Back to Journals » Clinical Interventions in Aging » Volume 10
Sex differences of continuous positive airway pressure treatment on flow-mediated dilation in patients with obstructive sleep apnea syndrome
Authors Kallianos A, Panoutsopoulos A, Mermigkis C, Kostopoulos K, Papamichail C, Kokkonouzis I, Kostopoulos C, Nikolopoulos I, Papaiwannou A, Lampaki S, Organtzis J, Pitsiou G, Zarogoulidis P, Trakada G
Received 8 March 2015
Accepted for publication 5 June 2015
Published 19 August 2015 Volume 2015:10 Pages 1361—1367
DOI https://doi.org/10.2147/CIA.S84199
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Walker
Anastasios Kallianos,1 Athanasios Panoutsopoulos,1 Christoforos Mermigkis,2 Konstantinos Kostopoulos,1 Chrysanthi Papamichail,1 Ioannis Kokkonouzis,3 Christoforos Kostopoulos,1 Ioannis Nikolopoulos,4 Antonis Papaiwannou,5 Sofia Lampaki,5 John Organtzis,5 Georgia Pitsiou,5 Paul Zarogoulidis,5 Georgia Trakada1
1Sleep Disorders Unit, Department of Clinical Therapeutics, “Alexandra” General Hospital, Athens School of Medicine, 2Sleep Disorders Unit, Pulmonary Department, 401 General Army Hospital, 3Pulmonary Department, 251 Hellenic Air Force General Hospital, 4Sleep Disorders Unit, “Sotiria” Regional Chest Diseases Hospital of Athens, Athens, Greece; 5Pulmonary Department – Oncology Unit, George Papanikolaou General Hospital of Thessaloniki, Aristotle University of Thessaloniki, Thessaloniki, Greece
Introduction: There is growing research evidence suggesting the presence of endothelial dysfunction and systemic inflammation in patients with obstructive sleep apnea syndrome (OSAS). Continuous positive airway pressure (CPAP) is the most effective method for treating OSAS; nonetheless, the effects of CPAP on the aforementioned pathophysiologic pathways as well as on the systemic disease that result or coexist with the OSAS remain elusive.
Aim: To assess the effect of 3-month CPAP therapy on endothelial-dependent dilation, plasma levels of inflammatory markers, blood pressure (BP), and glucose control on male and female patients with OSAS.
Methods: Our study group consisted of 40 (24 males and 16 females) patients with no prior history of cardiovascular disease, with an apnea–hypopnea index ≥15, who were assigned to receive CPAP treatment. Measurements of flow-mediated dilation (FMD), 24-hour ambulatory BP, and blood analysis were performed at baseline and 3 months after CPAP therapy.
Results: Baseline FMD values were negatively correlated with the apnea–hypopnea index (r=−0.55, P=0.001). After 3 months of CPAP, there was an increase in the FMD values (5.40%±2.91% vs 3.13%±3.15%, P<0.05) and a significant reduction in the patients’ 24-hour systolic BP (122.82±11.88 mmHg vs 130.24±16.75 mmHg, P<0.05), diastolic BP (75.44±9.14 mmHg vs 79.68±11.09 mmHg, P<0.05), and pulse pressure (47.38±9.77 mmHg vs 52.72±11.38 mmHg, P<0.05); daytime systolic BP (125.76±12.69 mmHg vs 132.55±17.00 mmHg, P<0.05) and diastolic BP (77.88±10.39 mmHg vs 82.25±11.01 mmHg, P<0.05); nighttime systolic BP (118.17±13.16 mmHg vs 126.22±17.42 mmHg, P<0.05) and pulse pressure (46.61±10.76 mmHg vs 52.66±11.86 mmHg, P<0.05); and C-reactive protein and HbA1c levels (0.40 [0.40–0.70] mg/L vs 0.60 [0.40–0.84] mg/L and 5.45%±0.70% vs 5.95%±1.08%, respectively; P<0.05). When divided by sex, only male patients produced similar statistically significant results, while female patients failed to show such associations.
Conclusion: Our results suggest that CPAP therapy improves the endothelial function, the BP, and the glucose control in male patients with OSAS. Further research is warranted in order to verify these results and to further elucidate the impact of CPAP on the cardiovascular risk of male and female patients with OSAS.
Keywords: obstructive sleep apnea syndrome, CPAP, CRP, blood pressure, glucose control
Introduction
Obstructive sleep apnea syndrome (OSAS) is a high prevalent sleep disorder characterized by recurrent episodes of upper airway obstruction and subsequent recurrent arousal during sleep.1 It is estimated that up to 5% of adults in western countries have OSAS.2 According to published data, OSAS is an independent risk factor for hypertension and coronary artery disease.3,4 Emerging studies suggest that the repetitive episodes of hypoxia and reoxygenation, in manner similar to that of the ischemia/reperfusion injury model, promote the activation of proinflammatory pathways and disrupt the normal endothelial function.5,6 These mechanisms play a crucial role in the development of adverse cardiovascular events.
Continuous positive airway pressure (CPAP) is the most effective method for treating OSAS and alleviating the patients’ symptoms.7 Nonetheless, the effects of CPAP therapy in the aforementioned pathophysiologic pathways as well as in the systemic disease that result or coexist with the OSAS require further elucidation. Furthermore, due to the various effects of sex hormones on the endothelial function in women, the majority of studies addressing the vascular endothelial function in OSAS have focused on men. The aim of this study was to assess the effect of 3-month CPAP therapy on endothelial-dependent dilation, plasma levels of inflammatory markers, and blood pressure (BP) and glucose control on male and female patients with OSAS.
Methods
This study was conducted in the sleep laboratory of the department of clinical therapeutics in the “Alexandra” Hospital, Athens Medical School. Informed consent was obtained from each patient, and approval was also obtained for the conduct of this study from the Investigational Review Board (IRB) of “Alexandra” General Hospital. Patients with suspected OSAS, who were referred to our department from March 2008 to March 2009, were recruited in the study.
All patients underwent attended overnight polysomnography. The determination of sleep stages, arousals, and respiratory events was performed according to the American Academy of Sleep Medicine (AASM) 2007 criteria.8 Subjects with an apnea–hypopnea index (AHI) ≥15 were assigned to receive CPAP treatment and formed our study group. Patients with known cardiovascular, neuromuscular, or pulmonary disease were excluded from the study. All female patients were in the postmenopausal phase of life, and during our study period, they received no hormone replacement therapy.
A high-resolution 12.0 MHz transducer ultrasound was used to measure right and left common carotid artery diameter as well as brachial artery diameter at rest during reactive hyperemia. All patients fasted for at least 8 hours prior to the measurement. The assessment was carried out in a quiet room, at a stable temperature of 22°C–24°C, by an experienced physician, who was blinded to the patient’s sleep recordings and blood analysis. Reactive hyperemia was induced by inflation of a BP cuff, which was placed on the lower part of the arm and inflated to 250 mmHg followed by release after 5 minutes. Brachial artery diameter was measured 40 seconds and 60 seconds after cuff deflation. Flow-mediated dilation (FMD) was calculated as the ratio of change in diameter (maximum [of two measurements] - baseline) over baseline value. Endothelial function measurements were reassessed after 3 months of CPAP therapy.
At baseline and 3 months after CPAP treatment, all patients had a complete physical examination, including measurement of the body mass index (BMI; calculated as kilograms per meter squared). Full blood analysis, including blood cell count, C-reactive protein (CRP), erythrocyte sedimentation rate, glucose, total cholesterol, triglycerides, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and the concentration of glycated hemoglobin (HbA1c), was also performed prior and 3 months after CPAP.
Furthermore, all subjects underwent 24-hour ambulatory BP monitoring. Measurements were performed on a working day, and the subjects were asked to continue their normal daily activities. BP and heart rate readings were obtained every 30 minutes. Patients were instructed to take a diary of their activities and time of retiring to bed. BP variability was calculated as the standard deviation (SD) of the systolic and diastolic BP, during the whole day, but separately during daytime and nighttime. The recordings were repeated after 3 months of CPAP treatment.
Statistical analysis
A standard statistical software package SPSS (SPSS Inc., Chicago, IL, USA) was used in the analysis. Descriptive statistics were calculated for all variables. Categorical variables were analyzed with the chi-square test or Fisher’s exact test as appropriate. The one-sample Kolmogorov–Smirnov test was used to test if a variable was normally distributed. Normally distributed data were analyzed with the t-test, while in the absence of normal distribution, the Mann–Whitney test was used. Normally distributed data are shown as mean ± SD, while in the absence of normal distribution, values are presented as medians (25th percentile, 75th percentile). Paired-samples t-test and Wilcoxon test were used for comparison of the patients’ endothelium-dependent values, 24-hour ambulatory BP readings, and blood analysis at baseline and 3 months after CPAP therapy. The correlations between continuous variables were assessed with the Pearson’s or Spearman’s correlation test. P-values less than 0.05 were considered statistically significant. All statistical tests were also performed for male and female subjects separately.
Results
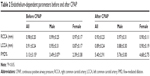
A total of 40 patients (24 males and 16 females) with a mean age of 53±12 years were recruited to the study. Patients had a mean BMI of 32±3 kg/m2, which did not change significantly during our study period. Correlation coefficients between baseline values of CRP and FMD and patients’ age, BMI, and sleep parameters are presented in Table 1. Pearson’s correlation showed that baseline FMD values were negatively correlated with AHI (r=−0.55, P=0.001). When divided by sex, only male patients produced similar statistically significant results, while female patients failed to show such associations. Patients’ endothelium-dependent values, 24-hour ambulatory BP readings, and blood analysis as a whole and separately for each sex at baseline and 3 months after CPAP therapy are presented in Tables 2–4, respectively. Overall statistical analysis revealed a significant increase in the FMD values (5.40%±2.91% vs 3.13%±3.15%, P<0.05), and the percent change in FMD values after CPAP treatment was positively correlated with the percent of time <90% oxygen saturation (r=0.53, P=0.02) (Table 5). The patients’ 24-hour systolic BP (122.82±11.88 mmHg vs 130.24±16.75 mmHg), diastolic BP (75.44±9.14 mmHg vs 79.68±11.09 mmHg), and pulse pressure (PP) (47.38±9.77 mmHg vs 52.72±11.38 mmHg); daytime systolic BP (125.76±12.69 mmHg vs 132.55±17.00 mmHg) and diastolic BP (77.88±10.39 mmHg vs 82.25±11.01 mmHg); and nighttime systolic BP (118.17±13.16 mmHg vs 126.22±17.42 mmHg) and PP (46.61±10.76 mmHg vs 52.66±11.86 mmHg) showed a significant decrease (P<0.05). Furthermore, after 3 months of CPAP treatment, the values of CRP and HbA1c were significantly decreased (0.40 [0.40–0.70] mg/L vs 0.60 [0.40–0.84] mg/L and 5.45%±0.70% vs 5.95%±1.08%, respectively; P<0.05). In sex-stratified analysis, male patients showed a significant increase in the FMD values (5.76%±3.00% vs 3.49%±3.07%, P<0.05) and the percent change of FMD after CPAP treatment was positively correlated with the AHI (r=0.53, P=0.04). Moreover, after 3 months of CPAP treatment, patients’ 24-hour systolic BP (124.24±5.68 mmHg vs 131.14±12.83 mmHg) and diastolic BP (77.72±9.10 mmHg vs 82.06±11.18 mmHg) and nighttime systolic BP (117.02±9.47 mmHg vs 126.67±16.21 mmHg) and PP (44.87±9.37 mmHg vs 49.38±11.13 mmHg), as well as the values of CRP (0.40 [0.40–0.47] mg/L vs 0.48 [0.40–0.70] mg/L) and HbA1c (5.31%±0.65% vs 5.71%±0.68%) were significantly decreased (P<0.05). Female patients showed a trend toward increased levels of FMD values and decreased levels of BP measurements and HbA1c and CRP values; nonetheless, these associations were not statistically significant.
Discussion
This single-center study demonstrated that overall CPAP treatment increases the FMD, decreases the levels of CRP and HbA1c, and improves BP measurements in obstructive sleep apnea (OSA) patients. Previous studies have excluded women with OSAS due to the various effects of the menstrual cycle on the endothelial function. In order to avoid any potential bias caused by the different hormonal status, we choose to study only postmenopausal female patients. In sex-stratified analysis, only male patients provided with statistically significant results. Female patients showed a trend toward improved FMD, CRP, HbA1c values, and BP measurements; yet this trend failed to reach statistical significance, a finding possibly attributed to the small number of enrolled patients.
The measurement of endothelium-dependent dilation in response to reactive hyperemia is a noninvasive and validated method for the assessment of the endothelial function.9 Alterations in endothelium-dependent dilation have been documented in patients with coronary artery disease and diabetes and recently OSAS.10–13 The regulation of vasomotor tone, as measured by the change in the forearm blood flow after transient ischemia, appears to be regulated by the availability of nitric oxide. Researchers have shown that in the OSAS, the pathophysiologic stressors that result from the repetitive episodes of hypoxemia/reoxygenation downregulate the activity of the endothelial nitric oxide synthase and upregulate the expression of various vasoactive substances, such as endothelin-1 and angiotensin II.14–16
In concordance with our results, previous studies have shown that endothelial dysfunction, as measured by FMD, was correlated with the severity of OSAS in terms of AHI.17–19 Nonetheless, multivariable regression analysis by Chung et al revealed that the oxygen desaturation index was the only significant variable that correlated with the FMD, a finding not supported by our results.19 Recently, data have emerged regarding the potential beneficial effects of CPAP on the endothelial function. A study by Ip et al demonstrated that the FMD was significantly improved after 4 weeks of CPAP treatment, while Bayram et al revealed that the improvement in the endothelial function was sustained after 6 months of treatment in complaint patients.20 Our results confirm the beneficial impact of CPAP and revealed a positive correlation between the percent change of FMD and the percent of time <90% oxygen saturation, suggesting a greater benefit in patients with more severe OSAS.
Over the last years, a large body of evidence has demonstrated the pivotal role of inflammation in cardiovascular disease. In view of this notion, CRP has gained increasing attention as an independent risk factor for coronary disease.21 In patients with OSAS, circulating levels of inflammatory markers appeared to be elevated, a finding suggestive of the presence of systemic inflammation.22,23 Previous reports have shown that CRP levels were correlated with the OSAS severity and the BMI; nevertheless, our study failed to show such an association.19 Furthermore, although both FMD and CRP are involved in the pathophysiology of cardiovascular complications, the association between these two parameters in OSAS remains elusive. In agreement with our results, two separate studies by Chung et al and Verma et al failed to find a relationship between FMD and CRP in healthy subjects.19,24 Nonetheless, a study by Nystrom et al in patients with coronary artery disease revealed a correlation between endothelial dysfunction (measured with FMD) and the levels of CRP.25 The different inclusion criteria among these studies may account for the observed discrepancies regarding the relationship between FMD and CRP. Even less data have been published regarding the impact of CPAP therapy on the inflammation markers. In this study, the CRP levels were significantly decreased 3 months after CPAP treatment. A previous study by Yokoe et al had also demonstrated that levels of CRP were decreased after 1 month of CPAP.26 These results denote the possible beneficial role of CPAP in reducing the vascular inflammation.
The relationship between OSAS and hypertension has been well-established by many researchers. According to published literature, the use of CPAP can lead to minor improvement in BP control.27 This study showed a significant decrease in 24-hour, daytime, and nighttime BP measurements after 3 months of CPAP treatment. Furthermore, current data suggest that patients with refractory hypertension and more severe OSAS appear to benefit more from CPAP treatment in terms of BP reduction.28,29 Our analysis provided statistically significant results in a population consisting of patients with no prior history of hypertension.
According to previous studies, OSA patients, independently of obesity, have higher prevalence of impaired glucose metabolism.30 The etiology of this metabolic deregulation may be associated with the intermittent episodes of hypoxia and subsequent activation of the sympathetic nervous system and the hypothalamic-pituitary axis.31 This study showed an improvement in blood glucose control, as estimated by the levels of HbA1c after CPAP treatment in nondiabetic patients. A similar study in euglycemic patients, by Papanas et al revealed that good adherence to CPAP treatment can significantly reduce HbA1c levels.32 These findings denote a considerable beneficial effect of CPAP in glucose control, even in patients with no prior history of diabetes.
The limitations of this study should be noted. We recruited a rather small number of patients; this could account for the inability of this study to provide with more statistically significant associations between the examined variables. The study population consisted of patients with no prior history of cardiovascular or pulmonary disease; thus, the study results should be interpreted with caution in patients beyond this particular group.
In summary, our results suggest that in male OSA patients, CPAP therapy reverses to some degree the endothelial dysfunction and vascular inflammation and can ultimately lead to better BP and glucose control. Female patients presented with similar trends, nonetheless not statistically significant. Further well-designed prospective studies are needed to elucidate the effect of CPAP on the reduction of cardiovascular risk in male and female OSA patients.
Acknowledgment
The abstract of this paper was presented at the ERS Conference 2011 as a poster presentation with interim findings. The poster’s abstract was published in “Poster Abstracts” in European Respiratory Journal (http://erj.ersjournals.com/content/38/Suppl_55/p2227).
Disclosure
The authors report no conflicts of interest in this work.
References
Patil SP, Schneider H, Schwartz AR, Smith PL. Adult obstructive sleep apnea: pathophysiology and diagnosis. Chest. 2007;132(1):325–337. | ||
Young T, Peppard PE, Gottlieb DJ. Epidemiology of obstructive sleep apnea: a population health perspective. Am J Respir Crit Care Med. 2002;165(9):1217–1239. | ||
Peppard PE, Young T, Palta M, Skatrud J. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med. 2000;342(19):1378–1384. | ||
Marin JM, Carrizo SJ, Vicente E, Agusti AG. Long-term cardiovascular outcomes in men with obstructive sleep apnoea-hypopnoea with or without treatment with continuous positive airway pressure: an observational study. Lancet. 2005;365(9464):1046–1053. | ||
Wang P, Zweier JL. Measurement of nitric oxide and peroxynitrite generation in the postischemic heart. Evidence for peroxynitrite-mediated reperfusion injury. J Biol Chem. 1996;271(46):29223–29230. | ||
Liao JK, Zulueta JJ, Yu FS, Peng HB, Cote CG, Hassoun PM. Regulation of bovine endothelial constitutive nitric oxide synthase by oxygen. J Clin Invest. 1995;96(6):2661–2666. | ||
Bradley TD, Floras JS. Obstructive sleep apnoea and its cardiovascular consequences. Lancet. 2009;373(9657):82–93. | ||
Iber K, Ancoli-Israel S, Chesson AL, Quan SF. The AASM Manual for the Scoring of Sleep and Associated Events. Westchester, IL: American Academy of Sleep Medicine; 2007. | ||
Corretti MC, Anderson TJ, Benjamin EJ, et al; International Brachial Artery Reactivity Task Force. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol. 2002;39(2):257–265. | ||
Simova I, Katova T, Denchev S. Diagnostic accuracy of flow-mediated dilatation and intima-media thickness for the presence of significant coronary artery disease. J Am Soc Hypertens. 2009;3(6):388–394. | ||
Yeboah J, Folsom AR, Burke GL, et al. Predictive value of brachial flow-mediated dilation for incident cardiovascular events in a population-based study: the multi-ethnic study of atherosclerosis. Circulation. 2009;120(6):502–509. | ||
Su Y, Liu XM, Sun YM, Wang YY, Luan Y, Wu Y. Endothelial dysfunction in impaired fasting glycemia, impaired glucose tolerance, and type 2 diabetes mellitus. Am J Cardiol. 2008;102(4):497–498. | ||
Kato M, Roberts-Thomson P, Phillips BG, et al. Impairment of endothelium-dependent vasodilation of resistance vessels in patients with obstructive sleep apnea. Circulation. 2000;102(21):2607–2610. | ||
Shirai M, Pearson JT, Shimouchi A, et al. Changes in functional and histological distributions of nitric oxide synthase caused by chronic hypoxia in rat small pulmonary arteries. Br J Pharmacol. 2003;139(5):899–910. | ||
Phillips BG, Narkiewicz K, Pesek CA, Haynes WG, Dyken ME, Somers VK. Effects of obstructive sleep apnea on endothelin-1 and blood pressure. J Hypertens. 1999;17(1):61–66. | ||
Moller DS, Lind P, Strunge B, Pedersen EB. Abnormal vasoactive hormones and 24-hour blood pressure in obstructive sleep apnea. Am J Hypertens. 2003;16(4):274–280. | ||
Nieto FJ, Herrington DM, Redline S, Benjamin EJ, Robbins JA. Sleep apnea and markers of vascular endothelial function in a large community sample of older adults. Am J Respir Crit Care Med. 2004;169(3):354–360. | ||
Bayram NA, Ciftci B, Keles T, et al. Endothelial function in normotensive men with obstructive sleep apnea before and 6 months after CPAP treatment. Sleep. 2009;32(10):1257–1263. | ||
Chung S, Yoon IY, Shin YK, et al. Endothelial dysfunction and C-reactive protein in relation with the severity of obstructive sleep apnea syndrome. Sleep. 2007;30(8):997–1001. | ||
Ip MS, Tse HF, Lam B, Tsang KW, Lam WK. Endothelial function in obstructive sleep apnea and response to treatment. Am J Respir Crit Care Med. 2004;169(3):348–353. | ||
Emerging Risk Factors Collaboration; Kaptoge S, Di Angelantonio E, et al. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet. 2010;375(9709):132–140. | ||
Shamsuzzaman AS, Winnicki M, Lanfranchi P, et al. Elevated C-reactive protein in patients with obstructive sleep apnea. Circulation. 2002;105(21):2462–2464. | ||
Schulz R, Mahmoudi S, Hattar K, et al. Enhanced release of superoxide from polymorphonuclear neutrophils in obstructive sleep apnea. Impact of continuous positive airway pressure therapy. Am J Respir Crit Care Med. 2000;162(2 pt 1):566–570. | ||
Verma S, Wang CH, Lonn E, et al; FATE Investigators. Cross-sectional evaluation of brachial artery flow-mediated vasodilation and C-reactive protein in healthy individuals. Eur Heart J. 2004;25(19):1754–1760. | ||
Nystrom T, Nygren A, Sjoholm A. Persistent endothelial dysfunction is related to elevated C-reactive protein (CRP) levels in Type II diabetic patients after acute myocardial infarction. Clin Sci. 2005;108(2):121–128. | ||
Yokoe T, Minoguchi K, Matsuo H, et al. Elevated levels of C-reactive protein and interleukin-6 in patients with obstructive sleep apnea syndrome are decreased by nasal continuous positive airway pressure. Circulation. 2003;107(8):1129–1134. | ||
Das AM, Khayat R. Hypertension in obstructive sleep apnea: risk and therapy. Expert Rev Cardiovasc Ther. 2009;7(6):619–626. | ||
Martinez-Garcia MA, Gomez-Aldaravi R, Soler-Cataluna JJ, Martinez TG, Bernacer-Alpera B, Roman-Sanchez P. Positive effect of CPAP treatment on the control of difficult-to-treat hypertension. Eur Respir J. 2007;29(5):951–957. | ||
Pepperell JC, Ramdassingh-Dow S, Crosthwaite N, et al. Ambulatory blood pressure after therapeutic and subtherapeutic nasal continuous positive airway pressure for obstructive sleep apnoea: a randomised parallel trial. Lancet. 2002;359(9302):204–210. | ||
Punjabi NM, Sorkin JD, Katzel LI, Goldberg AP, Schwartz AR, Smith PL. Sleep-disordered breathing and insulin resistance in middle-aged and overweight men. Am J Respir Crit Care Med. 2002;165(5):677–682. | ||
Van Cauter E, Holmback U, Knutson K, et al. Impact of sleep and sleep loss on neuroendocrine and metabolic function. Horm Res. 2007;67(suppl 1):2–9. | ||
Papanas N, Steiropoulos P, Nena E, et al. HbA1c is associated with severity of obstructive sleep apnea hypopnea syndrome in nondiabetic men. Vasc Health Risk Manag. 2009;5:751–756. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.