Back to Journals » OncoTargets and Therapy » Volume 11
Role of palliative resection of the primary pancreatic neuroendocrine tumor in patients with unresectable metastatic liver disease: a systematic review and meta-analysis
Authors Zhou B , Zhan C, Ding Y, Yan S, Zheng S
Received 27 November 2017
Accepted for publication 9 January 2018
Published 23 February 2018 Volume 2018:11 Pages 975—982
DOI https://doi.org/10.2147/OTT.S158171
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Carlos E Vigil
Bo Zhou,1 Canyang Zhan,2 Yuan Ding,1 Sheng Yan,1 Shusen Zheng1
1Division of Hepatobiliary and Pancreatic Surgery, Department of Surgery, First Affiliated Hospital, 2Department of Neonatology, Children’s Hospital, School of Medicine, Zhejiang University, Hangzhou, People’s Republic of China
Background: Treatment for pancreatic neuroendocrine tumors (PNETs) in patients with unresectable metastatic liver disease has long been a controversial issue. This systematic review aims to summarize the existing evidence concerning the value of primary tumor resection in this group of patients.
Methods: A systematic review of the literature and a meta-analysis were performed. The PubMed and Cochrane databases were searched to identify articles that compared palliative primary tumor resection and nonsurgical regimens in patients with PNETs and unresectable liver metastases. Relevant articles were identified in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The primary outcome was overall survival. The included studies were evaluated for heterogeneity and publication bias.
Results: Overall, 10 studies were included in the analysis. No randomized controlled trials (RCTs) were identified. These studies included 1,226 patients who underwent a resection of the primary tumor and 1,623 patients who did not undergo surgery. The median overall survival was 36–137 and 13.2–65 months in the surgical and nonsurgical groups, respectively, and the 5-year overall survival rate was 35.7–83 and 5.4%–50%, respectively, in these two groups. The meta-analysis demonstrated that there was no significant difference in liver tumor burden (odds ratio [OR] =1.51, 95% CI: 0.59–3.89, P=0.39) or tumor grade (OR =2.88, 95% CI: 0.92–9.04, P=0.07) among patients who underwent surgery and nonsurgical therapy. Furthermore, patients who underwent an aggressive surgical approach appeared to have a higher tumor grade. However, the meta-analysis demonstrated that patients who underwent primary tumor resection had better overall survival (P<0.001), with a pooled hazard ratio of 0.36 (95% CI: 0.30–0.45). No publication bias was detected.
Conclusion: This meta-analysis demonstrates that the palliative resection of the primary tumor in patients with PNETs and unresectable liver metastases can increase survival, although a bias toward a more aggressive surgical approach in patients with better performance status, less advanced disease, or a tumor located in the body or tail of the pancreas appears likely. RCTs with longer follow-up periods are required to confirm the advantages of palliative primary tumor resection for PNETs.
Keywords: pancreatic neuroendocrine tumors, surgery, liver metastases, prognosis
Introduction
The incidence of pancreatic neuroendocrine tumors (PNETs), which were previously considered rare heterogeneous tumors, has almost doubled to 10% over the past two decades.1,2 PNETs account for ~1%–2% of all pancreatic neoplasms and 7.0% of all neuroendocrine tumors.3 Surgery remains the only potentially curative treatment for PNETs, but its indication is often limited by the fact that most patients with PNETs present with metastatic disease at diagnosis. Several studies have demonstrated that liver metastases are found in >50% of patients affected by PNETs (80% of cases are bilobar).4 However, the clinical course of the disease is usually characterized by a relatively indolent history and it has a 5-year survival rate exceeding 60%.5 This feature is due to the tendency to promote aggressive surgery, even in the presence of liver metastases. This aggressive surgical approach seems justified whenever radical resection may be achieved on both the primary tumor and the liver metastases.6–8 However, in patients who are unsuitable for radical resection due to local vascular infiltration or liver metastases that extend to both liver lobes, or due to extrahepatic metastases, the indication for surgery remains highly controversial.9,10
Cytoreductive surgery, also termed debulking, refers to the greatest possible reduction of the tumor mass in the context of the persistence of metastases and is generally carried out when it is possible to remove 70%–90% of the disease.11 However, the indication for primary PNET removal in the presence of unresectable liver metastases is controversial: studies have shown that this practice could provide relief from hormonal and local tumor-related symptoms and increase disease control,12–15 but data on the possible amelioration of survival rates are scanty and affected by selection bias.
We therefore designed a systematic review to compare the effects of the primary tumor resection with those of nonsurgical regimens in patients with PNETs and unresectable liver metastases.
Methods
Search strategy
An in-depth literature search was performed with the following search terms: (neuroendocrine tumor OR endocrine tumor OR neuroendocrine tumor OR endocrine tumor OR islet cell carcinoma OR beta-cell tumor OR pancreatic endocrine tumor OR pancreatic endocrine tumor OR islet cell tumor) AND (operative surgical procedure OR operative surgical OR operative procedure OR pancreatectomy OR pancreatectomies OR pancreaticoduodenectomy OR pancreatoduodenectomies OR duodenopancreatectomy OR duodenopancreatectomies OR primary resection OR splenopancreatectomy OR primary tumor resection OR primary tumor resection) AND (metastatic OR metastasis OR secondary OR advanced OR residual neoplasm OR residual cancer OR residual tumor) AND (liver OR hepatic). We searched the computerized databases PubMed and Cochrane to identify eligible studies in English-language journals before June 2017.
The references of each of the included studies were also screened for any additionally relevant articles. The titles of all identified articles were screened to ascertain their relevance. Abstracts and/or full texts of selected potentially relevant papers were further evaluated.
Selection criteria
Only studies that reported the survival of patients with PNETs and unresectable liver metastases (regardless of the presence or absence of extrahepatic disease, tumor grade, or functional status) were considered. Studies were included regardless of study type, publication status, or sample size. We intended to analyze randomized controlled trials (RCTs), quasi-RCTs, and non-RCTs, but given the likely paucity of high-quality research on the topic, prospective or retrospective cohort studies were also considered. Case–control studies, case series, case reports, and other observational studies were excluded.
Studies that reported survival outcomes following a direct comparison between patients who underwent primary tumor resection and those who did not undergo resection were included. In both the surgical and nonsurgical groups, other medical (somatostatin analogs, chemotherapy, peptide receptor radiotherapy) or ablative (transarterial chemoembolization, radiofrequency ablation, cryoablation) treatments were permitted. In the case of duplicate publications, the most recent or most complete data were included.
Two independent reviewers (BZ and YD) carried out the study identification, selection, and data extraction. If there was no consensus, the principal investigator (SZ) made the final decision on study eligibility and data extraction.
Data extraction
From the studies that met the eligibility criteria, the following data were collected: 1) study: publication year, study design, and study location; 2) cases: number of included patients, age, gender, site of primary tumor, and function; 3) intervention: type of surgery in the surgical group ± additional treatments, postoperative complications, and mortality; type of nonsurgical interventions; 4) median follow-up; and 5) survival outcomes were extracted as hazard ratios (HRs) from univariate or multivariate Cox regression models or as the median overall survival time (MOS) or overall survival rate at specific time points, as applicable.
Statistical analysis
Data management and analyses were performed with RevMan 5.3 via the following methods: by calculating the relative risk with 95% confidence interval for dichotomous variables and by calculating the mean difference for continuous variables. Studies that reported HRs for survival in the surgical vs nonsurgical groups were pooled with a fixed-effect meta-analysis model, and a random-effect meta-analysis was conducted to investigate the liver tumor burden and tumor grade among patients who received surgery or nonsurgical therapy.
Additionally, heterogeneity measured as I2 was used to assess the percentage of total variation from all of the studies to define heterogeneity. A high value of I2 indicates heterogeneity. Publication bias was evaluated via Egger’s test in this statistical analysis.
Results
Literature search
The flowchart of the included studies is shown in Figure 1. Overall, 1,689 records were identified through the PubMed and Cochrane search. According to the predefined inclusion and exclusion criteria, 10 studies published from 2001 to 2017 were included in the meta-analysis. Unfortunately, none of the identified studies were RCTs or quasi-RCTs. All were retrospective cohort studies based on either single-center cohort (n=8) or national database cohort (n=2).
  | Figure 1 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram illustrating study selection. |
Study characteristics
Descriptive characteristics of the included studies are listed in Table 1.9,16–24 These studies included 1,226 patients who underwent palliative surgical resection of their primary tumor and 1,623 patients who did not receive surgery. Five studies were conducted in Italy:16–20 three in the United States,9,21,23 one in China,22 and one in Germany.24 The number of included patients ranged from 43 to 2,158, and the median age ranged from 49 to 60 years. However, the median age was not explicitly stated in three publications.16,18,24 When the gender distributions were reported, most studies had similar numbers of males and females (males ranged from 47 to 66.9%).
In two studies, PNETs were mixed with neuroendocrine tumors (NETs) from sites other than the pancreas, including the ileum, lung, stomach, esophagus, rectum, and unknown primary.17,22 Furthermore, four studies included a certain proportion of patients without liver metastasis.9,21,23,24 Three of the 10 studies included only the nonfunctional PNET,9,19,23 and the study by Citterio et al17 reported the effect of primary tumor resection in patients with functional NET with liver metastases. The postoperative mortality was 0, while the rate of postoperative abdominal complications, including pancreatic fistula, abdominal fluid collections, and bleeding, ranged from 15.9 to 42.1% (Table 2).
Tumor characteristics in the surgical and nonsurgical groups
As several studies suggested that patients who underwent an aggressive surgical approach had a higher possibility of advanced disease or a heavy burden of liver metastasis, we further compared these differences between the groups (Table 3). The meta-analysis demonstrated that there were no significant differences in liver tumor burden (odds ratio [OR] =1.51, 95% CI: 0.59–3.89, P=0.39) (Figure 2) or tumor grade (OR =2.88, 95% CI: 0.92–9.04, P=0.07) (Figure 3) among patients who received surgery or nonsurgical therapy; however, patients who underwent an aggressive surgical approach appeared to have a higher tumor grade. No significant heterogeneity was observed in the models for liver tumor burden (P=0.09, I2=54%), but there was statistically significant heterogeneity in the models for tumor grade (P<0.001, I2=86%).
  | Figure 2 Meta-analysis of the liver tumor burden among patients who received surgery and nonsurgical therapy. |
  | Figure 3 Meta-analysis of the tumor grade among patients who received surgery and nonsurgical therapy. |
Survival outcomes
Most of the 10 studies showed a trend toward better overall survival in patients who underwent primary tumor resection compared with those who did not receive surgery, expressed as the MOS and/or 5-year overall survival rate (Table 4). Additionally, the MOS was 36–137 and 13.2–65 months in the surgical and nonsurgical groups, respectively, and the 5-year overall survival rate was 35.7–83 and 5.4%–50%, respectively, in these two groups. The single exception was the study by Bettini et al,19 who reported no difference in survival between the groups (5-year overall survival: 40% in the surgical group vs 42% in the nonsurgical group; P=0.74), although the median survival was longer in the surgical group (54.3 vs 39.5 months). All studies included in the meta-analysis were combined using a fixed effects model, which demonstrated that survival was significantly improved in patients who underwent primary tumor resection (P<0.001), with a pooled HR of 0.36 (95% CI: 0.30–0.45) (Figure 4). No significant heterogeneity was detected across the studies (I2=0%, P=0.83). Publication bias was examined via Egger’s regression test (Figure 5), which indicated that there was no significant publication bias (P>0.05) in the outcomes of this meta-analysis.
  | Table 4 Overall survival data |
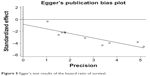  | Figure 5 Egger’s test results of the hazard ratio of survival. |
Discussion
The aim of the present study was to evaluate, through a systematic literature review, the potential benefits of primary tumor resection in patients with PNETs and unresectable liver metastases compared to other treatments that did not include surgery. Although there were no RCTs, 10 retrospective studies were included. Nine studies demonstrated that surgery yielded a significant improvement in survival, but only one study reported a nonsignificant tendency toward improved survival (Table 4). Furthermore, all of the studies included in the meta-analysis showed that patients who underwent primary tumor resection had better overall survival compared with patients who did not undergo resection (P<0.001), with a pooled HR of 0.36 (95% CI: 0.30–0.45) (Figure 4).
Given the relatively low incidence, the heterogeneity, and different clinical presentation of PNETs, it is difficult to develop standardized treatment strategies and start multicentre randomized trials, particularly in patients with PNETs and unresectable liver metastases. Recent guidelines from the European Neuroendocrine Tumor Society, North American Neuroendocrine Tumor Society, and National Comprehensive Cancer Network suggest that the resection of the primary tumor and the hepatic metastases should be performed when radical resection is achievable.25–28 Unfortunately, this is possible only in ~10% of patients with metastatic PNETs. For the vast majority of these patients, the impact of palliative resection on the primary tumor is still debated. Regarding primary resection, international guidelines distinguish tumors that arise from the small intestine from those that arise from the pancreas. Resection of the primary tumor is strongly recommended for intestinal NETs, even in the presence of liver or lymph node metastasis,29,30 but resection of the primary tumor in patients with metastatic PNETs is not recommended, except for selected low-risk patients with life-threatening symptoms due to complications of the primary lesion.31
Recently, three retrospective population-based observational studies used the surveillance, epidemiology, and end results database to identify patients with metastatic PNETs who underwent palliative resection of the primary tumor. Franko et al23 included patients who were treated between 1973 and 2003, while the study by Hüttner et al24 included patients who were treated from 2004 to 2011. To prevent the duplication of data, the study by Keutgen et al,32 who included patients across both time periods, was excluded from the present analysis. Keutgen et al found that the median survival of patients who underwent resection of the primary site was 65 months (95% CI: 60–86) vs 10 months (95% CI: 8–12) for patients who did not undergo resection (P<0.001), and all three studies reported highly consistent multivariate HRs that demonstrated prolonged overall survival in the surgical group.
In the present study, overall survival appeared to be longer in patients who underwent a resection of the primary tumor. However, no RCTs evaluated the outcomes of palliative primary PNET resection in patients with stage IV disease. The majority of included studies were retrospective cohort series, which might have therefore been subject to publication bias, although Egger’s regression test indicated that there was no significant publication bias (P>0.05) in the outcomes of this meta-analysis (Figure 5). A bias toward a more aggressive surgical approach in patients with better performance status, less advanced disease, or a tumor located in the body or tail of the pancreas appears likely. Keutgen et al32 demonstrated that patients in the surgery group were significantly younger (<65 years) and had more grade I tumors and more tumors located in the body/tail of the pancreas compared with patients who did not undergo surgery. Patients with body or tail tumors had a longer overall survival (HR =1.71, 95% CI: 1.43–2.04, P<0.001), although this was not significantly different in the multivariate analysis. Additionally, Franko et al23 found that the likelihood of resection of the primary tumor was highly dependent on tumor grade: 79% of grade I and II primary tumors were resected compared to 25% of grade III and IV tumors (P<0.001), which was also observed in our study.
Another confounding factor is the different types of patients included in the studies. First, some studies included patients other than patients with PNETs, with some of the patients having ileum, lung, stomach, rectum, and unknown primary NETs.17,22 Patients with midgut NETs were the main group of people together with patients with PNETs in the studies, and more and more evidence confirmed a survival benefit for primary tumor resection among midgut NET patients. Second, the inclusion of patients with liver metastasis and other distant metastasis or metastasis of sites other than the liver, such as bone metastasis, retroperitoneum metastasis, and distant lymph node metastasis,9,23 is another variable to address. Since the liver is the most common site of distant metastasis in patients with PNETs, it was not surprising to find little data available concerning primary tumor resection in the presence of other distant metastases. Bertani et al reported that the presence of extrahepatic disease was not associated with a worse survival in a univariate analysis (P=0.25), but the numbers were small, and the statistical power was limited. Furthermore, the multivariate analysis revealed a survival advantage for patients who underwent primary tumor resection, after excluding these patients with extrahepatic metastases from the analysis. Third, four studies included patients without liver metastases at the time of surgery.9,21,23,24 Finally, data from patients who underwent resection of the primary tumor alone were often not separated from data related to patients who also underwent liver resection.17,21,22 Therefore, the clinical relevance of the survival advantage reported for patients who underwent resection of the primary tumor should be considered with caution.
The present meta-analysis did not attempt to evaluate symptom response or quality of life implications following surgery, due to the insufficient number of studies that reported these outcomes. In addition, the role of nonsurgical medical therapies and the value of synchronous or delayed surgical debulking of liver metastases were not evaluated.
Compared with midgut NETs, PNETs have a worse prognosis and more operation complications. Surgical morbidity and mortality following PNET resection were not reported consistently.23,24,32 In our study, the postoperative mortality was 0, while the rate of postoperative abdominal complications, including pancreatic fistula, abdominal fluid collections, and bleeding, ranged from 15.9 to 42.1% (Table 2). Compared to the operative mortality, which ranged from 0 to 3.5% in other studies, the operative mortality was lower in our study. This finding supported the safety of primary tumor resection in patients with PNETs.
Conclusion
Our review highlights the possible benefits of an aggressive surgical approach in patients with metastatic PNETs, although a bias toward a more aggressive surgical approach in patients with better performance status, less advanced disease, or a tumor located in the body or tail of the pancreas appears likely. Nevertheless, in the absence of other data, resection of the primary tumor in patients who have PNETs and unresectable liver metastases should be considered in high-volume referral centers, with strict selection criteria and in a multidisciplinary setting of patient care, apart from resections needed to resolve life-threatening complications.
Acknowledgments
This study was supported by grants from the General Research Projects of the Department of Education of Zhejiang Province (no Y201737812), Zhejiang Province Department of Laboratory Animal Science and Technology Plan Projects (no 2018C37115), and Key Research and Development Project of Science and Technology Department of Zhejiang Province (no 2015C03053).
Disclosure
The authors report no conflicts of interest in this work.
References
Yao JC, Eisner MP, Leary C, et al. Population-based study of islet cell carcinoma. Ann Surg Oncol. 2007;14(12):3492–3500. | ||
Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97(4):934–959. | ||
Bilimoria KY, Talamonti MS, Tomlinson JS, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors: analysis of 3851 patients. Ann Surg. 2008;247(3):490–500. | ||
Chamberlain RS, Canes D, Brown KT, et al. Hepatic neuroendocrine metastases: does intervention alter outcomes? J Am Coll Surg. 2000;190(4):432–445. | ||
Panzuto F, Nasoni S, Falconi M, et al. Prognostic factors and survival in endocrine tumor patients: comparison between gastrointestinal and pancreatic localization. Endocr Relat Cancer. 2005;12(4):1083–1092. | ||
Yuan CH, Wang J, Xiu DR, et al. Meta-analysis of liver resection versus nonsurgical treatments for pancreatic neuroendocrine tumors with liver metastases. Ann Surg Oncol. 2016;23(1):244–249. | ||
Carty SE, Jensen RT, Norton JA. Prospective study of aggressive resection of metastatic pancreatic endocrine tumors. Surgery. 1992;112(6):1024–1031. | ||
Sarmiento JM, Farnell MB, Que FG, Nagorney DM. Pancreaticoduodenectomy for islet cell tumors of the head of the pancreas: long-term survival analysis. World J Surg. 2002;26(10):1267–1271. | ||
Solorzano CC, Lee JE, Pisters PW, et al. Nonfunctioning islet cell carcinoma of the pancreas: survival results in a contemporary series of 163 patients. Surgery. 2001;130(6):1078–1085. | ||
Frilling A, Li J, Malamutmann E, Schmid KW, Bockisch A, Broelsch CE. Treatment of liver metastases from neuroendocrine tumours in relation to the extent of hepatic disease. Br J Surg. 2009;96(2):175–184. | ||
Wong RJ, DeCosse JJ. Cytoreductive surgery. Surg Gynecol Obstet. 1990;170(3):276–281. | ||
Bruzoni M, Parikh P, Celis R, et al. Management of the primary tumor in patients with metastatic pancreatic neuroendocrine tumor: a contemporary single-institution review. Am J Surg. 2009;197(3):376–381. | ||
Capurso G, Bettini R, Rinzivillo M, Boninsegna L, Delle Fave G, Falconi M. Role of resection of the primary pancreatic neuroendocrine tumour only in patients with unresectable metastatic liver disease: a systematic review. Neuroendocrinology. 2011;93(4):223–229. | ||
Hill JS, McPhee JT, McDade TP, et al. Pancreatic neuroendocrine tumors: the impact of surgical resection on survival. Cancer. 2009;115(4):741–751. | ||
Almond LM, Hodson J, Ford SJ, et al. Role of palliative resection of the primary tumour in advanced pancreatic and small intestinal neuroendocrine tumours: a systematic review and meta-analysis. Eur J Surg Oncol. 2017;43(10):1808–1815. | ||
Bertani E, Fazio N, Botteri E, et al. Resection of the primary pancreatic neuroendocrine tumor in patients with unresectable liver metastases: possible indications for a multimodal approach. Surgery. 2014;155(4):607–614. | ||
Citterio D, Pusceddu S, Facciorusso A, et al. Primary tumour resection may improve survival in functional well-differentiated neuroendocrine tumours metastatic to the liver. Eur J Surg Oncol. 2017;43(2):380–387. | ||
Bertani E, Fazio N, Radice D, et al. Resection of the primary tumor followed by peptide receptor radionuclide therapy as upfront strategy for the treatment of G1–G2 pancreatic neuroendocrine tumors with unresectable liver metastases. Ann Surg Oncol. 2016;23(suppl 5):981–989. | ||
Bettini R, Mantovani W, Boninsegna L, et al. Primary tumour resection in metastatic nonfunctioning pancreatic endocrine carcinomas. Dig Liver Dis. 2009;41(1):49–55. | ||
Bertani E, Fazio N, Radice D, et al. Assessing the role of primary tumour resection in patients with synchronous unresectable liver metastases from pancreatic neuroendocrine tumour of the body and tail. A propensity score survival evaluation. Eur J Surg Oncol. 2017;43(2):372–379. | ||
Nguyen SQ, Angel LP, Divino CM, Schluender S, Warner RR. Surgery in malignant pancreatic neuroendocrine tumors. J Surg Oncol. 2007;96(5):397–403. | ||
Du S, Wang Z, Sang X, et al. Surgical resection improves the outcome of the patients with neuroendocrine tumor liver metastases: large data from Asia. Medicine (Baltimore). 2015;94(2):e388. | ||
Franko J, Feng W, Yip L, Genovese E, Moser AJ. Non-functional neuroendocrine carcinoma of the pancreas: incidence, tumor biology, and outcomes in 2,158 patients. J Gastrointest Surg. 2010;14(3):541–548. | ||
Hüttner FJ, Schneider L, Tarantino I, et al. Palliative resection of the primary tumor in 442 metastasized neuroendocrine tumors of the pancreas: a population-based, propensity score-matched survival analysis. Langenbecks Arch Surg. 2015;400(6):715–723. | ||
Jensen RT, Cadiot G, Brandi ML, et al. ENETS Consensus Guidelines for the management of patients with digestive neuroendocrine neoplasms: functional pancreatic endocrine tumor syndromes. Neuroendocrinology. 2012;95(2):98–119. | ||
Falconi M, Bartsch DK, Eriksson B, et al. ENETS Consensus Guidelines for the management of patients with digestive neuroendocrine neoplasms of the digestive system: well-differentiated pancreatic non-functioning tumors. Neuroendocrinology. 2012;95(2):120–134. | ||
Kulke MH, Anthony LB, Bushnell DL, et al. NANETS treatment guidelines: well-differentiated neuroendocrine tumors of the stomach and pancreas. Pancreas. 2010;39(6):735–752. | ||
Kulke MH, Benson AB 3rd, Bergsland E, et al. Neuroendocrine tumors. J Natl Compr Canc Netw. 2012;10(6):724–764. | ||
Niederle B, Pape UF, Costa F, et al. ENETS consensus guidelines update for neuroendocrine neoplasms of the jejunum and ileum. Neuroendocrinology. 2016;103(2):125–138. | ||
Guo J, Zhang Q, Bi X, et al. Systematic review of resecting primary tumor in MNETs patients with unresectable liver metastases. Oncotarget. 2017;8(10):17396–17405. | ||
Falconi M, Eriksson B, Kaltsas G, et al. ENETS consensus guidelines update for the management of patients with functional pancreatic neuroendocrine tumors and non-functional pancreatic neuroendocrine tumors. Neuroendocrinology. 2016;103(2):153–171. | ||
Keutgen XM, Nilubol N, Glanville J, et al. Resection of primary tumor site is associated with prolonged survival in metastatic nonfunctioning pancreatic neuroendocrine tumors. Surgery. 2016;159(1):311–318. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.