Back to Journals » International Medical Case Reports Journal » Volume 14
Scleral Buckle Infection Caused by Mycobacterium chelonae: A Case Report
Authors Kusaka M, Kagitani Y, Hama S, Kita M
Received 18 December 2020
Accepted for publication 15 January 2021
Published 5 February 2021 Volume 2021:14 Pages 51—53
DOI https://doi.org/10.2147/IMCRJ.S294627
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Supplementary video of "Mycobacterium chelonae buckle infection" [ID 294627].
Views: 1284
Mami Kusaka, Yu Kagitani, Sachiyo Hama, Mihori Kita
Department of Ophthalmology, National Hospital Organization Kyoto Medical Center, Kyoto, Japan
Correspondence: Mihori Kita
Department of Ophthalmology, National Hospital Organization Kyoto Medical Center, 1-1 Mukaihata-Cho, Fukakusa, Fushimi-Ku, Kyoto-city, Kyoto, 612-8555, Japan
Tel +81-75-641-9161
Fax +81-75-643-4325
Email [email protected]
Abstract: A 63-year-old female with a history of retinal detachment repair with scleral buckle from 28 years prior presented with exposure of the buckle in her right eye. After removing the buckle and anchoring sutures, the operation field was washed with 0.25% polyvinyl alcohol-iodine, vancomycin, and ceftazidime. Cultures revealed Mycobacterium chelonae. Systemic meropenem was discontinued on postoperative day 5. The retina remained attached, and there was no recurrence over a 1-year follow-up. We report a case of NTM buckle infection that responded well to thorough washings with polyvinyl alcohol-iodine and antibiotic solutions instead of using a prolonged course of systemic antibiotics.
Keywords: scleral buckle, infection, Mycobacterium chelonae, nontuberculous mycobacteria, NTM, polyvinyl alcohol-iodine, artificial device
Introduction
Scleral buckle infection is rare, with a reported incidence varying from 0.5% to 5.6%.1 However, this infection can potentially lead to severe complications such as endophthalmitis, panophthalmitis, and orbital cellulitis.2,3 Nontuberculosis mycobacterium infections are typically hard to manage due to poor drug penetration and a slow response to treatment.4 As a result, prolonged administration of systemic antibiotics is often necessary.
This report presents a case of Mycobacterium chelonae (M.chelonae) scleral buckle infection in which systemic antibiotics were successfully stopped a few days after removal of the buckle followed by thorough washings with 0.25% polyvinyl alcohol-iodine and antibiotics.
Case Report
This study was approved by the Ethics Committee of the National Hospital Organization Kyoto Medical Center, and all procedures involved adhered to the tenets of the Declaration of Helsinki. Written informed consent for the procedure and participation in the study including case details, images, and videos published was obtained from the patient after receiving an explanation of the therapeutic procedures.
A healthy 63-year-old female with a history of retinal detachment repair of her right eye including four scleral buckling surgeries 28 years ago was referred to our clinic. She had been suffering from a discharge in her right eye for 2 weeks. Slit-lamp examination revealed conjunctival injection of the right eye without any sign of iridocyclitis or keratitis, and exposure of the scleral buckle (Figure 1). She had high myopia with an axial length of 33.55 mm in her right eye. The best-corrected visual acuity was hand motion in the right eye, which did not change after the buckling surgery, and 20/20 in the left eye, for which the axial length was 25.33 mm. Systemic examination, which included a chest X-ray, showed no abnormal findings.
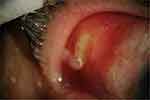 |
Figure 1 Exposed sponge buckle at the superior-temporal area at 8mm from the limbus, and injection of conjunctiva were observed in her right eye. |
After the culture of the conjunctival swab and subsequently being diagnosed with scleral buckle infection, the patient was started on a topical levofloxacin treatment along with 1000 mg of intravenous meropenem three times a day. On the following day, the encircling sponge buckle and the anchoring sutures were removed. The operation field was then washed with 50 mL of 0.25% polyvinyl alcohol-iodine, and the same amount of vancomycin (10mg/mL) and ceftazidime (20mg/mL) through the conjunctival tunnel that was formed during the buckle removal. (Supplementary Video 1) During the final step of the surgery, a subconjunctival injection of vancomycin and ceftazidime was performed. After the conjunctival injection and discharge disappeared on postoperative day 5, the systemic antibiotic was then discontinued. The retina remained attached, and there has been no recurrence of the inflammation over the 1-year follow-up.
On postoperative day 7, all of the culture results for the conjunctiva swab obtained at the first visit, and for the removed buckle and sutures identified the presence of M.chelonae (Gaffky scale 2). On postoperative days 6 and 8, there was no longer any growth found for the subsequent cultures of the conjunctival swab.
Discussion
One of the postoperative complications of scleral buckling surgery is buckle infection. It was reported that repeated washing of the ocular surface with 0.25% povidone-iodine during the scleral buckling surgery was able to minimize the ocular surface bacterial contamination at the completion of the procedure.5 The usual treatment for buckle infection is the removal of the buckle combined with systemic antibiotic treatment. The most commonly found organisms in these cases are coagulase-negative staphylococci, followed by Mycobacterium chelonae,1 which was identified in our current case.
Mycobacterium chelonae belongs to a nontuberculous mycobacteria species (NTM). Although these ubiquitous environmental organisms are found in the soil, dust, and water, they rarely cause infections. However, NTM have been recently recognized as being responsible for the increased morbidity of infections associated with artificial devices such as pacemakers, prosthetic cardiac valves, joint implants, intravenous catheters, and scleral buckle in not only immunosuppressed but also in immunocompetent patients.6–9
NTM infections are typically challenging to manage due to delayed laboratory identification, poor drug penetration, and slow response to therapy. Moreover, silicone sponge buckle elements can promote the development of a biofilm, which is thought to be responsible for scleral buckle infections even without exposure to the buckle.10,11 Fortunately, there could be no biofilm development found in our current case, which would have demonstrated a good response to our therapy.
NTM, especially the rapidly growing mycobacteria (RGM), which include Mycobacterium chelonae, are not sensitive to first-line antituberculous agents. NTM infections in and around the eye, which have been shown to be clinically recalcitrant to treatment, recur after cessation of therapy, and require long-term therapy of antibiotics including amikacin, clarithromycin, and ciprofloxacin.11,12
In our current case, we were able to successfully withdraw the use of systemic antibiotics during the early phase of buckle removal, without any recurrence of the inflammation over the 1-year follow-up. Thus, washing of the operation field with 0.25% polyvinyl alcohol-iodine, along with the administration of antibiotics at the time of the removal of the scleral buckle and sutures as soon as possible after the buckle exposure, can potentially lead to good results in Mycobacterium chelonae buckle infections in immunocompetent patients.
NTM infections are rare, and often require lengthy treatment. We report a case of NTM buckle infection that responded well to thorough washings with 0.25% polyvinyl alcohol-iodine and antibiotic solutions instead of using a prolonged course of systemic antibiotics.
Patient Consent
Obtained.
Disclosure
No conflicting relationship exists for all authors.
References
1. Chhablani J, Nayak S, Jindal A, et al. Scleral buckle infections: microbiological spectrum and antimicrobial susceptibility. J Ophthalmic Inflamm Infect. 2013;3(1):67. doi:10.1186/1869-5760-3-67
2. Tsui I. Scleral buckle removal: indications and outcomes. Surv Ophthalmol. 2012;57(3):253–263. doi:10.1016/j.survophthal.2011.11.001
3. Nemet AY, Ferencz JR, Segal O, Meshi A. Orbital cellulitis following silicone-sponge scleral buckles. Clin Ophthalmol. 2013;7:2147–2152. doi:10.2147/OPTH.S50321
4. Moorthy RS, Valluri S, Rao NA. Nontuberculous mycobacterial ocular and adnexal infections. Surv Ophthalmol. 2012;57(3):202–235. doi:10.1016/j.survophthal.2011.10.006
5. Shimada H, Nakashizuka H, Hattori T, et al. Prophylaxis for acute scleral buckle infection using 0.25 % povidone-iodine ocular surface irrigation during surgery. Int Ophthalmol. 2014;34(2):211–216. doi:10.1007/s10792-013-9816-5
6. Yuan SM. Mycobacterial Endocarditis: a Comprehensive Review. Braz J Cardiovasc Surg. 2015;30:93–103.
7. Hooda A, Pati PK, John B, George PV, Michael JS. Disseminated mycobacterium chelonae infection causing pacemaker lead endocarditis in an immunocompetent host. BMJ Case Rep. 2014;22:bcr2014–206042.
8. Roest S, Bax HI, Verkaik NJ, et al. Mycobacterium Chelonae, an ‘Atypical’ Cause of an LVAD Driveline Infection. Int J Infect Dis. 2020;92:127–129. doi:10.1016/j.ijid.2020.01.002
9. Iordache SD, Daneman N, Axelrod TS. Mycobacterium chelonae infection following silicone arthroplasty of the metacarpophalangeal joints: a case report. Hand. 2009;4(2):129–133. doi:10.1007/s11552-008-9138-7
10. Churgin DS, Tran KD, Gregori NZ, Young RC, Alabiad C, Flynn HW
11. Shah M, Relhan N, Kuriyan AE, et al. Endophthalmitis caused by nontuberculous mycobacterium: clinical features, antimicrobial susceptibilities, and treatment outcomes. Am J Ophthalmol. 2016;168:150–156. doi:10.1016/j.ajo.2016.03.035
12. Kim AY, Davis AS, Moreau A, Drevets DA, Melendes DP. Management of nontuberculous mycobacterial infections of the eye and orbit: a retrospective case series. Am J Ophthalmol Case Rep. 2020;20:100971. doi:10.1016/j.ajoc.2020.100971
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
