Back to Journals » Journal of Asthma and Allergy » Volume 12
Severe uncontrolled asthma with bronchiectasis: a pilot study of an emerging phenotype that responds to mepolizumab
Authors Carpagnano GE, Scioscia G , Lacedonia D, Curradi G, Foschino Barbaro MP
Received 27 November 2018
Accepted for publication 5 February 2019
Published 5 March 2019 Volume 2019:12 Pages 83—90
DOI https://doi.org/10.2147/JAA.S196200
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Charlotte Suppli Ulrik
Giovanna E Carpagnano,1 Giulia Scioscia,1 Donato Lacedonia,1 Giacomo Curradi,2 Maria Pia Foschino Barbaro1
1Department of Medical and Surgical Sciences, Institute of Respiratory Diseases, University of Foggia, Foggia, Italy; 2Medical and Scientific Department, GlaxoSmithKline, Verona, Italy
Background: Asthma and bronchiectasis are different conditions that frequently coexist. The prevalence of bronchiectasis rises considerably in subjects with severe asthma (25%–51%).
Objective: We evaluated the clinical and biological efficacy of mepolizumab on our pilot population of severe uncontrolled asthmatics with bronchiectasis not related to other pathologies.
Patients and methods: Four patients with severe uncontrolled asthma and diagnosed as bronchiectasis were recruited and started biological treatment with mepolizumab. Standard investigations were performed in all four patients at baseline (T0), after 3 months (T1) and after 1 year (T2) of treatment.
Results: After 1 year (T2) of therapy with mepolizumab, patients showed a significant increment of asthma control test value (12±1.1 vs 24.5±0.3, P<0.01), a reduction of the number of exacerbations/year (5±0.7 vs 0.75±0.75, P<0.01), an increase of pre-bronchodilator FEV1 (1,680±500 vs 1,860±550 mL, P<0.01) and a reduction of eosinophils in blood (0.75±0.14 vs 0.12±0.02 cells/µL, P<0.01), in the sputum (9.6%±2.1% vs 5.6%±2.7%, P<0.05) and in nasal cytology (++ vs +).
Conclusion: The efficacy of mepolizumab in terms of reduction of inflammation and increase of control that we observed in our patients might suggest that targeting the IL-5 in severe eosinophilic asthma with bronchiectasis may be a good therapeutic strategy.
Keywords: severe asthma, bronchiectasis, mepolizumab, phenotype
Introduction
Severe asthma is reported, to date, in 5%–15% of the total asthmatic population and is estimated to account for >60% of the total costs of the disease, primarily required for medications and asthma exacerbations. Severe asthma is frequently accompanied by a difficult and often unachievable control that requires critical management. However, today, the availability of biological drugs allows for a more modern therapy tailored to the patient. Although severe asthma still remains a challenge for specialists, the target therapy gave interesting clinical results when prescribed for the selected patients. Today, the phenotypic characterization of severe asthmatic patients has, therefore, become crucial in view of a different management and therapeutic approach.1 The extreme clinical, biological and inflammatory heterogeneity of severe asthma has required the application of cluster analyses that, considering multiple comorbidities and risk factors, divide accordingly severe asthmatic patients into homogenous groups. The result is the identification of several phenotypes of severe asthma as those associated to obesity, exercise, smoking habit, aspirin, allergens, nasal polyposis, menopause, infections and others.2 The investigation and treatment of comorbidities is of major importance in patients with severe asthma, as they contribute to poor disease control.
Severe uncontrolled asthma associated with bronchiectasis is an emerging phenotype. Recent epidemiological studies showed an unexpected prevalence of severe asthma with bronchiectasis that ranges from 2.2%3 to 77%.4 Almost one-third of severe asthmatic patients showed the presence of bronchiectasis on the chest computed tomography (CT) scan. Some groups have described bronchiectasis as a comorbidity associated to asthma,5 while others have identified asthma as being one of many etiologies of bronchiectasis.6
This phenotype has been described in the Type 2 (T2) endotype and particularly in patients with severe, late-onset eosinophilic asthma. It has been mainly observed in women and the elderly, in non-smokers, and it is often associated with an impaired lung function. Identification of bronchiectasis in asthmatic subjects could lead significantly to modifications to both therapy and prognosis.7,8
At the moment, to our knowledge, no study exists suggesting one specific therapeutic approach in these patients, leaving the choice to the specialist, absolutely personal. These patients usually have a severe and uncontrolled asthma that does not respond to the maximum pharmacological therapy as indicated by steps 4 and 5 of Global Initiative for Asthma (GINA) guidelines; thus, a step up with biologics could be appropriately proposed.9
In this study, we focused on our small population of patients with severe asthma and bronchiectasis that we started on biological therapy with the anti-IL5, mepolizumab.
Mepolizumab is a humanized monoclonal antibody used for the treatment of severe eosinophilic asthma (SEA), which recognizes and blocks IL-5, a signaling protein of the immune system.
The aim of this study was to evaluate the clinical and biological efficacy of mepolizumab in patients with severe uncontrolled asthma and bronchiectasis not related to other pathologies.
Patients and methods
Patients
This prospective pilot study included four consecutive patients with severe asthma and diagnosed as having bronchiectasis by means of chest CT scan and a compatible clinical history consistent with chronic sputum production and/or frequent respiratory infections, who were followed up at the accredited outpatient pulmonary clinic for severe asthma of the Institute of Respiratory Diseases of the University of Foggia, Italy during a period of 1 year. Written informed consent was obtained from all subjects, and the Institutional Ethics Committee of the University of Foggia approved the study, which was carried out according to the Declaration of Helsinki. Asthmatics were classified and treated according to GINA guidelines.9 Severe asthmatics were diagnosed according to the European Respiratory Society/American Thoracic Society taskforce criteria10 on severe asthma. All four patients of the study were treated with high-dose inhaled corticosteroids (ICS) with long-acting β-agonists (LABAs) and long-acting muscarinic receptor antagonists (LAMAs) and started biological treatment with mepolizumab at enrollment, respecting the prescribing criteria. Once the diagnosis was confirmed, additional standard investigations were performed on all four patients with severe asthma (familiar, personal and pathological anamnesis, lung function tests, prick tests, serum eosinophils count, total serum IgE, induced sputum and nasal cytology analysis, fractional exhaled NO measurement). We used the asthma control test (ACT) questionnaire to evaluate the control of symptoms. All measurements were done at baseline (T0), after 3 months (T1) and after 1 year of treatment with mepolizumab (T2).
All subjects enrolled underwent all tests foreseen by the guidelines8 to identify the etiology of bronchiectasis, including high-resolution chest CT (HRCT): α1-antitripsin deficiency, allergic bronchopulmonary aspergillosis, cystic fibrosis (CF), immunodeficiency and systemic disease. Patients with these conditions, smokers and former smokers were excluded because in this research, we were looking for the presence of bronchiectasis related to asthma and not associated to other pathologies.
Asthmatics enrolled were assessed during a period of stability and at least 4 weeks after an upper respiratory tract infection. No patient at enrollment was treated with oral corticosteroids.
At the first visit, a complete baseline questionnaire requesting information on medical history was administered to all subjects, who were then given a physical examination, atopy assessment and spirometry with bronchodilator reversibility test. During the second visit, subjects underwent blood analysis for eosinophil count and total IgE levels, fractional exhaled nitric oxide (FeNO) measurement, ACT and, finally, sputum induction and nasal cytology.
Atopic status
The skin prick test (SPT) was performed for a panel of inhalant allergens as previously described for common aero-allergens (Lofarma, Milan, Italy). SPT results were considered positive when eliciting a wheal diameter ≥3 mm, using negative (saline) and positive (histamine 10 mg/mL) controls for interpretation.11
Lung function
Pulmonary function tests were performed. FEV1 and FVC were measured using a spirometer (Sensormedics, Milan, Italy). The best value of three maneuvers was expressed as a percentage of the predicted normal value. After baseline evaluation, spirometry was repeated 15 minutes after the subjects had inhaled 400 µg of salbutamol. Reversibility of airway obstruction was expressed in terms of the percentage changes from baseline of the FEV1.12
Induced sputum collection and processing
According to the method described by Toungoussova et al,13 sputum was induced through inhalation of hypertonic saline solution (4.5%) with an ultrasonic nebulizer (DeVilbiss 65; DeVilbiss Corporation, Somerset, PA, USA) and analyzed after selection of mucus plugs. In two subjects, we used the spontaneous sputum.
All subjects enrolled were able to produce adequate sputum samples (defined as containing at least 500 non-squamous cells). The sputum (spontaneous or induced) was used for cytological analysis.
Measurement of FeNO
Measurement of FeNO was performed according to recent guidelines.14 The Medisoft FeNO device (HYPAIR; Medisoft, Dinant, Belgium), which is semi-portable for repeatable multi-flow measurement of exhaled NO with off-line measurement, was used. It has a software package that provides step-by-step online quality control. The measurement range is 0–600 ppb. Exhaled NO (FeNO) was measured using a previously described restricted breath technique, which employed expiratory resistance and positive mouth pressure to close the velum and exclude nasal NO, and a constant expiratory flow of 50 and 350 mL/s. Repeated exhalations were performed until three plateaus agreed within 5% of inter-observation difference.
Nasal cytology
A scraping of the nasal mucosa on the middle third of the inferior turbinate was performed. After sampling, the material was laid on a microscope slide, air dried and stained using the May–Grunwald–Giemsa method. The smear was observed under a common light microscope equipped with 1,000× objective. In order to analyze the rhino-cytogram, we performed a reading for fields (not <50), in order to observe the cellular elements that composed the nasal mucosa (eosinophils, mast cells, neutrophils, lymphocytes, bacteria, spores, biofilms and so on). To calculate the percentage of each cellular element, semi-quantitative grading counts of each cell type, according to the previously published studies, were performed (grade 1+: occasional groups, grade 2+: moderate number, grade 3+: easily visible, grade 4+: elevated number).15,16
Statistical analysis
Descriptive statistics (ie, means, SDs, percentages) were applied to summarize the continuous and categorical variables. In order to assess the difference between T0 and T2 for all variables analyzed, we performed a Wilcoxon matched-pairs signed rank test, considering a P-value of <0.05 statistically significant. Data were analyzed using GraphPad for MAC.
Results
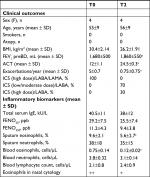
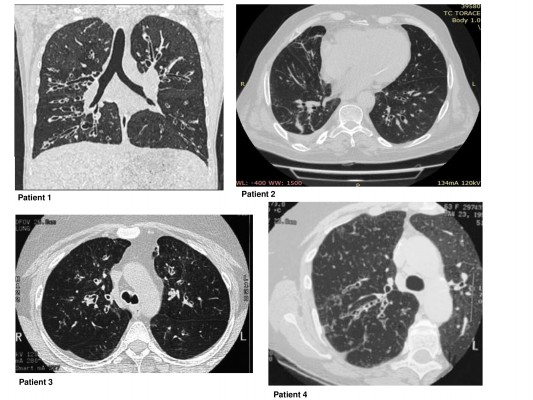
Demographic, clinical, functional and biological data are shown in Table 1. The patients enrolled were four females, with late-onset severe asthma and diagnosed as bronchiectasis (Figure 1). At enrollment (T0), the FENO50 was 29.2±7.5 ppb, FENO350 was 11.2±4.3 ppb, blood eosinophil count was 750±141.6 cells/mm3, sputum eosinophils were 9.6%±2.1%, FEV1 was 1,680±500 mL and the ACT questionnaire was 12±1.1. The nasal cytology showed a moderate degree of eosinophils (++) and the absence of nasal polyps or rhinosinusitis.
  | Figure 1 Chest CT scan before treatment (T0). Abbreviation: CT, computed tomography. |
After 1 year (T2) of mepolizumab treatment, patients showed a significant increment of ACT (12±1.1 vs 24.5±0.3, P<0.01; Figure 2), a reduction of number of exacerbations/year (5±0.7 vs 0.75±0.75, P<0.01; Figure 3), an increase of pre-bronchodilator FEV1 (1,680±500 vs 1,860±550 mL, P<0.01; Figure 4) and a reduction of eosinophils in blood (0.75±0.14 vs 0.12±0.02 cells/µL, P<0.01; Figure 5), in the sputum (9.6%±2.1% vs 5.6%±2.7%, P<0.05; Figure 6) and in nasal cytology (++ vs+; Table 1).
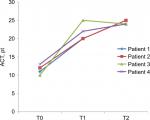  | Figure 2 ACT value before treatment (T0), after 3 months (T1) and after 1 year (T2) of treatment with mepolizumab. Abbreviation: ACT, asthma control test. |
  | Figure 3 Number of exacerbations/year of asthma before treatment (T0), after 3 months (T1) and after 1 year (T2) of treatment with mepolizumab. |
  | Figure 4 ΔFEV1 preBD vs baseline (mL, %), before treatment (T0), after 3 months (T1) and after 1 year (T2) of treatment with mepolizumab. Abbreviation: BD, bronchodilator. |
  | Figure 5 Eosinophils in the sputum (%) before treatment (T0), after 3 months (T1) and after 1 year (T2) of treatment with mepolizumab. |
  | Figure 6 Eosinophils in blood (cells/µL) before treatment (T0), after 3 months (T1) and after 1 year (T2) of treatment with mepolizumab. |
All patients, during treatment with mepolizumab, underwent a step down of inhalation therapy, suspending LAMA (100%) and reducing ICS to lower dose (70%).
Discussion
In this pilot study, we demonstrated the efficacy of mepolizumab in an emerging phenotype of patients with severe uncontrolled asthma and diagnosed bronchiectasis not related to other pathologies in terms of reduction of eosinophils and increasing control of the disease.
As previously reported, we confirmed prevalence of female gender in this phenotype as all subjects enrolled were women. Girón et al17 demonstrated that bronchiectasis of unknown etiology and secondary to asthma was more common in women. Gender differences in bronchiectasis are reported in CF disease: females with CF have a more severe form of the disease, poorer lung function and earlier Pseudomonas aeruginosa colonization and conversion to its more aggressive mucoid form. Chotirmall et al18 showed that immune responses in the CF airway are further compromised by estrogen which supresses the protective acute inflammatory burst necessary to clear bacterial infection and prevent exacerbation. As non-CF bronchiectasis usually presents in females who have undergone menopause, the role of estrone, the major estrogen of menopause, should be investigated in future studies as currently no evidence exists to link hormones to the severity of non-CF bronchiectasis in affected females.19
We further confirmed that the coexistence of severe asthma and bronchiectasis is typical of elderly patients with late-onset non-atopic severe uncontrolled eosinophilic asthma.20 Accordingly to Olveira et al,21 the prevalence of bronchiectasis rises considerably in subjects with more severe asthma, as evident from the poorer lung function and lower control of the disease of the four subjects enrolled in our study. As in this study, Coman et al20 further proved the higher incidence of exacerbations in patients with this phenotype, reporting more hospitalizations in severe asthmatics with bronchiectasis. This suggests that the coexistence of both pathologies worsens outcomes.
While early studies documented a relationship between asthma, bronchiectasis and atopy,22 many have not,23 even when specifically looking at subjects with severe asthma, as in our study. Furthermore, our patients did not have rhinosinusitis and nasal polyps and we could explain it with the hypothesis of Shteinberg et al24 that the involvement of the upper airway in patients with bronchiectasis is associated with an early-onset and atopic asthma.
All patients enrolled showed an eosinophilic inflammatory pattern. Although bronchiectasis is usually associated with neutrophilic inflammation,25,26 we know that when there is a coexistence of asthma, the inflammation might be eosinophilic or mixed (neutrophilic–eosinophilic).
The presence of bronchiectasis in patients with an eosinophilic profile has also been described in patients without asthma with a prevalence of 17.5%.27 Those patients with eosinophilic bronchiectasis also showed higher sputum IL-13, further proving the T2 endotype pattern, where this interleukin plays a key role. In their study, Tsikrika et al27 further described low concentration of FENO in bronchiectasis patients, although they had T2 endotype with increased eosinophils in airways and blood. We, therefore, were not surprised to find exactly the same normal level of FENO in our population despite an increased eosinophilic profile. Our explanation agrees with the hypothesis that eosinophilic inflammation in asthma with bronchiectasis is not primarily Th2 driven and possibly another T2-high pathway through Group 2 Innate Lymphoid Cells (ILC2s) plays a role in eosinophilic inflammation.28
As known, environmental stimuli, including pollutants, viruses, helminths and bacterial infections, such as the ones that characterized the history of bronchiectasis, trigger ILC2s-mediated T2 airways inflammation related to airway inflammation. These stimuli induce airway epithelial cell death and production of IL-25, IL-33 and thymic stromal lymphopoietin. These cytokines promote massive expansion of ILC2s. ILC2s produce Th2 cytokines, including IL-5, IL-9 and IL-13 that are involved in the activation/proliferation of eosinophils and mucus hypersecretion/tissue remodeling, respectively.29
The recent expansion in our understanding of the context of IL-5 and IL-5-producing ILC2s in eosinophil activation and the pathogenesis of eosinophil-dependent inflammatory diseases has led to advances in therapeutic options.
Mepolizumab was the first therapeutic humanized monoclonal antibody to human IL-5 to be approved for any indication globally. Mepolizumab acts by preventing IL-5 from binding to IL-5Ra complex expressed on the eosinophil cell surface. This action inhibits IL-5 signaling and the overexpressing functions of peripheral blood and tissue eosinophils.
For the first time, to our knowledge, we demonstrated the efficacy of mepolizumab in patients not only with SEA but also with diagnosed bronchiectasis, in terms of reduction of eosinophils and increasing control of the disease. We could explain these results with the hypothesis that the pathogenesis of this phenotype of patients is driven by ILC2s related to airway inflammation that respond well to the biological therapy with mepolizumab, and it is not influenced by other inflammatory cells such as neutrophils.
It is difficult to determine if the severity of asthma in subjects with coexistent bronchiectasis is due to the presence of bronchiectasis, or if the presence of bronchiectasis is the cause of the severity of asthma itself. As we observed higher levels of peripheral eosinophils in our patients with coexistent bronchiectasis, we suggest that the latter hypothesis is the most probable.20
Our findings suggest that the presence of bronchiectasis could be another criterion to identify an emerging phenotype of SEA that has been attracting increased interest for the unexpected high prevalence observed in asthmatic population and that we need more attention with a large dedicated study designed to better explore it.
Bronchiectasis non-CF is treated in clinical practice with therapy based on very little evidence.30 In our opinion, development of new treatments is urgently needed. Identifying different phenotypes of bronchiectasis may help us in directing therapy toward specific target with better results compared to that actually expected by traditional therapy. The efficacy of mepolizumab that we observed in patients with SEA and bronchiectasis might suggest that targeting the IL-5 in eosinophilic bronchiectasis may be a good therapeutic strategy and may support further larger studies in this direction.
The limitations of our study are mainly related to the small number of cases and to the absence of a comparison with a control group of patients with SEA without bronchiectasis and a group of male subjects. The bronchiectasis documented by HRCT scan was furthermore not classified or scored according to severity and was all included in the analysis, clinically significant or not.
According to Coman et al,20 we believe the extent of the immune–pathological roles of the eosinophils in SEA has yet to be fully understood, and prospective studies on routine CT scan and active treatment of bronchiectasis in subjects with severe asthma are needed to help guide management in this population.
Acknowledgment
Giovanna E Carpagnano and Giulia Scioscia are co-first authors for this work.
Author contributions
GEC, GS and GC designed the study. GS and DL contributed to the clinical and laboratory work for the study. GEC, GS, GC and MPFB drafted the article and revised it critically for important intellectual content. GEC, GS, GC and DL contributed to final approval of the version to be published. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
GC is a full-time employee for GlaxoSmithKline, Italy. The authors report no other conflicts of interest in this work.
References
Kupczyk M, Wenzel S. U.S. and European severe asthma cohorts: what can they teach us about severe asthma? J Intern Med. 2012;272(2):121–132. | ||
Chung KF, Adcock IM. How variability in clinical phenotypes should guide research into disease mechanisms in asthma. Ann Am Thorac Soc. 2013;(10 Suppl):S109–S117. | ||
Kang HR, Choi G-S, Park SJ, et al. The effects of bronchiectasis on asthma exacerbation. Tuberc Respir Dis (Seoul). 2014;77(5):209–214. | ||
Lynch DA, Newell JD, Tschomper BA, Cink TM, Newman LS, Bethel R. Uncomplicated asthma in adults: comparison of CT appearance of the lungs in asthmatic and healthy subjects. Radiology. 1993;188(3):829–833. | ||
Bisaccioni C, Aun MV, Cajuela E, Kalil J, Agondi RC, Giavina-Bianchi P. Comorbidities in severe asthma: frequency of rhinitis, nasal polyposis, gastroesophageal reflux disease, vocal cord dysfunction and bronchiectasis. Clinics. 2009;64(8):769–773. | ||
Dimakou K, Triantafillidou C, Toumbis M, Tsikritsaki K, Malagari K, Bakakos P. Non CF-bronchiectasis: aetiologic approach, clinical, radiological, microbiological and functional profile in 277 patients. Respir Med. 2016;116:1–7. | ||
Patel IS, Seemungal TAR, Wilks M, Lloyd-Owen SJ, Donaldson GC, Wedzicha JA. Relationship between bacterial colonisation and the frequency, character, and severity of COPD exacerbations. Thorax. 2002;57(9):759–764. | ||
Martínez-García M-A, de La Rosa Carrillo D, Soler-Cataluña J-J, et al. Prognostic value of bronchiectasis in patients with moderate-to-severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2013;187(8):823–831. | ||
(*NEW) 2018 GINA Report: Global Strategy for Asthma Management and Prevention | Global Initiative for Asthma – GINA. Available from: https://ginasthma.org/gina-reports/. Accessed February 19, 2019. | ||
Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–373. | ||
Bousquet J, Heinzerling L, Bachert C, et al. Practical guide to skin prick tests in allergy to aeroallergens. Allergy. 2012;67(1):18–24. | ||
Standardization of Spirometry, 1994 Update. American Thoracic Society. Am J Respir Crit Care Med. 1995;152(3):1107–1136. | ||
Toungoussova O, Migliori GB, Foschino Barbaro MP, et al. Changes in sputum composition during 15 min of sputum induction in healthy subjects and patients with asthma and chronic obstructive pulmonary disease. Respir Med. 2007;101(7):1543–1548. | ||
American Thoracic Society/European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am J Respir Crit Care Med. 2005;171(8):912–930. | ||
Gelardi M, Fiorella ML, Leo G, Incorvaia C. Cytology in the diagnosis of rhinosinusitis. Pediatr Allergy Immunol. 2007;18(s18):50–52. | ||
Meltzer EO. Evaluating rhinitis: clinical, rhinomanometric, and cytologic assessments. J Allergy Clin Immunol. 1988;82(5 Pt 2):900–908. | ||
Girón RM, de Gracia Roldán J, Olveira C, et al. Sex bias in diagnostic delay in bronchiectasis: an analysis of the Spanish historical registry of bronchiectasis. Chron Respir Dis. 2017;14(4):360–369. | ||
Chotirmall SH, Greene CM, Oglesby IK, et al. 17beta-estradiol inhibits IL-8 in cystic fibrosis by up-regulating secretory leucoprotease inhibitor. Am J Respir Crit Care Med. 2010;182(1):62–72. | ||
Vidaillac C, Yong VFL, Jaggi TK, Soh M-M, Chotirmall SH. Gender differences in bronchiectasis: a real issue? Breathe. 2018;14(2):108–121. | ||
Coman I, Pola-Bibián B, Barranco P, et al. Bronchiectasis in severe asthma. Ann Allergy Asthma Immunol. 2018;120(4):409–413. | ||
Olveira C, Padilla A, Martínez-García Miguel-Ángel, et al. Etiología de las bronquiectasias en Una cohorte de 2.047 pacientes. Análisis del registro histórico español. [Etiology of bronchiectasis in a cohort of 2,047 patients. An analysis of the spanish historical bronchiectasis registry]. Arch Bronconeumol. 2017;53(7):366–374. | ||
Säynäjäkangas O, Keistinen T, Tuuponen T, Kivelä SL. Links between hospital diagnoses of bronchiectasis and asthma. Allergy. 1997;52(11):1120–1122. | ||
Oguzulgen IK, Kervan F, Ozis T, Turktas H. The impact of bronchiectasis in clinical presentation of asthma. South Med J. 2007;100(5):468–471. | ||
Shteinberg M, Nassrallah N, Jrbashyan J, Uri N, Stein N, Adir Y. Upper airway involvement in bronchiectasis is marked by early onset and allergic features. ERJ Open Res. 2018;4(1):00115-2017. | ||
Jatakanon A, Lim S, Kharitonov SA, Chung KF, Barnes PJ. Correlation between exhaled nitric oxide, sputum eosinophils, and methacholine responsiveness in patients with mild asthma. Thorax. 1998;53(2):91–95. | ||
Cole PJ. Inflammation: a two-edged sword – the model of bronchiectasis. Eur J Respir Dis Suppl. 1986;147:6–15. | ||
Tsikrika S, Dimakou K, Papaioannou AI, et al. The role of non-invasive modalities for assessing inflammation in patients with non-cystic fibrosis bronchiectasis. Cytokine. 2017;99:281–286. | ||
Padilla-Galo A, Olveira C, Fernández de Rota-Garcia L, et al. Factors associated with bronchiectasis in patients with uncontrolled asthma; the NOPES score: a study in 398 patients. Respir Res. 2018;19(1):43. | ||
Yanagibashi T, Satoh M, Nagai Y, Koike M, Takatsu K. Allergic diseases: from bench to clinic – contribution of the discovery of interleukin-5. Cytokine. 2017;98:59–70. | ||
Chalmers JD, Chotirmall SH. Bronchiectasis: new therapies and new perspectives. Lancet Respir Med. 2018;6(9):715–726. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.