Back to Journals » Cancer Management and Research » Volume 11
Skin graft using MatriDerm® for plantar defects after excision of skin cancer
Authors Kang SW , Park JK , Shon HC, Choi ES , Kim DS , Min KT
Received 17 December 2018
Accepted for publication 4 March 2019
Published 10 April 2019 Volume 2019:11 Pages 2947—2950
DOI https://doi.org/10.2147/CMAR.S198568
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Chien-Feng Li
Sang-Woo Kang, Ji-Kang Park, Hyun-Chul Shon, Eui-Sung Choi, Dong-Soo Kim, Kyoung-Tae Min
Department of Orthopaedic Surgery, Chungbuk National University Hospital, Cheongju, Korea
Abstract: Skin cancer should be excised with sufficient margin to reduce recurrence rate. However, the surgeon always has to worry about the reconstruction method of skin defects after excision. In particular, defects in the plantar surface of the foot are difficult to reconstruct due to their position and structure, and various methods are applied by each surgeon. Surgeons think which methods are easier to apply to patients and less morbidity. To alleviate these concerns, we applied artificial dermal substitute to skin defects after skin cancer. Bowen’s disease (squamous cell carcinoma in situ) and melanoma in situ on the plantar surface of the foot were subjected to wide excision with sufficient margin. After excision, a skin defect with exposed plantar fascia was applied with a matrix defect and vacuum. A granulation tissue (dermal matrix) was formed and a split-thickness skin graft was performed. Both patients had good functional results and no problems with skin donor sites. Thus, we report a skin graft method that is relatively easy to apply after skin cancer excision on the plantar surface of the foot.
Keywords: plantar reconstruction, Bowen’s disease, melanoma, dermal substitute
Introduction
Surgical treatment of skin cancer, such as melanoma and squamous cell carcinoma, should be excised with sufficient margin and reconstructed for skin defects if necessary. However, skin defects after wide excision of skin cancer on the plantar side of the foot are difficult to reconstruct due to weight-bearing applied to the plantar side of the foot and the thick skin layer covering the plantar fascia. Various flap methods have been used to resolve plantar skin defects.1–4 However, the flap shows high donor site morbidity and it is difficult to apply if it is not performed by microsurgeon. MatriDerm® (MedSkin Solution Dr. Suwelack AG, Billerbeck, Germany) is an artificial dermis composed of collagen and elastin and can be used for reconstruction using split-thickness skin grafts (STSG). We previously obtained good results using MatriDerm for skin defects; defects as the result of trauma, burns or diabetic skin ulcers, and this was applied to reconstruction of skin defect after excision of plantar skin cancer.
Case report
Case 1
A 66-year-old Korean woman was treated at the local clinic with chronic ulceration on the plantar surface of the right foot for 8 months previously. At the time of admission, a 3×2 cm erythematous reticular chronic ulcer lesion was observed in the mid-plantar surface. The patient had never been exposed to radiation or arsenic, and had no history of skin malignancies. Skin biopsy was performed and the patient was diagnosed with Bowen’s disease. Photodynamic therapy was considered at first in consultation with a dermatologist, but wide excision was determined to reduce recurrence rate.
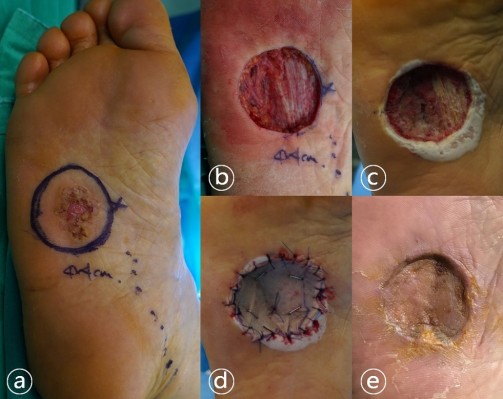
Surgery was performed with a figure four position on the supine position under spinal anesthesia. Wide excision with a margin of at least 1 cm (Figure 1A), and secondary reconstruction of the skin defect were planned. After wide excision, margin clearance was confirmed by frozen section. Plantar fascia and lateral plantar neurovascular bundle were exposed through skin defect (Figure 1B). The exposed structures were covered with MatriDerm, and a vacuum device was applied additionally. Vacuum change was performed weekly at the operation room until the permanent biopsy result was obtained and the skin graft was planned after confirming the formation of the dermal matrix (Figure 1C). Skin was harvested using a dermatome on the ipsilateral lateral thigh and split-thickness skin graft was performed (Figure 1D). Complete healing of the skin graft was confirmed at 8 weeks after surgery (Figure 1E). There were no complications such as infection in the donor site and graft during recovery, and there were no functional problems of the foot. The only complication observed was numbness on the distal lateral side of the foot and it gradually improved.
Case 2
A 69-year-old Korean male presented with a color change in the left foot that occurred 4 months ago. At the time of admission, a 3×2 cm pigmented skin lesion was observed on the lateral side of the mid plantar surface. The patient had no previous history and family history of melanoma. On skin biopsy, lesions were diagnosed as melanoma in situ. Therefore, wide excision was determined except for sentinel lymph node biopsy. Surgery was performed in prone position under spinal anesthesia. Wide excision with a margin of at least 1 cm and a medial plantar artery flap for skin defect were planned (Figure 2A). Wide excision was performed and margin clearance was confirmed by frozen section. The medial plantar artery flap was attempted as planned, but the distance between the lesion and the flap was too far to be performed. Plantar fascia was exposed through skin defect and secondary reconstruction was performed (Figure 2B). The exposed structures were covered with MatriDerm, and a vacuum device was applied additionally. One week after surgery, a dermal matrix was formed and a split-thickness skin graft was performed (Figure 2C and D). Complete healing of the skin graft was confirmed through outpatient follow-up at 6 weeks postoperatively (Figure 2E). There were no complications at the donor site and graft, and there were no functional problems in the foot. There was a tingling sense in the incision site for medial plantar artery flap, but it improved gradually.
Discussion
It is difficult for the surgeon to determine sufficient margin for wide excision of skin cancer in the plantar surface of the foot. Low flexibility of the skin due to thick plantar fascia located beneath the skin and the weight bearing applied to the plantar surface are reasons why soft tissue reconstruction of the plantar surface is difficult. Also, due to limited number of reconstruction methods, the larger the margin needed for wide excision, the fewer options the surgeon can determine. Several methods have been used depending on the location of the defect to reconstruct the plantar skin defect. Distal plantar skin defect can be treated by local flap such as medial plantar artery flap or V-Y flap,1 and island perforator flap or free flap can be applied to heel and mid-plantar skin defect.2,5 Wu and Gottlieb performed a skin graft or local flap on non-weight bearing skin defect.3 However, flap surgery is difficult to perform and the larger the defect size, the greater the donor site morbidity. Skin grafts have a limited survival rate when applied directly to fibrous tissue.
To overcome these drawbacks, we applied artificial dermal substitute for the reconstruction of skin defect by trauma or burn, to skin cancer. Wollina reported good results with single-stage reconstruction using MatriDerm for nose, lower leg, knee, and foot skin defects caused by chronic ulcers, bursitis, diabetic foot and pressure sore including squamous cell carcinoma.6 MatriDerm is an acellular dermal substitute with porous membrane three-dimensional structure composed of collagen and elastin from bovine ligament and dermis, applicable for full-thickness skin defects.7,8 MatriDerm is a thin, 1 mm thick, paper-like water-soluble membrane that acts as a scaffold to stimulate the formation of a dermal matrix, helping to restore skin and inhibiting scar formation.9 This action allows single-stage surgical treatment through STSG for skin defects due to trauma, surgical wound, and IIb–III degrees burn.7,10 In addition, since the elasticity and flexibility of the MatriDerm are similar to those of surrounding tissues, functional problems such as tendon excursion and range of motion recovery of joints can be compensated.11 However, MatriDerm cannot be used for infected tissue. Hever and Cavale reported that MatriDerm was the best in a single-stage procedure for patients with large skin defects, comparing STSG alone with STSG using MatriDerm and Integra (Integra Life Sciences Corp., NJ, USA).12 This is because MatriDerm is based on elastin, unlike other templates, and elastin acts as a major role in increasing elasticity and resilience in the skin. For this reason, we selected MatriDerm as the material in these cases.
In our cases, the plantar fascia and neurovascular bundle were exposed through the defect after excision of the skin cancer on the plantar surface. When the fascia is exposed to the weight-bearing position, flap is usually the chosen method to maintain the previous elasticity. Instead, we applied MatriDerm to reduce the adhesion between skin and basal structure and to increase the healing potential. Although this method is easily applicable, more studies are required to be applied to deep tissue cancers with high stage.
Conclusion
Wide excision with sufficient margin is necessary to prevent recurrence of skin cancer. However, additional surgery should be considered after excision in areas where skin reconstruction is difficult, such as plantar surface of the foot. We report cases of skin graft using MatriDerm for plantar skin defect, which is relatively easy to reconstruct, regardless of position and size.
Consent
Written informed consent was obtained from the patients for publication of this case report and any accompanying images.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Evans GR, Robb GL. Cutaneous foot malignancies: outcome and options for reconstruction. Ann Plast Surg. 1995;34(4):396–401.
2. Koshima I, Narushima M, Mihara M, et al. Island medial plantar artery perforator flap for reconstruction of plantar defects. Ann Plast Surg. 2007;59(5):558–562. doi:10.1097/SAP.0b013e3180315528
3. Wu LC, Gottlieb LJ. Glabrous dermal grafting: a 12-year experience with the functional and aesthetic restoration of palmar and plantar skin defects. Plast Reconstr Surg. 2005;116(6):1679–1685.
4. Kang HG, Kim JH, Cho HS, Han I, Oh JH, Kim H-S. Soft tissue reconstruction of the foot using the distally based island pedicle flap after resection of malignant melanoma. Clin Orthop Surg. 2010;2(4):244–249. doi:10.4055/cios.2010.2.4.244
5. Hong JP, Kim EK. Sole reconstruction using anterolateral thigh perforator free flaps. Plast Reconstr Surg. 2007;119(1):186–193. doi:10.1097/01.prs.0000244856.98170.9c
6. Wollina U. One-stage reconstruction of soft tissue defects with the sandwich technique: collagen-elastin dermal template and skin grafts. J Cutan Aesthet Surg. 2011;4(3):176–182. doi:10.4103/0974-2077.91248
7. van Zuijlen PP, van Trier AJ, Vloemans JF, Groenevelt F, Kreis RW, Middelkoop E. Graft survival and effectiveness of dermal substitution in burns and reconstructive surgery in a one-stage grafting model. Plast Reconstr Surg. 2000;106(3):615–623.
8. Wainwright D, Madden M, Luterman A, et al. Clinical evaluation of an acellular allograft dermal matrix in full-thickness burns. J Burn Care Rehabil. 1996;17(2):124–136. doi:10.1097/00004630-199603000-00006
9. Min JH, Yun IS, Lew DH, Roh TS, Lee WJ. The use of matriderm and autologous skin graft in the treatment of full thickness skin defects. Arch Plast Surg. 2014;41(4):330–336. doi:10.5999/aps.2014.41.4.330
10. De Vries HJ, Mekkes JR, Middelkoop E, Hinrichs WL, Wildevuur CR, Westerhof W. Dermal substitutes for full-thickness wounds in a one-stage grafting model. Wound Repair Regen. 1993;1(4):244–252. doi:10.1046/j.1524-475X.1993.10410.x
11. Choi JY, Kim SH, Oh GJ, Roh SG, Lee NH, Yang KM. Management of defects on lower extremities with the use of matriderm and skin graft. Arch Plast Surg. 2014;41(4):337–343. doi:10.5999/aps.2014.41.4.337
12. Cavale P. Matriderm vs integradermal regeneration template single layer as a dermal substitute for synchronous autologous skin grafting in necrotising fasciitis – a case study. Advanc Plast Reconstr Surg. 2017;1(1):1–4.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.