Back to Journals » Infection and Drug Resistance » Volume 12
Shigella: Antibiotic-Resistance Mechanisms And New Horizons For Treatment
Authors Ranjbar R , Farahani A
Received 19 June 2019
Accepted for publication 28 August 2019
Published 7 October 2019 Volume 2019:12 Pages 3137—3167
DOI https://doi.org/10.2147/IDR.S219755
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Eric Nulens
Reza Ranjbar, Abbas Farahani
Molecular Biology Research Center, Systems Biology and Poisonings Institute, Baqiyatallah University of Medical Sciences, Tehran, Iran
Correspondence: Reza Ranjbar
Molecular Biology Research Center, Systems Biology and Poisonings Institute, Baqiyatallah University of Medical Sciences, Iran
Tel +98 21 8803 9883
Email [email protected]
Abstract: Shigella spp. are a common cause of diarrheal disease and have remained an important pathogen responsible for increased rates of morbidity and mortality caused by dysentery each year around the globe. Antibiotic treatment of Shigella infections plays an essential role in reducing prevalence and death rates of the disease. However, treatment of these infections remains a challenge, due to the global rise in broad-spectrum resistance to many antibiotics. Drug resistance in Shigella spp. can result from many mechanisms, such as decrease in cellular permeability, extrusion of drugs by active efflux pumps, and overexpression of drug-modifying and -inactivating enzymes or target modification by mutation. Therefore, there is an increasing need for identification and evolution of alternative therapeutic strategies presenting innovative avenues against Shigella infections, as well as paying further attention to this infection. The current review focuses on various antibiotic-resistance mechanisms of Shigella spp. with a particular emphasis on epidemiology and new mechanisms of resistance and their acquisition, and also discusses the status of novel strategies for treatment of Shigella infection and vaccine candidates currently under evaluation in preclinical or clinical phases.
Keywords: Shigella, antibiotics, resistance, drug resistance, mechanism, treatment, biofilm, efflux pumps, prevention, vaccine
Introduction
Shigella spp. are a Gram-negative, rod-shaped, immotile, and non-spore–forming bacteria and a causative agent of acute diarrhea that may progress to bloody mucoid diarrhea, also known as bacillary dysentery (or shigellosis).1 Shigella is the most common cause of diarrheal disease and has remained a major pathogen responsible for increased rates of morbidity and mortality caused by dysentery each year around the globe, particularly affecting children aged <5 years in developing countries.2 The four types of Shigella spp. comprise subgroup A (S. dysenteriae), subgroup B (S. flexneri), subgroup C (S. boydii), and subgroup D (S. sonnei). Each subgroup contains several serotypes. Shigellosis can occur in pandemic, epidemic, and sporadic forms. Epidemiological reports have shown that the epidemic subgroup of diarrhea typically occurs as a result of infection with S. flexneri in developing countries and S. sonnei in industrialized countries.1,3 Shigella spp. are categorized by the World Health Organization (WHO) as bacteria mainly causing infections in the community.4 Shigellosis is a great public health threat, because its infective dose is on the order of 10–100 organisms compared to other enteric pathogens (usually it is 105–108 for Salmonella and Vibrio, respectively).5,6
Antibiotic treatment of common bacterial infections plays an important role in reducing prevalence and death rates of the disease. However, incorrect antibiotic use or overuse in treating diarrhea increases antibiotic resistance. Shigella spp. are resistant to most antibiotics, and drug treatment related to these bacteria is costly, time-consuming, and sometimes problematic, particularly in areas with limited medical care.7,8 About half the strains of Shigella in many parts of the world are now resistant to multiple drugs. Recently, various antibiotic-resistance mechanisms have been described by researchers, and these antibiotic-resistance mechanisms limit therapeutic options for treatment of Shigella infections.8,9
Drug resistance in Shigella spp. can result from many mechanisms, such as extrusion of drugs by active efflux pumps, decrease in cellular permeability, and overexpression of drug-modifying and -inactivating enzymes or target modification by mutation.6,10,11 The current study was done to review various antibiotic-resistance mechanisms of Shigella spp., with a particular focus on epidemiology and new mechanisms of resistance and their acquisition, and also to discuss treatment and prevention measures for diseases caused by these organisms.
Search Strategy
We searched the biomedical electronic databases Web of Science, Scopus, PubMed (Medline), Embase, Cochrane Library, and Google Scholar for articles on Shigella published in English between 1990 and May 2019, using the key terms (alone and in combination) “Shigella”, “drug resistance”, “mechanism”, “biofilm”, “efflux pump”, “vaccine”, and “treatment”. We excluded case reports and some studies on the implementation of established techniques. Articles published before 1990 were also excluded, except when necessary. A total of 193 relevant articles were identified from the databases and included in this review.
Drug-Resistance Mechanisms In Shigella Spp
Role Of Outer-Membrane Permeability
Natural resistance to antimicrobial drugs by various mechanisms preventing the drug from being absorbed is capable of transforming the drug, its biotransformation into the cell, or reducing affinity with the drugs’ target.12 Cell walls of microorganisms are the first barrier against penetration of the drug. Some modifications of membrane permeability or changes in the membrane lead to porin loss, which can result in an increase in minimum inhibitory concentration(MIC) for antimicrobial agents.13 Most antibiotics used in treatment of Shigella infection should be able to penetrate the cell membrane to reach intracellular accumulation and target sites. For example, quinolone antibacterial agents, such as nalidixic acid, ofloxacin, and ciprofloxacin, interfere with DNA replication by inhibiting DNA topoisomerase IV and gyrase. Aminoglycoside antibiotics, such as streptomycin and spectinomycin, mediate inhibition of protein synthesis by binding to ribosomal subunits and reaching intracellular targets. β-Lactam antibiotics, eg, penicillin and cephalosporin, are a class of antibiotics containing a β-lactam ring in their molecular structures and inhibit cell-wall biosynthesis by targeting penicillin-binding proteins. Mutation or absence of ∼39 kDa porin in the membrane of such Gram-negative bacteria as Shigella spp. mainly influences susceptibility to slow penetration of β-lactams, such as aztreonam and dianionic moxalactam, and also low permeability of hydrophilic antibiotics, such as penicillin and piperacillin.6,13 Indeed, resistance toward β-lactam antibiotics is associated with modification of the outer-membrane porins OmpF (∼38 kDa) and OmpC (∼42 kDa) and cytosolic proteins of ∼26 kDa, OmpR as a transcriptional regulator.6 In a study, three imipenem-resistant mutants of S. dysenteriae were obtained from India and showed lower levels of both major OMPs (∼38 and ∼43 kDa). Increasing imipenem resistance in mutants was associated with permeability of outer-membrane proteins.14 Lipopolysaccharides (LPSs) have been recognized as an essential outer-membrane component needed for assembly of trimeric PhoE porin and confer colicin E2 resistance in S. flexneri strains,15 and have also been reported to be linked with the rise in resistance toward imipenem in S. dysenteriae.14 Some outer-membrane components, such as IcsA molecules, are not only associated with bile salts resistance but are also related to promotion in development of biofilm by mediating bacterial cell–cell interactions. Consequently, they produce resistant phenotypes.16
Efflux Systems
Active efflux pumps play a significant role in antibiotic-resistance phenotypes of Gram-negative bacteria and expelling toxic compounds from their cells. Efflux systems are grouped into five families: the major facilitator superfamily (MFS), resistance–nodulation–division family, small multidrug resistance (MDR) family, ATP-binding cassette superfamily, and multidrug and toxic compound extrusion family.17 AcrAB–TolC pump is involved in antibiotic resistance phenotype of Escherichia spp., Enterobacter spp., Salmonella spp., and Shigella spp isolates. The AcrAB–TolC system is a tripartite complex comprising TolC (outer-membrane channel), AcrB (inner-membrane transporter protein), and periplasmic AcrA involved in assembly and maintenance of these two integral membrane proteins. AcrAB–TolC belongs to resistance–nodulation–division family of efflux pumps, associated with efflux of quinolones, and one of factors responsible for development of resistance among Shigella isolates. Indeed, overexpression of AcrAB–TolC results in overall decreased accumulation of quinolones inside bacterial cells, also resulting in reduced susceptibility to them.18 AcrAB-associated to bile-salt resistance has been found in some strains of S. flexneri. Their expression has been shown to increase after exposure to bile salts, and enabled Shigella to resist bactericidal effects of bile. Researchers believe that this phenomenon may confer resistance to other antimicrobial agents. Furthermore, overexpression of AcrB has been found to be linked to multiple drug-resistance phenotypes in some Gram-negative bacteria.19
Synergistic action regarding activation of acrAB–tolC efflux pumps has been shown to decrease expression of outer-membrane porins, gyrase, and topoisomerase target–gene mutations toward fluoroquinolone resistance in Shigella isolates.20,21 Drug-efflux pumps, such as marA, tolC, ydhE, and mdfA, confer quinolone resistance. Kim et al demonstrated that resistance to fluoroquinolone is due to increased expression level of MdfA efflux pump in Shigella spp. This efflux is a member of the MFS antibiotic-efflux system, and MdfA efflux pump–mediated fluoroquinolone resistance was first identified among MDR Escherichia coli.22 Tetracycline efflux and resistance is associated with the MFS antibiotic–efflux system encoded by various tet genes in Gram-negative bacteria, such as Shigella spp. and Klebsiella spp. Among tet efflux systems, it seems that tetA and tetB are mediated by resistance to tetracycline in S. sonnei and S. flexneri, respectively (for more details, refer to “Tetracycline Resistance” section).11
Resistance To β-Lactam Antibiotics
Class A β-Lactamases
Class A β-lactamases can hydrolyze narrow-spectrum penicillin, but not carbapenems or cephalosporins, and are inhibited by tazobactam and clavulanic acid. Extended-spectrum β-lactamases (ESBLs) belong to Ambler class A. ESBLs conferring resistance to third-generation cephalosporins have been found in Shigella isolates. The first report documenting identification of ESBL-producing Shigella strains was from Bangladesh in 2004.23 Emergence of ESBLs in Shigella spp. is a global major health threat affecting both developed and developing countries. Different β-lactamases belonging to Ambler class A have been reported among Shigella isolates, such as TEM, SHV, and CTX-M enzymes. The first isolate of S. flexneri producing an ESBL and harboring a plasmid encoding the blaSHV-2 gene was reported from France in 1995.24
To date, several reports from Argentina, Israel, Canada, Turkey, Lebanon, Japan, Iran, South Korea, China, and other various regions in Asia have identified Shigella spp. harboring different types of ESBL genes (Table 1).23,25–32 Although most ESBLs are derivatives of TEM and SHV β-lactamase families, which were first identified, Shigella spp. can also express the CTX-M family, among which CTX-M-15 is one of the most relevant findings associated with the current epidemiology of ESBLs, which has been predominantly identified in commensal and pathogenic ESBL-producing Shigella isolates around the world. Despite many surveillance studies and investigations, the reason for this epidemiological shift remains unknown.24,33 These enzymes are responsible for selective hydrolysis of ceftriaxone and cefotaxime and even more distinctly for ceftazidime, although some types of CTX-M, such as CTX-M-15, may hydrolyze ceftazidime.33 In general, CTX-M-15 has been found to have great catalytic efficiency (high kcat/Km) against piperacillin, benzylpenicillin, ceftriaxone, and cefotaxime, as reported for other of CTX-M-types, such as CTX-M-3, CTX-M-16, and CTX-M-18.
 |  |  |
Table 1 Prevalence Of Antimicrobial Resistance Genes In Shigella Spp. Isolated From Different Regions Of The World |
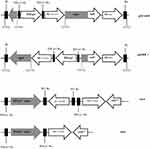
To date, CTX-M-15 has been detected in Shigella isolates from various countries across the world, including Canada, Russia, Poland, the UK, France, Bulgaria, Turkey, and Iran.33,34 CTX-M-type β-lactamases contain at least 40 enzymes, and these can be readily transferred among Shigella isolates by conjugative plasmids belonging to IncF, IncZ, and Incl groups.34,35 Li et al revealed that ISEcp1 was present adjacently to all blaCTX-M genes in Shigella strains, meaning that it plays an important role in mobilizing blaCTX-M genes (Figure 1).36 A large study conducted in Vietnam analyzed IncI1 plasmid pKHSB1 carrying blaCTX-M-15 collected from a clonal population of S. sonnei.37 Another study reported full sequence of IncI1 plasmid pSH4469 carrying blaCTX-M-15 in a clinical isolate of S. sonnei isolated from an outbreak in South Korea.34 The overall structure of plasmid pSH4469 was nearly identical to pKHSB1, and it seemed that both were responsible for dissemination of blaCTX-M-15 among Shigella isolates (Figure 1).
Recently, a new hybrid of CTX-M-9 and CTX-M-1 β-lactamases named CTX-M-123 was identified among S. flexneri isolates from patients in China. In this study, blaCTX-M-123 was carried by two conjugative plasmids named IncHI2 and IncF, and these conjugatable plasmids were responsible for dissemination of blaCTX-M-123 in S. flexneri isolates.38 Moreover, several new β-lactamase subtypes — CTX-M-79, CTX-M-27, CTX-M-24, CTX-M-15, CTX-M-14, CTX-M-64, CTX-M-65, CTX-M-55, and CTX-M-3 — have been found in clinical Shigella strains isolated from different provinces in China.36,39,40 ESBL genes among Shigella isolates might be transferred from E. coli isolates to Shigella spp., especially S. sonnei isolates, through conjugation in human gut.41,42 The increase in MDR and emergence of ESBL in Shigella spp. may be the cause of treatment failures and accordingly limitation in therapeutic options.41
Class B β-Lactamases
Class B β-lactamase enzyme can hydrolyze carbapenem and other β-lactams, except for aztreonam, and classical β-lactamase inhibitors, such as tazobactam and clavulanic acid, do not inhibit them. Metallo-β-Lactamase was first detected in a transferable plasmid from Pseudomonas aeruginosa, and also IMP-1 was first identified from many kinds of Gram-negative rods in Japan.43,44 O’Hara et al reported a novel type of metallo-β-lactamase named MET-1 mediated by a S. flexneri plasmid. They believed that MET-1 was a derivative of IMP-1 β-lactamase.44 This plasmid conferred resistance against sulfonamide and kanamycin, in addition to β-lactamase.44 Lyobe et al found that MET-1 experienced two amino-acid changes from IMP-1. The gene was renamed IMP-3, and thus IMP-3 could be considered an ancestor of IMP-1 β-lactamase. They also showed that this gene was located on a cassette inserted in a class I integron, widely disseminated among other species of Shigella, and conferred resistance to almost all β-lactam antibiotics.45
Carbapenem resistance conferred by blaVIM and blaIMP genes has recently been detected in S. sonnei and S. flexneri isolates from pediatric patients with diarrhea in the Andaman and Nicobar islands in India.43 In this study, after analysis of nucleotide sequencing of blaIMP and blaVIM genes by BLAST, 100% similarity with sequences of these genes isolated from Acinetobacter baumannii and P. aeruginosa available at the NCBI database was confirmed. After spread of carbapenem resistance to Shigella in another part of the world, an indication of a potential public health challenge, treatment options will be limited, and infection-control measures remain of high importance.43 Finally, although class A β-lactamase KPC is one of the most commonly identified carbapenemases among other Gram-negative bacteria in some parts of the world, it has not yet been identified in Shigella isolates, except for a single blaKPC-carrying S. flexneri strain isolated from the National Senegalese Enterobacteriaceae Center located at the Pasteur Institute in Dakar.46
Class C β-Lactamases
Ceftriaxone is recommended for treatment of ciprofloxacin-resistant Shigella isolates. However, today some strains of Shigella spp. have a resistance gene to cephalosporins. Class C β-lactamases, also known as AmpC-type enzymes, confer high-level resistance against cephalosporins. AmpC β-lactamase is encoded by both plasmid and chromosomal genes, and the first report of plasmid-encoded CMY-2-type AmpC β-lactamase was detected among ceftriaxone-resistant S. sonnei isolates obtained from an outbreak of bacillary dysentery in Taiwan.47 CMY-2 enzymes have been reported in China, Taiwan, Costa Rica, Iran, and India from several epidemic strains.47–50 Zhang et al found two AmpC β-lactamase producers with blaCMY-2 and blaDHA-1 in Shigella strains recovered from diarrhea patients in China.51
Tajbakhsh et al reported on the first AmpC β-lactamase (blaCMY-2) producers in S. sonnei isolated from patients in Tehran, Iran.48 In recent years, other studies conducted in Iran have indicated spread of resistance to extended-spectrum cephalosporins among Shigella isolates.31,50 In a similar study carried out in Iran, studying cephalosporin-resistant Shigella isolates, the researchers identified gene CMY-59 in one S. sonnei isolate from pediatric patients aged <12 years.31 However, the majority of AmpC-positive isolates studied in other parts of the world belonging to the CMY-2 genotype and other AmpC genes (blaMOX, blaFOX, blaMIR(ACT-1), blaCIT, and blaACC) have been identified in Shigella isolates; however, so far there has not a study on them.52,53 Indeed, few reports have described the presence of AmpC β-lactamases among Shigella isolates worldwide (see Table 1 for more details).47,48
Class D β-Lactamases
Class D β-lactamases or OXA-type β-lactamases confer resistance to ampicillin and cephalothin and can hydrolyze oxacillin and cloxacillin, as well as benzylpenicillin, but they are not inhibited by tazobactam or other inhibitors.52,54 Initially, blaOXA β-lactamases were reported among P. aeruginosa isolates, although now blaOXA genes have been identified in integrons and plasmids in many Gram-negative bacteria.55 In Shigella spp., resistance to ampicillin is mainly associated with an OXA-type β-lactamase.54,56 blaOXA-30 was initially described in ampicillin-resistant S. flexneri strains from China in 2000.57 Results of some studies have shown that S. flexneri isolates are a probable host specific for blaOXA-type β-lactamase.52,58 Another study from Iran showed that all blaOXA-positive isolates carried blaOXA-1 and many of them were present in S. flexneri, suggesting an individual host preference of these enzymes in S. flexneri isolates.50
blaOXA-1 and blaOXA-30 genes, containing Tn2603 and Tn1409 transposons, respectively, differ from each other by having one mutation at codon 131. A gene encoding blaOXAβ-lactamases is carried on integron.58 Furthermore, other studies have shown that blaOXA-30 and aadA1 are located in the gene cassettes of class 1 integrons (Figure 2). Therefore, class 1 integrons carry resistance traits for β-lactams (blaOXA) and trimethoprim (dfrA1).52
 |
Figure 2 Physical map of Shigella atypical class 1 integron and locations of blaOXA-30 and aadA genes. |
Quinolone And Fluoroquinolone Resistance
Resistance To (Fluoro)quinolones Due To Chromosomal Target–Site Mutations
Corresponding subunits for DNA gyrase and topoisomerase IV are gyrA, gyrB, parC, and parE, encoded by genes gyrA, gyrB, parC, and parE genes, respectively. DNA gyrase consists of two gyrA subunits and two gyrB subunits, and topoisomerase IV contains two parC and two parE subunits. The most mutations have been found in a small region near the start of the gyrA gene termed a quinolone resistance–determining region (QRDR), between Ala67 and Gln107, and as reported in several studies (Table 2), most frequently mutations occur at codons 83, 87, and 211, while mutations in gyrB were detected with lower frequencies in different studies.59,60 Some researchers believe that when a single mutation occurs in gyrA, it may confer resistance to quinolones, but for decreased susceptibility to fluoroquinolones, a number of further mutations in parC and gyrA regions are needed.61,62 parC gene mutations most frequently occur at codons 80 among Shigella isolates. gyrA gene mutations have been confirmed to be much more prevalent than mutations in the gyrB gene.63,64 Most nucleotide and amino-acid changes in QRDRs of gyrA, gyrB, parC, and parE among Shigella spp. are shown in Table 2. Novel mutations in QRDRs have also been identified from different regions of the world. Two novel mutations at codons 86 and 129 in parC and a mutation in codon 211 of gyrA were first reported in S. sonnei strains recovered from China in 2009.65
 |
Table 2 Frequency Of Amino-Acid And Nucleotide Changes In The Quinolone Resistance–Determining Regions Of ShigellaIsolates In Different Parts Of The World |
Finally, two novel mutations at codons 408 and 458 in parE have recently been discovered among Shigellaspp., isolated in India in 2013 and Jiangsu Province in China in 2016.10,63 Mutation in codon 458 is believed to result in resistance to ciprofloxacin and nalidixic acid, while a single isolate with a mutation at codon 408 in parE is related to resistance to nalidixic acid, but susceptible to ciprofloxacin. It seems that both the novel mutations in parE of S. flexneri isolates may be correlated with the increased MIC for ciprofloxacin and mediate fluoroquinolone resistance. Also, in neither study were parE mutations identified among quinolone-sensitive isolates.10,63
Data presented in Table 2 show that mutation patterns in gyrA and parC genes, particularly common mutations, are similar to those reported by other studies, reflecting a universal pattern among Shigella spp. A direct contribution to quinolone and fluoroquinolone resistance by each of these mutations in chromosomal target–site mutations (QRDRs) remains unknown. However, other mechanisms may be present in Shigella isolates, and further investigations are needed.
Resistance To (Fluoro)quinolones Due To Plasmid-Mediated Resistance Mechanisms
Distribution of plasmid genes called plasmid-mediated quinolone-resistance regions (PMQRs) namely qnr (qnrA, qnrB, qnrC, qnrD, qnrS, qep, aac[6ʹ]-lb-cr) genes is the main reason for resistance to quinolones among Shigella isolates, and they are usually associated with transposable or mobile elements on plasmids.64,66 qnr genes, which are often incorporated into integrons, may allow for dissemination among Shigella and possibly other members of the Enterobacteriaceae family, and then quinolone-resistance isolates may spread across geographic regions and even across countries with population mobility.65,67 The aac(6′)-Ib-cr gene encodes an acetyltransferase associated with reduced quinolone activity, and is identified in many members of the Enterobacteriaceae family.68 PMQRs have been identified widely among human and animal isolates, and have become a pressing issue worldwide. In a study conducted in China, fluoroquinolone-resistance rates in animal isolates of S. flexneri were reported to be higher than those in human strains.68
In the US, the Shigella resistance rate to fluoroquinolones reached 87% during 2014–2015.69 Resistance of Shigella isolates to fluoroquinolone is mainly due to mutational alterations in QRDRs of DNA gyrase and topoisomerase IV genes, but PMQRs may facilitate in selection of isolates exhibiting higher levels of resistance through extrachromosomally encoded mechanisms and confer reduced susceptibility to quinolones (or fluoroquinolones).68 aac(6′)-Ib-cr and qnrS genes were first identified in isolates of S. flexneri 2a in 1998 and S. flexneri serotype 1a in 2002, respectively.70,71 Furthermore, the qnrS gene was identified in isolates of Shigella flexneri 2b in 2005 in Japan.70 Also, as reported in two previous studies, aac(6′)-Ib-cr and qnrS were predominant PMQR determinants across two provinces in China, conferring high levels of fluoroquinolone resistance.72,73 These studies indicated that aac(6′)-Ib-cr–positive Shigella isolates have been present in China for many years.70,72,73 S. flexneri serotypes 1a, 2a, 2b, 4c, and S. sonnei carrying the qnrS gene have been reported worldwide.67
Importantly, qnrS-positive isolates of Shigella, especially S. flexneri strains, show high-level resistance to fluoroquinolones, and many researchers from different part ofs the world suggest that the plasmid-mediated quinolone-resistance gene qnrS plays an essential role in reduced susceptibility of Shigella strains to fluoroquinolones.67,72,73 Indeed, qnrS plasmid could change fluoroquinolone susceptibility of S. flexneri isolates containing both gyrA83 and parC80 mutations into ciprofloxacin-resistant isolates.74 Overall, aac(6ʹ)-Ib-cr is the most prevalent gene, followed by qnrS detected in isolates of S. flexneri from the US, India, Japan, China, and Iran, and also most studies have highlighted an increased prevalence of PMQR determinants through the years.50,70,75–77 A recent study conducted in China reported that aac(6)-Ib-cr–positive isolates and qepA-positive isolates expressed high levels of quinolone resistance. This finding indicates that other mechanisms, such as reduced outer-membrane permeability, active efflux pumps, and harboring of different resistance genes, may be responsible for resistance to quinolones.18,77
Fosfomycin Resistance
Fosfomycin (Fom) is a broad-spectrum antibiotic inhibiting bacterial cell-wall biogenesis by inactivating the MurA enzyme.38,78 Despite the use of Fom in treatment of microbial infections for four decades, Fom has remained effective against common uropathogens, and Fom resistance has remained rare throughout the world.79 However, Fom resistance was observed among E. coli strains to harbor novel transferable fosfomycin-resistance determinants named FosC2 and FosA3.80 Two primary resistance mechanisms have been described for fosfomycin resistance: mutations in uhpA/T and glpT genes encoding proteins for two carrier-dependent systems responsible for fosfomycin uptake, and attainment of fosfomycin-modifying enzymes containing two kinases, FomA and FomB, and three types of metalloenzymes: FosX, FosA, and FosB.81
Fosfomycin-modifying enzymes were discovered for the first time among S. flexneri strains isolated from patients in China.38 Some studies have suggested that increasing prevalence of fosA3 was due to dissemination of IncN and IncI plasmids, facilitating its quick dispersal.38,80 Indeed, ESBL (blaCTX-M-123, blaCTX-M-55, or blaCTX-M-15) and fosA3 genes were cocarried by transconjugant plasmids from diverse incompatibility groups, and all of them contained determinants encoding resistance to cefotaxime, ceftriaxone, and fosfomycin.38,80 In this regard, conjugatable plasmids are likely to play an essential role in dissemination of fosA3 and ESBL genes among Shigella isolates with high clonal diversity, and they should be closely monitored.38
Aminoglycoside Resistance
Aminoglycosides are used to treat a wide range of infections. Aminoglycosides mediate inhibition of protein synthesis.82 Resistance to aminoglycosides is associated with enzymatic inactivation, ribosomal modification, and active efflux pumps. Among these mechanisms, aminoglycoside-modifying enzymes are the most common in the clinical setting.83–85 These enzymes are activated through three general reactions, resulting in adenylation, acetylation, or phosphorylation. Aminoglycoside adenyltransferase (aadA gene cassettes) are very common in Enterobacteriaceae, especially among Salmonella and Shigella isolates, conferring resistance to streptomycin and spectinomycin.84,86 Indeed, streptomycin resistance is strongly associated with integrons because of the high prevalence of aadA gene cassettes within class 1 and 2 integrons. A typical class 2 integron has a gene cassette of 2.2 kb with a resistance-gene arrangement (dfrA1–sat–aadA1) conferring resistance to trimethoprim, streptothricin, and spectinomycin/streptomycin, respectively, while aadA1 was absent in atypical class 2 integrons.87,88 Class 2 integrons have been identified in transposon Tn7 and predominantly inserted into chromosomes with high frequency.89 An atypical class 1 integron with an unusual 3ʹ conserved sequence carrying a estX–psp–aadA2–cmlA-aadA1–qacH cassette array has been detected among different Gram-negative species (E. coli, Shigella, and Salmonella) from different hosts (human, animal, and food), periods and geographical regions. Accordingly, horizontal transfer of these integrons by plasmids promotes spread of multiple-resistance genes in sporadic and outbreak isolates of Shigella.89–91
Many types of aadA gene cassettes have been identified among Enterobacteriaceae, but types aadA1 and aadA2 have high prevalence among Shigella isolates.84,89,92 Aminoglycoside phosphotransferases encoded by strA and strB are the most common genes dispersed among Shigella isolates by plasmids, such as IncFII and pNV-Y394.83,87,93,94 The gene encoding strA has been identified in 42.1% of Shigella isolates recovered from diarrheal patients in Pakistan.95 In a study conducted in India, 100% and 88% of S. dysenteriae type 1 and S. sonnei strains harbored strA genes, encoding resistance to streptomycin.96 The majority of Shigella isolates harbored strA and strB, along with unrelated resistance determinants, which are coded by blaTEM, blaCTX-M, qnrS, aadA1, tet(A), tet(B), catA, and catP.95,96 A 6.3 kb plasmid has been detected in the S. flexneri 3a strain and is involved in a streptomycin-resistance phenotype. This plasmid is likely to cause acquired resistance to streptomycin.97 Also, a study conducted in South Korea showed that resistance to streptomycin was mediated by strA or strB among S. sonnei isolates obtained there and revealed that tetA, strA–strB, and sul1 were encoded and present in 8.4 kb of untransferable R plasmid.98
Occurrence of aminoglycoside-resistance genes among Shigella isolates does not occur, because these drugs have long been excluded for treatment of shigellosis in different geographical areas.85 However, there is still a concern, because class 1 integrons containing the gene-cassette array of blaOXA-30 + aadA1 with complete 3′CS have been reported on plasmids in Shigella spp. isolates, Salmonella enterica serovar Typhimurium and E. coli strains, and transferable plasmids may enhance spread of resistance genes within integrin, establishing arole of plasmids in horizontal transfer of resistance genes.99
Tetracycline Resistance
Tetracyclines are used against a wide variety of diseases in humans and animals.100 Tetracycline-resistant bacteria are found in opportunistic pathogens and normal flora species. According to the study by Roberts101 and nomenclature for tetracycline resistance genes(https://faculty.washington.edu/marilynr), five tetracycline-efflux genes — tet(A), tet(B), tet(C), tet(D), and tet(G) — and one ribosomal protection protein encoded by tet(M) have been identified among Shigella isolates, most of which are encoded in transmittable elements, with extensive dissemination in different groups of bacteria. In a study, a 20.4 kb genomic island was identified encoding MDR genes, such as a wide variety of tet genes flanked by transposases.102 This identical MDR cassette was first identified in S. flexneri serotype 2a strain YSH6000 and was referred to as Shigella resistance locus–pathogenicity island.103 Interestingly, these MDR genes have recently been found in an E. coli plasmid, pRSB225, with a similar arrangement.104 The IncB/O/K/Z-type plasmid, termed p866, carrying resistance genes tet(A) and tet(B) have been identified in S. sonnei strains.105 These findings suggest that tet genes might be dispersed among other species by horizontal gene transfer.
Among 154 tetracycline-resistant isolates recovered as confirmed causes of traveler’s diarrhea in Spain, 79.2% (n=122) harbored at least tet(A) or tet(B). Combinations of tet(A) + tet(B), tet(A) + tet(G), and tet(B) + tet(G) were found in five, one, and seven isolates, respectively.106 Results of two studies revealed that tet(A) was more frequent among S. sonnei strains, whereas tet(B) was more frequent among S. flexneri strains.106,107 Also, S. sonnei and S. flexneri differed from each other in terms of prevalence of plasmid Inc groups.106 In 50 isolates of Shigella spp. identified from stool samples collected from children with diarrhea in Iran, 90% and 18% of isolates carried tetA and tetB, respectively, and no positive results were identified for tet(C) or tet(D) in this study.11 Results of the same study conducted in Iran revealed that tet(A) and tet(B) were present in 75.7% and 21.42% of Shigella spp. and that tet(A) was more frequent in S. flexneri and S. sonnei populations.108
A study carried out in Mexico identified genes tet(A), tet(B), and tet(C) in 1, 6, and 18 S. sonnei isolates, and 2, 7, and 1 S. flexneri isolates, respectively, whereas tet(D) was observed only in S. sonnei isolates (8%).109 Among 20 S. dysenteriae isolates from dysentery outbreaks obtained from different parts of India, tet(B) was more common (90%) than tet(A) (10%). In the same study, genes tet(A) and tet(B) were detected in 15% and 79% of Shigella isolates, respectively, samples of which were obtained from children with diarrhea in southern Mozambique.107 Based on recent studies,107,108,110 it seems that efflux-mediated tetracycline resistance to tetracycline in S. sonnei and S. flexneri strains may be related to expression of tet(A) and tet(B), respectively. According to tetracycline resistance–gene nomenclature (https://faculty.washington.edu/marilynr), tet(B) is able to confer resistance to minocycline, while other efflux pumps encoded in a transferable tet gene do not have such a property and may cause clonal dissemination of tet(B)-positive Shigella isolates worldwide. In general, tet(A) and tet(B) are the most prevalent tetracycline-resistance genes in Shigella spp. Other tetracycline-efflux genes, such as tet(C) or tet(D), are rarely detected alone.
Phenicol Resistance
Phenicols have used to treat Shigella infections during the past few years around the world, and application of them led to strong selective pressure for resistance to these antibiotics.111 Resistance in Shigella is associated with enzymatic inactivation of unfluorinated phenicols by chloramphenicol acetyltransferase genes encoded by variants of catA (catA1, catA2, catA3) and catB (catB2, catB3, catB7, catB8), as well as active efflux by cmlA (cmlA1, cmlA4, cmlA9) and/or fluorinated and unfluorinated phenicols (floR) by major facilitator–superfamily proteins.112
Chloramphenicol resistance in Shigella isolates is mainly associated with the presence of cat genes. Among 95 Shigella isolates collected from diarrheal patients in Pakistan, at least 69 (72.6%) were resistant to chloramphenicol. catA and catP were detected in 32 (33.68%) and 24 (25.26%) isolates, respectively, from chloramphenicol-resistant Shigella isolates.95 Among 103 S. sonnei isolates associated with several waterborne outbreaks in Taiwan, in 84% of sporadic isolates, tetB and catA genes were transferred along with 130 kb plasmids.89 The presence of catA encoding chloramphenicol O-acetyltransferase was detected and confirmed in S. flexneri strains with strA or aadA1 genes or both.96
Colistin Resistance
Colistin (polymyxin E) is a polypeptide antimicrobial agent interacting with outer membranes of Gram-negative bacteria. Since the first report of the plasmid-mediated polymyxin-resistance gene mcr-1 was published in an E. coli isolate in November 2016 in China,113 this gene has also been identified in Salmonella enterica and Klebsiella pneumoniae, but is rarely reported in other Enterobacteriaceae members here.114 This gene has now been identified in other Enterobacteriaceae genera, such as Shigella, Cronobacter, Kluyvera, and Enterobacter isolated from vegetables, the environment, food, animals, and human beings.115,116 The mechanism of resistance of mcr-1 is produced by a phosphatidylethanolamine transferase, leading to modification of lipid A present as a result of addition of phosphoethanolamine to lipid A in cell membranes of Gram-negative bacteria, resulting in more cationic LPSs and lower affinity for colistin and related polymyxins and consequently reduced antimicrobial activity.117 The presence of mcr-1 in Shigella isolates leads to a four- to eightfold increase in the MIC of polymyxin B.118 mcr-1 is located on various plasmid backbones, including IncI2, IncFI, IncHI2, IncFIB, IncP, IncY, and IncX4, sized 58–252 kb (Figure 3).114 ESBL-encoding genes or other resistance genes might coexist with it. Also, plasmid-mediated colistin-resistant S. flexneri isolates recovered from animal feces on a farm showed that it might be circulated via the fecal–oral route, at least between animals on that farm, and possibly distributed via the food-production network.118 In most reports, mcr-1 is known to be the only resistance gene for related plasmids, indicating that selective pressure associated with polymyxin is responsible for mcr-1 acquisition.114,118 It means that other plasmids conferring MDR phenotypes can be achieved from resistant S. flexneri strains. Mobile elements, such as IS and integrons, could also help isolates acquiring other resistance elements from the environment.118 A novel transposon, Tn6390 has been detected in S. flexneri C960, in which two copies of ISApl1 bracketed to the mcr-1 gene play a pivotal role in transposition of mcr-1.118 Recently, mcr-1 was identified in S. sonnei isolates from Shanghai (2010–2012) with polymyxin B resistance (MIC 4–8 μg/mL).118
 |
Figure 3 Structure of genes surrounding mcr-1 in S. flexneri. |
Sulfonamide And Trimethoprim Resistance
After the spread of trimethoprim–sulfonamide resistance in different parts of the world, these agents are currently considered ineffective for empirical therapy of shigellosis.119 Acquired resistance mechanisms have frequently been identified, mostly due to mutational or recombinational changes in target enzymes (dihydropteroate synthase and dihydrofolate reductase, respectively) or acquired resistance by drug-resistant target enzymes, such as acquired sul genes coding for drug-resistant dihydropteroate synthases or dfr genes coding for drug-resistant dihydrofolate reductases.120,121
Resistance To Trimethoprim
At least 42 dfr genes conferring trimethoprim resistance have been detected in different groups of bacteria worldwide, 12 of which have been identified in Shigella spp. Resistance to trimethoprim might be explained by the presence of integron-borne dfr genes (Figure 4). Gene cassettes within class 1 integrons detected on plasmids or chromosomes in Shigella isolates often encode resistance to trimethoprim (dfrA), streptomycin (aadA), and ampicillin (oxa-1).58,85 Class 2 integrons borne on Tn7 have often been found in Shigella spp., and gene-cassette arrays of them usually contain dfrA1, sat1, and aadA1.122
 |
Figure 4 Variable regions of class 1 (A) and class 2 (B) integrons reported in different geographic area. Horizontal arrows indicate transcriptional orientation of genes. |
The presence of dfrA1 genes among Shigella isolates is the main mechanism of trimethoprim resistance, occurring in a cassette in both class 1 and class 2 integrons (Figure 4). Two types of class 2 integrons among S. sonnei strains have been identified in Japan. One of them was typical type of class 2 integrons (2,158 bp) with dfrA1, sat1, and aadA1 cassette arrays, and the other was an atypical type of class 2 integrons (1,313 bp) carried only two gene-cassette arrays with dfrA1 and sat1.123 This integron‐associated antibiotic resistance can be transferred to other species via plasmid conjugation. dfr12–orfF–aadA2, dfr17–aadA5, and aadA1 cassette arrays carried by class 1 integrons have been recognized in S. sonnei isolates recovered from South Korea, China, Vietnam, and Australia.84,85,99 Also, dfrA1–sat1–aadA1–orfX, free aadA1, or free orfX cassette arrays carried by class 2 integrons have been detected in Senegalese Shigella spp. isolates.92 In contrast to dfrA genes, dfrB genes have not been identified among Shigella isolates. Recently, trimethoprim-resistance clones were sequenced by primer walking, and a native 6,779 bp plasmid was identified with presence of the dfrA14 gene in a sul2–strA’–dfrA14–‘strA–strB gene arrangement in S. sonnei strains, suggesting that dfrA14 was associated with a small nonconjugative plasmid.124 Class 2 integrons within a dfr1–sat2–aadA1 cassette array were predominant in S. sonnei isolates from outbreaks cases in Taiwan, while class 2 integrons were absent in sporadic cases.89
A large study conducted in South Korea analyzed 122 S. sonnei isolates collected from stool samples in different parts of the country from 1991 to 2000. Resistance to trimethoprim was associated with dfrA1 and dfrA12. dfrA1 was found as a gene cassette of Tn7 located in chromosomes, while dfrA12 was located in conjugative R plasmids as a gene cassette of class 1 integrons. Tn7 was not detected in S. sonnei isolates recovered from the 1980s, while in this study Tn7 was found in all S. sonnei isolates. The authors proposed that S. sonnei isolates carrying Tn7 were responsible for outbreaks of shigellosis in different parts of South Korea in the 2000s.98
Resistance To Sulfonamides
Since the first report of resistance to sulfonamides was found in both S. sonnei and S. flexneri isolates recovered from the early 1970s in South Korea,98,125 resistance to this antibiotic has been identified in 94 % of S. sonnei strains from the 1980s and 100% of isolates from the 2000s and 2010s in different parts of the world.2,50,118 Sulfonamide resistance is mediated by sul1, sul2, and sul3 genes, and they are common in Shigella.89 The sul1 gene is highly frequent among Shigella isolates, because it is part of the 3ʹ-conserved sequence region of class 1 integrons. Also, sul3, linked to an unusual 3ʹ-conserved segment, is associated with class 1 integrons.89,126 According to one study, sul2 is one of the three sulfonamide-resistance genes, and is usually located on large transmissible plasmids or small non-conjugative plasmids and was first detected on a small nonconjugative plasmid of E. coli.90 Since then, this gene has been found mostly on plasmids in Shigella isolates from humans in South Korea,98 Taiwan,89 Australia,127 and Bangladesh.128
Several studies have suggested that sul genes are linked to other resistance genes. The sul1 gene is often identified together with other antimicrobial-resistance genes located on gene-cassette arrays in variable regions of class 1 integrons. Class 1 integrons differ from class 2 in their excise gene cassettes, capacity to integrate, and presence of sul1 in 3′ conserved region (3′CS). In Brazil, sul1 (sulfonamide-resistant) was identified in two (3%) MDR Shigella samples, which were also positive for class 1 integrons.122 In previous studies on MDR S. sonnei isolates obtained in South Korea, resistance to sulfamethoxazole was mainly associated with sul1, located in 8.4 kb of nonconjugative R plasmid.98
Antibiotic-resistance gene clusters containing strA, strB, and sul2 are widespread among Gram-negative bacteria, particularly in Shigella isolates.124 Iqbal et al128 found sul1, sul2, sul3, integron 1, and integron 2 genes in all MDR S. flexneri 2a strains, and also found that sul2 was absent in all sulfamethoxazole-sensitive strains (n= 54), while it was present in all sulfamethoxazole-resistant strains (n=146). However, in this study, no change was observed in expressions of sul1, sul3, integron 1, or integron 2 genes in sulfamethoxazole-resistant and -sensitive S. flexneri 2a strains. Interestingly, curing of this 4.3 MDa plasmid resulted in loss of sul2 and susceptibility to sulfamethoxazole in paired strains, suggesting involvement of sul2 and this plasmid in resistance to sulfamethoxazole. In the same study, 24 S. sonnei isolates were detected to carry an atypical class 1 integron associated with sul3. The estX–psp–aadA2–cmlA1–aadA1–qacH cassette array of sul3-associated class 1 integron has been found to encode an esterase, a lipase, putative phosphoserine phosphatase/resistance to streptomycin, chloramphenicol, and quaternary amines.23
Macrolide Resistance
The American Academy of Pediatrics and the Infectious Diseases Society of America have recommended azithromycin as a medication for treatment of shigellosis in children, and also the WHO introduced it as second-line treatment in adults.129 The Centers for Disease Control and Prevention has observed resistance to azithromycin in approximately 3% of Shigella cases tested. Resistant outbreaks involving Shigella spp. isolates with reduced susceptibility to azithromycin (RSA) are more recent phenomena and continually detected in Asia, North America, the US, Australia130,131 and other geographic regions.132–134 According to updated Clinical and Laboratory Standards Institute guidelines, if MICmeasured by broth microdilution is ≤16 and ≤8 µg/mL, then epidemiological cutoff values denote susceptibility for S. sonnei and S. flexneri wild-type, and if MIC is ≥32 and ≥16 µg/mL, then susceptibility is confirmed for non–wild-type, respectively.135 Recently, several reports have suggested that resistance to azithromycin in Shigella spp. isolates is associated with presence of mphA or ermB plasmid-mediated genes or by both genes.133,136 Macrolide resistance is mediated by four main mechanisms: enzymatic inactivation by phosphotransferases encoded by mph genes or esterases encoded by ere determinants; target-site modification by an rRNA methylase encoded by erm genes; punctual mutations in rplV encoding L22 ribosomal protein, rplD encoding L4 ribosomal protein, and rrlH (23S rRNA); and drug-resistance mediated by efflux pumps, such as OmpA, OmpW, mefA, and msrA.137,138 All macrolide-resistance mechanisms can mediate resistance to azithromycin and erythromycin. The mph(A) gene was first identified in an E. coli isolate from Japan.139 Since then, this gene has been recognized among Pseudomonas spp., Aeromonas spp., Stenotrophomonas spp., Shigella spp., and other enteropathogens.137 Dissemination and acquisition of macrolide mphA resistance mechanisms in Shigella spp. has been shown to be mainly due to spread of plasmids from E. coli.140 All the discovered mphA-associated plasmids have been identified in E. coli isolates, indicating their role as a repository from which antimicrobial resistance to Shigella spp. may appear.135 This phenomenon has been previously been described in E. coli donating mphA to S. sonnei.141
Additionally, Shigella strains with RSA have been found mostly in strains recovered from men who have sex with men (MSM; 68.8% or higher) from the Montreal region. In this study, complete sequence analysis of six selected plasmids from different serotypes of S. flexneri and S. sonnei emphasized the role of IS26 in dispersal of RSA.130 Also, in a study conducted in Taiwan, a series of clonally related azithromycin insusceptible Shigella spp. isolates was reported in relation to MSM.136 Various gene-transfer systems (mobile genetic element acquisitions) are involved in acquiring antibiotic-resistance genes, such as transposons, integrons, and conjugative plasmids. The IS26–mphA–mrx–mphR (A)–IS6100 gene cassette has been characterized in a clinical strain of S. boydii carrying the p2246–CTXM plasmid. Insertion sequences of IS6100 and IS26 have been found in the neighborhood of mphA in an S. sonnei strain recovered from France. Indeed, in addition to plasmid mobilization, dissemination, and acquisition of RSA among Shigella spp. and serotypes has also potentially occurred through IS26 mobilization.130,137
IncFI and IncFII plasmids have been shown to carry the azithromycin-resistance gene erm.135 The IncFI plasmid containing an ISCR3 insertion sequence surrounds both ermC and ermB genes and carries a blaCTX-M-24 gene downstream of an ISEcp1 element. The IncFII plasmid carries ermB and ermC genes downstream of an IS6 transposase. Both plasmids share significant DNA homology with other previously sequenced plasmids found in S. sonnei and S. flexneri serotype 3a isolates associated with development of the disease in MSM.135
Recently, a plasmid carrying azithromycin-resistance genes, namely pKSR100 (conjugative R-plasmid) in S. flexneri serotype 3a has been described to be involved in intercontinental spread of RSA among MSM-associated outbreak lineage.142 pKSR100-like plasmids have been found to be predominantly related to MSM-associated outbreak lineage in Australia and elsewhere.131 There are a considerable number of studies on the prevalence of RSA among Shigella isolates throughout the world, particularly in MSM, and demonstrated global dissemination of a multi-resistant plasmid, highly associated with MSM, which is present across different continents.131,142
Biofilm Formation–Mediated Resistance
Recently, much attention has been given to biofilm formation in bacteria, because microbial cells grown in biofilms are less sensitive to antimicrobial agents and more resistant to environmental stress such as dehydration and oxidation. Microbial infections caused by biofilm-associated Shigella spp. are global health challenges.143 Biofilm formation is regulated by a multifactorial process, with cellular adherence, exopolysaccharide secretion, and numerous gene regulations controlling detachment of bacteria from mature biofilm, and is mainly related to quorum sensing and social networking in the microbial world.144,145 Ellafi et al investigated biofilm formation by Shigella strains grown in different NaCl concentrations, and they showed that all the isolates produced biofilm. According to their study, biofilm formation is a protective system under different environmental stress conditions.146 Recent studies have demonstrated that bile salts increase the capacity of S. flexneri strains to adhere to and penetrate epithelial cells.147,148 Indeed, extended exposure of Shigella to bile salts occurred in cases of increased biofilm formation, and thus it is an important resistance mechanism for Shigella sp. Similar biofilm phenotypes have been observed for Campylobacter, Listeria, and Vibrio, demonstrating that bile salt–induced biofilm production is conserved among members of the Enterobacteriaceae family.19,148 Also, biofilm formation has been shown to require the presence of glucose, whereas biofilm diffusion requires the elimination of bile salts from the medium.19 Bacteria in the form of biofilm can be 100,000 times more resistant to antimicrobial agents than planktonic forms of bacteria in the same species.143 During biofilm formation, the effect of shf, mdoH, VpsT, and LuxR-like genes and OpgH protein expression has been confirmed among enteric bacteria, as well as Shigella.143,145 Another study described biofilm-formation potentials and pathological behaviors of various mutants S. flexneri strains with an incomplete inner core of LPS containing only Kdo moieties.
Interestingly, 1rfaC (also called waaC) mutant, with an incomplete inner core of LPS due to deficiency in Hep biosynthesis, shows strong biofilm-formation ability and considerably high invasiveness and adhesiveness to human epithelial cells compared LPS-mutant strains. However, this strategy is successful in conferring high-level resistance only in bacterial species with a deficiency in Hep synthesis of LPS.149 The relationship between biofilm formation and pathogenicity, as well as virulence factors and antimicrobial properties, has not been thoroughly studied in Shigella spp., and further studies are needed.
Therapeutics
Treatment for Shigella infections is recommended to prevent spread of infection to others and to shorten disease duration. According to current WHO guidelines and a systematic review, the use of fluoroquinolones (first-line, preferably ciprofloxacin), cephalosporins (second-line), and β-lactams (second-line) for 7–10 days is recommended for treatment of shigellosis.4,111 In regions known to have high rates of resistance to ciprofloxacin, azithromycin may be considered appropriate second-line therapy. Cefixime is also a good alternative, although its use should be balanced with respect to risk of developing antimicrobial resistance and spread of ESBL.111 Conventional antibiotic therapeutics against shigellosis have become increasingly inefficient, due to the increase in number of MDR strains. However, no well-designed in vitro or in vivo combinations of antimicrobial agents have been performed to evaluate different antibiotic-class regimens for treating infections caused by Shigella. There has only been a related study on an antimicrobial-sensitivity case series reporting resistance in different regions and treatment outcomes for infections caused by Shigella.4,111 In general, it has been observed that there is a decrease in susceptibility to first- and second-line agents. As such, there is an urgent need for development of novel therapeutic strategies for treatment of MDR Shigella infections.111
Alternative Therapeutics
Natural And Organic Products
Natural products are small molecules produced naturally by microbial agents, plants, and animals that have been demonstrated to be useful in treating Shigella infections. Biotherapeutic agents (preferably probiotics) have been suggested in prevention of antibiotic-induced diarrhea, and are also an alternative therapeutic choice for treatment of gastroenteritis infectious.150 Bacteria and yeast are the most frequent microorganisms used as probiotics. Several mechanisms have been suggested for antimicrobial activity of bacteria toward enteric bacterial pathogens, including production of undissociated organic acids, organic acid molecules, and bacteriocin, competition for adhesion sites, and coaggregation with pathogens.151
Pretreatment of cells with bacterial components and products obtained from Lactobacillus rhamnosus and L. acidophilus results in interference with Shigella adherence and internalization into host cells and leads to an absence of IL8 expression, substantiating attenuation of inflammatory response during aggregate pretreatment. Lactobacilli have been shown to regulate cytokine production and stimulate the immune system.152 Time-kill methodology has shown that viability of S. sonnei decreased after contact with cell-free culture supernatants of lactobacilli, which could be potentially used as probiotic strains in food industry.151 Zhang et al selected a total of 91 lactobacilli for antimicrobial activity against Shigella isolates, among which 16 lactobacilli displayed potent antibacterial activity against S. sonnei strains. The nature of these antimicrobial agents was studied and found to be dependent on production of organic acids.153 Also, other studies have indicated that lactobacilli and lactic and acetic acid bacteria possess high activity against MDR Shigella pathogenic strains, and that they can be the best candidate for probiotics.152,154,155 Saccharomyces boulardii is a thermophilic and nonpathogenic yeast showing antagonistic activity against several bacterial pathogens, such as enterohemorrhagic and enteropathogenic E. coli, Vibrio cholera, Salmonella typhimurium, and S. flexneri.156
In one study, the effect of aqueous ethanol extract of Euphorbia prostrata was investigated in vitro and in vivo on bacterial growth of S. dysenteriae type 1 and found to be effective against Shigella isolates, with MIC and minimal bactericidal concentration of 3,500–12,000 µg/mL.157 The preventive role of orally administered Aloe barbadensis Miller (Aloe vera)–supplemented probiotic lassi (APL) was determined for S. dysenteriae infection in mice, with a significant (P<0.05) decrease found in Shigella counts (log CFU/mL) and immunoprotective effects of APL against S. dysenteriae.158 Antivirulence activity of a boiling black tea (Camellia sinensis) extract was shown to reduce expression of virulence traits by S. dysenteriae, as shown by decreased bacterium-survival strategies, and also an enhancement was found in innate immunoresponse against Shigella isolates.159 Antishigellosis activity of Picralima nitida Stapf (Apocynaceae) extract has been found to be effective against S. dysenteriae type I strains, and MIC and minimal bactericidal concentration were 800 and 6,400 μg/mL, respectively.160 In vitro antibacterial activity of methyl gallate isolated from Terminalia chebula has been shown to cause total disintegration of outer and inner membranes and leakage of cytoplasmic contents of MDR S. dysenteriae. Viable intracellular S. dysenteriae reduced in a time-dependent manner in the presence of methyl gallate and decreased to zero within 20 hours.161 In another study, antimicrobial activity of thyme oil and ciprofloxacin and their synergistic effects were evaluated, and the combination displayed differing degrees of effects on microbial cell formation based on results obtained from scanning electron microscopy and transmission electron microscopy. In vitro and in vivo synergy between them showed maximum growth inhibition in S. flexneri. Bacterial loads in infected colons reduced as a result of treatment with thyme oil, while the conventional drug failed to heal colon ulcers. Also, it decreased penetration of lamina propria by inflammatory cells.162
Antishigellosis activity of a Crinum jagus water–ethanol extract was found to be effective against S. flexneri, with inhibition of diameter by 18.90 (0.39 mg/mL) and 25.36 (200 mg/mL) mm, respectively. Indeed, Crinum jagus extract drastically decreased (P<0.01) diarrheal stool emission and microbial load and also reduced IFNγ, IL2, IgM, IgA, and motilin blood levels in S. flexneri–induced diarrheic rats.163 An in vitro study on bovine lactoferrin recognized it as a coadministered adjuvant therapeutic in antibiotic therapy against Shigella isolates. Some strains of Shigella show a twofold or more decrease in their ampicillin MIC values in the presence of bovine lactoferrin.164 In vitro data showed that antibacterial activity of gallic acid inhibited the effect on biofilm formation and reduced the number of viable S. flexneri strains. Indeed, gallic acid inhibited biofilm formation in S. flexneri by regulating expression of the mdoH gene. mdoH is essential for glucosyltransferase activity and osmoregulated periplasmic glucans synthesis, as they both contribute to biofilm formation and develop antibiotic resistance in pathogenesis. Inhibition of mdoH can help in treatment of S. flexneri biofilm.143 Organic acids, such as citric, acetic, lactic, and malic acid, are natural substances categorized as “generally recognized as safe” according to the US Food and Drug Administration. They have antimicrobial activity and are widely used to inactivate bacteria. Results of a study showed that organic acids, carvacrol, and their combination were useful against S. sonnei. However, S. sonnei was shown to decrease to 4.53 and 3.25 log CFU/mL using 0.5% w:v malic, lactic acid, respectively, indicating the synergistic effect of combination therapy.165 A previous study reported the effects of dithiocarbamate transition-metal complexes on survival and recovery of pathogenic bacteria, and they are very attractive and novel pharmaceutical targets for control and management of antibiotic-resistant bacteria, as well as Shigella isolates.166
Novel Therapeutic Strategies For Shigella Treatment
Nanoparticles
Nanoparticles (NPs) have gained growing importance in recent years and shown broad-spectrum antibacterial activity against pathogenic bacteria, due to their bactericidal characteristics.167 NPs usually destroy bacterial targets with a damaging effect on membrane load cells and their integrity, along with generation of free oxygen radicals. Commonly, they can be delivered efficiently as antimicrobial agents. Recently, copper oxide NPs have been recognized as an antimicrobial agent for treatment of Shigella. MIC and minimum bactericidal concentration of copper oxide NPs were 2,500 μg/mL and ≤5,000 IU/mL, respectively, in treatment of S. sonnei using 33 nm NPs. The study also showed that smaller copper oxide NPs had stronger antibacterial effects than larger NPs at a specific time and concentration.168 Iron is a biocompatible element, and can be directly used for treatment of many types of microbial pathogens. In one study, antimicrobial properties of Fe2O3 and Ag–Fe2O3 NPs against S. dysenteriae strains was evaluated, and both Fe2O3 and Ag–Fe2O3 NPs were shown to have antimicrobial effects, with antimicrobial activity of Ag–Fe2O3 NPs much more than that of Fe2O3 NPs alone.169 The bactericidal effect of iron oxide NPs has been determined, with values of 50–100 μg/mL against Gram-positive and Gram-negative bacteria, as well as S. dysenteriae and E. coli.167 The use of nanoantibiotic formulations is another strategy for treatment of drug-resistant Shigella. Mukherjee et al reported on the synthesis of a nanosized form of tetracycline by loading it in calcium phosphate NPs and showed that this treatment significantly decreased incidence of colon-length shortening, mushy-stool excretion, weight loss, and microbial colonization in gastrointestinal tracts of Shigella-infected mice. Immunohistological research has shown that as a result of tetracycline–calcium phosphate NP treatment, changes in morphology and level of inflammatory cytokines IL1β, IFNγ, and TNFα in intestinal tissue of mice caused by shigellosis were reverted to almost normal characteristics.170 Silver NPs (AgNPs) are characterized by their broad-spectrum bactericidal toxic effects against broad-spectrum bacterial pathogens. Omara et al tested AgNPs against pathogenic Salmonella and Shigella strains recovered from layer-poultry farms. AgNPs at a concentration of 16 μg/mL were found to have both bacteriostatic and bactericidal effects against Salmonella and Shigella isolates.171 Although NPs have shown high antibacterial activity during in -vitro and in -vivo experiments, future studies and judiciously performed clinical trials are required to achieve a better understanding of their potential side effects and clear regulatory guidelines.
Phage Therapy
Bacteriophages (phages) are the most abundant organisms killing bacteria a through lysis mechanism. They can be recovered from various environments. Phage therapy has earned increasing attention due to several advantages, including high specificity to target bacteria without effects on normal microflora of the human body, replication at infection site, bactericidal activity against antibiotic-resistant bacteria, and fewer side effects than other therapies. Phages are self-limiting, because phages remain at a shallow level on target sites after killing bacterial targets.172 Phage therapy of shigellosis was discovered by a French microbiologist named d'Herelle, who described efficacy of phages in curing symptoms of dysentery.173 Use of phages for treatment of MDR S. dysenteriae recovered from wastewater has been investigated as an alternative to antibiotics.172 Another study showed that a virulent phage named pSb1 was able to infect all the S. boydii strains and had productive lytic activity against them. Also, results indicated that pSb1 might be a member of an N4-like phage group and might have potential applications as an alternative option for treatment of shigellosis.174 Regular targeting of only a subgroup of strains within one bacterial species or closely related species without causing distortions in the gut microbiota is one of the benefits of phage treatment over antibiotic therapy in treatment of shigellosis.175 Numerous animal have studies demonstrated that phages are able to survive in experimental animals with dysentery. Despite no reports on significant undesirable reactions during the long history of phage therapy in humans,176 phage treatments still need to overcome admission constraints in the main medical repertoire.
Vaccine Strategies
Varieties of candidate vaccines have been developed to prevent infection by Shigella spp., most of which are currently under evaluation for safety and immunogenicity. However, there is no licensed vaccine available against this pathogen. At present, studies in humans and animals have shown that protection by vaccination is possible. Potential candidates for Shigella vaccines include glycoconjugate vaccines, such as recombinant glycoconjugate, synthetic glycoconjugate, O-polysaccharide covalently linked to immunogenic carrier proteins,177,178 virG-based live attenuated (WRSS1, WRSs3, WRSf3, WRSf2G12, WRSf2G15and WRSd1),179,180 recombinant outer-membrane proteins,181 live attenuated vaccines,182,183 invasion-plasmid antigens B, C, and D,184 DNA-based vaccines, Ty21a typhoid vaccine expressing Shigella LPS,185 recombinant probiotic–based candidates,186 and whole-cell-killed and Shigella trivalent inactivated whole-cell and heat-killed multiserotype Shigella,187,188 as well as novel antigen candidates, such as triacylated S-LPS, subcellular complexes purified from virulent cultures (Invaplex),189 GMMA protein particles,190 and an OMV-NP vaccine (Table 3).191
 |  |  |  |
Table 3 Overview Of Shigella Vaccines In A Aifferent Phase |
Live attenuated strains such as S. dysenteriae type 1 WRSd1, S. sonnei WRSS1, WRSs2, WRSs3, and some whole-cell-killed and novel antigen candidates are now developing, and were safe and immunogenic in a phase I trial (more details are presented in Table 3). Among possible Shigella candidate vaccines, GMMA protein particles, live attenuated Shigella flexneri 2a SC602, and S. dysenteriae type 1 SC599 strains have entered phase II clinical evaluation, and only glycoconjugate candidates have already undergone Phase III trials, with other formulations still under development in patients with shigellosis.192,193 As shown in Table 3, many studies have demonstrated humoral response as a main value of an immunoresponse to vaccination, and also fever and transient diarrhea have been repaorted as the most frequent complications in relation to some vaccine candidates in clinical investigations (Table 3). Finally, it seems that development and evaluation of multivalent candidates may provide a means for protection against serogroups/serotypes of Shigella in future.
Conclusion
In this review, antibiotic-resistance mechanisms and therapeutic strategies have been summarized regarding Shigella infection. Antimicrobial resistance in Shigella spp. is multifactorial in that it can occur through innate, acquired, or adaptive mechanisms, and infections resulting from it are exceedingly difficult to treat. Drug resistance among Shigella spp. occurs as a result of selective pressure and horizontal resistance-gene transmission. Accordingly, there is an urgent need to comprehensively learn and understand mechanisms of drug resistance among Shigella isolates, in order to develop antishigellosis drugs. The multifarious nature of antibiotic-resistance mechanisms contributes in an increase in the number of MDR strains and causes conventional antibiotic therapeutics to be highly inefficient against shigellosis. Despite intensive research efforts in the last few decades related to determination of antimicrobial resistance, researchers have not been able to find the best solution to control MDR isolates. Also, direct contributions to antibiotic resistance by many antimicrobial-resistant mechanisms remains unknown, showing a need for continuous monitoring of a broader range of associated mechanisms contributing to development of MDR strains. Most candidate vaccines cause an improvement in host immunity along with prevention of infection, but some have low efficiency and host challenge, which must be thoroughly overcome before being used against Shigella infection. More understanding on the host–microbe relationship is needed to develop novel therapeutic strategies to fight shigellosis. Development of innovative therapeutic and alternative strategies is also required for prevention and treatment of Shigella infections.
Author Contributions
Both authors conceptualized and designed the review, contributed towards data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kahsay AG, Muthupandian S. A review on sero diversity and antimicrobial resistance patterns of Shigella species in Africa, Asia and South America, 2001–2014. BMC Res Notes. 2016;9(1):422. doi:10.1186/s13104-016-2236-7
2. Puzari M, Sharma M, Chetia P. Emergence of antibiotic resistant Shigella species: a matter of concern. J Infect Public Health. 2018;11(4):451–454. doi:10.1016/j.jiph.2017.09.025
3. Qu F, Bao C, Chen S, et al. Genotypes and antimicrobial profiles of Shigella sonnei isolates from diarrheal patients circulating in Beijing between 2002 and 2007. Diagn Microbiol Infect Dis. 2012;74(2):166–170. doi:10.1016/j.diagmicrobio.2012.06.026
4. Organization WH. Antimicrobial Resistance: Global Report on Surveillance. World Health Organization; 2014.
5. Grimont F, Lejay-Collin M, Talukder KA, et al. Identification of a group of shigella-like isolates as Shigella boydii 20. J Med Microbiol. 2007;56(6):749–754. doi:10.1099/jmm.0.46818-0
6. Raja SB, Murali MR, Devaraj SN. Differential expression of ompC and ompF in multidrug-resistant Shigella dysenteriae and Shigella flexneri by aqueous extract of Aegle marmelos, altering its susceptibility toward beta-lactam antibiotics. Diagn Microbiol Infect Dis. 2008;61(3):321–328. doi:10.1016/j.diagmicrobio.2008.02.006
7. Taneja N, Mewara A. Shigellosis: epidemiology in India. Indian J Med Res. 2016;143(5):565–576. doi:10.4103/0971-5916.187104
8. Qiu S, Wang Y, Xu X, et al. Multidrug-resistant atypical variants of Shigella flexneri in China. Emerg Infect Dis. 2013;19(7):1147. doi:10.3201/eid1909.130682
9. Traa BS, Walker CLF, Munos M, Black RE. Antibiotics for the treatment of dysentery in children. Int J Epidemiol. 2010;39(suppl_1):i70–i84. doi:10.1093/ije/dyq024
10. Bhattacharya D, Bhattacharya H, Thamizhmani R, et al. Shigellosis in Bay of Bengal Islands, India: clinical and seasonal patterns, surveillance of antibiotic susceptibility patterns, and molecular characterization of multidrug-resistant Shigella strains isolated during a 6-year period from 2006 to 2011. Eur J Clin Microbiol Infect Dis. 2014;33(2):157–170. doi:10.1007/s10096-013-1937-2
11. Shahsavan S, Owlia P, Lari AR, Bakhshi B, Nobakht M. Investigation of efflux-mediated tetracycline resistance in Shigella isolates using the inhibitor and real time polymerase chain reaction method. Iran J Pathol. 2017;12(1):53.
12. Poole K. Outer membranes and efflux: the path to multidrug resistance in gram-negative bacteria. Curr Pharm Biotechnol. 2002;3(2):77–98. doi:10.2174/1389201023378454
13. Kar AK, Ghosh AS, Chauhan K, et al. Involvement of a 43-kilodalton outer membrane protein in beta-lactam resistance of Shigella dysenteriae. Antimicrob Agents Chemother. 1997;41(10):2302–2304.
14. Ghosh AS, Kar AK, Kundu M. Impaired imipenem uptake associated with alterations in outer membrane proteins and lipopolysaccharides in imipenem-resistant Shigella dysenteriae. J Antimicrob Chemother. 1999;43(2):195–201. doi:10.1093/jac/43.2.195
15. Tran EN, Papadopoulos M, Morona R. Relationship between O-antigen chain length and resistance to colicin E2 in Shigella flexneri. Microbiology. 2014;160(Pt 3):589–601. doi:10.1099/mic.0.074955-0
16. Koseoglu VK, Hall CP, Rodriguez-Lopez EM, Agaisse H. The autotransporter IcsA promotes Shigella flexneri biofilm formation in presence of bile salts. Infect Immun. 2019. doi:10.1128/iai.00861-18
17. Sun J, Deng Z, Yan A. Bacterial multidrug efflux pumps: mechanisms, physiology and pharmacological exploitations. Biochem Biophys Res Commun. 2014;453(2):254–267. doi:10.1016/j.bbrc.2014.05.090
18. Yang H, Duan G, Zhu J, et al. The AcrAB-TolC pump is involved in multidrug resistance in clinical Shigella flexneri isolates. Microb Drug Resist. 2008;14(4):245–249. doi:10.1089/mdr.2008.0847
19. Nickerson KP, Chanin RB, Sistrunk JR, et al. Analysis of Shigella flexneri resistance, biofilm formation, and transcriptional profile in response to bile salts. Infect Immun. 2017;85(6). doi:10.1128/iai.01067-16
20. Kim JY, Kim SH, Jeon SM, Park MS, Rhie HG, Lee BK. Resistance to fluoroquinolones by the combination of target site mutations and enhanced expression of genes for efflux pumps in Shigella flexneri and Shigella sonnei strains isolated in Korea. Clin Microbiol Infect. 2008;14(8):760–765. doi:10.1111/j.1469-0691.2008.02033.x
21. Taneja N, Mishra A, Kumar A, Verma G, Sharma M. Enhanced resistance to fluoroquinolones in laboratory-grown mutants & clinical isolates of Shigella due to synergism between efflux pump expression & mutations in quinolone resistance determining region. Indian J Med Res. 2015;141(1):81–89.
22. Edgar R, Bibi E. MdfA, an Escherichia coli multidrug resistance protein with an extraordinarily broad spectrum of drug recognition. J Bacteriol. 1997;179(7):2274–2280. doi:10.1128/jb.179.7.2274-2280.1997
23. Rahman M, Shoma S, Rashid H, Siddique AK, Nair GB, Sack DA. Extended-spectrum beta-lactamase-mediated third-generation cephalosporin resistance in Shigella isolates in Bangladesh. J Antimicrob Chemother. 2004;54(4):846–847. doi:10.1093/jac/dkh413
24. Fortineau N, Naas T, Gaillot O, Nordmann P. SHV-type extended-spectrum beta-lactamase in a Shigella flexneri clinical isolate. J Antimicrob Chemother. 2001;47(5):685–688. doi:10.1093/jac/47.5.685
25. Matar GM, Jaafar R, Sabra A, et al. First detection and sequence analysis of the bla-CTX-M-15 gene in lebanese isolates of extended-spectrum-beta-lactamase-producing Shigella sonnei. Ann Trop Med Parasitol. 2007;101(6):511–517. doi:10.1179/136485907x193860
26. Sabra AH, Araj GF, Kattar MM, et al. Molecular characterization of ESBL-producing Shigella sonnei isolates from patients with bacilliary dysentery in Lebanon. J Infect Dev Ctries. 2009;3(4):300–305.
27. Alici O, Acikgoz ZC, Gocer S, Gamberzade S, Karahocagil MK. [Short communication: prevalence of extended spectrum beta-lactamases in gram negative rods: data of 2001–2004 period]. Mikrobiyol Bul. 2006;40(4):355–361.
28. Kim S, Kim J, Kang Y, Park Y, Lee B. Occurrence of extended-spectrum beta-lactamases in members of the genus Shigella in the Republic of Korea. J Clin Microbiol. 2004;42(11):5264–5269. doi:10.1128/jcm.42.11.5264-5269.2004
29. Xiong Z, Li T, Xu Y, Li J. Detection of CTX-M-14 extended-spectrum beta-lactamase in shigella sonnei isolates from China. J Infect. 2007;55(5):e125–e128. doi:10.1016/j.jinf.2007.07.017
30. Vasilev V, Japheth R, Yishai R, et al. Extended-spectrum beta-lactamase-producing Shigella strains in Israel, 2000–2004. Eur J Clin Microbiol Infect Dis. 2007;26(3):189–194. doi:10.1007/s10096-007-0263-y
31. Ranjbar R, Ghazi FM, Farshad S, et al. The occurrence of extended-spectrum beta-lactamase producing Shigella spp. in Tehran, Iran. Iran J Microbiol. 2013;5(2):108–112.
32. Andres P, Petroni A, Faccone D, et al. Extended-spectrum beta-lactamases in Shigella flexneri from Argentina: first report of TOHO-1 outside Japan. Int J Antimicrob Agents. 2005;25(6):501–507. doi:10.1016/j.ijantimicag.2005.02.016
33. Bialvaei AZ, Pourlak T, Aghamali M, Asgharzadeh M, Gholizadeh P, Kafil HS. The prevalence of CTX-M-15 extended-spectrum beta-lactamases among Salmonella spp. and Shigella spp. isolated from three Iranian hospitals. Eur J Microbiol Immunol (Bp). 2017;7(2):133–137. doi:10.1556/1886.2017.00004
34. Kim JS, Kim J, Jeon SE, et al. Complete nucleotide sequence of the IncI1 plasmid pSH4469 encoding CTX-M-15 extended-spectrum beta-lactamase in a clinical isolate of Shigella sonnei from an outbreak in the Republic of Korea. Int J Antimicrob Agents. 2014;44(6):533–537. doi:10.1016/j.ijantimicag.2014.08.007
35. Kim JS, Kim S, Park J, et al. Plasmid-mediated transfer of CTX-M-55 extended-spectrum beta-lactamase among different strains of Salmonella and Shigella spp. in the Republic of Korea. Diagn Microbiol Infect Dis. 2017;89(1):86–88. doi:10.1016/j.diagmicrobio.2017.03.014
36. Li J, Li B, Ni Y, Sun J. Molecular characterization of the extended-spectrum beta-lactamase (ESBL)-producing Shigella spp. in Shanghai. Eur J Clin Microbiol Infect Dis. 2015;34(3):447–451. doi:10.1007/s10096-014-2244-2
37. Holt KE, Thieu Nga TV, Thanh DP, et al. Tracking the establishment of local endemic populations of an emergent enteric pathogen. Proc Natl Acad Sci U S A. 2013;110(43):17522–17527. doi:10.1073/pnas.1308632110
38. Liu Y, Cheng Y, Yang H, et al. Characterization of extended-spectrum beta-lactamase genes of Shigella flexneri isolates with fosfomycin resistance from patients in China. Ann Lab Med. 2017;37(5):415–419. doi:10.3343/alm.2017.37.5.415
39. Zhang R, Zhou HW, Cai JC, et al. Serotypes and extended-spectrum beta-lactamase types of clinical isolates of Shigella spp. from the Zhejiang province of China. Diagn Microbiol Infect Dis. 2011;69(1):98–104. doi:10.1016/j.diagmicrobio.2010.08.027
40. Liu G, Qian H, Tang B, et al. Prevalence and characterisation of third-generation cephalosporin-resistant Shigella flexneri isolates from Jiangsu Province, China, 2013–2015. J Glob Antimicrob Resist. 2018;15:283–287. doi:10.1016/j.jgar.2018.08.012
41. Rashid H, Rahman M. Possible transfer of plasmid mediated third generation cephalosporin resistance between Escherichia coli and Shigella sonnei in the human gut. Infect Genet Evol. 2015;30:15–18. doi:10.1016/j.meegid.2014.11.023
42. Qu F, Ying Z, Zhang C, et al. Plasmid-encoding extended-spectrum beta-lactamase CTX-M-55 in a clinical Shigella sonnei strain, China. Future Microbiol. 2014;9(10):1143–1150. doi:10.2217/fmb.14.53
43. Thamizhmani R, Rhagavan R, Sugunan AP, Vijayachari P. VIM- and IMP-type metallo-beta-lactamase-producing Shigella spp. in childhood diarrhea from Andaman Islands. Infect Dis (Lond). 2015;47(10):749–750. doi:10.3109/23744235.2015.1022874
44. O’Hara K, Haruta S, Sawai T, Tsunoda M, Iyobe S. Novel metallo beta-lactamase mediated by a Shigella flexneri plasmid. FEMS Microbiol Lett. 1998;162(2):201–206. doi:10.1111/j.1574-6968.1998.tb12999.x
45. Iyobe S, Kusadokoro H, Ozaki J, et al. Amino acid substitutions in a variant of IMP-1 metallo-beta-lactamase. Antimicrob Agents Chemother. 2000;44(8):2023–2027. doi:10.1128/aac.44.8.2023-2027.2000
46. Sambe-Ba B, Seck A, Wane AA, Fall-Niang NK, Gassama-Sow A. [Sensitivity to antibiotics and genetic support to resistance of Shigella flexneri strains isolated in Dakar from 2001 to 2010]. Bull Soc Pathol Exot. 2013;106(2):89–94. doi:10.1007/s13149-013-0283-z
47. Huang IF, Chiu CH, Wang MH, Wu CY, Hsieh KS, Chiou CC. Outbreak of dysentery associated with ceftriaxone-resistant Shigella sonnei: first report of plasmid-mediated CMY-2-type AmpC beta-lactamase resistance in S. sonnei. J Clin Microbiol. 2005;43(6):2608–2612. doi:10.1128/jcm.43.6.2608-2612.2005
48. Tajbakhsh M, Garcia Migura L, Rahbar M, et al. Antimicrobial-resistant Shigella infections from Iran: an overlooked problem? J Antimicrob Chemother. 2012;67(5):1128–1133. doi:10.1093/jac/dks023
49. Ayala AT, Acuna HM, Calvo MT, Morales JL, Chacon EC. [Emergence of CMY-2-type plasmid-mediated AmpC beta-lactamase in Shigella sonnei and Salmonella spp. in Costa Rica, 2003–2015]. Rev Panam Salud Publica. 2016;40(1):70–75.
50. Zamanlou S, Ahangarzadeh Rezaee M, Aghazadeh M, Ghotaslou R, Babaie F, Khalili Y. Characterization of integrons, extended-spectrum beta-lactamases, AmpC cephalosporinase, quinolone resistance, and molecular typing of Shigella spp. from Iran. Infect Dis (Lond). 2018;50(8):616–624. doi:10.1080/23744235.2018.1455222
51. Zhang CL, Liu QZ, Wang J, Chu X, Shen LM, Guo YY. Epidemic and virulence characteristic of Shigella spp. with extended-spectrum cephalosporin resistance in Xiaoshan District, Hangzhou, China. BMC Infect Dis. 2014;14:260. doi:10.1186/1471-2334-14-260
52. Cui X, Wang J, Yang C, et al. Prevalence and antimicrobial resistance of shigella flexneri serotype 2 variant in China. Front Microbiol. 2015;6:435. doi:10.3389/fmicb.2015.00435
53. Anandan S, Muthuirulandi Sethuvel DP, Gajendiren R, Verghese VP, Walia K, Veeraraghavan B. Molecular characterization of antimicrobial resistance in clinical Shigella isolates during 2014 and 2015: trends in South India. Germs. 2017;7(3):115–122. doi:10.18683/germs.2017.1116
54. Toro C, Farfán M, Contreras I, et al. Genetic analysis of antibiotic-resistance determinants in multidrug-resistant Shigella strains isolated from Chilean children. Epidemiol Infect. 2005;133(1):81–86. doi:10.1017/S0950268804003048
55. Pfeifer Y, Cullik A, Witte W. Resistance to cephalosporins and carbapenems in gram-negative bacterial pathogens. Int J Med Microbiol. 2010;300(6):371–379. doi:10.1016/j.ijmm.2010.04.005
56. Cui X, Yang C, Wang J, et al. Antimicrobial resistance of Shigella flexneri serotype 1b isolates in China. PLoS One. 2015;10(6):e0129009. doi:10.1371/journal.pone.0129009
57. Siu LK, Lo JY, Yuen KY, Chau PY, Ng MH, Ho PL. beta-lactamases in Shigella flexneri isolates from Hong Kong and Shanghai and a novel OXA-1-like beta-lactamase, OXA-30. Antimicrob Agents Chemother. 2000;44(8):2034–2038. doi:10.1128/aac.44.8.2034-2038.2000
58. Navia MM, Capitano L, Ruiz J, et al. Typing and characterization of mechanisms of resistance of Shigella spp. isolated from feces of children under 5 years of age from Ifakara, Tanzania. J Clin Microbiol. 1999;37(10):3113–3117.
59. Azmi IJ, Khajanchi BK, Akter F, et al. Fluoroquinolone resistance mechanisms of Shigella flexneri isolated in Bangladesh. PLoS One. 2014;9(7):e102533. doi:10.1371/journal.pone.0102533
60. Hu LF, Li JB, Ye Y, Li X. Mutations in the GyrA subunit of DNA gyrase and the ParC subunit of topoisomerase IV in clinical strains of fluoroquinolone-resistant Shigella in Anhui, China. J Microbiol. 2007;45(2):168–170.
61. Ramana J, Ramana J. Structural Insights into the fluoroquinolone resistance mechanism of Shigella flexneri DNA gyrase and topoisomerase IV. Microb Drug Resist. 2016;22(5):404–411. doi:10.1089/mdr.2015.0018
62. Nuesch-Inderbinen M, Heini N, Zurfluh K, Althaus D, Hachler H, Stephan R. Shigella antimicrobial drug resistance mechanisms, 2004–2014. Emerg Infect Dis. 2016;22(6):1083–1085. doi:10.3201/eid2206.152088
63. Xue C, Cai J, Kang H, et al. Two novel mutations in parE among Shigella flexneri isolated from Jiangsu Province of China, 2016. Ann Transl Med. 2018;6(15):306. doi:10.21037/atm.2018.07.11
64. Dutta S, Kawamura Y, Ezaki T, Nair GB, Iida K, Yoshida S. Alteration in the GyrA subunit of DNA gyrase and the ParC subunit of topoisomerase IV in quinolone-resistant Shigella dysenteriae serotype 1 clinical isolates from Kolkata, India. Antimicrob Agents Chemother. 2005;49(4):1660–1661. doi:10.1128/aac.49.4.1660-1661.2005
65. Gu B, Qin TT, Fan WT, et al. Novel mutations in gyrA and parC among Shigella sonnei strains from Jiangsu Province of China, 2002–2011. Int J Infect Dis. 2017;59:44–49. doi:10.1016/j.ijid.2017.03.023
66. Das A, Natarajan M, Mandal J, Herman C. The emergence of quinolone resistant Shigella sonnei, Pondicherry, India. PLoS One. 2016;11(8):e0160290. doi:10.1371/journal.pone.0160290
67. Zhang WX, Chen HY, Tu LH, Xi MF, Chen M, Zhang J. Fluoroquinolone resistance mechanisms in Shigella isolates in Shanghai, China, between 2010 and 2015. Microb Drug Resist. 2019;25(2):212–218. doi:10.1089/mdr.2018.0113
68. Zhu Z, Cao M, Zhou X, Li B, Zhang J. Epidemic characterization and molecular genotyping of Shigella flexneri isolated from calves with diarrhea in Northwest China. Antimicrob Resist Infect Control. 2017;6:92. doi:10.1186/s13756-017-0252-6
69. Bowen A, Hurd J, Hoover C, et al. Importation and domestic transmission of Shigella sonnei resistant to ciprofloxacin - United States, May 2014-February 2015. MMWR Morb Mortal Wkly Rep. 2015;64(12):318–320.
70. Pu XY, Pan JC, Wang HQ, Zhang W, Huang ZC, Gu YM. Characterization of fluoroquinolone-resistant Shigella flexneri in Hangzhou area of China. J Antimicrob Chemother. 2009;63(5):917–920. doi:10.1093/jac/dkp087
71. Hata M, Suzuki M, Matsumoto M, et al. Cloning of a novel gene for quinolone resistance from a transferable plasmid in Shigella flexneri 2b. Antimicrob Agents Chemother. 2005;49(2):801–803. doi:10.1128/aac.49.2.801-803.2005
72. Qin T, Qian H, Fan W, et al. Newest data on fluoroquinolone resistance mechanism of Shigella flexneri isolates in Jiangsu Province of China. Antimicrob Resist Infect Control. 2017;6:97. doi:10.1186/s13756-017-0249-1
73. Pu XY, Pan JC, Zhang W, Zheng W, Wang HQ, Gu YM. Quinolone resistance-determining region mutations and the plasmid-mediated quinolone resistance gene qnrS played important roles in decreased susceptibility to fluoroquinolones among Shigella isolates in southeast China between 1998 and 2013. Int J Antimicrob Agents. 2015;45(4):438–439. doi:10.1016/j.ijantimicag.2014.12.004
74. Pu XY, Gu Y, Li J, Song SJ, Lu Z. Characterization of the complete sequences and stability of plasmids carrying the genes aac(6ʹ)-Ib-cr or qnrS in Shigella flexneri in the Hangzhou area of China. World J Microbiol Biotechnol. 2018;34(6):72. doi:10.1007/s11274-018-2454-3
75. Ferjani S, Saidani M, Amine FS, Boutiba-Ben Boubaker I. Prevalence and characterization of plasmid-mediated quinolone resistance genes in extended-spectrum beta-lactamase-producing Enterobacteriaceae in a Tunisian hospital. Microb Drug Resist. 2015;21(2):158–166. doi:10.1089/mdr.2014.0053
76. Yaghoubi S, Ranjbar R, Soltan Dallal MM, Shirazi MH, Sharifi-Yazdi MK. Frequency of mutations in quinolone resistance-determining regions and plasmid-mediated quinolone resistance in Shigella isolates recovered from pediatric patients in Tehran, Iran: an Overlooked Problem. Microb Drug Resist. 2018;24(6):699–706. doi:10.1089/mdr.2017.0155
77. Yang H, Duan G, Zhu J, Zhang W, Xi Y, Fan Q. Prevalence and characterisation of plasmid-mediated quinolone resistance and mutations in the gyrase and topoisomerase IV genes among Shigella isolates from Henan, China, between 2001 and 2008. Int J Antimicrob Agents. 2013;42(2):173–177. doi:10.1016/j.ijantimicag.2013.04.026
78. Perea EJ, Torres MA, Borobio MV. Synergism of fosfomycin-ampicillin and fosfomycin-chloramphenicol against Salmonella and Shigella. Antimicrob Agents Chemother. 1978;13(5):705–709. doi:10.1128/aac.13.5.705
79. Falagas ME, Kastoris AC, Kapaskelis AM, Karageorgopoulos DE. Fosfomycin for the treatment of multidrug-resistant, including extended-spectrum beta-lactamase producing, Enterobacteriaceae infections: a systematic review. Lancet Infect Dis. 2010;10(1):43–50. doi:10.1016/s1473-3099(09)70325-1
80. Wachino J, Yamane K, Suzuki S, Kimura K, Arakawa Y. Prevalence of fosfomycin resistance among CTX-M-producing Escherichia coli clinical isolates in Japan and identification of novel plasmid-mediated fosfomycin-modifying enzymes. Antimicrob Agents Chemother. 2010;54(7):3061–3064. doi:10.1128/aac.01834-09
81. Silver LL. Fosfomycin: mechanism and resistance. Cold Spring Harb Perspect Med. 2017;7(2):a025262. doi:10.1101/cshperspect.a025262
82. Fourmy D, Yoshizawa S, Puglisi JD. Paromomycin binding induces a local conformational change in the A-site of 16 S rRNA. J Mol Biol. 1998;277(2):333–345. doi:10.1006/jmbi.1997.1551
83. Shaw PC, Liang AC, Kam KM, Ling JM. Presence of strA-strB gene within a streptomycin-resistance operon in a clinical isolate of Shigella flexneri. Pathology. 1996;28(4):356–358. doi:10.1080/00313029600169344
84. McIver CJ, White PA, Jones LA, et al. Epidemic strains of Shigella sonnei biotype g carrying integrons. J Clin Microbiol. 2002;40(4):1538–1540. doi:10.1128/jcm.40.4.1538-1540.2002
85. Iversen J, Sandvang D, Srijan A, Cam PD, Dalsgaard A. Characterization of antimicrobial resistance, plasmids, and gene cassettes in Shigella spp. from patients in vietnam. Microb Drug Resist. 2003;9(Suppl 1):S17–S24. doi:10.1089/107662903322541856
86. Michael GB, Schwarz S. Antimicrobial resistance in zoonotic nontyphoidal Salmonella: an alarming trend? Clin Microbiol Infect. 2016;22(12):968–974. doi:10.1016/j.cmi.2016.07.033
87. Dutta S, Jain P, Nandy S, Matsushita S, Yoshida S. Molecular characterization of serologically atypical provisional serovars of Shigella isolates from Kolkata, India. J Med Microbiol. 2014;63(Pt 12):1696–1703. doi:10.1099/jmm.0.081307-0
88. Ranjbar R, Aleo A, Giammanco GM, Dionisi AM, Sadeghifard N, Mammina C. Genetic relatedness among isolates of Shigella sonnei carrying class 2 integrons in Tehran, Iran, 2002–2003. BMC Infect Dis. 2007;7:62. doi:10.1186/1471-2334-7-62
89. Chang CY, Lu PL, Lin CC, Lee TM, Tsai MY, Chang LL. Integron types, gene cassettes, antimicrobial resistance genes and plasmids of Shigella sonnei isolates from outbreaks and sporadic cases in Taiwan. J Med Microbiol. 2011;60(Pt 2):197–204. doi:10.1099/jmm.0.022517-0
90. Antunes P, Machado J, Sousa JC, Peixe L. Dissemination of sulfonamide resistance genes (sul1, sul2, and sul3) in Portuguese Salmonella enterica strains and relation with integrons. Antimicrob Agents Chemother. 2005;49(2):836–839. doi:10.1128/aac.49.2.836-839.2005
91. Bischoff KM, White DG, Hume ME, Poole TL, Nisbet DJ. The chloramphenicol resistance gene cmlA is disseminated on transferable plasmids that confer multiple-drug resistance in swine Escherichia coli. FEMS Microbiol Lett. 2005;243(1):285–291. doi:10.1016/j.femsle.2004.12.017
92. Gassama Sow A, Aidara-Kane A, Barraud O, Gatet M, Denis F, Ploy MC. High prevalence of trimethoprim-resistance cassettes in class 1 and 2 integrons in Senegalese Shigella spp isolates. J Infect Dev Ctries. 2010;4(4):207–212.
93. Muthuirulandi Sethuvel DP, Anandan S, Devanga Ragupathi NK, et al. IncFII plasmid carrying antimicrobial resistance genes in Shigella flexneri: vehicle for dissemination. J Glob Antimicrob Resist. 2019;16:215–219. doi:10.1016/j.jgar.2018.10.014
94. Parajuli P, Deimel LP, Verma NK. Genome analysis of Shigella flexneri serotype 3b strain SFL1520 reveals significant horizontal gene acquisitions including a multidrug resistance cassette. Genome Biol Evol. 2019;11(3):776–785. doi:10.1093/gbe/evz026
95. Tariq A, Haque A, Ali A, et al. Molecular profiling of antimicrobial resistance and integron association of multidrug-resistant clinical isolates of Shigella species from Faisalabad, Pakistan. Can J Microbiol. 2012;58(9):1047–1054. doi:10.1139/w2012-085
96. Pazhani GP, Niyogi SK, Singh AK, et al. Molecular characterization of multidrug-resistant Shigella species isolated from epidemic and endemic cases of shigellosis in India. J Med Microbiol. 2008;57(Pt 7):856–863. doi:10.1099/jmm.0.2008/000521-0
97. Barman S, Chatterjee S, Chowdhury G, et al. Plasmid-mediated streptomycin and sulfamethoxazole resistance in Shigella flexneri 3a. Int J Antimicrob Agents. 2010;36(4):348–351. doi:10.1016/j.ijantimicag.2010.06.037
98. Seol SY, Kim YT, Jeong YS, et al. Molecular characterization of antimicrobial resistance in Shigella sonnei isolates in Korea. J Med Microbiol. 2006;55(Pt 7):871–877. doi:10.1099/jmm.0.46441-0
99. Pan JC, Ye R, Meng DM, Zhang W, Wang HQ, Liu KZ. Molecular characteristics of class 1 and class 2 integrons and their relationships to antibiotic resistance in clinical isolates of Shigella sonnei and Shigella flexneri. J Antimicrob Chemother. 2006;58(2):288–296. doi:10.1093/jac/dkl228
100. Hartman AB, Essiet II, Isenbarger DW, Lindler LE. Epidemiology of tetracycline resistance determinants in Shigella spp. and enteroinvasive Escherichia coli: characterization and dissemination of tet(A)-1. J Clin Microbiol. 2003;41(3):1023–1032. doi:10.1128/jcm.41.3.1023-1032.2003
101. Roberts MC. Tetracycline resistance determinants: mechanisms of action, regulation of expression, genetic mobility, and distribution. FEMS Microbiol Rev. 1996;19(1):1–24. doi:10.1111/j.1574-6976.1996.tb00251.x
102. Parajuli P, Rajput MI, Verma NK. Plasmids of Shigella flexneri serotype 1c strain Y394 provide advantages to bacteria in the host. BMC Microbiol. 2019;19(1):86. doi:10.1186/s12866-019-1455-1
103. Luck SN, Turner SA, Rajakumar K, Sakellaris H, Adler B. Ferric dicitrate transport system (Fec) of Shigella flexneri 2a YSH6000 is encoded on a novel pathogenicity island carrying multiple antibiotic resistance genes. Infect Immun. 2001;69(10):6012–6021. doi:10.1128/iai.69.10.6012-6021.2001
104. Wibberg D, Szczepanowski R, Eikmeyer F, Puhler A, Schluter A. The IncF plasmid pRSB225 isolated from a municipal wastewater treatment plant’s on-site preflooder combining antibiotic resistance and putative virulence functions is highly related to virulence plasmids identified in pathogenic E. coli isolates. Plasmid. 2013;69(2):127–137. doi:10.1016/j.plasmid.2012.11.001
105. Allue-Guardia A, Koenig SSK, Quiros P, Muniesa M, Bono JL, Eppinger M. Closed genome and comparative phylogenetic analysis of the clinical multidrug resistant Shigella sonnei strain 866. Genome Biol Evol. 2018;10(9):2241–2247. doi:10.1093/gbe/evy168
106. Pons MJ, Torrents de la Pena A, Mensa L, et al. Differences in tetracycline resistance determinant carriage among Shigella flexneri and Shigella sonnei are not related to different plasmid Inc-type carriage. J Glob Antimicrob Resist. 2018;13:131–134. doi:10.1016/j.jgar.2017.12.015
107. Mandomando I, Jaintilal D, Pons MJ, et al. Antimicrobial susceptibility and mechanisms of resistance in Shigella and Salmonella isolates from children under five years of age with diarrhea in rural Mozambique. Antimicrob Agents Chemother. 2009;53(6):2450–2454. doi:10.1128/aac.01282-08
108. Alizadeh-Hesar M, Bakhshi B, Najar-Peerayeh S. Clonal dissemination of a single Shigella sonnei strain among Iranian children during Fall 2012 in Tehran, I.R. Iran. Infect Genet Evol. 2015;34:260–266. doi:10.1016/j.meegid.2015.06.024
109. Martinez-Salazar JM, Alvarez G, Gomez-Eichelmann MC. Frequency of four classes of tetracycline resistance determinants in Salmonella and Shigella spp. clinical isolates. Antimicrob Agents Chemother. 1986;30(4):630–631. doi:10.1128/aac.30.4.630
110. Ledov VA, Golovina ME, Markina AA, et al. Highly homogenous tri-acylated S-LPS acts as a novel clinically applicable vaccine against Shigella flexneri 2a infection. Vaccine. 2019;37(8):1062–1072. doi:10.1016/j.vaccine.2018.12.067
111. Williams PCM, Berkley JA. Guidelines for the treatment of dysentery (shigellosis): a systematic review of the evidence. Paediatr Int Child Health. 2018;38(sup1):S50–s65. doi:10.1080/20469047.2017.1409454
112. Schwarz S, Kehrenberg C, Doublet B, Cloeckaert A. Molecular basis of bacterial resistance to chloramphenicol and florfenicol. FEMS Microbiol Rev. 2004;28(5):519–542. doi:10.1016/j.femsre.2004.04.001
113. Liu YY, Wang Y, Walsh TR, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis. 2016;16(2):161–168. doi:10.1016/s1473-3099(15)00424-7
114. Pham Thanh D, Thanh Tuyen H, Nguyen Thi Nguyen T, et al. Inducible colistin resistance via a disrupted plasmid-borne mcr-1 gene in a 2008 vietnamese Shigella sonnei isolate. J Antimicrob Chemother. 2016;71(8):2314–2317. doi:10.1093/jac/dkw173
115. Schwarz S, Johnson AP. Transferable resistance to colistin: a new but old threat. J Antimicrob Chemother. 2016;71(8):2066–2070. doi:10.1093/jac/dkw274
116. Poirel L, Jayol A, Nordmann P. Polymyxins: antibacterial activity, susceptibility testing, and resistance mechanisms encoded by plasmids or chromosomes. Clin Microbiol Rev. 2017;30(2):557–596. doi:10.1128/cmr.00064-16
117. Hinchliffe P, Yang QE, Portal E, et al. Insights into the mechanistic basis of plasmid-mediated colistin resistance from crystal structures of the catalytic domain of MCR-1. Sci Rep. 2017;7:39392. doi:10.1038/srep39392
118. Ma Q, Huang Y, Wang J, et al. Multidrug-resistant Shigella sonnei carrying the plasmid-mediated mcr-1 gene in China. Int J Antimicrob Agents. 2018;52(1):14–21. doi:10.1016/j.ijantimicag.2018.02.019
119. Niyogi SK. Shigellosis. J Microbiol. 2005;43(2):133–143.
120. Skold O. Resistance to trimethoprim and sulfonamides. Vet Res. 2001;32(3–4):261–273. doi:10.1051/vetres:2001123
121. Huovinen P. Resistance to trimethoprim-sulfamethoxazole. Clin Infect Dis. 2001;32(11):1608–1614. doi:10.1086/320532
122. Peirano G, Agerso Y, Aarestrup FM, dos Prazeres Rodrigues D. Occurrence of integrons and resistance genes among sulphonamide-resistant Shigella spp. from Brazil. J Antimicrob Chemother. 2005;55(3):301–305. doi:10.1093/jac/dki012
123. Ahmed AM, Furuta K, Shimomura K, Kasama Y, Shimamoto T. 1Genetic characterization of multidrug resistance in Shigella spp. from Japan. J Med Microbiol. 2006;55(Pt 12):1685–1691. doi:10.1099/jmm.0.46725-0
124. Miranda A, Avila B, Diaz P, et al. Emergence of plasmid-borne dfrA14 trimethoprim resistance gene in Shigella sonnei. Front Cell Infect Microbiol. 2016;6:77. doi:10.3389/fcimb.2016.00077
125. Chun D-K, Seol S-Y. Drug resistance of Shigella and Salmonella and the inhibition and elimination of drug resistance. J Korean Soc Microbiol. 1979;14(1):27–37.
126. Summers AO. Genetic linkage and horizontal gene transfer, the roots of the antibiotic multi-resistance problem. Anim Biotechnol. 2006;17(2):125–135. doi:10.1080/10495390600957217
127. Nigro SJ, Hall RM. GIsul2, a genomic island carrying the sul2 sulphonamide resistance gene and the small mobile element CR2 found in the enterobacter cloacae subspecies cloacae type strain ATCC 13047 from 1890, Shigella flexneri ATCC 700930 from 1954 and acinetobacter baumannii ATCC 17978 from 1951. J Antimicrob Chemother. 2011;66(9):2175–2176. doi:10.1093/jac/dkr230
128. Iqbal MS, Rahman M, Islam R, et al. Plasmid-mediated sulfamethoxazole resistance encoded by the sul2 gene in the multidrug-resistant Shigella flexneri 2a isolated from patients with acute diarrhea in Dhaka, Bangladesh. PLoS One. 2014;9(1):e85338. doi:10.1371/journal.pone.0085338
129. Salah M, Shtayeh I, Ghneim R, et al. Evaluation of Shigella species azithromycin CLSI epidemiological cutoff values and macrolide resistance genes. J Clin Microbiol. 2019;57(4). doi:10.1128/jcm.01422-18
130. Yousfi K, Gaudreau C, Pilon PA, et al. Genetic mechanisms behind the spread of reduced susceptibility to azithromycin in Shigella strains isolated from men who have sex with men in Quebec, Canada. Antimicrob Agents Chemother. 2019;63(2). doi:10.1128/aac.01679-18
131. Ingle DJ, Easton M, Valcanis M, et al. Co-circulation of multidrug-resistant Shigella among men who have sex with men, Australia. Clin Infect Dis. 2019. doi:10.1093/cid/ciz005
132. Borg ML, Modi A, Tostmann A, et al. Ongoing outbreak of Shigella flexneri serotype 3a in men who have sex with men in England and Wales, data from 2009–2011. Euro Surveill. 2012;17(13):20137.
133. Gaudreau C, Barkati S, Leduc JM, Pilon PA, Favreau J, Bekal S. Shigella spp. with reduced azithromycin susceptibility, Quebec, Canada, 2012–2013. Emerg Infect Dis. 2014;20(5):854–856. doi:10.3201/eid2005.130966
134. Sjolund Karlsson M, Bowen A, Reporter R, et al. Outbreak of infections caused by Shigella sonnei with reduced susceptibility to azithromycin in the United States. Antimicrob Agents Chemother. 2013;57(3):1559–1560. doi:10.1128/aac.02360-12
135. Darton TC, Tuyen HT, The HC, et al. Azithromycin resistance in Shigella spp. in Southeast Asia. Antimicrob Agents Chemother. 2018;62(4). doi:10.1128/aac.01748-17
136. Liao YS, Liu YY, Lo YC, Chiou CS. Azithromycin-nonsusceptible Shigella flexneri 3a in men who have sex with men, Taiwan, 2015–2016. Emerg Infect Dis. 2016;23(2):345–346. doi:10.3201/eid2302.161260
137. Boumghar-Bourtchai L, Mariani-Kurkdjian P, Bingen E, et al. Macrolide-resistant Shigella sonnei. Emerg Infect Dis. 2008;14(8):1297–1299. doi:10.3201/eid1408.080147
138. Zhang C, Zhang R, Yu Q, Chu X, Sun J, Liu Q. Decreased susceptibility to azithromycin among clinical Shigella isolates from China. Microb Drug Resist. 2017;23(5):596–601. doi:10.1089/mdr.2016.0134
139. Healy M, Huong J, Bittner T, et al. Microbial DNA typing by automated repetitive-sequence-based PCR. J Clin Microbiol. 2005;43(1):199–207. doi:10.1128/jcm.43.1.199-207.2005
140. Mannion AJ, Martin HR, Shen Z, et al. Plasmid-mediated quinolone resistance in Shigella flexneri isolated from Macaques. Front Microbiol. 2018;9:311. doi:10.3389/fmicb.2018.00311
141. Phuc Nguyen MC, Woerther PL, Bouvet M, Andremont A, Leclercq R, Canu A. Escherichia coli as reservoir for macrolide resistance genes. Emerg Infect Dis. 2009;15(10):1648–1650. doi:10.3201/eid1510.090696
142. Baker KS, Dallman TJ, Ashton PM, et al. Intercontinental dissemination of azithromycin-resistant shigellosis through sexual transmission: a cross-sectional study. Lancet Infect Dis. 2015;15(8):913–921. doi:10.1016/s1473-3099(15)00002-x
143. Kang J, Liu L, Liu M, Wu X, Li J. Antibacterial activity of gallic acid against Shigella flexneri and its effect on biofilm formation by repressing mdoH gene expression. Food Control. 2018;94:147–154. doi:10.1016/j.foodcont.2018.07.011
144. Agle ME, Blaschek HP. Shigella: Survival on Produce and Biofilm Formation. Oxford: Blackwell Publishing Ltd. 2006:19–46.
145. Dorman MJ, Dorman CJ. Regulatory hierarchies controlling virulence gene expression in Shigella flexneri and vibrio cholerae. Front Microbiol. 2018;9:2686. doi:10.3389/fmicb.2018.02686
146. Ellafi A, Abdallah FB, Lagha R, Harbi B, Bakhrouf A. Biofilm production, adherence and morphological alterations of Shigella spp. Under salt conditions. Annals of microbiology. 2011;61(4):741–747. doi:10.1007/s13213-010-0190-5
147. Olive AJ, Kenjale R, Espina M, Moore DS, Picking WL, Picking WD. Bile salts stimulate recruitment of IpaB to the Shigella flexneri surface, where it colocalizes with IpaD at the tip of the type III secretion needle. Infect Immun. 2007;75(5):2626–2629. doi:10.1128/iai.01599-06
148. Barta ML, Guragain M, Adam P, et al. Identification of the bile salt binding site on IpaD from Shigella flexneri and the influence of ligand binding on IpaD structure. Proteins. 2012;80(3):935–945.
149. Xu D, Zhang W, Zhang B, Liao C, Shao Y. Characterization of a biofilm-forming Shigella flexneri phenotype due to deficiency in Hep biosynthesis. PeerJ. 2016;4:e2178. doi:10.7717/peerj.2178
150. Filho-Lima JV, Vieira EC, Nicoli JR. Antagonistic effect of Lactobacillus acidophilus, Saccharomyces boulardii and Escherichia coli combinations against experimental infections with Shigella flexneri and Salmonella enteritidis subsp. typhimurium in gnotobiotic mice. J Appl Microbiol. 2000;88(3):365–370. doi:10.1046/j.1365-2672.2000.00973.x
151. Zhang Y, Zhang L, Du M, et al. Antimicrobial activity against Shigella sonnei and probiotic properties of wild lactobacilli from fermented food. Microbiol Res. 2011;167(1):27–31. doi:10.1016/j.micres.2011.02.006
152. Moorthy G, Murali MR, Niranjali Devaraj S. Lactobacilli inhibit Shigella dysenteriae 1 induced pro-inflammatory response and cytotoxicity in host cells via impediment of Shigella-host interactions. Dig Liver Dis. 2010;42(1):33–39. doi:10.1016/j.dld.2009.04.021
153. Zhang YC, Zhang LW, Ma W, et al. Screening of probiotic lactobacilli for inhibition of Shigella sonnei and the macromolecules involved in inhibition. Anaerobe. 2012;18(5):498–503. doi:10.1016/j.anaerobe.2012.08.007
154. Mirnejad R, Vahdati AR, Rashidiani J, Erfani M, Piranfar V. The antimicrobial effect of lactobacillus casei culture supernatant against multiple drug resistant clinical isolates of Shigella sonnei and Shigella flexneri in vitro. Iran Red Crescent Med J. 2013;15(2):122–126. doi:10.5812/ircmj.7454
155. Ayeni AO, Ayeni FA. Antagonistic effects of lactic and acetic acid bacteria on Shigella sp. SS10 in co-culture. TAF Preventive Medicine Bulletin. 2016;15(1):27–31. doi:10.5455/pmb.1-1438753866
156. Mumy KL, Chen X, Kelly CP, McCormick BA. Saccharomyces boulardii interferes with Shigella pathogenesis by postinvasion signaling events. Am J Physiol Gastrointest Liver Physiol. 2008;294(3):G599–G609. doi:10.1152/ajpgi.00391.2007
157. Rene K, Hortense G, Pascal W, et al. Activity of aqueous ethanol extract of Euphorbia prostrata ait on Shigella dysenteriae type 1-induced diarrhea in rats. Indian J Pharmacol. 2007;39(5):240–244. doi:10.4103/0253-7613.37275
158. Hussain SA, Patil GR, Reddi S, et al. Aloe vera (Aloe barbadensis Miller) supplemented probiotic lassi prevents Shigella infiltration from epithelial barrier into systemic blood flow in mice model. Microb Pathog. 2017;102:143–147. doi:10.1016/j.micpath.2016.11.023
159. Kiran S, Ratho RK, Sharma P, Harjai K, Capalash N, Tiwari RP. Effect of black tea (Camellia sinensis) on virulence traits of clinical isolates of Shigella dysenteriae and Escherichia coli EPEC P2 1265 strain. Eur Food Res Technol. 2010;231(5):763–770. doi:10.1007/s00217-010-1328-1
160. Kouitcheu LB, Tamesse JL, Kouam J. The anti-shigellosis activity of the methanol extract of picralima nitida on Shigella dysenteriae type I induced diarrhoea in rats. BMC Complement Altern Med. 2013;13:211. doi:10.1186/1472-6882-13-211
161. Acharyya S, Sarkar P, Saha DR, Patra A, Ramamurthy T, Bag PK. Intracellular and membrane-damaging activities of methyl gallate isolated from Terminalia chebula against multidrug-resistant Shigella spp. J Med Microbiol. 2015;64(8):901–909. doi:10.1099/jmm.0.000107
162. Allam NG, Eldrieny EA, Mohamed AZ. Effect of combination therapy between thyme oil and ciprofloxacin on ulcer-forming Shigella flexneri. J Infect Dev Ctries. 2015;9(5):486–495. doi:10.3855/jidc.6302
163. Noubissi PA, Fokam Tagne MA, Fankem GO, Ngakou Mukam J, Wambe H, Kamgang R. Effects of crinum jagus water/ethanol extract on Shigella flexneri-induced diarrhea in rats. Evid Based Complement Alternat Med. 2019;2019:9537603. doi:10.1155/2019/9537603
164. Mosquito S, Zegarra G, Villanueva C, Ruiz J, Ochoa TJ. Effect of bovine lactoferrin on the minimum inhibitory concentrations of ampicillin and trimethoprim-sulfamethoxazole for clinical Shigella spp. strains. Biochem Cell Biol. 2012;90(3):412–416. doi:10.1139/o11-066
165. Chai C, Lee S, Kim J, Oh S-W. Synergistic antimicrobial effects of organic acids in combination with carvacrol against Shigella Sonnei. J Food Saf. 2016;36(3):360–366. doi:10.1111/jfs.12251
166. Balakrishnan S, Duraisamy S, Kasi M, Kandasamy S, Sarkar R, Kumarasamy A. Syntheses, physicochemical characterization, antibacterial studies on potassium morpholine dithiocarbamate nickel (II), copper (II) metal complexes and their ligands. Heliyon. 2019;5(5):e01687. doi:10.1016/j.heliyon.2019.e01687
167. Saqib S, Munis MFH, Zaman W, et al. Synthesis, characterization and use of iron oxide nano particles for antibacterial activity. Microsc Res Tech. 2019;82(4):415–420. doi:10.1002/jemt.23182
168. Babaei S, Bajelani F, Mansourizaveleh O, Abbasi A, Oubari F. A study of the bactericidal effect of copper oxide nanoparticles on Shigella Sonnei and Salmonella Typhimurium. J Babol Univ Med Sci. 2017;19(11):76–81. doi:10.18869/acadpub.jbums.19.11.76
169. Kareem ZH, Shareef HK, Alkaim AF. Evaluation of antibacterial activity of Fe2 O3 nanoparticles against Shigella dysenteriae. J Pharm Sci Res. 2018;10(8):1980–1982.
170. Mukherjee R, Dutta D, Patra M, Chatterjee B, Basu T. Nanonized tetracycline cures deadly diarrheal disease ‘shigellosis’ in mice, caused by multidrug-resistant Shigella flexneri 2a bacterial infection. Nanomedicine. 2018. doi:10.1016/j.nano.2018.11.004
171. Omara ST, Zawrah MF, Samy AA. Minimum bactericidal concentration of chemically synthesized silver nanoparticles against pathogenic Salmonella and Shigella strains isolated from layer poultry farms. J Appl Pharm Sci. 2017;7(8):214–221. doi:10.7324/JAPS.2017.70829
172. Jamal M, Chaudhry WN, Hussain T, Das CR, Andleeb S. Characterization of new myoviridae bacteriophage WZ1 against multi-drug resistant (MDR) Shigella dysenteriae. J Basic Microbiol. 2015;55(4):420–431. doi:10.1002/jobm.201400688
173. Goodridge LD. Bacteriophages for managing Shigella in various clinical and non-clinical settings. Bacteriophage. 2013;3(1):e25098. doi:10.4161/bact.25098
174. Jun JW, Yun SK, Kim HJ, Chai JY, Park SC. Characterization and complete genome sequence of a novel N4-like bacteriophage, pSb-1 infecting Shigella boydii. Res Microbiol. 2014;165(8):671–678. doi:10.1016/j.resmic.2014.09.006
175. Mai V, Ukhanova M, Reinhard MK, Li M, Sulakvelidze A. Bacteriophage administration significantly reduces Shigella colonization and shedding by Shigella-challenged mice without deleterious side effects and distortions in the gut microbiota. Bacteriophage. 2015;5(4):e1088124. doi:10.1080/21597081.2015.1088124
176. Tang SS, Biswas SK, Tan WS, Saha AK, Leo BF. Efficacy and potential of phage therapy against multidrug resistant Shigella spp. PeerJ. 2019;7:e6225. doi:10.7717/peerj.6225
177. Pozsgay V, Kubler-Kielb J, Schneerson R, Robbins JB. Effect of the nonreducing end of Shigella dysenteriae type 1 O-specific oligosaccharides on their immunogenicity as conjugates in mice. Proc Natl Acad Sci U S A. 2007;104(36):14478–14482. doi:10.1073/pnas.0706969104
178. Phalipon A, Costachel C, Grandjean C, et al. Characterization of functional oligosaccharide mimics of the Shigella flexneri serotype 2a O-antigen: implications for the development of a chemically defined glycoconjugate vaccine. J Immunol. 2006;176(3):1686–1694. doi:10.4049/jimmunol.176.3.1686
179. Ranallo RT, Fonseka S, Boren TL, et al. Two live attenuated Shigella flexneri 2a strains WRSf2G12 and WRSf2G15: a new combination of gene deletions for 2nd generation live attenuated vaccine candidates. Vaccine. 2012;30(34):5159–5171. doi:10.1016/j.vaccine.2012.05.003
180. Raqib R, Sarker P, Zaman K, et al. A phase I trial of WRSS1, a Shigella sonnei live oral vaccine in Bangladeshi adults and children. Hum Vaccin Immunother. 2019:1–12. doi:10.1080/21645515.2019.1575165.
181. Heine SJ, Diaz-McNair J, Andar AU, et al. Intradermal delivery of Shigella IpaB and IpaD type III secretion proteins: kinetics of cell recruitment and antigen uptake, mucosal and systemic immunity, and protection across serotypes. J Immunol. 2014;192(4):1630–1640. doi:10.4049/jimmunol.1302743
182. DeLaine BC, Wu T, Grassel CL, et al. Characterization of a multicomponent live, attenuated Shigella flexneri vaccine. Pathog Dis. 2016;74(5). doi:10.1093/femspd/ftw034
183. Toapanta FR, Bernal PJ, Kotloff KL, Levine MM, Sztein MB. T cell mediated immunity induced by the live-attenuated Shigella flexneri 2a vaccine candidate CVD 1208S in humans. J Transl Med. 2018;16(1):61. doi:10.1186/s12967-018-1439-1
184. Mitobe J, Sinha R, Mitra S, et al. An attenuated Shigella mutant lacking the RNA-binding protein Hfq provides cross-protection against Shigella strains of broad serotype. PLoS Negl Trop Dis. 2017;11(7):e0005728. doi:10.1371/journal.pntd.0005728
185. Wu Y, Chakravarty S, Li M, Wai TT, Hoffman SL, Sim BK. Development of a live attenuated bivalent oral vaccine against Shigella sonnei Shigellosis and Typhoid Fever. J Infect Dis. 2017;215(2):259–268. doi:10.1093/infdis/jiw528
186. Yagnik B, Sharma D, Padh H, Desai P. Immunization with r-Lactococcus lactis expressing outer membrane protein A of Shigella dysenteriae type-1: evaluation of oral and intranasal route of administration. J Appl Microbiol. 2017;122(2):493–505. doi:10.1111/jam.13353
187. Kaminski RW, Wu M, Turbyfill KR, et al. Development and preclinical evaluation of a trivalent, formalin-inactivated Shigella whole-cell vaccine. Clin Vaccine Immunol. 2014;21(3):366–382. doi:10.1128/cvi.00683-13
188. Nag D, Sinha R, Mitra S, et al. Heat killed multi-serotype Shigella immunogens induced humoral immunity and protection against heterologous challenge in rabbit model. Immunobiology. 2015;220(11):1275–1283. doi:10.1016/j.imbio.2015.07.002
189. Turbyfill KR, Clarkson KA, Vortherms AR, Oaks EV, Kaminski RW, Pasetti MF. Assembly, biochemical characterization, immunogenicity, adjuvanticity, and efficacy of Shigella artificial invaplex. mSphere. 2018;3(2). doi:10.1128/mSphere.00583-17
190. Obiero CW, Ndiaye AGW, Scire AS, et al. A phase 2a randomized study to evaluate the safety and immunogenicity of the 1790GAHB generalized modules for membrane antigen vaccine against Shigella sonnei administered intramuscularly to adults from a Shigellosis-endemic Country. Front Immunol. 2017;8:1884. doi:10.3389/fimmu.2017.01884
191. Camacho AI, Irache JM, de Souza J, Sanchez-Gomez S, Gamazo C. Nanoparticle-based vaccine for mucosal protection against Shigella flexneri in mice. Vaccine. 2013;31(32):3288–3294. doi:10.1016/j.vaccine.2013.05.020
192. Barel LA, Mulard LA. Classical and novel strategies to develop a Shigella glycoconjugate vaccine: from concept to efficacy in human. Hum Vaccin Immunother. 2019. doi:10.1080/21645515.2019.1606972
193. Passwell JH, Ashkenzi S, Banet-Levi Y, et al. Age-related efficacy of Shigella O-specific polysaccharide conjugates in 1–4-year-old Israeli children. Vaccine. 2010;28(10):2231–2235. doi:10.1016/j.vaccine.2009.12.050
194. Taneja N, Mewara A, Kumar A, Verma G, Sharma M. Cephalosporin-resistant Shigella flexneri over 9 years (2001-09) in India. J Antimicrob Chemother. 2012;67(6):1347–1353. doi:10.1093/jac/dks061
195. Nagano Y, Nagano N, Wachino J, Ishikawa K, Arakawa Y. Novel chimeric beta-lactamase CTX-M-64, a hybrid of CTX-M-15-like and CTX-M-14 beta-lactamases, found in a Shigella sonnei strain resistant to various oxyimino-cephalosporins, including ceftazidime. Antimicrob Agents Chemother. 2009;53(1):69–74. doi:10.1128/aac.00227-08
196. Schumacher H, Nir M, Mansa B, Grassy A. beta-lactamases in Shigella. Apmis. 1992;100(10):954–956.
197. Qin T, Bi R, Fan W, Kang H, Ma P, Gu B. Novel mutations in quinolone resistance-determining regions of gyrA, gyrB, parC and parE in Shigella flexneri clinical isolates from eastern Chinese populations between 2001 and 2011. Eur J Clin Microbiol Infect Dis. 2016;35(12):2037–2045. doi:10.1007/s10096-016-2761-2
198. Chowdhury FM, Rahman MZ, Sarkar MMH, et al. Protection against shigellosis caused by Shigella dysenteriae serotype 4 in guinea pigs using Escherichia albertii DM104 as a live vaccine candidate strain. Acta Microbiol Immunol Hung. 2017;64(2):151–164. doi:10.1556/030.64.2017.015
199. McKenzie R, Venkatesan MM, Wolf MK, et al. Safety and immunogenicity of WRSd1, a live attenuated Shigella dysenteriae type 1 vaccine candidate. Vaccine. 2008;26(26):3291–3296. doi:10.1016/j.vaccine.2008.03.079
200. Rahman KM, Arifeen SE, Zaman K, et al. Safety, dose, immunogenicity, and transmissibility of an oral live attenuated Shigella flexneri 2a vaccine candidate (SC602) among healthy adults and school children in Matlab, Bangladesh. Vaccine. 2011;29(6):1347–1354. doi:10.1016/j.vaccine.2010.10.035
201. Launay O, Sadorge C, Jolly N, et al. Safety and immunogenicity of SC599, an oral live attenuated Shigella dysenteriae type-1 vaccine in healthy volunteers: results of a phase 2, randomized, double-blind placebo-controlled trial. Vaccine. 2009;27(8):1184–1191. doi:10.1016/j.vaccine.2008.12.021
202. Noriega FR, Losonsky G, Wang JY, Formal SB, Levine MM. Further characterization of delta aroA delta virG Shigella flexneri 2a strain CVD 1203 as a mucosal Shigella vaccine and as a live-vector vaccine for delivering antigens of enterotoxigenic Escherichia coli. Infect Immun. 1996;64(1):23–27.
203. Nag D, Koley H, Sinha R, et al. Immunization of mice with a live transconjugant Shigella hybrid strain induced Th1 and Th17 cell-mediated immune responses and confirmed passive protection against heterologous Shigellae. Scand J Immunol. 2016;83(2):92–101. doi:10.1111/sji.12394
204. Formal SB, Hale TL, Kapfer C, et al. Oral vaccination of monkeys with an invasive Escherichia coli K-12 hybrid expressing Shigella flexneri 2a somatic antigen. Infect Immun. 1984;46(2):465–469.
205. Kim MJ, Moon Y-H, Kim H, et al. Cross-protective Shigella whole-cell vaccine with a truncated O-polysaccharide chain. Front Microbiol. 2018;9:2609. doi:10.3389/fmicb.2018.02609
206. Chakraborty S, Harro C, DeNearing B, et al. Evaluation of the safety, tolerability, and immunogenicity of an oral, inactivated whole-cell Shigella flexneri 2a vaccine in healthy adult subjects. Clin Vaccine Immunol. 2016;23(4):315–325. doi:10.1128/cvi.00608-15
207. Chitradevi STS, Kaur G, Sivaramakrishna U, Singh D, Bansal A. Development of recombinant vaccine candidate molecule against Shigella infection. Vaccine. 2016;34(44):5376–5383. doi:10.1016/j.vaccine.2016.08.034
208. Hatz CF, Bally B, Rohrer S, et al. Safety and immunogenicity of a candidate bioconjugate vaccine against Shigella dysenteriae type 1 administered to healthy adults: a single blind, partially randomized phase I study. Vaccine. 2015;33(36):4594–4601. doi:10.1016/j.vaccine.2015.06.102
209. Pozsgay V, Chu C, Pannell L, Wolfe J, Robbins JB, Schneerson R. Protein conjugates of synthetic saccharides elicit higher levels of serum IgG lipopolysaccharide antibodies in mice than do those of the O-specific polysaccharide from Shigella dysenteriae type 1. Proc Natl Acad Sci U S A. 1999;96(9):5194–5197. doi:10.1073/pnas.96.9.5194
210. Mitra S, Sinha R, Mitobe J, Koley H. Development of a cost-effective vaccine candidate with outer membrane vesicles of a tolA-disrupted Shigella boydii strain. Vaccine. 2016;34(15):1839–1846. doi:10.1016/j.vaccine.2016.02.018
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.