Back to Journals » OncoTargets and Therapy » Volume 15
Remission from the 5-Fu-Based Chemotherapy to Gemcitabine-Based Chemotherapy-Based on the Pathological Classification of Periampullary Carcinoma: A Case Report and Literature Review
Authors Hu W , Duan Z, Zhang Y, Liu J, Bao J, Gao R, Tang Y, Liu T, Xiong H, Li W, Fu X , Liao S, Fang L, Liang B
Received 2 May 2022
Accepted for publication 3 August 2022
Published 25 August 2022 Volume 2022:15 Pages 891—896
DOI https://doi.org/10.2147/OTT.S372053
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Gaetano Romano
Wei Hu,1,* Zhiqing Duan,1,* Yinuo Zhang,1,* Jing Liu,2,* Jing Bao,1 Ruqing Gao,1 Yajie Tang,1 Tiande Liu,1 Hu Xiong,1 Wen Li,1 Xiaowei Fu,1 Shousheng Liao,2 Lu Fang,1 Bo Liang1
1Department of General Surgery, The Second Affiliated Hospital of Nanchang University, Nanchang, Jiangxi, 330006, People’s Republic of China; 2Department of Pathology, The Second Affiliated Hospital of Nanchang University, Nanchang, Jiangxi, 330006, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Bo Liang; Lu Fang, Email [email protected]; [email protected]
Background: Periampullary carcinoma, which includes ampullary carcinoma, pancreatic head cancer, distal common bile duct cancer, and duodenal papillary cancer, is a relatively rare malignancy with uncertain therapeutic options. Although several studies have investigated the efficacy of multiple adjuvant chemotherapy regimens for periampullary carcinoma treatment, the optimal regimen remains to be determined. The inherent heterogeneity of the mucosal origin divides periampullary carcinoma into intestinal and pancreaticobiliary types. Therefore, the selection of chemotherapy regimens based on pathological type may have potential therapeutic significance.
Case Presentation: A 72-year-old woman with moderately differentiated periampullary adenocarcinoma experienced disease progression after receiving FOLFOX regimen. Subsequently, the sample was subtyped first by H&E evaluation and then by the evaluation of an IHC panel composed of CK20, CDX2, MUC1, MUC2, and MUC5AC. The pathologists concluded that the patient’s sample was of the pancreaticobiliary (PB) subtype. The subsequent change to gemcitabine plus S-1 adjuvant therapy achieved remission of liver metastases based on the pathological classification of the cancer.
Conclusion: Based on the pathological classification, adjuvant chemotherapy with gemcitabine may be beneficial for patients with PB subtype periampullary carcinoma. 5-Fu-based adjuvant chemotherapy may be beneficial for patients with intestinal subtype periampullary carcinoma.
Keywords: periampullary carcinoma, adjuvant chemotherapy, pathological classification, S-1, gemcitabine
Background
Periampullary carcinoma, which comprises ampulla of Vater, pancreatic head, distal common bile duct, and duodenal papillary cancers, is a malignant tumor originating 2 cm from the ampulla of Vater.1,2,38 It constitutes 5% of all gastrointestinal tract malignancies and ranks eighth to ninth in incidence and the fourth to fifth most common cause of mortality in gastrointestinal tract malignancies.3,4
For patients with localized disease, radical surgical resection is regarded as the mainstay of curative treatment and is feasible in approximately 50% of ampullary cancer cases.5,6 However, the 5-year survival rate after surgery has been reported to range from 5% to 68%,7–11 which results from the high risk of recurrence and metastasis.12,13 Moreover, due to limited adjuvant treatments such as chemoradiotherapy, metastatic and advanced ampullary adenocarcinomas are characterized by poor prognosis, with a 2-year overall survival rate ranging from 5% to 10%.14 To further improve treatment outcomes, many clinicians now treat patients with periampullary cancer with adjuvant chemotherapy.15 At present, there are no clear evidence-based guidelines to aid in selecting both the modality and regimen of adjuvant therapy for resected ampullary carcinoma; therefore, the regimens have been inconclusive and are largely based on treatments of other gastrointestinal tract malignancy types, which are mainly fluorouracil-based and gemcitabine-based chemotherapy. However, the optimal choice is still to be determined.6,16
In addition, many studies have shown distinctly variable treatment outcomes after chemotherapy, which is assumed to be caused by the inherent heterogeneity of the mucosal origin of periampullary cancer. Based on heterogeneous mucosal origin, periampullary cancer can be divided into pancreaticobiliary (PB) subtypes, including pancreatic cancer, distal bile duct cancer, and some ampullary carcinomas, and intestinal (IN) subtypes, including duodenal carcinoma and some ampullary carcinomas; the two subtypes with distinct tissue phenotypes show a significant difference in prognosis and survival rate.17 As a result, histological subtype is considered an independent prognostic factor affecting overall survival, thus indicating a potential therapeutic direction.18,19 At present, clinicians tend to treat patients with the IN subtype of periampullary cancer with fluorouracil-based chemotherapy, which is mainly applied in colorectal cancer treatment, and treat PB subtype patients with gemcitabine-based chemotherapy regimens, the standard therapy for pancreatic cancer.20,21
Herein, we report a case of a patient diagnosed with moderately differentiated periampullary carcinoma. The patient showed a decrease in hepatic metastases when undergoing a change from a fluorouracil-based to gemcitabine-based chemotherapy regimen after pathological classification.
Case Presentation
A 72-year-old woman presented with upper abdominal pain for more than 3 days and was admitted to the hospital. The patient had a history of hypertension and pacemaker implantation; therefore, the use of oral antihypertensive drugs and blood pressure control were acceptable Physical examination revealed that the patient’s skin and sclera were moderately yellowish, with radiating pain in the shoulder and back. Obstructive jaundice was diagnosed upon admission. Abnormal prothrombin, cancer antigen 19-9, and alpha-fetoprotein levels were within the normal range; however, the carcinoembryonic antigen (CEA) level increased to 8.41 ng/mL; total bilirubin level increased to 180.14 μmol/L, and direct bilirubin level increased to 125.84 μmol/L. Computed tomography (CT) enhancement of the upper abdomen revealed gallstones and dilated bile ducts. Endoscopic nasobiliary drainage was considered due to high total bilirubin levels; however, duodenal stenosis resulted in the failure of nasobiliary duct placement. An electronic duodenoscope showed irregular bulging of the nipple, poor biopsy elasticity, and nipple occupancy. Biopsy pathology suggested ampullary adenocarcinoma. After PTCD was placed and treated for obstructive jaundice, laparoscopic-assisted pancreaticoduodenectomy was performed under general anesthesia on June 22, 2020. The tumor tissue was sent for pathological examination, which indicated moderately differentiated adenocarcinoma (Figure 1A). The size of the tumor is 2.0*1.5 cm, and the tumor is infiltrated into the full thickness of the duodenal wall, pancreas, and common bile duct. Therefore, the patient was stratified as T3bN0M0 and IIB stage. Immunohistochemical analysis revealed the following: HER-2 (-), MLHI (+), MSH2 (+), MSH6 (+), PMS2 (+), EP-CAM (+), P53(-), Ki-67 (40%+), CK (+), CEA (+), CK7 (+), CK19 (+), CK20 (-), and CDX2 (weak+).
 |
Figure 1 H&E and IHC of the patient’s tumor. (A) H&E stain of tumor; (B) CK20 (-); (C) CDX2 (-); (D) MUC1 (+); (E), MUC2 (-); (F), MUC5AC (+). |
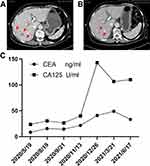
The adjuvant chemotherapy regimen of FOLFOX was started on August 24, 2020, postoperatively. The regimen was oxaliplatin (150 mg intravenous [IV], D1) + calcium levofolinate (100 mg IV, D1–D5) + fluorouracil (750 mg IV, D1–D5) for two cycles. The patient was admitted to our department on November 15, 2020, to continue the FOLFOX regimen. Re-examination on December 27, 2020, revealed multiple new liver metastases on enhanced CT of the upper abdomen (Figure 2A).
Owing to the poor effect of the patient’s chemotherapy, we tested the patient’s sample again. The sample was subtyped first by H&E staining and then by the evaluation of an IHC panel composed of CK20, CDX2, MUC1, MUC2, and MUC5AC (Figure 1). The IHC results are CK20 (-), CDX2 (-), MUC1 (-), MUC2 (-), and MUC5AC (-) (Figure 1B–F). Based on the H&E and IHC evaluations, the pathologists concluded that the patient’s sample was of the PB subtype.
Therefore, the chemotherapy regimen was changed to gemcitabine and S-1 for 3 weeks as a cycle. Gemcitabine (1000 mg/m2) was administered intravenously on the first and the eighth day of each cycle, and S-1 (40 mg) was administered orally, once in the morning and once in the evening. The medication was stopped for 1 week after 14 consecutive days of oral administration. After the patient regularly received three cycles of chemotherapy, CT enhancement on March 22, 2021, showed that the lesions had significantly decreased in size (Figure 2B). The CEA and CA125 levels continued to decline after changing the chemotherapy regimen (Figure 2C). Neutropenia was present during the chemotherapy, but the situation improved with G-CSF treatment.
Discussion
Periampullary carcinoma is a relatively rare malignancy, and ampullary cancer, which represents a subset of periampullary cancer, comprises only 0.2% of all gastrointestinal malignancies and 6–7% of periampullary tumors.22,23 Currently, pancreaticoduodenectomy (Whipple procedure) is the only curative modality for periampullary cancer. Despite similarities in anatomical position and histological appearance, there is a significant variation in the survival rate after resection of patients with distinct periampullary cancer subtypes derived from different anatomical locations.17 The 5-year survival rates after surgical resection are 33–68% for ampullary carcinoma, 23–30% for bile duct, and 25–59% for duodenal adenocarcinoma, but only 5–20% for pancreatic carcinoma.7–11 Therefore, adjuvant chemotherapy is often used in periampullary carcinoma owing to the disappointing long-term cure rate, and the optimal regimen remains inconclusive.
In recent years, the selection of chemotherapeutic drugs for periampullary carcinoma under the guidance of pathological subtyping has emerged. IN subtype patients tend to use fluorouracil-based regimens, which are efficacious for colorectal carcinoma, while PB subtype patients tend to use gemcitabine-based regimens, which are commonly used for pancreatic carcinoma.11 The conventional typing method involves observing the morphology and structure of tissues processed using H&E staining. This typing method is accurate in most cases, but there are still some subjective deviations. In addition, IHC staining, which can distinguish cancer subtypes based only on histomorphology, is advocated for typing, as even poorly differentiated cancers retain their histological marker profile in mucosal origin.17 According to the system proposed by Ang et al, tumors that stain positive for CK20, CDX2, or MUC2 and negative for MUC1, or positive for all of the first three, regardless of MUC1, are classified as IN. Tumors that stained positive for MUC1 in the absence of both CDX2 and MUC2 were classified as PB, regardless of CK20. The other combinations were classified as ambiguous. It is described as positive when more than 25% of the tumor cells in the sample are stained.24 The combination of H&E and IHC staining provides a more effective method for classifying periampullary carcinomas.
Some studies have reported the benefits of the subtype classification for the treatment of periampullary cancer. A retrospective study of 95 patients with ampullary adenocarcinoma resection by Schiergens et al showed that gemcitabine-based adjuvant chemotherapy provided improved overall survival (32 months vs 13 months, P = 0.013) only for patients with PB subtype cancers. Patients with IN subtype cancers tended to have worse survival outcomes after gemcitabine therapy (35 months vs 112 months, P = 0.193).21 Similarly, an international multicenter cohort study of 976 patients with ampullary adenocarcinoma published by Moekotte et al showed that patients with PB or AM subtypes had better survival after gemcitabine-based adjuvant chemotherapy (median survival not reached vs 32 months, P=0.20), while patients with the IN subtype did not show any survival benefit from adjuvant chemotherapy.25 Another multicenter retrospective cohort study published by Bolm et al also verified the effectiveness of gemcitabine-based adjuvant therapy in prolonging the survival of PB or AM patients (median survival 85 vs 65 months, P =0.005) and the difference in patients with the IN type.26
Therefore, the poor clinical outcomes after adjuvant chemotherapy may result from the absence of stratification by histological subtype, and it is necessary to further explore the role of histological subtype classification in selecting optimal treatment strategies.
Concurrently, radiotherapy is often used in combination with chemotherapy due to its effectiveness in controlling recurrence. A cohort study of 4190 patients with ampullary carcinoma in the United States revealed the significance of adjuvant chemoradiotherapy in survival, which was a retrospective study using the National Cancer Database without a consistent chemotherapy regimen and radiotherapy dosage. After matching 1:1 propensity score, the median overall survival of the observation group and adjuvant chemoradiotherapy group were 31.0 and 38.1 months respectively (p=0.02).27 In addition, Erdmann et al published a single-center randomized trial that revealed that adjuvant intra-arterial chemotherapy (IAC) plus radiotherapy with a total dose of 54 Gy can prolong the median overall survival in patients with non-pancreatic ampullary cancer, and that IAC effectively reduced the probability of liver metastases (from 17 to 8 cases, p =0.038). The median survival times of the adjuvant chemoradiation and observation groups were 37 and 28 months, respectively. Although there was no survival benefit in the univariate analysis (p=0.077), multivariate analysis after adjusting for variables suggested a statistically significant survival benefit (p=0.011).28 In combination with the relevant literature, the survival benefit of adjuvant chemoradiotherapy may be limited to cancer patients characterized by advanced T3/4 stages or lymph node positivity.29,30
Currently, targeted therapy and immunotherapy are being explored as emerging adjuvant treatment strategies. Targeted therapy mainly focuses on potential targets, such as the wingless pathway, lipid metabolism pathway, and cyclooxygenase (COX).31–33 However, there are no reliable targeted drugs for clinical applications, and studies on promising drugs, mainly panitumumab and bevacizumab, are underway. Some studies have suggested good prospects, such as the combination of bevacizumab and the CAPOX regimen, revealing therapeutic effectiveness.34,35 In contrast, immunotherapy, an important advancement in cancer treatment, aims to strengthen the immune response to kill cancer cells, which is distinct from traditional cancer therapy. Similarly, there is no existing immune-based treatment plan for periampullary carcinoma except for some potential treatment points with unascertainable feasibility.36,37 Thus, further studies are required.
Conclusion
Here, we report a case of a medially differentiated periampullary carcinoma patient with the PB subtype whose liver metastases were relieved after gemcitabine plus S-1-based adjuvant chemotherapy. According to existing literature reports, surgery is still the only reliable curative treatment, which is often followed by subtype-based personalized adjuvant chemoradiotherapy regimen decision-making with the basic principle that patients with the IN type tend to use fluorouracil-based treatment options and patients with the PB subtype tend to be treated with gemcitabine-based options. This histology-based typing principle may provide novel opportunities for improving survival.
Reporting Checklist
The authors have completed the reporting checklist.
Ethical Statement
This study was approved by the medical ethics committee of the Second Affiliated Hospital of Nanchang University (The Examination and approval NO. Review [2021] NO. (091)) and obtained the written informed consent of patients to publish clinical details and images.
Funding
This study was funded by the National Natural Science Foundation of China (No. 82160578), Natural Science Foundation of Jiangxi Province (Nos. 20212BCJ23024, 20202BAB216029 and 20202BABL206095), Health Department of Jiangxi Province, China (No. 20198020), and the Education Department of Jiangxi Province, China (Grant No. GJJ190019).
Disclosure
The authors have no conflicts of interest to declare.
The patient signed the informed consent form approved by the institutional review committee and agreed to the publication of the journal.
References
1. Hester CA, Dogeas E, Augustine MM, et al. Incidence and comparative outcomes of periampullary cancer: a population-based analysis demonstrating improved outcomes and increased use of adjuvant therapy from 2004 to 2012. J Surg Oncol. 2019;119(3):303–317. doi:10.1002/jso.25336
2. Hugenschmidt H, Labori KJ, Brunborg C, et al. Circulating tumor cells are an independent predictor of shorter survival in patients undergoing resection for pancreatic and periampullary adenocarcinoma. Ann Surg. 2020;271(3):549–558. doi:10.1097/SLA.0000000000003035
3. Sun J, Liu D. Surgical treatment of periampullary carcinoma. Chin J Pract Surg. 2005;09:571–573.
4. Romiti A, Barucca V, Zullo A, et al. Tumors of ampulla of Vater: a case series and review of chemotherapy options. World J Gastrointest Oncol. 2012;4(3):60–67. doi:10.4251/wjgo.v4.i3.60
5. Ko AH, Nakakura EK. Adjuvant therapy for ampullary cancer. JAMA Surg. 2019;154(8):715. doi:10.1001/jamasurg.2019.1171
6. Al-Jumayli M, Batool A, Middiniti A, et al. Clinical outcome of ampullary carcinoma: single cancer center experience. J Oncol. 2019;2019:3293509. doi:10.1155/2019/3293509
7. Feretis M, Wang T, Iype S, et al. Development of a prognostic model that predicts survival after pancreaticoduodenectomy for ampullary cancer. Pancreas. 2017;46(10):1314–1321. doi:10.1097/MPA.0000000000000929
8. Berberat PO, Kunzli BM, Gulbinas A, et al. An audit of outcomes of a series of periampullary carcinomas. Eur J Surg Oncol. 2009;35(2):187–191. doi:10.1016/j.ejso.2008.01.030
9. Riall TS, Cameron JL, Lillemoe KD, et al. Resected periampullary adenocarcinoma: 5-year survivors and their 6-to 10-year follow-up. Surgery. 2006;140(5):764–772. doi:10.1016/j.surg.2006.04.006
10. O’Connell JB, Maggard MA, Manunga J
11. Schnelldorfer T, Ware AL, Sarr MG, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Ann Surg. 2008;247(3):456–462. doi:10.1097/SLA.0b013e3181613142
12. Tella SH, Mahipal A. The future of adjuvant therapy in ampullary cancer: should we offer it to our patients? Hepatobiliary Surg Nutr. 2020;9(3):368–370. doi:10.21037/hbsn.2019.11.06
13. Seo HK, Hwang DW, Lee JH, et al. Role of systemic inflammation in predicting the prognosis of ampulla of Vater carcinoma. Surg Oncol. 2019;29:33–40. doi:10.1016/j.suronc.2019.02.006
14. Key C, Meisner ALW. Cancers of the liver and biliary tract. In: Ries LAG, Young JL, Keel GE, Eisner MP, Lin YD, Horner MJ, editors. SEER Survival Monograph: Cancer Survival Among Adults: US SEER Program, 1988–2001, Patient and Tumor Characteristics. Bethesda, MD: National Cancer Institute, SEER Program; 2007:49–58.
15. Kim JH, Jeong JH, Ryoo BY, et al. Adjuvant chemotherapy for resected ampulla of vater carcinoma: retrospective analysis of 646 Patients. Cancer Res Treat. 2020;53:424–435.
16. Al Abbas AI, Falvello V, Zenati M, et al. Impact of adjuvant chemotherapy regimen on survival outcomes in immunohistochemical subtypes of ampullary carcinoma. J Surg Oncol. 2019. doi:10.1002/jso.25808
17. Chandrasegaram MD, Gill AJ, Samra J, et al. Ampullary cancer of intestinal origin and duodenal cancer - A logical clinical and therapeutic subgroup in periampullary cancer. World J Gastrointest Oncol. 2017;9(10):407–415. doi:10.4251/wjgo.v9.i10.407
18. Heby M, Lundgren S, Nodin B, Elebro J, Eberhard J, Jirström K. Relationship between mismatch repair immunophenotype and long-term survival in patients with resected periampullary adenocarcinoma. J Transl Med. 2018;16(1):66. doi:10.1186/s12967-018-1444-4
19. Ecker BL, Vollmer CM
20. Overman MJ, Varadhachary GR, Kopetz S, et al. Phase II study of capecitabine and oxaliplatin for advanced adenocarcinoma of the small bowel and ampulla of Vater. J Clin Oncol. 2009;27(16):2598–2603. doi:10.1200/JCO.2008.19.7145
21. Schiergens TS, Reu S, Neumann J, et al. Histomorphologic and molecular phenotypes predict gemcitabine response and overall survival in adenocarcinoma of the ampulla of Vater. Surgery. 2015;158(1):151–161. doi:10.1016/j.surg.2015.02.001
22. Albores-Saavedra J, Schwartz AM, Batich K, Henson DE. Cancers of the ampulla of vater: demographics, morphology, and survival based on 5625 cases from the SEER program. J Surg Oncol. 2009;100(7):598–605. doi:10.1002/jso.21374
23. Kim RD, Kundhal PS, McGilvray ID, et al. Predictors of failure after pancreaticoduodenectomy for ampullary carcinoma. J Am Coll Surg. 2006;202(1):112–119. doi:10.1016/j.jamcollsurg.2005.08.002
24. Ang DC, Shia J, Tang LH, Katabi N, Klimstra DS. The utility of immunohistochemistry in subtyping adenocarcinoma of the ampulla of vater. Am J Surg Pathol. 2014;38(10):1371–1379. doi:10.1097/PAS.0000000000000230
25. Moekotte AL, Malleo G, van Roessel S, et al. Gemcitabine-based adjuvant chemotherapy in subtypes of ampullary adenocarcinoma: international propensity score-matched cohort study. Br J Surg. 2020;107(9):1171–1182. doi:10.1002/bjs.11555
26. Bolm L, Ohrner K, Nappo G, et al. Adjuvant therapy is associated with improved overall survival in patients with pancreatobiliary or mixed subtype ampullary cancer after pancreatoduodenectomy - A multicenter cohort study. Pancreatology. 2020;20(3):433–441. doi:10.1016/j.pan.2020.01.009
27. Nassour I, Hynan LS, Christie A, et al. Association of adjuvant therapy with improved survival in ampullary cancer: a national cohort study. J Gastrointest Surg. 2018;22(4):695–702. doi:10.1007/s11605-017-3624-6
28. Erdmann JI, Morak MJ, Duivenvoorden HJ, et al. Long-term survival after resection for non-pancreatic periampullary cancer followed by adjuvant intra-arterial chemotherapy and concomitant radiotherapy. HPB (Oxford). 2015;17(7):573–579. doi:10.1111/hpb.12401
29. Kim HS, Jang JY, Yoon YS, et al. Does adjuvant treatment improve prognosis after curative resection of ampulla of Vater carcinoma? A multicenter retrospective study. J Hepatobiliary Pancreat Sci. 2020;27(10):721–730. doi:10.1002/jhbp.801
30. Parikh P, Waters JA, Pitt HA, et al. Ampullary carcinoma: adjuvant chemoradiation improves survival in node-positive patients. Gastroenterology. 2010;138(5):S875–S875. doi:10.1016/S0016-5085(10)64044-9
31. Perrone G, Santini D, Verzì A, et al. COX-2 expression in ampullary carcinoma: correlation with angiogenesis process and clinicopathological variables. J Clin Pathol. 2006;59(5):492–496. doi:10.1136/jcp.2005.030098
32. Hayata Y, Nakagawa H, Kurosaki S, et al. Axin2(+) peribiliary glands in the periampullary region generate biliary epithelial stem cells that give rise to ampullary carcinoma. Gastroenterology. 2021;160:2133–2148.e6. doi:10.1053/j.gastro.2021.01.028
33. Wang C-Y, Chao Y-J, Chen Y-L, et al. Upregulation of peroxisome proliferator-activated receptor-alpha and the lipid metabolism pathway promotes carcinogenesis of ampullary cancer. Int J Med Sci. 2021;18(1):256–269. doi:10.7150/ijms.48123
34. Gulhati P, Raghav K, Shroff RT, et al. Bevacizumab combined with capecitabine and oxaliplatin in patients with advanced adenocarcinoma of the small bowel or ampulla of vater: a single-center, open-label, Phase 2 study. Cancer. 2017;123(6):1011–1017. doi:10.1002/cncr.30445
35. Gulhati P, Raghav K, Shroff R, et al. Phase II study of panitumumab in RAS wild-type metastatic adenocarcinoma of small bowel or ampulla of vater. Oncologist. 2018;23(3):277–e226. doi:10.1634/theoncologist.2017-0568
36. Lundgren S, Micke P, Elebro J, et al. Topographical distribution and spatial interactions of innate and semi-innate immune cells in pancreatic and other periampullary adenocarcinoma. Front Immunol. 2020;11:558169. doi:10.3389/fimmu.2020.558169
37. Kim MH, Jang M, Kim H, Lee WJ, Kang CM, Choi HJ. Distinct immunological properties of the two histological subtypes of adenocarcinoma of the ampulla of Vater. Cancer Immunol Immunother. 2019;68(3):443–454. doi:10.1007/s00262-018-02293-6
38. Duan Z, Zhang Y, Tang Y, Gao R, Bao J, Liang B. Adjuvant therapy for periampullary carcinoma and the significance of histopathological typing: a systematic review. Transl Oncol. 2022;20:101414. doi:10.1016/j.tranon.2022.101414
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.