Back to Journals » International Journal of General Medicine » Volume 16
Serum Cortistatin Level in Type 2 Diabetes Mellitus and Its Relationship with Nonalcoholic Fatty Liver Disease
Authors Sun T, Wang C , Huo L, Wang Y, Liu K, Wei C, Zhao H, Chen S, Ren L
Received 10 November 2022
Accepted for publication 23 January 2023
Published 21 February 2023 Volume 2023:16 Pages 631—639
DOI https://doi.org/10.2147/IJGM.S396315
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tiantian Sun,1,* Chang Wang,1,2,* Lijing Huo,3 Yichao Wang,1 Ke Liu,1 Changmei Wei,3 Hang Zhao,1 Shuchun Chen,1 Luping Ren1
1Department of Endocrinology, Hebei General Hospital, Shijiazhuang, People’s Republic of China; 2North China University of Science and Technology, Tangshan, People’s Republic of China; 3Department of Clinical Laboratory, Hebei General Hospital, Shijiazhuang, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Luping Ren, No. 348 Heping West Road, Shijiazhuang, Hebei Province, People’s Republic of China, Tel +86 0311-85988975, Email [email protected]
Purpose: To evaluate serum cortistatin (CST) levels in type 2 diabetes mellitus (T2DM) patients with or without non-alcoholic fatty liver disease (NAFLD) and to examine the relationship between CST and NAFLD.
Methods: A total of 90 T2DM patients, which included 56 NAFLD patients (referred to as DM+NAFLD group) and 34 patients without NAFLD (DM-only group), and 83 non-diabetes individuals that included 39 NAFLD patients (NAFLD-only group) and 44 without NAFLD that acted as the normal-control group (NC group). The differences in the serum CST levels between the groups were compared, and the correlations between CST and other variables were calculated by applying both correlational analysis and multiple linear regression analysis.
Results: The mean serum CST levels were significantly lower in the DM+NAFLD and DM groups than in the NC group (P < 0.05). In addition, the CST levels were lower in the DM group relative to that in the NAFLD group (P < 0.05). However, no statistical difference was noted in the serum CST between diabetic patients with and without NAFLD (P > 0.05). Similarly, in the non-diabetic group, the serum CST level was not significantly different between individuals with and without NAFLD (P > 0.05). Furthermore, the serum CST levels were negatively associated with the levels of total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), fasting plasma glucose (FPG), homeostasis model assessment-insulin resistance (HOMA-IR), and insulin cell function index (HOMA-β). Conversely, the serum CST levels were positively associated with high-density lipoprotein cholesterol (HDL-C). The data obtained through multiple linear regression implied that LDL-C and HOMA-β, but not HOMA-IR, were closely related to serum CST levels.
Conclusion: T2DM was related to decreased serum CST. However, serum CST was correlated with HOMA-β in T2DM patients, while HOMA-IR was not. There was no correlation between CST and NAFLD.
Keywords: type 2 diabetes mellitus, cortistatin, nonalcoholic fatty liver disease
Introduction
Type 2 diabetes mellitus (T2DM) is a major global health issue and poses a great human health threat.1 The pathogenesis of T2DM mainly involves the dysfunction of islets beta cells and insulin resistance. Meanwhile, with the pandemic of obesity, the prevalence of nonalcoholic fatty liver disease (NAFLD) has increased in the past several decades. Whereas the incidence of NAFLD in the overall population is 20–30%,2 and its incidence in T2DM patients has reached to approximately 55%.3 NAFLD is proposed to occur as a manifestation of hepatic insulin resistance and is a common comorbidity in patients with T2DM.4 Although insulin resistance plays a role in the link between NAFLD and T2DM, the underlying mechanism connecting these two diseases remains largely elusive.5
Bioactive peptides have received considerable attention in the prevention and treatment of diabetes. Cortistatin (CST) is a neuropeptide that shares structural similarity with somatostatin (SST).6 Past studies have demonstrated that CST mRNA is widely present in the endocrine tissues, particularly in the pancreatic tissues.7 Accumulated evidences suggest that CST can affect insulin secretion.8 While examining the dose-dependent effect of CST or SST on growth hormone and insulin secretion, Broglio et al reported that CST, like SST, has an inhibitory influence on the secretion of growth hormone and insulin. Similarly, Grottoli et al reported that CST inhibited insulin secretion in patients with acromegaly and prolactinoma.9 In addition, some studies have found that CST can affect insulin sensitivity. A study by Cordoba-Chacon et al reported insulin resistance in CST-deficient male mice.10 In addition to animals, the link between CST and T2DM has been reported in human studies. For instance, Chen et al11 discovered that the serum CST levels were significantly low in newly diagnosed T2DM patients compared to that in healthy controls. Furthermore, the serum CST levels were negatively associated with the fasting plasma glucose (FPG), fasting insulin (FINS), and homeostasis model assessment-insulin resistance (HOMA-IR). Cumulatively, these studies indicate that CST may be involved in the regulation of glucose metabolism through its effect on insulin secretion and insulin resistance. Accordingly, CST might play a potential role in the development of T2DM and may thus serve as a new therapeutic target.
Although CST is found in the liver,12,13 there are no studies that correlate the serum CST level with NAFLD. Also, until date, no study has yet investigated CST changes in T2DM combined with NAFLD, which presents a situation of more profound insulin resistance. In addition, the association between CST and insulin sensitivity and its role in regulating glucose metabolism in patients with T2DM deserves detailed studies. In this study, we investigated the serum levels of CST in T2DM patients with or without NAFLD. We further analyzed the correlation of serum CST levels with insulin secretion, insulin sensitivity, and NAFLD. Through this study, we aimed to achieve a better understanding of the role of CST in the development of T2DM as well as its association with NAFLD.
Materials and Methods
Participants
In the present study, we randomly selected 90 T2DM patients and 83 non-diabetic individuals. Among 90 T2DM patients, 56 had NAFLD whereas 34 had no NAFLD. These patients were admitted to the Hebei General Hospital between March 2019 and November 2019 as a DM group. In addition, among 83 non-diabetic patients, 39 had NAFLD and 44 had no NAFLD. These individuals were examined during the same period and were categorized as the non-DM group. The diagnosis of T2DM met the diagnostic criteria of the World Health Organization in 1999 and excluded type 1 DM, chronic hepatic and/or renal dysfunction, acute infection, nutritional disorders, chronic vascular diseases, cancer and other specified diseases that can lead to fatty liver, such as hepatitis B virus infection, hypo-lipoproteinemia, hepatolenticular degeneration, congenital lipid atrophy, and celiac diseases. The present study upheld the privacy rights of human subjects, obtained their informed consent, and implemented all ethical procedures. Furthermore, the study was approved by Hebei General Hospital Ethics Committee (ethical approval number: 2022053) and is consistent with the Declaration of Helsinki.
Clinical Data Collection
Clinical Data
The basic information, past medical history, and duration of the diabetes of all the subjects who participated in the study were gathered. Body mass index (BMI) was counted. The venous blood drawn from all patients was drawn in the morning after at least 8 h of fasting. Glucose metabolism-related indicators, including fasting plasma glucose (FPG), fasting insulin (FINS), and fasting c-peptide were recorded. The biochemical indexes, including liver function, blood lipid, and serum uric acid were also recorded. Furthermore, homeostasis model assessment-insulin resistance (HOMA-IR) and insulin cell function index (HOMA-β) were calculated for each individual. The serum CST and C-reactive protein (CRP) levels were measured by electrochemiluminescence immunoassays (ECLA) using Roche commercial kits (COBAS e 601, Roche, Germany).
Definitions
NAFLD diagnostic criteria: All subjects having no history of heavy drinking (male <30 g/day, female <20 g/day), no drug intake that can cause liver steatosis, no history of obesity surgery, and no history of exposure to liver toxins in the past six months. The severity of NAFLD was classified based on the results of B-ultrasound.14 Based on the ultrasound findings, patients with NAFLD were classified as mild, moderate, or severe according to their degree of disease.15 The formulas used for calculating HOMA-IR and HOMA β are as follows: HOMA-IR = fasting glucose (mmol/L) × fasting insulin (mIU/L)/22.5, HOMA-β= (20 × fasting insulin [mIU/L])/(fasting glucose [mmol/L] - 3.5).16,17
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 (IBM, Inc., Armonk, NY, USA). The classified data were represented as rate or percentage. If the continuous data exhibited normal distribution, it was represented as mean ±standard deviation, otherwise as median (interquartile range). While comparing the data of each group, one-way ANOVA was used if the data were normally distributed in the four groups; otherwise, a nonparametric rank test was applied for analysis of inter-group differences. In addition, correlation analysis was conducted to observe the correlation between the serum CST level and other indicators in the T2DM group. The indicators of normal distribution were analyzed by Pearson’s correlational analysis, while skewed distribution was analyzed by Spearman correlation analysis. Multiple linear regression analysis was applied to calculate the relationship between CST and related variables, but only after other potential factors were controlled. According to the Kolmogorov–Smirnov test, the regression residuals obeyed a normal distribution. P<0.05 was considered as statistically significant.
Results
Clinical and Laboratory Features in the Four Groups
The Basic Index in Different Groups
The age of each group did not show any significant statistical difference (P > 0.05). The BMI was significantly lower in the two groups of DM and NC compared to that in the DM+NAFLD group, while the BMI of the NAFLD group was greater than that of the NC group, with statistically significant difference (P < 0.05). The diastolic blood pressure was significantly greater in the DM+NAFLD and NAFLD groups compared to that in the NC group (P < 0.05). There were statistical variations in gender composition in different groups (P < 0.05). The liver function in different groups: the direct bilirubin (DBIL) was significantly greater in the DM+NAFLD and DM groups when compared to that in the NC group (P < 0.05). The serum DBIL level was significantly upregulated in the DM+NAFLD group when compared to that in the NAFLD group (P < 0.05). The serum alanine transaminase (ALT) level in the DM+NAFLD and NAFLD groups was higher than that in the DM and NC groups, while the serum aspartate transaminase (AST) level in the DM+NAFLD group and the NAFLD group was greater than that in DM group (P < 0.05) (Table 1).
 |
Table 1 Clinical Characteristics of All Subjects |
Blood Lipids, Uric Acid, and Inflammation Indicators in Different Groups
The low-density lipoprotein cholesterol (LDL-C) of the DM+NAFLD and DM groups was greater than that of the NAFLD and NC groups (P < 0.05). TC and high-density lipoprotein cholesterol (HDL-C) levels were lower than those of the NAFLD and NC groups (P < 0.05). The level of triglyceride (TG) in the DM and NC groups was markedly lower than that in the DM+NAFLD and NAFLD groups (P < 0.05). The level of apolipoprotein B (ApoB) in the DM+NAFLD and DM groups was lower than that in the NAFLD group (P < 0.05). Uric acid in the DM+NAFLD group and NAFLD group was higher than that in the NC group (P < 0.05). Compared with those in the NAFLD and NC groups, the CRP levels in the DM+NAFLD and DM groups were significantly higher (P < 0.05) (Table 1).
Islet Functions in Different Groups
Compared with the NAFLD group and NC group, FPG in the DM+NAFLD group and DM group was higher (P < 0.05). FINS in the DM+NAFLD group, DM group, and NC group were significantly lower than that in the NAFLD group. The C-peptide level in the DM group and NC group was lower than that in the NAFLD group (P < 0.05). HOMA-IR in the DM+NAFLD and NAFLD group was higher than that in the NC group. HOMA-β in the NAFLD and NC group was significantly higher than that in the DM group (P < 0.05) (Table 1).
Serum CST in Different Groups
As shown in Figure 1, the mean serum CST levels was 212 (IQR 128–289) ng/mL in the DM+ NAFLD group, 152 (IQR 121–229) ng/mL in the DM group, 245 (IQR 163–349) ng/mL in the NAFLD group and 275 (IQR 204–361) ng/mL in the NC group, and further nonparametric rank tests were performed to obtain statistically significant differences in CST levels between groups (P < 0.01). Post hoc multiple comparisons showed that the levels of CST in the DM+NAFLD and DM group were significantly lower than that in the NC group (P < 0.05). The CST levels in the DM group were significantly lower than that in the NAFLD group (P < 0.01). However, there was no significant statistical difference in the serum CST levels in diabetic patients with and without NAFLD (P > 0.05). Similarly, in non-diabetic group with or without NAFLD, there was no significant statistical difference in the serum CST levels (P > 0.05).
 |
Figure 1 Serum CST levels in four groups. Note: *P<0.05, **P<0.01. |
Correlation Analyses of Serum CST
The correlational analysis revealed that the serum CST level was negatively associated with total cholesterol (TC), LDL-C, FPG, HOMA-IR, and HOMA-β (r = −0.229, r = −0.214, r = −0.159, r = −0.151, and r = −0.154, respectively, P < 0.05), but positively correlated with HDL-C (r = 0.194, P < 0.05)(Table 2). The result of regression analysis demonstrated that LDL-C and HOMA-β were independent factors influencing the serum CST level (Table 3).
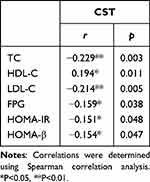 |
Table 2 Correlation of Plasma CST with the Clinical and Laboratory Characteristics |
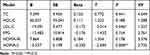 |
Table 3 Using CST as the Dependent Variable to Perform Multiple Linear Regression Analysis |
Discussion
This study is first of its kind that investigated plasma CST changes in T2DM with or without NAFLD. Our results revealed that the CST levels were lower in both the DM and DM+NAFLD groups in comparison to that in the NC group. Our findings corroborate with the data published previously by other groups. For instance, Chen et al demonstrates that the serum CST levels in newly diagnosed T2DM patients were lower than those in healthy people.11 Similarly, while studying the changes in the CST levels in GDM and normal pregnant women, Murat et al18 reported that the serum CST levels decreased in the GDM patients.
While studying the association of serum CST with insulin secretion, we demonstrated that the serum CST level was negatively associated with FBG and HOMA-β. Importantly, linear regression analysis revealed that the CST levels were significantly related to the decrease of HOMA-β. Together, these results indicated that CST was associated with insulin secretion and might play a role in abnormal glucose metabolism in T2DM patients. These results are supported by several previous studies carried out in both animals and humans. Studies carried out by Sergi Soriano et al in islets isolated from mice demonstrated that CST reduced glucose-stimulated insulin secretion.19 This effect was eliminated in the presence of SST receptor antagonists, indicating that CST works mainly through somatostatin receptor subtypes (SSTRs). Moreover, CST was reported to affect the insulin levels through pancreatic β-cell inhibition. Papotti et al20 reported the presence of CST in the human endocrine pancreas and demonstrated its participation in the regulation of insulin and glucagon levels through local autocrine or paracrine circuits. An exogenous injection of CST could reduce insulin levels in healthy volunteers and patients with prolactinoma, acromegaly, or Cushing’s disease.8,9,21 Benso et al also reported that CST inhibited insulin secretion.22 In the current study, we demonstrated that the serum CST level was negatively correlated with HOMA-β. Therefore, our observations coupled with those of the previous studies suggest that CST may be partly involved in the development of T2DM by inhibiting insulin secretion.
The current study shows that CST was negatively associated with FBG and HOMA-IR. However, as per the linear regression analysis, no relationship was found between CST and HOMA-IR. In the past studies, Chen et al11 reported that serum CST level was negatively associated with HOMA-IR and FBG in newly diagnosed T2DM patients. Similarly, Murat et al reported that the serum CST level in GDM patients was negatively associated with HOMA-IR, FBG, and FINS.18 However, linear regression analysis between CST and HOMA-IR were not assessed in both these studies. Since linear regression analysis is more convincing than correlation analysis in these aspects, our results suggesting no involvement of CST in the regulation of insulin sensitivity holds more weightage. Collectively, our study demonstrates that serum CST level is primarily involved in insulin secretion rather than in insulin sensitivity. Further investigation is, however, warranted to clarify the role of CST in the development of T2DM.
To our knowledge, there have not been either animal or human studies before on the association of CST with NAFLD. Our study is the first to examine the serum CST level changes in NAFLD in both T2DM and non-DM subjects. As the results showed, although serum CST is lower in diabetic patients, no change is observed in T2DM combined with NAFLD. Furthermore, the serum CST level was not significantly different in non-DM subjects either with or without NAFLD. These results suggested that CST was related to the development of diabetes, but not with NAFLD. This result is rational as it is consistent with the finding that the serum CST level is not associated with HOMA-IR. As is already known, one of the important pathogenic mechanisms of NAFLD is related to impaired insulin response and insulin resistance.23 NAFLD is presumed to be the hepatic manifestation of insulin resistance. Our research showed that CST is not correlated with HOMA-IR in multiple linear regression analysis, suggesting that CST does not mediate the development of insulin resistance. This conclusion is consistent with the findings that partly demonstrate the involvement of CST in the pathogenesis of T2DM, but not NAFLD.
It is widely accepted that T2DM is a chronic inflammatory disease mediated by cytokines. Previous researches have demonstrated that the inflammatory response activated in T2DM patients may reduce the circulating CST levels. An in vitro study showed that tumor necrosis factor-α (TNF-α) significantly reduced the CST expression in human coronary artery endothelial cells and ring cells.24 Similarly, the serum CST levels in the skin of patients with psoriasis were significantly decreased, which was featured by increased skin expression of proinflammatory mediators.25 Based on these observations, we measured the serum CRP levels and determined the correlation between the levels of serum CRP and CST. We discovered that CRP in the DM group was significantly higher than that in the non-DM group, suggesting a low-grade inflammatory response in T2DM. However, no significant correlation between CRP and CST was observed. The possible relationship of CST to inflammation needs to be further investigated.
T2DM patients are commonly associated with the disorders of lipid metabolism.26 Thus, we investigated the relationship of serum CST to lipid constituents. Our study suggests that CST is negatively correlated with TC and LDL-C, but positively correlated with HDL-C. Nonetheless, regression analysis of CST and blood lipids showed that only LDL-C is related to serum CST, while TC and HDL-C have no relationship with serum CST. Meanwhile, the DM group exhibited higher LDL-C levels when compared to the non-diabetic group. The correlation of CST with lipid composition is different from the results reported previously. Past studies conducted by Chen et al indicated that the serum CST levels were positively related to HDL-C in newly diagnosed T2DM.11 Contrarily, Tian et al showed that the serum CST levels in patients with coronary artery disease were negatively correlated not only with TC but also with TG.27 These different observations may be attributed to the difference in the study populations. However, all studies indicate that serum CST is related to the disorders of lipid metabolism. The relationship between lipid metabolism and CST warrants further investigation.
The study we performed has some limitations. First, NAFLD was evaluated by ultrasonography; as a result, the liver fat content and NAFLD grade could not be accurately determined. Second, we conducted a cross-sectional analyses using a relatively small sample size. Therefore, the time association and causal relationship of CST with T2DM could not be determined. Third, this study did not measure the plasma SST levels, which is structurally homologous to CST and can regulate glucagon secretion and insulin secretion.28,29 Accordingly, we plan to further analyze the plasma levels of SST in patients with T2DM combined with NAFLD and explore its relationship with CST in the future.
Conclusion
The current study confirms that CST is correlated with T2DM and may mediate the development of T2DM. However, the serum CST level is mainly correlated with HOMA-β, but not HOMA-IR in T2DM patients. Moreover, there is no correlation between CST and NAFLD either in DM or non-DM individuals. The underlying mechanism needs to be further investigated.
Abbreviations
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; TBIL, total bilirubin; DBIL, direct bilirubin; IBIL, indirect bilirubin; ALT, alanine aminotransferase; AST, aspartate transaminase; TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; ApoA1, apolipoproteinA1; ApoB, apolipoprotein B; SUA, serum uric acid; CRP, C-reactive protein; FPG, fasting plasma glucose; FINS, fasting insulin; HOMA-IR, homeostasis model assessment-insulin resistance; HOMA-β, insulin cell function index; CST, cortistatin; SST, somatostatin; T2DM, type 2 diabetes mellitus; NAFLD, nonalcoholic fatty liver disease; NC, normal control; GDM, gestational diabetes mellitus; TNF-α, tumor necrosis factor-α; IL-10, interleukin-10; GSIS, glucose-stimulated insulin secretion; SSTRs, somatostatin receptor subtypes; KATP, ATP-sensitive potassium; GIRK, G protein gated inwardly rectifying K; PCI, percutaneous coronary intervention.
Data Sharing Statement
The data can be obtained by email request ([email protected]).
Ethics Approval
The study was approved by the Ethics Committee of Hebei General Hospital.
Acknowledgments
The authors would like to thank all the reviewers who participated in the review, as well as MJEditor (www.mjeditor.com) for its English editing services during the preparation of this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Aliyeva IJ. Variability of the type II diabetes mellitus incidence rate of population of cities in the Republic of Azerbaijan. J Chem Health Risks. 2021;11:233–239.
2. Younossi ZM, Koenig AB, Abdelatif D, et al. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64(1):73–84. doi:10.1002/hep.28431
3. Bhala N, Younes R, Bugianesi E. Epidemiology and natural history of patients with NAFLD. Curr Pharm Des. 2013;19(29):5169–5176. doi:10.2174/13816128113199990336
4. Tretyakova VA, Zhernakova NI, Arisheva OS, et al. Meta-analysis of non-alcoholic fatty liver disease and electromechanical reconstruction of myocardium. Arch Razi Inst. 2022;77(1):123–128. doi:10.22092/ARI.2021.356678.1891
5. Li FP, Zhang SQ, Wang F, et al. Insulin resistance and islet beta cell function in type 2 diabetes mellitus and non alcoholic fatty liver disease. Zhonghua Nei Ke Za Zhi. 2009;48(11):940–943.
6. Spier AD, de Lecea L. Cortistatin: a member of the somatostatin neuropeptide family with distinct physiological functions. Brain Res Brain Res Rev. 2000;33(2–3):228–241. doi:10.1016/S0165-0173(00)00031-X
7. Dalm VA, Van Hagen PM, de Krijger RR, et al. Distribution pattern of somatostatin and cortistatin mRNA in human central and peripheral tissues. Clin Endocrinol (Oxf). 2004;60(5):625–629. doi:10.1111/j.1365-2265.2004.02024.x
8. Broglio F, Arvat E, Benso A, et al. Endocrine activities of cortistatin-14 and its interaction with GHRH and ghrelin in humans. J Clin Endocrinol Metab. 2002;87(8):3783–3790. doi:10.1210/jcem.87.8.8749
9. Grottoli S, Gasco V, Broglio F, et al. Cortistatin-17 and somatostatin-14rvatplay the same effects on growth hormone, prolactin, and insulin secretion in patients with acromegaly or prolactinoma. J Clin Endocrinol Metab. 2006;91(4):1595–1599. doi:10.1210/jc.2005-1837
10. Córdoba-Chacón J, Gahete MD, Pozo-Salas AI, et al. Cortistatin is not a somatostatin analogue but stimulates prolactin release and inhibits GH and ACTH in a gender-dependent fashion: potential role of ghrelin. Endocrinology. 2011;152(12):4800–4812. doi:10.1210/en.2011-1542
11. Chen W, Fu Y, Yin X, et al. Circulating levels of cortistatin are correlated with metabolic parameters in patients with newly diagnosed type 2 diabetes mellitus. Peptides. 2017;94:86–90. doi:10.1016/j.peptides.2017.05.008
12. Benitez R, Caro M, Andres-Leon E, et al. Cortistatin regulates fibrosis and myofibroblast activation in experimental hepatotoxic- and cholestatic-induced liver injury. Br J Pharmacol. 2022;179(10):2275–2296. doi:10.1111/bph.15752
13. Xidakis C, Mastrodimou N, Notas G, et al. RT-PCR and immunocytochemistry studies support the presence of somatostatin, cortistatin and somatostatin receptor subtypes in rat Kupffer cells. Regul Pept. 2007;143(1–3):76–82. doi:10.1016/j.regpep.2007.03.005
14. National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association. Guidelines of prevention and treatment for nonalcoholic fatty liver disease: a 2018 update. Zhonghua Gan Zang Bing Za Zhi. 2018;26(3):195–203. doi:10.3760/cma.j.issn.1007-3418.2018.03.008
15. Li Y, Wang J, Tang Y, et al. Bidirectional association between nonalcoholic fatty liver disease and type 2 diabetes in Chinese population: evidence from the Dongfeng-Tongji cohort study. PLoS One. 2017;12(3):e0174291. doi:10.1371/journal.pone.0174291
16. Katsuki A, Sumida Y, Gabazza EC, et al. Homeostasis model assessment is a reliable indicator of insulin resistance during follow-up of patients with type 2 diabetes. Diabetes Care. 2001;24(2):362–365. doi:10.2337/diacare.24.2.362
17. Bermu´dez V, Cano R, Cano C, et al. Homeostasis model assessment (HOMA) as surrogate insulinization criteria in patients with type 2 diabetes. Am J Ther. 2008;15(4):409–416. doi:10.1097/MJT.0b013e318160b909
18. Akbas M, Koltan SO, Koyuncu FM, et al. Decreased maternal serum cortistatin levels in pregnancies with gestational diabetes mellitus. J Matern Fetal Neonatal Med. 2020;33(7):1239–1244. doi:10.1080/14767058.2019.1627321
19. Soriano S, Castellano-Muñoz M, Rafacho A, et al. Cortistatin regulates glucose-induced electrical activity and insulin secretion in mouse pancreatic beta-cells. Mol Cell Endocrinol. 2019;479:123–132. doi:10.1016/j.mce.2018.09.009
20. Papotti M, Tarabra E, Allia E, et al. Presence of cortistatin in the human pancreas. J Endocrinol Invest. 2003;26(8):RC15–8. doi:10.1007/BF03347348
21. Giordano R, Picu A, Bonelli L, et al. The activation of somatostatinergic receptors by either somatostatin-14 or cortistatin-17 often inhibits ACTH hypersecretion in patients with Cushing’s disease. Eur J Endocrinol. 2007;157(4):393–398. doi:10.1530/EJE-07-0147
22. Benso A, Gottero C, Prodam F, et al. Effects of cortistatin-14 and somatostatin-14 on the endocrine response to hexarelin in humans. J Endocrinol Invest. 2003;26(7):599–603. doi:10.1007/BF03347014
23. Oseini AM, Sanyal AJ. Therapies in non-alcoholic steatohepatitis (NASH). Liver Int. 2017;37(Suppl1):97–103. doi:10.1111/liv.13302
24. Gruber HE, Hoelscher GL, Bethea SF, et al. Cortistatin is endogenous to the human intervertebral disc and exerts in vitro mitogenic effects on annulus cells and a downregulatory effect on TNF-α expression. Spine J. 2014;14(12):2995–3001. doi:10.1016/j.spinee.2014.06.002
25. Li D, He HL, Yao MZ, et al. Cortistatin is dysregulated in skin tissue of patients with psoriasis vulgaris and suppresses keratinocyte proliferation in vitro. Int J Dermatol. 2015;54(8):e309–314. doi:10.1111/ijd.12836
26. Henning RJ. Type-2 diabetes mellitus and cardiovascular disease. Future Cardiol. 2018;14(6):491–509. doi:10.2217/fca-2018-0045
27. Tian QP, Feng XR, Pang YZ, et al. Relationship between plasma cortistatin and coronary heart disease. Beijing Da Xue Xue Bao Yi Xue Ban. 2009;41(5):537–540.
28. Liu Y, Yin XH, Qi YF. Progress in biological effects of cortistatin. Sheng Li Ke Xue Jin Zhan. 2009;40(3):219–224.
29. Chanclon B, Martinez-Fuentes AJ, Gracia-Navarro F. Role of SST, CORT and ghrelin and its receptors at the endocrine pancreas. Front Endocrinol. 2012;3:114.
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
