Back to Journals » Clinical Ophthalmology » Volume 10
Safety and comfort evaluation of a new formulation of Visine® lubricant eye drops containing HydroBlend™ and GentlePur™
Authors Torkildsen G, Frisch S, Bai M, Gentner III L, Doshi U, Zhang J
Received 13 March 2015
Accepted for publication 3 September 2015
Published 19 February 2016 Volume 2016:10 Pages 331—336
DOI https://doi.org/10.2147/OPTH.S84529
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Gail Torkildsen,1 Sherryl Frisch,2 Mingqi Bai,2 Louis Gentner III,2 Uday Doshi,2 Jane Zhang2
1Andover Eye Associates, Andover, MA, USA; 2Johnson & Johnson Consumer and Personal Products Worldwide, Division of Johnson & Johnson Consumer Companies, Inc, Morris Plains, NJ, USA
Purpose: To evaluate the clinical safety and comfort of a new benzalkonium chloride-free Visine® lubricant eye drop formulation (Hydroblend™ and GentlePur™) in healthy and dry eye subjects.
Methods: This was a single-site, open-label clinical study comprised of 22 healthy and 22 dry eye subjects. Subjects were instructed to instill 1–2 drops of the test product four times a day for 2 weeks and were examined at visit 1 (day 0), visit 2 (day 7), and visit 3 (day 14). Assessments at each visit included postdosing product usage comfort scores, predosing fluorescein corneal staining score, predosing visual acuity, and pre- and postdosing ocular structure change using slit-lamp biomicroscopy. Adverse events were monitored throughout the course of the study.
Results: Throughout the 14 days of the trial period, subjects from both healthy and dry eye groups rated the eye drops as “very comfortable”. For dry eye group, the mean product usage comfort scores for the first 3 minutes postdosing ranged from 8.5 to 8.8 at visit 1 and 9.2 to 9.6 at visit 3 on a 0–10 point scale, with 0 being very uncomfortable and 10 being very comfortable. The mean corneal staining scores over five corneal regions changed from 0.65 at visit 1 to 0.39 at visit 3 for dry eye group. The individual region corneal staining scores were also decreased from visits 1 to 3 for dry eye group. All subjects maintained pretreatment means visual acuity at visits 2 and 3. Biomicroscopic examination indicated no structural changes at all visits. There were no significant adverse events reported during the course of the study.
Conclusion: The study confirms that GentlePur™ is an appropriate choice as a preservative for ocular application. The new formulation was safe and comfortable when used four times a day in healthy and dry eye subjects.
Keywords: lubricant eye drop, preservative, safety, polyquaternium-42, GentlePur™, benzalkonium chloride (BAK), Hydroblend™
Introduction
According to the 2007 Report of the International Dry Eye Workshop,1 dry eye is a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort,2–4 visual disturbance,5–7 and tear film instability8–10 with potential damage to the ocular surface.11 It has been estimated that approximately 5 million Americans 50 years and older have moderate-to-severe forms of dry eye, with millions more affected by less severe forms of the disease.12,13 While dry eye has traditionally affected older patients, the population of dry eye sufferers has been trending toward a younger age group in recent years. This is likely due to factors such as increases in laser vision correction procedures, chronic ocular allergies, and the growing numbers of persons who perform visual tasks over long period of time.14
The varying severity and complexity of dry eye make it a challenging disease to treat. Without proper treatment, dry eye disease can adversely affect the quality of life,15 visual function,16 contact lens tolerance,17 and refractive surgery outcomes.18 Lubricant eye drop (also known as artificial tear) remain as the first line of treatment for dry eye and are commonly used to supplement for deficient natural tears.19 Like most multidose topical formulations for ocular use, these tear substitutes contain preservatives to inhibit microbial contamination. While preservatives are not required in unit dose vials, these nonpreserved preparations are more costly for the patients to purchase and less convenient to use than ocular lubricants in multidose packaging. Among common ophthalmic preservatives, benzalkonium chloride (BAK, BZK, or BAC) was the first ophthalmic preservative and has been in use for over 60 years.20 BAK is a quaternary ammonium surfactant preservative and is currently used in over 70% of ophthalmic products on the market.21 While the preservative efficacy (PE) of BAK is well established, it has also come under scrutiny over the years for its potential to disrupt the precorneal tear film and damage the epithelial surface of the cornea, especially for dry eye patients who use the product frequently.22–27 To avoid the potential risks associated with BAK use in dry eye patients, many commercially available tear substitute products now offer preservative-free or BAK-free solutions.
A systematic development program was initiated to identify, characterize, and validate a novel, safe, effective, and gentle preservative as an alternative to BAK. The preservative was required to meet three fundamental criteria: 1) potency of PE; 2) potential lack of ophthalmic or systemic toxicity based on available literature data; 3) potential lack of reactivity to other commonly used ophthalmic ingredients.
After a thorough literature search, a list of preservatives that may have potential for ophthalmic use was generated. A preservative effectiveness test on all preservatives using a lubricant eye drop formula without BAK was carried out, and the products with adequate PE were selected. Subsequently, a series of in vitro studies including transepithelial permeability, fluorescein leakage, and the neutral red uptake cytotoxicity procedure were conducted using BAK as the negative control. Results from the neutral red uptake procedure were most discerning regarding the cytotoxicity of the preservatives. On the basis of all the in vitro test results, polyquaterium-42 (GentlePur™) demonstrated superior preservative effectiveness and much less irritability than BAK.
GentlePur™ is a quaternary ammonium cationic surfactant polymer that contains straight polyethylene chain segments. In comparison, BAK is a quaternary ammonium cationic surfactant monomer that contains benzyl rings segments. Although both are highly effective antimicrobial agents. GentlePur™ has extremely low ocular irritancy and has been used as a preservative in contact lens solutions and lubricant eye drops.28,29
The purpose of this study was to evaluate the comfort and safety of a new formulation of Visine® (Johnson & Johnson, New Brunswick, NJ, USA) lubricant eye drops containing GentlePur™ and 0.2% glycerin, 0.36% hypromellose, and 1% polyethylene glycol 400 “Hydroblend™” in healthy and dry eye subjects.
Methods
Study population
This study was conducted in compliance with the ethical principles of Good Clinical Practices, the Declaration of Helsinki, the International Conference on Harmonization guidelines, and all applicable local and federal requirements relevant to the use of investigational drugs. The study employed a single-site, open label, investigator-masked protocol. The two study groups enrolled 22 subjects each; one group of subjects was a healthy control group, the other group of subjects had a confirmed diagnosis of dry eye. Inclusion criteria for both groups were as follows: subjects needed to be at least 18 years old, follow all study directions, and attend all required visits, and provide written informed consent for study participation. All subjects had corrected visual acuity of logMar +0.5 (ETDRS, Early Treatment Diabetic Retinopathy Study) or better in each eye and good overall ocular health (with the exception of the dry diagnosis). Subjects were excluded if they had used contact lenses within 1 week of the study or were planning to wear contacts within the anticipated study timeframe; had any active ocular inflammation or allergy; had used any topical ophthalmic agents within 7 days of the study; had ever undergone laser-assisted in situ keratomileusis, surgery for neurotrophic keratitis or corneal transplant; had any ocular surgery within 6 months; or were currently using any systemic medication known to cause ocular dryness. Subjects with known sensitivities to any study components or those with any medical condition that could affect study parameters were also excluded, as were women of childbearing age who were pregnant or unwilling to use approved birth control for the duration of the study. Subjects who had participated in any other clinical trial in the previous 30 days were also ineligible. To qualify for the dry eye group, subjects were required to have a history of lubricant eye drop use in the previous 12 months, and fluorescein corneal staining score ≥1 (National Eye Institute [NEI] scale) in any of the five regions and a total score <7 in one eye; subjects who did not meet both of these criteria, or subjects with a history of lubricant eye drop use ≥6 times per day, were excluded from both control and dry eye groups.
Test product
The test product is a prototype lubricant eye drop formulation. It combines Hydroblend™ (0.2% glycerin, 0.36% hypromellose, and 1% polyethylene glycol 400) and the preservative GentlePur™ (polyquaternium-42).
Study protocol
The study duration was 2 weeks. Each subject attended three study visits: visit 1 (day 0, baseline), visit 2 (day 7±1), and visit 3 (day 14±1). Following baseline assessment on day 0, all subjects were provided with test lubricant eye drop and instructed to instill the drops into each eye (1–2 drops) four times a day for 14 days. Assessments conducted at each visit included visual acuity, ocular structure change using slit-lamp biomicroscopy, and fluorescein corneal staining (inferior, superior, central, temporal, nasal) based on the NEI scale of 0–330,31 (0= normal and 3= severe). Product usage comfort assessments were conducted at visits 1 and 3 for each eye immediately and at 1, 2, and 3 minutes postdosing using a scale ranging from 0 to 10, with 0 being very uncomfortable and 10 being very comfortable. The assessment schedule is summarized in Table 1. Additionally, adverse events were monitored and recorded throughout the entire course of the study.
  | Table 1 Safety assessment schedule |
Statistical analysis
Forty-four subjects, 22 healthy and 22 with dry eye, were enrolled in the study. One dry eye subject was discontinued after visit 1 due to noncompliance (unable to attend visit 2). Sex was summarized by numbers and percentages, and age was summarized by means and standard deviations. Scores for each eye were obtained for visual acuity, product usage comfort, and corneal staining (inferior, superior, central, temporal, nasal, and average over the five regions). These were averaged over the two eyes to give corresponding subject-level scores, and the subject-level scores were averaged over all subjects to give mean group scores.
Results
Study population
A total of 56 subjects were screened to identify 22 subjects for each study group. After the study began, one subject from the dry eye group was discontinued due to noncompliance. A summary of demographics is presented in Table 2. Overall, the dry eye group was somewhat older and had a larger number of women, as expected from the prevalence of the disease in the general population.
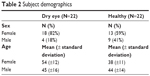  | Table 2 Subject demographics |
Product usage eye comfort score
At the first (visit 1) and last (visit 3) study visits, subjects were asked to provide assessments of drop comfort for each eye immediately after instillation and at 1, 2, and 3 minutes following instillation. The assessments used a 0–10 point scale, where 0 is very uncomfortable and 10 is very comfortable. Mean values of four time points at each visit, shown in Table 3 and Figure 1A and B, are all 8.50 or higher for all time points, visits, and subject groups.
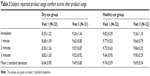  | Table 3 Subject reported product usage comfort scores after product usage |
  | Figure 1 Mean of subjects’ reported product usage eye comfort score for dry eye (A) and healthy eye (B) groups. |
Corneal staining score
Fluorescein staining of the corneal surface from inferior, superior, central, temporal, nasal region, and an average over the five regions based on the NEI scale of 0–3 (0= normal and 3= severe) was assessed and the results are included in Table 4.
  | Table 4 Mean corneal staining for dry eye and healthy eye groups from each region and average across five regions |
For the dry eye group, the five-region corneal staining scores improved from visits 1 to 3. The average staining scores over the five regions of the cornea were 0.65 at visit 1, 0.75 at visit 2, and 0.39 at visit 3. A decrease in corneal fluorescein staining from baseline (visit 1) to visit 3 for dry eye group is presented in Figure 2.
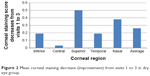  | Figure 2 Mean corneal staining decrease (improvement) from visits 1 to 3 in dry eye group. |
For the healthy group, the corneal staining scores at baseline were very low (0.15) and they showed very little changes from baseline at visits 2 and 3.
Visual acuity
Averaging over both eyes, pretreatment (baseline) visual acuity means on the logMAR scale were 0 and −0.02 for the dry eye subjects and healthy subjects, respectively. For both groups, visual acuity means were maintained at visit 2 and improved by 0.03 at visit 3.
Ocular structure using slit-lamp biomicroscopy
Slit-lamp biomicroscopy was conducted on four ocular structures (lid, lens, conjunctiva, and cornea) predose and postdose at all three visits for a total of six time points. For all structures, the five posttreatment findings showed no change from visit 1 pretreatment findings.
Adverse events
In addition to the acuity and slit-lamp examinations, a tabulation of all reported adverse events was used in the overall safety assessment. Reported adverse events were mild and did not require action to mitigate the effects observed. Two dry eye subjects had a total of four adverse events from the entire study population. Two adverse events (filmy vision upon instillation and blurred vision after instillation) were related to the study medication. The other two adverse events (ocular redness and increased tears) were possibly related to the study medication. All adverse events were assessed as mild and nonserious. There were no discontinuations due to the study medication.
Discussion
In order to determine the potential for ocular irritation by GentlePur™ and provide data to support its use in humans, a rabbit study was conducted. In this study, ten rabbits were dosed with lubricant eye drops preserved with GentlePur™ six times daily for 30 consecutive days. This study established that topical administration of the preservative was nontoxic and nonirritating in the rabbit eye test system.
In the current study, GentlePur™ preserved lubricant eye drops were tested for eye comfort and safety in both a healthy control group and in subjects with mild dry eye disease. At the beginning and the end of 2 weeks of treatment, all subjects found the product to be comfortable to use, with product usage eye comfort means of 8.5 or higher on a scale of 0–10, where 0 is very uncomfortable and 10 is very comfortable, indicating a high tolerance of the test product for both dry eye and healthy eye subjects. From visits 1 to 3, the eye comfort score at each time point (immediately, 1, 2, and 3 minutes after dosing) and mean eye comfort score over four time points increased numerically for dry eye subjects and maintained nearly no change for healthy eye subjects (Table 3 and Figure 1A and B).
Corneal fluorescein staining has been recognized as a reliable clinical measurement for ocular surface condition and is correlated with the severity of dry eye. Per study protocol inclusion criteria, dry eye subjects were selected with corneal staining score of ≥1 in any of the five regions on the NEI scale in at least one eye. Therefore, it is expected that dry eye group had higher baseline corneal fluorescein staining score than that of healthy subjects in all five corneal regions. Although the corneal staining score increased from visits 1 to 2, it decreased from visits 1 to 3 for all five regions in dry eye group, indicating an overall improvement from the new lubricant eye drop treatment for dry eye patients. For the healthy eye group, the mean baseline corneal staining score was very low (0.15) and remained very low (0.10) at the end of the 2-week treatment, indicating no increase of eye dryness for healthy eye subjects (Table 4 and Figure 2).
Over the trial period there was also no evidence of deteriorating outcomes in visual acuity following treatment. In fact, similar to corneal staining, there was a tendency for slight improvement from visits 1 to 3. Product usage comfort increased slightly with use for dry eye subjects and was fairly consistent for healthy subjects.
Slit-lamp biomicroscopic examination on four ocular regions did not find any structural change after 2 weeks of treatment from the baseline. While there were four adverse events reported in this study, all were considered to be mild and nonserious, as there were no discontinuations due to the study medication, and all four events resolved by the end of the study date or shortly thereafter.
Conclusion
The prototype lubricant eye drop formulation from the makers of Visine® combines a gentle and effective preservative GentlePur™ with unique Hydroblend™ technology to provide a new lubricant eye drop option for dry eye sufferers and can be used as often as needed as directed by label instructions.
Acknowledgment
Financial support for this study was provided by Johnson & Johnson Consumer and Personal Products Worldwide (Division of Johnson & Johnson Consumer Companies).
Disclosure
The authors report no conflicts of interest in this work.
References
International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):75–92. | ||
Begley CG, Chalmers RL, Abetz L, et al. The relationship between habitual patient-reported symptoms and clinical signs among patients with dry eye of varying severity. Invest Ophthalmol Vis Sci. 2003;44:4753–4761. | ||
Adatia FA, Michaeli-Cohen A, Naor J, et al. Correlation between corneal sensitivity, subjective dry eye symptoms and corneal staining in Sjogren’s syndrome. Can J Ophthalmol. 2004;39:767–771. | ||
Vitale S, Goodman LA, Reed GF, Smith JA. Comparison of the NEI-VFQ and OSDI questionnaires in patients with Sjogren’s syndrome-related dry eye. Health Qual Outcomes. 2004;2:44. | ||
Rieger G. The importance of the precorneal tear film for the quality of optical imaging. Br J Ophthalmol. 1992;76:157–158. | ||
Liu Z, Pflugfelder SC. Corneal surface irregularity and the effect of artificial tears in aqueous tear deficiency. Ophthalmology. 1999;106:936–943. | ||
Goto E, Yagi Y, Matsumoto Y, Tsubota K. Impaired functional visual acuity of dry eye patients. Am J Ophthalmol. 2002;133:181–186. | ||
Holly F, Lemp MA. Formation and rupture of the tear film. Exp Eye Res. 1973;15:515–525. | ||
Bron AJ. Diagnosis of dry eye. Surv Ophthalmol. 2001;45(Suppl 2):S221–S226. | ||
Goto T, Zheng X, Klyce SD, et al. A new method for tear film stability using videokeratography. Am J Ophthalmol. 2003;135:607–612. | ||
Stern ME, Siemasko KF, Gao J, Calonge M, Niederkorn JY, Pflugfelder SC. Evaluation of ocular surface inflammation in the presence of dry eye and allergic conjunctival disease. Ocul Surf. 2005;3(4 Suppl):S161–S164. | ||
Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003;136(2):318–326. | ||
Schaumberg DA, Dana R, Buring JE, Sullivan DA. Prevalence of dry eye disease among US men: estimates from the Physicians’ Health Studies. Arch Ophthalmol. 2009;127(6):763–768. | ||
Blehm C, Vishnu S, Khattak A, Mitra S, Yee RW. Computer vision syndrome: a review. Surv Ophthalmol. 2005;50(3):253–262. | ||
Mertzanis P, Abetz L, Rajagopalan K, et al. The relative burden of dry eye in patients’ lives: comparisons to a US normative sample. Invest Ophthalmol Vis Sci. 2005;46:46–50. | ||
Gulati A, Sullivan R, Buring JE, et al. Validation and repeatability of a short questionnaire for dry eye syndrome. Am J Ophthalmol. 2006;142:125–131. | ||
Nichols JJ, Ziegler C, Mitchell GL, Nichols KK. Self-reported dry eye disease across refractive modalities. Invest Ophthalmol Vis Sci. 2005;46:1911–1914. | ||
Albietz JM, Lenton LN, McLennan SB. Chronic dry eye and regression after laser in situ keratomileusis for myopia. J Cataract Refract Surg. 2004;30:675–684. | ||
Calonge M. The treatment of dry eye. Surv Ophthalmol. 2001;45(Suppl 2):S227–S239. | ||
McPherson S, Wood R. Self-sterilizing ophthalmic solutions. Am J Ophthalmol. 1949;32:675. | ||
Reeman PD, Kahook MY. Preservatives in topical ophthalmic medications: historical and clinical perspectives. Exp Rev Ophthalmol. 2009;4(1):59–64. | ||
Wilson WS, Duncan AJ, Jay JL. Effect of benzalkonium chloride on the stability of the precorneal tear film in rabbit and man. Br J Ophthalmol. 1975;59(11):667–669. | ||
Ishibashi T, Yokoi N, Kinoshita S. Comparison of the short-term effects on the human corneal surface of topical timolol maleate with and without benzalkonium chloride. J Glaucoma. 2003;12(6):486–490. | ||
Berdy GJ, Abelson MB, Smith LM, George MA. Preservative-free artificial tear preparations. Assessment of corneal epithelial toxic effects. Arch Ophthalmol. 1992;110(4):528–532. | ||
Debbasch C, Brignole F, Pisella PJ, Warnet JM, Rat P, Baudouin C. Quaternary ammoniums and other preservatives’ contribution in oxidative stress and apoptosis on Chang conjunctival cells. Invest Ophthalmol Vis Sci. 2001;42(3):642–652. | ||
de Jong C, Stolwijk T, Kuppens E, de Keizer R, van Best J. Topical timolol with and without benzalkonium chloride: epithelial permeability and autofluorescence of the cornea in glaucoma. Graefes Arch Clin Exp Ophthalmol. 1994;232(4):221–224. | ||
Collin HB, Grabsch BE. The effect of ophthalmic preservatives on the healing rate of the rabbit corneal epithelium after keratectomy. Am J Optom Physiol Opt. 1982;59(3):215–222. | ||
Boost M, Cho P, Lai S. Efficacy of multipurpose solutions for rigid gas permeable lenses. Ophthal Physiol Opt. 2006;26:468–475. | ||
Michelson P, Fox R. Overview of management of dry eye associated with Sjogren’s syndrome. In: Fox RI, Fox CM, editors. Sjogren’s Syndrome. New York, NY: Springer Science + Business Media, LLC; 2011. | ||
Lemp M. Report of the National Eye Institute/Industry Workshop on Clinical Trials in Dry Eyes. CLAO J. 1995;21(4):221–232. | ||
Versura P, Bavelloni A, Grillini M, Fresina M, Campos E. Diagnostic performance of a tear protein panel in early dry eye. Mol Vis. 2013;19:1247–1257. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
