Back to Journals » Cancer Management and Research » Volume 12
Regulation of E2F Transcription Factor 3 by microRNA-152 Modulates Gastric Cancer Invasion and Metastasis
Authors Shi L, Zhu H, Shen Y, Dou X, Guo H, Wang P, Zhang S, Zhou L , Zou X
Received 23 November 2019
Accepted for publication 30 January 2020
Published 14 February 2020 Volume 2020:12 Pages 1187—1197
DOI https://doi.org/10.2147/CMAR.S239752
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Ahmet Emre Eşkazan
Liangliang Shi, 1–3,* Hao Zhu, 1–3,* Yonghua Shen, 1–3,* Xiaotan Dou, 1–3 Huimin Guo, 1–3 Pin Wang, 1–3 Shu Zhang, 1–3 Lin Zhou, 1–3 Xiaoping Zou 1–3
1Department of Gastroenterology, Drum Tower Clinical Medical College of Nanjing Medical University, Nanjing 210008, People’s Republic of China; 2Department of Gastroenterology, Nanjing Drum Tower Hospital, The Affiliated Hospital of Nanjing University Medical School, Nanjing 210008, People’s Republic of China; 3Jiangsu Clinical Medical Center of Digestive Disease, Nanjing 210008, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaoping Zou; Lin Zhou
Department of Gastroenterology, Nanjing Drum Tower Hospital, No. 321 Zhongshan Road, Nanjing 210008, People’s Republic of China
Tel +86 25-68182222
Fax +86 25-83304616
Email [email protected]; [email protected]
Background: The transcription factor, E2F transcription factor 3 (E2F3), has been proved to modulate metastasis in multiple human cancers. The present study was aimed to expound the function and specific mechanism of E2F3 in gastric cancer (GC) progression.
Materials and Methods: The expression of E2F3, microRNA-152 (miR-152) and PLK1 (polo-like kinase 1) in GC cell lines was detected by quantitative RT-PCR and Western blot. The roles of E2F3 and miR-152 in GC metastasis were classified using gain-of-function and loss-of-function assays. The miRNAs directly targeting E2F3 were identified by bioinformatics analysis and luciferase reporter experiment. Chromatin immunoprecipitation was carried out to reveal the correlation between E2F3 and PLK1.
Results: E2F3 expression was frequently up-regulated in GC tissues, and its high expression might imply poor prognosis. Downregulation of E2F3 restrained GC migration and invasion in vitro and in vivo. Interestingly, we proved that miR-152 was an upstream regulator of E2F3. Moreover, miR-152 reduced E2F3 expression by directly targeting its 3ʹ-UTR, and then modulated GC metastasis via polo-like kinase 1 (PLK1) mediated protein kinase B (AKT) and extracellular signal-regulated kinase (ERK) signals.
Conclusion: E2F3 plays a crucial role in GC progression and the newly discovered miR-152/E2F3/PLK1 axis provides a new underlying target for therapy of metastasis in GC patients.
Keywords: miR-152, E2F3, PLK1, metastasis, gastric cancer
Introduction
Gastric cancer (GC) remains the third most frequent cancer and the fourth major cause of tumor-associated death around the world.1,2 The efficacy of GC treatment has been improved by early detection and targeted therapy for decades.2 However, the prognoses of GC patients with metastasis remain very poor. Hence, it is particularly vital to explore the underlying mechanisms of GC progression to improve patient outcome and survival.
E2F transcription factor 3 (E2F3), as a crucial transcription factor (TF) of E2F family, is found to be implicated in adjusting apoptosis, cell cycle and proliferation.3,4 It has been widely recognized that E2F3 regulates various genes which play vital parts in signal transduction and DNA synthesis.3,4 E2F3 is commonly dysregulated in numerous cancers, and acts as an oncogenic role.4 Overexpression of E2F3 has been indicated in bladder, breast and prostate cancers.5–7 Moreover, it has been reported that E2F3 takes part in the metastasis to lung and liver in thyroid carcinosis.8 Another significant study9 showed that E2F3 deletion in tumor-associated macrophages leads to reduced pulmonary metastasis through conditional knockout approaches. Nevertheless, the specific function of E2F3 in GC metastasis is still indistinct.
MicroRNAs (miRNAs) are non-coding RNAs of 18–25 nucleotides, which bind to the 3ʹ-untranslated regions (3ʹ-UTR) of target genes, leading to mRNA degradation or protein translation decrease.10,11 MiRNAs are an important manner to modulate gene expression at post-transcriptional level.10,11 What’ more, mountains of evidence proves that miRNAs are implicated in the regulation of GC progression.12–14 Therefore, we suppose that miRNAs targeting E2F3 might be a major cause of E2F3’s dysregulation in GC metastasis.
Herein, it was observed that E2F3 was up-regulated in GC tissues and closely correlated with GC patients’ survival. We next demonstrated knockdown of E2F3 suppressed GC cell migration and invasion. In addition, it was proved that miR-152 modulated E2F3 expression by binding to its 3ʹ-UTR. The following results further showed the suppression of miR-152 promoted GC metastasis, while the up-regulation of miR-152 showed opposite effects. Finally, this study indicated the miR-152/E2F3 axis regulated GC progression through polo-like kinase 1 (PLK1) mediated protein kinase B (AKT) and extracellular signal-regulated kinase (ERK) pathways.
Materials and Methods
Tissue Collection and Cell Culture
We collected GC tissues from GC patients who had undergone operation at Nanjing Drum Tower Hospital, China. The clinicopathologic characteristics of these patients are described in Supplemental Table 1. The present research was ratified by Ethics Committee of Nanjing Drum Tower Hospital, and all patient signed the informed consent. HEK293T cell (originally purchased from ATCC) was cultured in Dulbecco’s modified Eagle’s medium (Thermo Scientific HyClone, Beijing, China). MKN28M, MKN28NM, GC9811-P and GC9811 cell lines were obtained from the State Key Laboratory of Cancer Biology and Xijing Hospital of Digestive Diseases, and cultured with RPMI-1640 medium (HyClone). The use of all the cell lines was approved by Ethics Committee of Nanjing Drum Tower Hospital.
RNA Extraction and Quantitative Real-Time PCR
RNA extraction was conducted using Trizol reagent (Invitrogen). Quantitative real-time PCR (qRT-PCR) was performed as previously described.15 The reverse transcription (RT) and PCR (polymerase chain reaction) primers for U6 and miR-152 were synthesized in RiBoBio (Guangzhou, China). As shown in Supplemental Table 2, PCR primers for E2F3, GAPDH (glyceraldehyde 3-phosphate dehydrogenase) and PLK1 were listed.
Western Blot
Western blot was carried out as shown before.16 Anti-E2F3 (Abcam), β-actin (Sigma, St Louis, MO, USA), anti-PLK1 (Invitrogen), anti-ERK1/2 (Abcam), anti-p-ERK1/2 (Abcam), anti-AKT (Cell Signaling) and anti-p-AKT (Cell Signaling) were used as primary antibodies, respectively.
Bioinformatic Analyses
Data on E2F3 expression in GC were collected from Oncomine (https://www.oncomine.org/resource/login.html). GC survival data were obtained from Kaplan Meier plotter (http://www.kmplot.com/analysis/index.php?p=background). Two efficient computational approaches were used for the prediction of the regulatory miRNAs that target E2F3, including TargetScan (http://www.targetscan.org/) and Pictar (https://pictar.mdc-berlin.de). Targets were selected for further confirmation from the group of miRNAs that were common to the results generated from both approaches.
Lentivirus Infection and Oligonucleotide Transfection
MiR-152 mimics and inhibitors, and negative control (NC) oligonucleotides were obtained from RiboBio. E2F3 siRNAs and plasmids, and NC were purchased from Genechem (Shanghai, China). Cell transfection was performed with lipofectamine 2000 reagent (Invitrogen), following the manufacturer’s manual.
Migration and Invasion Assays
The migration and invasion assays were performed as previously described.15 The Boyden chambers were obtained from Corning (MA, USA).
In vivo Metastasis Assays
The animal experiments were conducted as previously described using GC9811-P cells infected with miR-152 or E2F3 siRNA lentivirus and NC.15 The animal assays were approved by the Nanjing Drum Tower Hospital Animal Care Committee, and all animal studies complied with Nanjing Drum Tower Hospital animal use guidelines.
Luciferase Reporter Assays
The luciferase assays were carried out as previously described.15,17 Plasmids containing wild-type Luc-E2F3, mutant Luc-E2F3-ß3′-UTR, wild-type PLK1 promoter and mutant PLK1 promoter were obtained from GeneCopoeia (Rockville, MD, USA).
Chromatin Immunoprecipitation (CHIP) Assays
CHIP assays were performed as previously described using an anti-E2F3 antibody.17 The non-specific antibody (rabbit anti-IgG; BD Biosciences) was used as a negative control. The precipitated DNAs were subjected to PCR in an attempt to amplify the E2F3-binding sites using primers that are specific for PLK1.
Statistical Analysis
For statistical analyses, SPSS 22.0 software (Chicago, IL, USA) was used. The Student’s unpaired t-test was applied for comparison between 2 groups. The correlation of E2F3 and miR-152 in GC tissues was examined applying the Spearman correlation test. P<0.05 was considered statistically significant.
Results
Up-Regulated E2F3 Expression Is Related with Progression and Poor Prognosis in GC
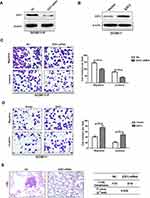
We extracted data of Kaplan Meier plotter and Oncomine to elucidate the expression and clinical value of E2F3 in GC. The results of 4 independent cohorts indicated that E2F3 was more frequently expressed in GC than normal gastric samples (Figure 1A). In addition, data from two cohorts including 876 and 641 GC individuals showed patients with higher expression of E2F3 exhibited decreased overall survival and progression-free survival (Figure 1B). In consequence, these investigations prove E2F3 expression is a crucial predictor of GC prognosis.
In order to classify E2F3’s function in GC metastasis, qRT-PCR and Western blot assays were conducted to examine E2F3 expression in GC9811-P, GC9811, MKN28M and MKN28NM, which are two pair of high and low invasive cell sublines characterised previously.18,19 Results indicated E2F3 expression was significantly down-regulated in GC9811 and MKN28NM compared with their counterparts (Figure 1C and D). Collectively, it can be suggestive that up-regulated E2F3 is notably associated with raised GC oncogenesis.
Up-Regulation of E2F3 Promotes GC Invasion and Metastasis
Next, we observed whether up-regulation of E2F3 promotes GC migration and invasion. For this purpose, GC9811-P cells were transfected with lentiviral containing E2F3 siRNA (Figure 2A). Transwell experiments showed that E2F3 knockdown markedly repressed the cell migration and invasion (Figure 2C). On the contrary, up-regulation of E2F3 facilitated GC cell migration and invasion (Figure 2B and D). In vivo metastasis experiments demonstrated that the silencing of E2F3 expression suppressed GC metastasis (Figure 2E). Thus, the above observations reveal that E2F3 serves as a carcinogenic role in the metastasis of GC.
miR-152 Suppresses E2F3 Expression by Directly Targeting Its 3ʹ-UTR
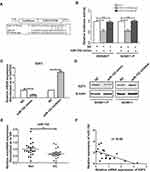
Bioinformatic analyses were performed to search for potential miRNAs targeting E2F3. According to the two bioinformatics programs (Targetscan and Pictar), a miR-152 binding sequence was predicted in the E2F3 3ʹ-UTR. To prove whether miR-152 suppressed E2F3 by directly binding to its 3ʹ-UTR, mutant or wildtype E2F3 3ʹ-UTR binding sites were, respectively, inserted into reporter vectors, as shown in Figure 3A. The results from luciferase reporter assays demonstrated miR-152 led to a distinct reduction in luciferase activity when the wild-type E2F3 3ʹ-UTR was present, whereas the luciferase activity was not markedly altered in the mutant 3ʹ-UTR (Figure 3B). These data suggested that miR-152 could inhibit E2F3 expression via targeting its 3ʹ-UTR. Furthermore, qRT-PCR and Western blot experiments indicated miR-152 silencing significantly raised E2F3 expression in GC9811 cells, while up-regulation of miR-152 dramatically reduced E2F3 expression in GC9811-P (Figure 3C and D). Collectively, it was indicative that miR-152 inhibits E2F3 expression by directly binding to the predicted site of its 3ʹ-UTR.
To further assess the clinical relevance of miR-152/E2F3 axis, we examined whether E2F3 expression was associated with miR-152 in GC samples. qRT-PCR experiments were performed to measure miR-152 expression in GC from 20 patients. It was found that miR-152 expression in GC samples was down-regulated, in contrast to the match non-tumor specimens (Figure 3E). The following statistical analysis suggested miR-152 expression was inversely correlated with E2F3 in GC tissues (Figure 3F).
miR-152 Is Involved in the Modulation of GC Metastasis
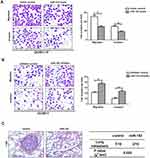
MiR-152 and inhibitor were, respectively, transfected into GC9811-P and GC9811 cells, to perform gain-of-function and loss-of-function tests. Transwell experiments revealed miR-152 up-regulation dramatically inhibited migration and invasion in GC9811-P cells (Figure 4A), whereas down-regulation of miR-152 enhanced migration and invasion in GC9811 cells (Figure 4B). Animal experiments further proved miR-152 up-regulation restrained GC metastasis (Figure 4C). Therefore, these investigations manifest that miR-152 plays a negative role on the GC invasion and metastasis.
miR-152/E2F3 Axis Regulates PLK1 Mediated AKT and ERK Pathways in GC Progression
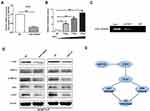
Next, we inspected the downstream mechanism of miR-152/E2F3 axis in GC progression. In a previous study,5 the expression pattern after E2F3 downregulation in bladder cancer was identified using cDNA microarray. Among the differentially expressed genes, PLK1 had been proved to be implicated in tumor metastasis and act as an oncogene.5,20 The data from Oncomine and Kaplan Meier plotter indicated that PLK1 was over-expressed in GC samples and high PLK1 expression predicted inferior prognosis in GC patients (Supplemental Figure 1). In consequence, we speculated that E2F3 adjusted GC progression through PLK1. In order to observe whether PLK1 expression was altered after E2F3 changed, qRT-PCR and Western blot experiments were carried out. The results demonstrated that knockdown of E2F3 expression in GC9811-P cells gave rise to increased expression of PLK1 (Figure 5A and D). Subsequently, luciferase assays were carried out to determine whether E2F3 was able to directly upregulate PLK1. HEK293T cells were co-transfected with 0.5μg PLK1 reporters together with 0.5μg, 1μg or 2μg E2F3 expression vectors. It was shown that PLK1 promoter was evidently activated by E2F3 (Figure 5B). The analyses of CHIP indicated that E2F3 could specifically bind to the promoter of PLK1 in GC9811-P cells, proving that E2F3 could directly modulate PLK1 (Figure 5C).
A large number of studies indicate that AKT and ERK signaling mediated by PLK1 play a crucial role in GC metastasis.20–22 Thus, we detected expression changes of primary molecules in AKT and ERK signal to next elucidate the downstream mechanism of miR-152/E2F3. Western blot assay showed that down-regulation of E2F3 or up-regulation of miR-152 notably improved PLK1 activity, and enhanced p-AKT (phospho-AKT) and p-ERK1/2 (phospho-ERK1/2) expressions (Figure 5D). Finally, these observations indicated that miR-152/E2F3 might modulate GC invasion and metastasis through accelerating PLK1 activity and subsequently enabling AKT and ERK signals (Figure 5E).
Discussion
Quite a few molecules and pathways are implicated in the process of cancer metastasis, and such complicated courses are fulfilled via modulation of metastasis-related genes by TFs, including serum response factor (SRF), nuclear factor kappa B (NF-κB), and activator protein 1 (AP1).17,23,24 However, the TF expression pattern related to GC metastasis has not been completely classified. Recent studies have shown that E2F3, a TF, contributes to tumor progression, including liver, prostate, lung and bladder cancer.5–7,25 Therefore, we are exploring the exact function and mechanism of E2F3 in GC invasion and metastasis, which is undefined as yet.
As a TF, E2F3 is widely reported to be a key regulator of cell cycle, leading to cell proliferation or apoptosis.3,4 Up-regulation of E2F3 is a common oncogenic factor during human carcinogenesis.4 E2F3 is frequently overexpressed in quite a few cancers and its decrease may block tumor progression.7,26 E2F3 has been found to be implicated in thyroid cancer metastasis to lung and liver, but the specific mechanism remained undetermined.8 The results from this study indicated that E2F3 was over-expressed in GC samples, and high level of E2F3 expression might tend to poor prognosis in GC folks. Furthermore, E2F3 was also closely correlated with metastasis of GC, and E2F3 knockdown could weaken the ability of GC invasion.
Over the past decade, growing evidence has proved miRNAs are closely related to human carcinogenesis, and play an oncogenetic or tumor suppressive role depending on the target genes.27 It has been distinct that miRNAs are implicated in metastasis of most tumors. miRNAs, including miR-7, miR-302c, miR-483, miR-371a-3p and miR-218, have been indicated to be involved in the GC progression.19,26,28–30 Our previous study demonstrated that miR-146a/b could markedly inhibit invasion and metastasis of GC cells.15 MiR-146a/b down-regulating UHRF1 (ubiquitin-like, containing PHD and RING finger domains 1) resulted in the activation of several tumor suppressor genes.15 Herein, miR-152 was recognized to play a significant part by directly targeting E2F3 3ʹ-UTR in GC metastasis.
E2F3 accomplished its multipurpose achievements by targeting the promoter of downstream genes. Libertini et al31 have shown that E2F3 could bind to the promoter of Interleukin 6 receptor in prostate cancer. It was also found that Aurora-A was transcriptionally targeted by E2F3 in ovarian cancer.32 A previous study5 using cDNA microarray showed PLK1 may be one of the E2F3 downstream molecules, and it motivated us to pay close attention to association between E2F3 and PLK1 in GC metastasis. PLK1 is a key regulator of cell cycle,33 and highly overexpressed in various cancer cells, including gastric cancer, breast cancer, and liver, and is categorized as an oncogene.20,34–36 In the subsequent research, we aimed to classify the specific mechanism of E2F3 regulating PLK1. CHIP and luciferase assays indicated that E2F3 directly targeted PLK1 to increase its activity. Numerous studies showed that PLK1 regulates multiple signals, which are involved in tumor proliferation, metastasis and drug-resistance.37 As is demonstrated, AKT and ERK pathways are activated by PLK1, and their activation stimulated EMT (epithelial-to-mesenchymal transition) in breast and prostate cancers.12,21 Moreover, Cai et al found that PLK1 accelerated the GC metastasis and EMT through regulating the AKT pathway.38 Consistent with these reports, our data further proved that miR-152/PLK1 axis could modulate GC invasion and metastasis by enhancing PLK1 activity and subsequent acceleration of AKT and ERK signals.
From the above, we come to a conclusion that high expression of E2F3 in GC progression might result from decreased level of miR-152, which could markedly promote GC invasion and metastasis. Moreover, miR-152/E2F3 link might modulate GC progression through AKT and ERK signaling mediated by PLK1. The novel identified miR-152/E2F3/PLK1 axis provides a new underlying target for early warning and therapy of metastasis in GC patients.
Acknowledgments
This study was supported by combined grants from the National Natural Science Foundation of China (No.81402337). This study was conducted in Nanjing Drum Tower Hospital from September 2013 to December 2019.
Author Contributions
Conceptualization, L.S., L.Z. and X.Z.; formal analysis, L.S., H.Z. and Y.S.; funding acquisition, L.Z.; investigation, Y.S., H.G. and X.D.; methodology, L.S., H.Z., P.W., and S.Z.; supervision, X.Z.; writing—original draft, L.S. and L.Z.; writing—review & editing, L.S., H.Z., Y.S., X.D., H.G., P.W., S.Z., L.Z. and X.Z. All the authors approved the version to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34. doi:10.3322/caac.v69.1
2. Van Cutsem E, Sagaert X, Topal B, Haustermans K, Prenen H. Gastric cancer. Lancet. 2016;388(10060):2654–2664. doi:10.1016/S0140-6736(16)30354-3
3. Ginsberg D. E2F3-a novel repressor of the ARF/p53 pathway. Dev Cell. 2004;6(6):742–743. doi:10.1016/j.devcel.2004.05.012
4. Gao Y, Feng B, Lu L, et al. MiRNAs and E2F3: a complex network of reciprocal regulations in human cancers. Oncotarget. 2017;8(36):60624–60639. doi:10.18632/oncotarget.17364
5. Olsson AY, Feber A, Edwards S, et al. Role of E2F3 expression in modulating cellular proliferation rate in human bladder and prostate cancer cells. Oncogene. 2007;26(7):1028–1037. doi:10.1038/sj.onc.1209854
6. Reimer D, Hubalek M, Kiefel H, et al. Regulation of transcription factor E2F3a and its clinical relevance in ovarian cancer. Oncogene. 2011;30(38):4038–4049. doi:10.1038/onc.2011.119
7. Johnson J, Thijssen B, McDermott U, Garnett M, Wessels LF, Bernards R. Targeting the RB-E2F pathway in breast cancer. Oncogene. 2016;35(37):4829–4835. doi:10.1038/onc.2016.32
8. Ziebold U, Lee EY, Bronson RT, Lees JA. E2F3 loss has opposing effects on different pRB-deficient tumors, resulting in suppression of pituitary tumors but metastasis of medullary thyroid carcinomas. Mol Cell Biol. 2003;23(18):6542–6552. doi:10.1128/MCB.23.18.6542-6552.2003
9. Trikha P, Sharma N, Pena C, et al. E2f3 in tumor macrophages promotes lung metastasis. Oncogene. 2016;35(28):3636–3646. doi:10.1038/onc.2015.429
10. Yates LA, Norbury CJ, Gilbert RJ. The long and short of microRNA. Cell. 2013;153(3):516–519. doi:10.1016/j.cell.2013.04.003
11. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215–233. doi:10.1016/j.cell.2009.01.002
12. Riquelme I, Letelier P, Riffo-Campos AL, Brebi P, Roa JC. Emerging role of miRNAs in the drug resistance of gastric cancer. Int J Mol Sci. 2016;17(3):424. doi:10.3390/ijms17030424
13. Song S, Ajani JA. The role of microRNAs in cancers of the upper gastrointestinal tract. Nat Rev Gastroenterol Hepatol. 2013;10(2):109–118. doi:10.1038/nrgastro.2012.210
14. Ishimoto T, Baba H, Izumi D, et al. Current perspectives toward the identification of key players in gastric cancer microRNA dysregulation. Int J Cancer. 2016;138(6):1337–1349. doi:10.1002/ijc.29627
15. Zhou L, Zhao X, Han Y, et al. Regulation of UHRF1 by miR-146a/b modulates gastric cancer invasion and metastasis. FASEB J. 2013;27(12):4929–4939. doi:10.1096/fj.13-233387
16. He L, Wang H, Jin H, et al. CIAPIN1 inhibits the growth and proliferation of clear cell renal cell carcinoma. Cancer Lett. 2009;276(1):88–94. doi:10.1016/j.canlet.2008.10.044
17. Zhao X, He L, Li T, et al. SRF expedites metastasis and modulates the epithelial to mesenchymal transition by regulating miR-199a-5p expression in human gastric cancer. Cell Death Differ. 2014;21(12):1900–1913. doi:10.1038/cdd.2014.109
18. Bai F, Guo X, Yang L, et al. Establishment and characterization of a high metastatic potential in the peritoneum for human gastric cancer by orthotopic tumor cell implantation. Dig Dis Sci. 2007;52(6):1571–1578. doi:10.1007/s10620-006-9570-x
19. Tie J, Pan Y, Zhao L, et al. MiR-218 inhibits invasion and metastasis of gastric cancer by targeting the Robo1 receptor. PLoS Genet. 2010;6(3):e1000879. doi:10.1371/journal.pgen.1000879
20. Jeong SB, Im JH, Yoon JH, et al. Essential role of Polo-like Kinase 1 (Plk1) oncogene in tumor growth and metastasis of tamoxifen-resistant breast cancer. Mol Cancer Ther. 2018;17(4):825–837. doi:10.1158/1535-7163.MCT-17-0545
21. Liao G, Wang R, Rezey AC, Gerlach BD, Tang DD. MicroRNA miR-509 regulates ERK1/2, the Vimentin network, and focal adhesions by targeting Plk1. Sci Rep. 2018;8(1):12635. doi:10.1038/s41598-018-30895-8
22. Gala K, Chandarlapaty S. Molecular pathways: HER3 targeted therapy. Clin Cancer Res. 2014;20(6):1410–1416. doi:10.1158/1078-0432.CCR-13-1549
23. Zhang S, Guo X, Wu H, et al. Wingless modulates activator protein-1-mediated tumor invasion. Oncogene. 2019;38(20):3871–3885. doi:10.1038/s41388-018-0629-x
24. Fusella F, Secli L, Busso E, et al. The IKK/NF-kappaB signaling pathway requires Morgana to drive breast cancer metastasis. Nat Commun. 2017;8(1):1636. doi:10.1038/s41467-017-01829-1
25. Kent LN, Bae S, Tsai S-Y, et al. Dosage-dependent copy number gains in E2f1 and E2f3 drive hepatocellular carcinoma. J Clin Invest. 2017;127(3):830–842. doi:10.1172/JCI87583
26. Yu FY, Zhou CY, Liu YB, Wang B, Mao L, Li Y. miR-483 is down-regulated in gastric cancer and suppresses cell proliferation, invasion and protein O-GlcNAcylation by targeting OGT. Neoplasma. 2018;65(3):406–414. doi:10.4149/neo_2018_170608N411
27. Esquela-Kerscher A, Slack FJ. Oncomirs - microRNAs with a role in cancer. Nat Rev Cancer. 2006;6(4):259–269. doi:10.1038/nrc1840
28. Zhao X, Dou W, He L, et al. MicroRNA-7 functions as an anti-metastatic microRNA in gastric cancer by targeting insulin-like growth factor-1 receptor. Oncogene. 2013;32(11):1363–1372. doi:10.1038/onc.2012.156
29. Chen L, Min L, Wang X, et al. Loss of RACK1 promotes metastasis of gastric cancer by inducing a miR-302c/IL8 signaling loop. Cancer Res. 2015;75(18):3832–3841. doi:10.1158/0008-5472.CAN-14-3690
30. Guo H, Ji F, Zhao X, et al. MicroRNA-371a-3p promotes progression of gastric cancer by targeting TOB1. Cancer Lett. 2019;443:179–188. doi:10.1016/j.canlet.2018.11.021
31. Libertini SJ, Chen H, al-Bataina B, et al. The interleukin 6 receptor is a direct transcriptional target of E2F3 in prostate tumor derived cells. Prostate. 2012;72(6):649–660. doi:10.1002/pros.v72.6
32. He L, Yang H, Ma Y, Pledger WJ, Cress WD, Cheng JQ. Identification of Aurora-A as a direct target of E2F3 during G2/M cell cycle progression. J Biol Chem. 2008;283(45):31012–31020. doi:10.1074/jbc.M803547200
33. de Carcer G, Manning G, Malumbres M. From Plk1 to Plk5: functional evolution of polo-like kinases. Cell Cycle. 2011;10(14):2255–2262. doi:10.4161/cc.10.14.16494
34. de Carcer G, Venkateswaran SV, Salgueiro L, et al. Plk1 overexpression induces chromosomal instability and suppresses tumor development. Nat Commun. 2018;9(1):3012. doi:10.1038/s41467-018-05429-5
35. Song R, Hou G, Yang J, et al. Effects of PLK1 on proliferation, invasion and metastasis of gastric cancer cells through epithelial-mesenchymal transition. Oncol Lett. 2018;16(5):5739–5744. doi:10.3892/ol.2018.9406
36. Zhang H, Diab A, Fan H, et al. PLK1 and HOTAIR accelerate proteasomal degradation of SUZ12 and ZNF198 during Hepatitis B virus-induced liver carcinogenesis. Cancer Res. 2015;75(11):2363–2374. doi:10.1158/0008-5472.CAN-14-2928
37. Cholewa BD, Liu X, Ahmad N. The role of polo-like kinase 1 in carcinogenesis: cause or consequence? Cancer Res. 2013;73(23):6848–6855. doi:10.1158/0008-5472.CAN-13-2197
38. Cai XP, Chen LD, Song HB, Zhang CX, Yuan ZW, Xiang ZX. PLK1 promotes epithelial-mesenchymal transition and metastasis of gastric carcinoma cells. Am J Transl Res. 2016;8(10):4172–4183.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.