and Control in our hospital usually supervises the diagnosing of infectious diseases, including IE, and chooses the appropriate antibiotics and treatment durations for all patients with positive blood culture results as well as when other departments request consultations. The general medicine department of this hospital manages the first-touch practice of outpatients without a reference from their primary doctors, in addition to treating both in- and outpatients with common diseases, fevers of unknown origin, diseases that are difficult to correctly diagnose, and complicated pathological situations requiring intensive care.4–7 In this study, patients suspected of having, or diagnosed with IE before admission would, in principle, be seen by the endocarditis team of the department of cardiology and cardiovascular surgery. In comparison, patients with non-specific symptoms and signs such as fever of unknown origin, consciousness disorder, paralysis, or rash, who were not suspected of having, or were not diagnosed with IE prior to referral, would initially be seen by the department of general medicine, emergency medicine, neurology, or dermatology or whatever department seemed appropriate. When such patients are diagnosed with, or are suspected of having, IE in our hospital, the Department of Cardiology or Cardiovascular Surgery is consulted to determine whether they should be treated by the specialist team or by each original department to which they were admitted. Thus, some patients are treated by the department of general medicine throughout their hospitalization and others by the department of cardiology or cardiovascular surgery later in the admission.
Data Sources
Three clinicians belonging to the department of general medicine in our hospital reviewed the medical charts and collected data on potentially eligible patients. Survey items are shown in Supplement 1. Nosocomial infection was defined as an infection acquired later than 48 hours after admission to our hospital caused by IE and manifesting new symptoms and signs. As for transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) findings, patients who fulfilled the major modified Duke’s criteria were considered echocardiography-positive. Causative bacteria were determined from the results of blood cultures and cultures or polymerase chain reaction tests performed on the operated valves. Presence of acute heart failure (AHF), central nervous system (CNS) disorders, extracranial embolism, disseminated infection, glomerulonephritis, and disseminated intravascular coagulation (DIC) was investigated, and the sum of the numbers of these complications was calculated (range 0–6). AHF was defined according to the European Society of Cardiology and American College of Cardiology Foundation/American Heart Association guidelines10,11 as a low left ventricular ejection fraction (<50%). Embolic stroke and cerebral hemorrhaging were defined as CNS disorders. Disseminated infections included pyogenic spondylitis, pyogenic arthritis, spinal epidural abscesses, deep-seated abscesses, and mycotic aneurysms. Complicated glomerulonephritis was defined as the following abnormal findings on urinalysis: ≥1+ blood and ≥1+ protein on the dipstick, or urinary blood cell casts, or dysmorphic hematuria. DIC was diagnosed according to the DIC scoring system of the Japanese Association for Acute Medicine.12
Data Analysis
Continuous variables are expressed as the median and interquartile range, and were compared with the Mann–Whitney U-test. Categorical variables are expressed as percentages and were compared with χ2 tests. Missing values were classified as “no abnormal findings” in each analysis. Statistical significance was set at p<0.05. IBM SPSS Statistics (version 25, IBM, Armonk, New York, USA) and Microsoft Excel 2016 software were used for the statistical analyses.
Ethical Consideration
All subjects provided consent as per the comprehensive agreement method of our hospital, and patients’ anonymity was protected. The Ethics Committee of Saga University Hospital (file number 2019–01-07) approved the study, which was conducted in accordance with the guidelines of the 1975 Declaration of Helsinki.
Results
Enrollment and Allocation of Study Patients
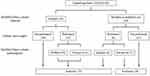
Of 110 patients identified as having IE using the ICD-10, 70 were diagnosed with “definite IE” according to the modified Duke’s clinical criteria after reviewing their medical charts. Nine of 40 patients initially diagnosed with “probable IE” or “not IE” underwent valvular operations; four of these were initially diagnosed with “definite IE” according to the modified Duke’s pathological criteria. Thus, 74 patients were diagnosed with “definite IE” according to either clinical or pathological criteria and accordingly enrolled and analyzed in our previous study4 (Figure 1). Seventy-four patients were admitted to 15 clinical departments: cardiology (23 cases, 31%), general medicine (14, 19%), cardiovascular surgery (9, 12%), emergency medicine (6, 8%), nephrology (5, 7%), neurology (4, 5%), gastroenterology (3, 4%), hepatology (3, 4%), and other departments (7, 10%). Three of 60 patients initially admitted to the departments of urology, orthopedics, and cardiology were subsequently moved to the department of general medicine. Consequently, 17 patients (23%) were allocated to the GM group, and the remaining 57 patients (77%) were allocated to the non-GM group (Figure 2).
Patient Characteristics
Table 1 shows the patients’ characteristics. The GM group had no patients diagnosed with definite or suspected IE before hospitalization to our hospital (0 [0%] vs 18 [32%], p=0.008). In addition, more patients in the GM group had back or joint pain within 2 months before the first visit to our hospital than did the non-GM group (41% vs 9%, p=0.001). Although more patients had chronic dermatological disorders as comorbidities in the GM group than in the non-GM group, the difference was not significant (24% vs 7%, p=0.054). Age, sex, transportation by ambulance, antibiotic treatment prior to blood culturing, dental disease on admission to our hospital, having visited a dental clinic within 6 months, invasive dental treatments within 6 months, valvular surgery for IE during the hospital stay, mortality within 30 days from admission to our hospital, and overall in-hospital mortality did not significantly differ between the two groups. Table 2 shows the laboratory findings on admission to our hospital, none of which differed significantly between the groups.
 |
Table 1 Patient Characters |
 |
Table 2 Laboratory Findings at the First Visit in Our Hospital |
Microbiology
Table 3 shows the microbiological characteristics. Blood cultures were performed on all 74 patients (100%), including those performed at the patients’ previous hospitals. Sixty-four patients (87%) were positive; 10 patients (14%) were negative. The rates of positive blood cultures did not significantly differ between the GM and non-GM groups (88% vs 86%, p=0.810), nor did they significantly differ in causative bacteria, ie, Staphylococcus aureus (24% vs 30%, p=0.613), viridans-group Streptococci (12% vs 21%, p=0.391), Streptococcus gallolyticus (0% vs 2%, p=0.582), and Enterococcus spp. (6% vs 7%, p=0.870), which fulfilled the major modified Duke’s criteria.
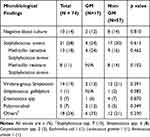 |
Table 3 Microbiological Findings |
Echocardiologic Data
In the present study, TTE was performed on all 74 patients (100%). TEE was performed on 26 patients (35%): 6/17 patients (35%) in the GM group and 20/57 (35%) in the non-GM group. TEE was not performed in 48 patients (65%), 43 of whom were diagnosed only by TTE without requiring TEE, and 2 of them died before TEE had been performed. The reasons for the remaining 3 patients not undergoing TEE could not be identified. The GM group had more negative results that did not fulfill the major modified Duke’s criteria than did the non-GM group via TTE or TEE (29% vs 2%, p<0.001), especially on the TTE (53% vs 2%, p<0.001). The infected valve type, presence or absence of a prosthetic valve, and presence or absence of valvular diseases more severe than grade I did not significantly differ between the groups (Table 4).
 |
Table 4 The Findings of Microbiology and Echocardiology |
Imaging Studies Other Than Echocardiography
Table 5 shows the rates for which imaging studies other than echocardiography were performed. Spinal magnetic resonance imaging (MRI) (47% vs 7%, p<0.001), especially lumbar MRIs (41% vs 4%, p<0.001) were performed more often in the GM group than in the non-GM group. The rates at which other imaging studies were performed did not significantly differ between the groups.
 |
Table 5 Performing of Imaging Tests Excluding Echocardiology |
Complications
Table 6 shows the complications in the present study. More patients in the GM group than in the non-GM group had glomerulonephritis (77% vs 37%, p=0.004), pyogenic spondylitis (35% vs 2%, p<0.001), pyogenic arthritis (18% vs 0%, p=0.001), and deep-seated abscesses (24% vs 5%, p=0.024). A higher frequency of urinalyses was performed in the GM group than in the non-GM group (94% vs 63%, p=0.014). CNS disorders including embolic stroke and cerebral hemorrhaging; extracranial embolism including splenic infarction, renal infarction, pulmonary embolism, pulmonary infarction, hepatic infarction and superior mesenteric artery embolism; DIC; and AHF did not significantly differ between the groups, nor did the total number of complications and embolisms per patient.
 |
Table 6 Complications |
Discussion
This is the first report to compare the clinical characteristics of patients with IE admitted to and treated in the general medicine department at a university hospital in Japan to those of patients admitted to other departments without a history of admission to the general medicine department. Cardiology and cardiovascular surgery are two representative departments involved in diagnosing and treating IE. The roles of the cardiovascular surgery department in treating IE are defined in the consensus guidelines for IE published by the American Association for Thoracic Surgery in 2016, which describe when and how to operate and how to manage patients after surgery.8 Furthermore, the roles of the cardiology department in Japan were described in a report of a multicenter study of cardiologists in 2013, which described the clinical features of IE treated by the cardiology department, such as the predisposing background, causative microorganisms, appropriate antibiotics, echocardiographic findings, and complications.9 However, as we previously reported, the general medicine department of our hospital treated the third-highest number of inpatients with IE,4 thus emphasizing the significance of the present study.
The nonspecific features, manifestations, and laboratory examination findings make suspecting and diagnosing IE itself challenging.2 Modified Duke’s criteria, whose two major criteria include consecutive positive blood cultures and presence of vegetation usually detected by echocardiography, are used worldwide to diagnose IE.3 Therefore, failure to detect the presence of vegetation by echocardiography could make IE more complicated to diagnose.3 The present study showed that many patients in the GM group were not suspected to IE or diagnosed with definite IE because of negative echocardiography results obtained before admission to our hospital. Surprisingly, in the present study, we even found a patient with IE who had negative results on eight consecutive blood cultures and two TEEs. The IE was conclusively diagnosed only via valvular surgery, showing the extreme difficulties in correctly diagnosing IE in the GM group.5 Accordingly, we conclude that our hospital’s department of general medicine correctly diagnosed inpatients with IE who were difficult to diagnose and treat and who had not been suspected of having, or been diagnosed with, IE on admission because of their atypical clinical manifestations. The combination of 18F-FDG positron emission tomography and computed tomographic imaging (18F-FDG PET/CT) could be useful tools for making diagnoses in patients in whom the diagnosis was obscure, such as IE complicating a prosthetic valve or intracardiac device, or detection of embolic complications, metastatic infections, or underlying cancers.13 However, the Japanese Health Insurance system reimburses the cost of 18F-FDG PET/CT only when this procedure is performed to detect a primary lesion of metastatic cancer of unknown origin, which may explain why many patients did not undergo 18F-FDG PET/CT in the present retrospective study.
While 27% of known pyogenic spondylitis patients were reported to be complicated with IE,14 5% and 21% of IE patients were conversely complicated with pyogenic spondylitis and pyogenic arthritis, respectively,15,16 indicating the importance of suspecting the complications of pyogenic spondylitis or pyogenic arthritis with IE and vice versa. We found more cases of complicated pyogenic spondylitis and pyogenic arthritis in the GM group than in the non-GM group in the present study. However, the risks of complicating pyogenic spondylitis or pyogenic arthritis with IE were considered equal between both groups because no significant differences were found in the incidences of diabetes mellitus or the administration of steroids or immunosuppressants, which were reported as risk factors of pyogenic spondylitis and pyogenic arthritis.17,18 In addition, the incidences of predisposing cardiac disease, presence of bacteremia by Gram-positive cocci including Staphylococcus aureus and Enterococcus spp., and complications of irreversible CNS disorders, which are often reported in patients with both IE and pyogenic spondylitis,14 were equal between both groups. Two reasons for the high incidences of complicating pyogenic spondylitis or pyogenic arthritis in the GM group despite these issues were considered: the larger number of patients admitted due to pyogenic spondylitis or pyogenic arthritis with undiagnosed baseline IE in the GM group and the carefulness of the physicians in the general medicine department when seeing patients with IE. These physicians actively detected the presence of back or joint pain, took patients’ histories, and conducted physical examinations, which enabled determination of complicating pyogenic spondylitis and pyogenic arthritis, regardless of the presence or absence of risk factors. Significantly more patients in the GM group had back or joint pain starting within 2 months before the first visit to our hospital; thus, spinal MRIs were performed more often. This could suggest that physicians actively suspected IE when patients had pyogenic spondylitis or pyogenic arthritis; thus, the general medicine department aggressively investigated the pyogenic spondylitis or pyogenic arthritis.
Although glomerulonephritis is a minor item on the modified Duke’s criteria,3 the reported incidence varied from almost null to approximately 26%.19,20 In the present study, the GM group had a higher incidence of glomerulonephritis. Although the rates of patients with diabetes mellitus, patients on hemodialysis, patients taking antibiotic treatment prior to blood culturing, and serum creatinine concentrations did not significantly differ between the groups, the GM group was given more urinalyses, indicating that clinicians in the general medicine department were actively testing for glomerulonephritis in patients with IE, which may have increased the rate of detecting glomerulonephritis in the GM group.
Even in this era of modernized antibiotics, mortality from IE and the rate of valvular surgery in patients with IE have commonly been reported to be 15%–30% and 25%–60%, respectively.9,21–25 It has even been reported that in-hospital mortality from IE is 11% in Japan.9 Various risk factors adversely influence the IE prognosis, including disadvantageous patient backgrounds (eg, old age, prosthetic valve infection, diabetes mellitus, administration of steroids or immunosuppressants, chronic hepatic disorder, and chronic kidney disease), serious complications of IE (eg, AHF, acute kidney injury, embolic stroke, cerebral hemorrhage, and septic shock), vicious causative microorganisms (eg, Staphylococcus aureus, multidrug-resistant organisms, and fungi), and undesirable echocardiographic findings (eg, vegetation on the left side of the heart, vegetation size >10 mm, annular abscesses, massive valve destruction, and decreased left ventricular function).26 However, these factors did not significantly differ, nor did the mortality rate within 30 days from admission to our hospital, the rate of overall in-hospital mortality, or the rate of performing valvar surgery between both groups. Thus, the general medicine department at our hospital treated patients with IE with equivalent seriousness and mortality rates, achieving similar prognoses to those of the other departments, including cardiology and cardiovascular surgery.
Limitations
The present study was a single-hospital based retrospective study at a university hospital, performed as a secondary analysis of a previous study with relatively few subjects. Therefore, the study setting could have introduced some selection bias. Some patients might have been inappropriately excluded because inpatients with IE in the present study were identified via ICD-10. Furthermore, a multicenter prospective study is needed to clarify the roles of general medicine departments throughout Japan when treating diagnostically challenging diseases or fevers of unknown origin on a daily basis and in diagnosing and treating IE.
Conclusion
The department of general medicine at our university hospital in Japan could have correctly diagnosed inpatients with IE who were not suspected to have IE or showed no typical findings on echocardiography before admission, especially with similar prognostic factors, and achieved similar prognoses as those of other departments, including cardiology and cardiovascular surgery. Clinicians in the department of general medicine actively suspected and diagnosed IE from the presence of pyogenic spondylitis, pyogenic arthritis, or glomerulonephritis.
Acknowledgment
We thank Traci Raley and Trish Reynolds from Edanz Group for editing a draft of this manuscript.
Author contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors state that they have no conflicts of interest.
References
1. Selton-Suty C, Célard M, Le Moing V, et al. Preeminence of Staphylococcus aureus in infective endocarditis: a 1-year population-based survey. Clin Infect Dis. 2012;54(9):1230–1239. doi:10.1093/cid/cis199
2. Thimas JC, Bernard DP. Infective endocarditis. Lancet. 2016;387:882–893. doi:10.1016/S0140-6736(15)00067-7
3. Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000;30(4):633–638. doi:10.1086/313753
4. Yamashita S, Tokushima M, Nakashima T, et al. Clinical status quo of infective endocarditis in a university hospital in Japan: a single-hospital-based retrospective cohort study. Intern Med. 2020;59(12):1497–1507. doi:10.2169/internalmedicine.4159-19
5. Yamashita S, Tago M, Katsuki NE, et al. Acute mitral regurgitation of unknown etiology associated with disseminated intravascular coagulation eventually diagnosed as enterococcus faecalis infective endocarditis by mitral valve surgery. Am J Case Rep. 2018;19:1467–1473. doi:10.12659/AJCR.912097
6. Yamashita S, Furukawa NE, Matsunaga T, Hirakawa Y, Tago M, Yamashita SI. Extremely high serum ferritin: an instrumental marker of masquerading adult-onset still’s disease with hemophagocytic syndrome. Am J Case Rep. 2017;18:1296–1301. doi:10.12659/AJCR.905684
7. Nishi TM, Yamashita S, Hirakawa YN, Katsuki NE, Tago M, Yamashita SI. Proliferative fasciitis/myositis involving the facial muscles including the masseter muscle: a rare cause of trismus. Am J Case Rep. 2019;20:1411–1417. doi:10.12659/AJCR.917193
8. Pettersson GB, Coselli JS, Hussain ST, et al. 2016 The American Association for Thoracic Surgery (AATS) consensus guidelines: surgical treatment of infective endocarditis: executive summary. J Thorac Cardiovasc Surg. 2017;153(6):1241–1258. doi:10.1016/j.jtcvs.2016.09.093
9. Nakatani S, Mitsutake K, Ohara T, et al. CADRE investigators. Recent picture of infective endocarditis in Japan—lessons from Cardiac Disease Registration (CADRE-IE). Circ J. 2013;77:1558–1564. doi:10.1253/circj.CJ-12-1101
10. Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. J Am Coll Cardiol. 2013;62(16):1495–1539. doi:10.1016/j.jacc.2013.05.020
11. Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2016;18(8):891–975. doi:10.1002/ejhf.592
12. Gando S, Saitoh D, Ogura H, et al. Natural history of disseminated intravascular coagulation diagnosed based on the newly established diagnostic criteria for critically ill patients: results of a multicenter, prospective survey. Crit Care Med. 2008;36(1):145–150. doi:10.1097/01.CCM.0000295317.97245.2D
13. Vincent LL, Otto CM. Infective endocarditis: update on epidemiology, outcomes, and management. Curr Cardiol Rep. 2018;20(10):86. doi:10.1007/s11886-018-1043-2
14. Koslow M, Kuperstein R, Eshed I, Perelman M, Maor E, Sidi Y. The unique clinical features and outcome of infectious endocarditis and vertebral osteomyelitis co-infection. Am J Med. 2014;127(7):
15. Pigrau C, Almirante B, Flores X, et al. Spontaneous pyogenic vertebral osteomyelitis and endocarditis: incidence, risk factors, and outcome. Am J Med. 2005;118(11):1287. doi:10.1016/j.amjmed.2005.02.027
16. Murillo O, Grau I, Gomez-Junyent J, et al. Endocarditis associated with vertebral osteomyelitis and septic arthritis of the axial skeleton. Infection. 2018;46(2):245–251. doi:10.1007/s15010-018-1121-9
17. Loibl M, Stoyanov L, Doenitz C, et al. Outcome-related co-factors in 105 cases of vertebral osteomyelitis in a tertiary care hospital. Infection. 2014;42(3):503–510. doi:10.1007/s15010-013-0582-0
18. Yoon SH, Chung SK, Kim KJ, Kim HJ, Jin YJ, Kim HB. Pyogenic vertebral osteomyelitis: identification of microorganism and laboratory markers used to predict clinical outcome. Eur Spine J. 2010;19(4):575–582. doi:10.1007/s00586-009-1216-1
19. Hisamatsu Y, Endo K, Hirata K, et al. A clinical investigation of infective endocarditis at a community hospital in Japan. J Infect Chemother. 2000;74(1):51–56. doi:10.11150/kansenshogakuzasshi1970.74.51
20. Majumdar A, Chowdhary S, Ferreira MA, et al. Renal pathological findings in infective endocarditis. Nephrol Dial Transplant. 2000;15:1782–1787. doi:10.1093/ndt/15.11.1782
21. Leone S, Ravasio V, Durante-Mangoni E, et al. Epidemiology, characteristics, and outcome of infective endocarditis in Italy: the Italian study on endocarditis. Infection. 2012;40:527–535. doi:10.1007/s15010-012-0285-y
22. García-Cabrera E, Fernández-Hidalgo N, Almirante B, et al. Group for the study of cardiovascular infections of the Andalusian Society of Infectious Diseases. Neurological complications of infective endocarditis: risk factors, outcome, and impact of cardiac surgery: a multicenter observational study. Circulation. 2013;127:2272–2284. doi:10.1161/CIRCULATIONAHA.112.000813
23. Prendergast BD, Tornos P. Surgery for infective endocarditis: who and when? Circulation. 2010;121(9):1141–1152. doi:10.1161/CIRCULATIONAHA.108.773598
24. Murdoch DR, Corey GR, Hoen B, et al. Clinical presentation, etiology and outcome of infective endocarditis in the 21st century: the International Collaboration on endocarditis-prospective cohort study. Arch Intern Med. 2009;169(5):463–473. doi:10.1001/archinternmed.2008.603
25. Mostaghim AS, Lo HYA, Khardori N. A retrospective epidemiologic study to define risk factors, microbiology, and clinical outcomes of infective endocarditis in a large tertiary-care teaching hospital. SAGE Open Med. 2017;5:205031211774177. doi:10.1177/2050312117741772
26. Habib G, Hoen B, Tornos P, et al. Guidelines on the prevention, diagnosis, and treatment of infective endocarditis (new version 2009). Eur Heart J. 2009;30:2369.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.