Back to Journals » Clinical Interventions in Aging » Volume 14
Recovery Of Bone And Muscle Mass In Patients With Chronic Kidney Disease And Iron Overload On Hemodialysis And Taking Combined Supplementation With Curcumin And Resveratrol
Authors Murillo Ortiz BO , Fuentes Preciado AR, Ramírez Emiliano J , Martínez Garza S , Ramos Rodríguez E, de Alba Macías L.A.
Received 19 July 2019
Accepted for publication 22 October 2019
Published 18 November 2019 Volume 2019:14 Pages 2055—2062
DOI https://doi.org/10.2147/CIA.S223805
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Blanca Olivia Murillo Ortiz,1 Alma Rosa Fuentes Preciado,2 Joel Ramírez Emiliano,2 Sandra Martínez Garza,1 Edna Ramos Rodríguez,3 Luz Angélica de Alba Macías2
1Clinical Epidemiology Research Unit, UMAE No. 1 Bajio, Mexican Institute Social Security, León, Guanajuato, Mexico; 2Department of Medical Sciences, University of Guanajuato, León, Guanajuato, Mexico; 3Regional General Hospital No. 58, Mexican Institute Social Security, León, Guanajuato, Mexico
Correspondence: Blanca Olivia Murillo Ortiz
Unidad de Investigación en Epidemiología Clínica, Unidad Médica de Alta Especialidad No. 1 Bajío, Instituto Mexicano del Seguro Social, Boulevard López Mateos e Insurgentes s/n, Colonia Los Paraísos, León, Guanajuato, México
Tel/Fax +52 4777183039
Email [email protected]
Introduction: Malnutrition is common in haemodialysis patients and closely related to morbidity and mortality. We evaluated the effect of twelve weeks of supplementation with resveratrol and curcumin on recovery of bone and muscle mass and protein oxidation, lipid peroxidation on patients with chronic kidney disease and iron overload undergoing hemodialysis, we performed a randomized, double-blind, placebo-controlled trial.
Methods: We included a total of 40 patients, were randomly assigned to two groups, 20 to the group with antioxidant supplementation (Resveratrol + Curcumin) (Group A), treated with a daily oral dose of 500 mg of Resveratrol and 500 mg of Curcumin, and 20 to the control group treated with placebo (Group B).
Results: Significant differences were found in the body composition of the patients between both groups. There was a significant difference in Body Mass Index (BMI) values (p = 0.002), fat percentage (p = 0.007), muscle mass (p = 0.01) bone mass (p = 0.01), as well as in the score of the subjective global evaluation (p = 0.03). Also differences were found between the basal and final serum levels of Triglycerides (TG) (p = 0.01), VLDL (p = 0.003). A significant decrease in the levels of serum ferritin (2003.69 ± 518.73 vs 1795.65 ± 519.00 ng/mL; p = 0.04). Nor were significant differences observed between the baseline and the final Thiobarbituric Acid Reactive Substances (TBARS) values (70.45 ± 69.21 vs 50.19 ± 32.62, p = 0.24). The same results was obtained for carbonyl values (2.67 ± 0.75 vs 2.50 ± 0.85; p = 0.50).
Discussion: The present study is the first assay on patients with chronic kidney disease and iron overload that demonstrates the beneficial effects of combined supplementation with Curcumin and Resveratrol on muscle and bone mass. There was a significant decrease in circulating levels of ferritin, to finding that remarkably novel.
Keywords: curcumin, resveratrol, chronic kidney disease, recovery of bone and muscle mass
Introduction
Mechanisms Of Muscle Wasting In Chronic Kidney Disease
Muscle wasting is especially relevant in patients with chronic kidney disease (CKD) because it is progressive, and increases morbidity and the risk of death1. CKD induced activation of catabolism is the most important cause of muscle wasting tan decreased protein synthesis.
The molecular pathways underlying muscle atrophy are complicated. Studies in humans and rodents have identified the ubiquitin proteasome system (UPS) as the main pathway for the degradation of skeletal muscle proteins.1 Nuclear factor KB (NFKB) is activated in a redox-sensitive manner during muscle contraction due to increased oxidant production. Increased NFKB signaling decreases insulin action and promotes muscle wasting.2–4
Loss Of Bone Mass And Mineral Density In Chronic Kidney Disease Patients
CKD has also been associated with low grade inflammation, and is a major cause of both local and systemic bone loss due to excessive bone resorption and impaired bone formation. This imbalance in bone remodeling is at least in part mediated by the activation of osteoclasts and the impairment of osteoblast function by cytokines. In subjects with osteopenia, this balance is affected by excessive bone resorption or inadequate bone formation during bone remodeling, resulting in low bone density.5,6
Iron overload is a common complication in patients with CKD in hemodialysis; it results from the transfusions of red cells that these patients receive to treat symptomatic anemia, as well as from the administration of enteral and/or parenteral iron supplements. In a male mice model of iron overload, the administration of iron dextran increased the level of Reactive Oxygen Species (ROS) and the phosphorylation of p66 in bone, and caused trabecular and cortical thinning, while bone loss was largely prevented by treatment with the antioxidant N-acetyl-L-cysteine (NAC).7 Recently, Eniko Balogh, et al concluded that iron and ferritin specifically inhibit osteogenic commitment and differentiation of human bone marrow both in vitro and in vivo.8
Some authors Durbin SM et al and Shakibaei M et al have demonstrated that Resveratrol supplementation can promote osteogenesis differentiation of mesenchymal stem cells by mediating the modulation of Sirt-1/Runx2; it can also increase osteocalcin and alkaline phosphatase (ALP) levels in plasma and mitigate the loss of femur strength in hindlimb-suspended old male rats.9,10
Effects Of Curcumin On Muscle Mass And Bone Density
Curcumin is a polyphenolic compound derived from the Indian spice curcumin longa; it has been studied for over 3 decades, and has been reported to have potential benefits for oxidative stress, cancer, diabetes and inflammatory disease.11,12 Approximately 65% of the orally administered curcumin is rapidly cleared from the body, mainly in the feces.
Kazim S, et al2 suggest that curcumin has the potential to help prevent muscle damage by downregulating the NFKB, and upregulating the nuclear factor (erythroid-derived 2) like 2. They also noted that curcumin activates Sirtuin inhibitors (SIRT1) and potentially enhances mitochondrial biogenesis and fatty acid oxidation in adipocytes and myotubes.
Some of the molecular targets of curcumin are involved in bone remodeling. Curcumin stimulates apoptosis in mature rabbit osteoclasts, having an inhibitory effect on two factors Activator protein 1 (AP-1) and NFKB involved in osteoclast survival.
Bharti et al demonstrated that curcumin suppresses Receptor Activator for Nuclear Factor K B Ligand (RANKL) signaling and osteoclastogenesis by interfering with the NFKB pathway.13 Hie et al reported the inhibitory effects of curcumin on osteoclastic activity in insulin-dependent diabetes mellitus using rats with streptozotocin-induced diabetes.14 In RAW 264.7 cells, curcumin also inhibited osteoclastogenesis through the inhibition of NF-KB.15
In ovariectomized female rats (OVX), oral administration of curcumin (10mg kg-1 per day for 4 weeks) decreased total serum cholesterol levels and improved some bone histomorphometric parameters, including the endosteal osteoid width in tibia affected by estrogen deficiency.16
In a study by Riva A et al on 57 healthy subjects with low bone density, an oral formulation based on turmeric phytosome (Meriva®) induced a significant increase (+ 7.1% and + 4.8% at week 24) in the bone density of the small fingers and the upper jaw, respectively, compared with the initial values.17
Effects Of Resveratrol On Muscle Mass And Bone Density
Resveratrol has been shown to have many beneficial biological effects, including inhibition of NFKB activity and activation of AMP-activated protein kinase (AMPK). Resveratrol has been reported to stimulate histone deacetylase activity (SIRT1), and this effect is possibly one of the main mechanisms of action of this drug.18 Sharma et al reported that treatment with resveratrol (5 mg or 10 mg/kg orally) for 2 weeks improved urinary protein excretion, renal dysfunction and renal oxidative stress in streptozotocin-induced diabetic rats.19 Kim et al showed that SIRT1 activation by resveratrol reduces cisplatin induced acetylation of p53, apoptosis, and cytotoxicity in the proximal tubular cells of mice.20 Other studies have shown that resveratrol attenuates renal injury caused by several drugs, including glycerol, gentamicin and cyclosporine, by reducing oxidative stress.21–25
Evidence indicates that Resveratrol prevented an increase in Muscle RING-finger protein-1 (MuRF1) expression and attenuated muscle atrophy in an in vivo model of CDK. Li Jing Sun et al found that phosphorylation of NFKB was also inhibited at the same time.26
To determine the effect of twelve weeks of supplementation with resveratrol and curcumin on patients with chronic kidney disease and iron overload undergoing hemodialysis, we performed a randomized, double-blind, placebo-controlled trial.
Materials And Methods
General Characteristics Of Patients
The study was designed as a randomized, double blind, placebo-controlled trial with follow up at 6 and 12 weeks. The study complies with the Helsinki declaration and was approved by the Institutional Ethical Committee of the Mexican Institute of Social Security (IMSS R-2015-785-109). All the patients were invited to participate and the consent was informed and signed their written informed consent. We included a total of 40 patients with nephropathy undergoing renal replacement therapy with hemodialysis at the High Specialty Medical Unit No. 1 Bajio, Institute Mexican Social Security. The patients were randomly assigned to two groups, 20 to the group with antioxidant supplementation (Resveratrol + Curcumin) (Group A), treated with a daily oral dose of 500 mg of Resveratrol and 500 mg of Curcumin, and 20 to the control group treated with placebo (Group B) over a period of 12 weeks. The patients were matched according to their age and gender.
Statistical Analysis
The data were analyzed using Student’s t-test. Repeated measurements were performed during the follow-up to determine the differences between the groups.
Results
Anthropometric And Clinical Characteristics Of Patients On Hemodialysis
An analysis of the etiology of chronic kidney disease in patients on hemodialysis showed that 40% of the patients included in this study had an etiology of arterial hypertension, 25% had Diabetes mellitus type II and 35% had other etiologies, including renal hypoplasia and chronic glomerulonephritis (Table 1). The etiology of kidney disease was distributed homogeneously between both groups.
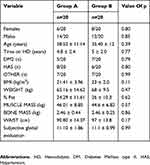 |
Table 1 Baseline Clinical And Anthropometric Characteristics Of Patients On Hemodialysis |
No significant differences of gender and age were found between the groups (Table 2). It can thus be inferred that both groups of patients were homogeneous in terms of the aforementioned variables, and therefore the differences found in the other measured parameters were not attributed to them.
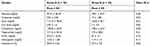 |
Table 2 Baseline Biochemical Markers Of Patients On Hemodialysis |
Effect On The Biochemical And Clinical Markers Of The Group Treated With Resveratrol And Curcumin
At the end of the study, we compared biochemical markers to evaluate the effect of Resveratrol and Curcumin supplements. Significant differences were found between the basal and final serum levels of Triglycerides (TG) (p = 0.01) and VLDL (p = 0.003), whereas no significant differences were found in glucose and uric acid levels (Table 3).
 |
Table 3 Effect On The Biochemical Markers Of The Group Treated With Resveratrol And Curcumin Supplementation (Group A) And Control (Group B) |
Effect Of Treatment With Resveratrol And Curcumin On Clinical And Anthropometric Parameters
Significant differences were found in the body composition of the patients between both groups. There was a significant difference in BMI values (p = 0.002), fat percentage (p = 0.007), muscle mass (p = 0.01) bone mass (p = 0.01), as well as in the score of the subjective global evaluation (p = 0.03) (Table 4).
 |
Table 4 Clinical And Anthropometric Characteristics Of The Group Treated With Resveratrol And Curcumin |
Oxidative Stress And Iron Overload
Iron Overload
A significant decrease in the levels of serum ferritin (−15.45%; 2003.69 ± 518.73 vs 1795.65 ± 519.00 ng/mL; p = 0.04) was observed in the patients treated with Curcumin and Resveratrol, while the control group showed no significant change (Table 5).
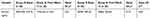 |
Table 5 Effect On The Ferritin Levels Of The Group Treated With Resveratrol And Curcumin Supplementation (Group A) And Control (Group B) |
Lipids And Oxidized Proteins In The Serum Of Patients On Hemodialysis
Serum TBARS levels were quantified to determine the oxidative damage to lipids. To determine the oxidative damage to oxidized proteins, serum carbonyls were quantified. These measurements showed that the markers of oxidative stress had no significant changes. Nor were significant differences observed between the baseline and the final TBARS values (70.45 ± 69.21 vs 50.19 ± 32.62, p = 0.24). The same results was obtained for carbonyl values (2.67 ± 0.75 vs 2.50 ± 0.85; p = 0.50).
Discussion
The findings of this study showed that supplementation with resveratrol and curcumin induced an increase in muscle mass and bone mass in hemodialysis patients. Several studies conducted in recent years reported possible health benefits of Resveratrol and Turmeric in cardiovascular and renal disease patients. Resveratrol is a powerful antioxidant agent that can act as a ROS scavenger.27 Moreover, Resveratrol can have several protective effects against age related disorders, including kidney diseases, through the activation of NAD dependent deacetylase and Sirtuin 1 (SIRT1).28 SIRT1 deacetylates several substrates and is an important regulator of a wide variety of cellular processes, including stress responses, cell survival, mitochondrial biogenesis and metabolism, occur in response to cellular energy levels and the redox state.28 Regarding Turmeric, a study by Shain et al (2016) in Wistar rats treated with 100 mg/Kg/day and exercise for 6 weeks showed that Turmeric helps prevent muscle wasting by regulating NFKB and Nuclear factor erythroid (Nrf2).2
Furthermore, by deacetylating target proteins SIRT1 can regulate multiple cellular functions, including apoptosis, mitochondrial biogenesis, inflammation, glucose/lipid metabolism, autophagy and cellular stress adaptations.7 An excess of ROS plays a role in a variety of diseases, as well as in the aging process, which involves numerous cellular response pathways.29,30
Patients with chronic kidney disease maintain inflammation and oxidative stress processes during renal replacement therapy with hemodialysis, which is why this disease is used as a model of premature aging.31 Oxidative stress is induced by an imbalance between ROS production and antioxidant defenses; therefore, exogenous antioxidants or the modulation of antioxidant enzymes can be expected to reduce oxidative stress. In this study we observed high levels of oxidative stress and high levels of lipid and protein peroxidation in patients with chronic kidney disease in hemodialysis; these metabolic conditions contribute to the accelerated loss of muscle and bone mass.
Muscle atrophy is a common complication of CKD and is associated with increased morbidity and mortality. The processes that lead to the loss of muscle mass are also present in various catabolic conditions. Understanding the pathogenesis of CKD and induced muscle loss could lead to the use of therapeutic interventions to prevent muscle wasting, and potentially other catabolic conditions, in CKD patients.32
Skeletal muscle atrophy is a common complication there is a decrease in protein synthesis in skeletal muscle, while protein degradation increases due to an increase in the activity and expression of the ubiquitin-proteasome proteolytic pathway. This pathway does not respond to simple nutritional intervention. It is belived that certain agents, including glucocorticoids, cytokines, the proteolysis inducing factor (PIF) and oxidative stress are responsible for the induction of the ubiquitin-proteasome pathway in skeletal muscle under catabolic conditions. Insulin suppresses the activation of this pathway, and so the loss of insulin action in diabetes leads to muscle wasting. It is thought that cytokines, PIF and ROS induce proteasome expression by the activation of NFKB.30
Targets for therapeutic intervention include antagonists of the inducers of proteasome expression, intracellular signaling pathways that lead to the activation of NFKB, and enzymes that induce ubiquitin conjugation to the substrate protein (myosin) and to proteasome.33 Anti-cytokine and anti-PIF antibodies are effective in attenuating muscle protein degradation in certain animal experimental models, while glucocorticoid receptor antagonists are effective in the treatment of sepsis. Agents that inhibit the activation of NFKB, such as resveratrol, thalidomide, ibuprofen, eicosapentaenoic acid and beta-hydroxy-beta-methylbutyrate are effective in preserving skeletal muscle mass in cachexia.30 These results suggest that the ubiquitin-proteasome pathway is an appropriate therapeutic target to prevent muscle wasting.34
Patients with chronic kidney disease (CKD) have an increased risk of mortality and morbidity that is associated with loss of body fat and lean mass.35 A study using an in vivo model of chronic kidney disease showed that Resveratrol prevents MURF1 from increasing and attenuates muscular atrophy. Li Jing Sun et al found that phosphorylation of NFKB is also inhibited at the same time.26
Dong Tao W et al (2014) showed that Resveratrol prevents muscle atrophy induced by TNFα by regulating Akt/mTOR/Foxo1 signaling in C2C12 myotubes. There are also reports that treatment with Resveratrol prevents degradation induced by the Proteolysis Factor (PIF).28 Previous studies have also shown that Resveratrol can directly eliminate ROS. Moreover, resveratrol has been reported to stimulate the activity of histone deacetylase SIRT1, which is probably one of the main mechanisms of action of this drug.18,26
In the present study, anthropometric parameters were evaluated at the beginning of the treatment to determine its effect on the percentage of muscle mass and bone mass. Both variables showed a significant increase after 12 weeks of supplementation (Muscle, 46.01 ± 8.85 kg versus 53.51 ± 9.81 kg, p= 0.01; Bone 2.46 ± 0.44 kg versus 2.85 ± 0.48 kg, p= 0.01), leading to a consequent increase in the BMI, which in turn had significant effect on the subjective global evaluation. The effect on the subjective global evaluation had an influence on the nutritional risk of renal patients, which has been mentioned as an important survival factor.
Several studies have analyzed the effect of supplementation on bone mass percentage; a randomized controlled trial with placebo showed that Resveratrol increases bone mineral density and bone alkaline phosphatase in obese men. That study aimed to evaluate the effects of Resveratrol treatment on the bones of men with metabolic syndrome.36 The patients received an oral treatment consisting of 1000 mg of Resveratrol (RSV, high), 150 mg of Resveratrol (RSV, low) or placebo daily for 16 weeks. Bone alkaline phosphatase (BAP) increased in a dose-dependent manner when the patients were treated with Resveratrol (R = 0.471, P <0.001), which resulted in a significantly greater increase in BAP in the RSV (high) group compared to the group receiving placebo (week 4, 16.4 ± 4.2%, P <0.001; week 8, 16.5 ± 4.1%, P <0.001; week 16, 15.2 ± 3.7%, P <0.001). The trabecular volumetric bone mineral density of the lumbar spine (LS vBMD-trab) also increased, showing a direct dose-dependent relationship with RSV (R = 0.26, P = 0.03), with a significant increase of 2.6 ± 1.3% in the RSV (high) group compared with the placebo group (P=0.04), our study with the combined supplementation of turmeric and resveratrol, an increase in bone mass was observed.
It has been experimentally shown in murine models that Resveratrol (RSV) has anti-inflammatory properties, and studies with rodents suggest it has protective effects for bones.37 An assay done on mice with iron overload and osteoporosis showed that Resveratrol protects against the destructive effects of iron on bones, and that there was a significant recovery of bone density and trabecular space. Other authors have described beneficial effects of Resveratrol such as promoting osteogenic differentiation by modulating Sirt1/Runx2 mediated by mesenchymal progenitor cells, increasing plasma levels of osteocalcin and alkaline phosphatase, and reducing the loss of diameter in the femur neck of male rats.37
Similarly, the study analyzed the effect of a combination of Turmeric 500mg/day + Resveratrol at 500 mg/day on the percentage of bone mass of CKD patients on hemodialysis. Amira Zaky et al, have demonstrated that the synergistic and therapeutic effects of resveratrol–curcumin combination, also show that both compounds exert beneficial effect either cooperatively or through differential molecular mechanisms in counteracting aluminum-induced neuroinflammation.38
In our study, the patients had high levels of ferritin, which produces a high level of inflammation and we were able to observe that at the end of the treatment the ferritin levels decreased significantly. There was a significant decrease in circulating levels of ferritin, a finding that is remarkably similar to what was reported in a murine model by Lu Zhao (2015). Tarantino G, et al, recently observed that transferrin levels are a predictor of adequate hemodialysis, a limitation of their study not having evaluated the transferrin levels.39 It is possible that the accumulated iron increases the bone response through the mentioned pathways. However, the relevant molecular mechanisms of Resveratrol, the regulation of FOXOS in bone, and osteoblast damage induced by iron overload remain unclear.
Conclusions
The present study is the first assay on patients with chronic kidney disease and iron overload that demonstrates the beneficial effects of combined supplementation with Curcumin and Resveratrol on muscle and bone mass. The subjective global evaluation had an influence on the nutritional risk of renal patients, as an important survival factor. Ferritin levels were monitored, showing a significant decrease in serum levels.
Abbreviations
ALP, Alkaline Phosphatas; AMPK, AMP-activated protein kinase; AP-1, Activator protein; BAP, Bone alkaline phosphatase; BMI, Body Mass Index; CKD, Chronic Kidney Disease; FOXO, Forkhead box O; LS vBMD-trab, Trabecular Volumetric Bone Mineral Density of the Lumbar Spine; MuRF1, Muscle RING-finger protein-1; NAC, Antioxidant N-acetyl-L-Cysteine; NAD, Nicotine Adenine Dinucleotide; NF-KB, Nuclear factor -kB; PIF1, Proteolysis Inducing Factor; RANKL, Receptor Activator for Nuclear Factor κ B Ligand; ROS, Reactive Oxygen Species; RSV, Resveratrol; SGE, Subjective global evaluation; SIRT1, Sirtuin inhibitors; TBARS, Thiobarbituric Acid Reactive Substances; UPS, Ubiquitin Proteasome System.
Acknowledgements
This work was supported by Mexican grants from FIS/IMSS/1561.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Workeneh BT, Mitch WE. Review of muscle wasting associated with chronic kidney disease. Am J Clin Nutr. 2010;91(4):1128S–1132S. doi:10.3945/ajcn.2010.28608B
2. Sahin K, Pala R, Tuzcu M, et al. Curcumin prevents muscle damage by regulating NF-kB and Nrf2 pathways and improves performance: an in vivo model. J Inflamm Res. 2016;9:147–154. doi:10.2147/JIR.S110873
3. George L, Asghar M, Lokhandwala MF. Exercise activates redox-sensitive transcription factors and restores renal D1 receptor function in old rats. Am J Physiol Renal Physiol. 2009;297(5):F1174–F1180. doi:10.1152/ajprenal.00397.2009
4. Ali S, Mann DA. Signal transduction via the NFkB pathway: a targeted treatment modality for infection, inflammation and repair. Cell Biochem Funct. 2004;22(2):67–79. doi:10.1002/cbf.1082
5. Raisz LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. J Clin Invest. 2005;115:3318–3325. doi:10.1172/JCI27071
6. Jilka RL, O’Brien CA. The role of osteocytes in age-related bone loss. Curr Osteoporos Rep. 2016;14:16–25. doi:10.1007/s11914-016-0297-0
7. Zhang Y, Zhai W, Zhao M, et al. Effects of iron overload on the bone marrow microenvironment in mice. PLoS One. 2015;10(3):e0120219. doi:10.1371/journal.pone.0120219
8. Balogh E, Tolnai E, Nagy BJ, et al. Iron overload inhibits osteogenic commitment and differentiation of mesenchymal stem cells via the induction of ferritin. Biochim Biophys Acta. 2016;1862(9):1640–1649. doi:10.1016/j.bbadis.2016.06.003
9. Durbin SM, Jackson JR, Ryan MJ, Gigliotti JC, Alway SE, Tou JC. Resveratrol supplementation influences bone properties in the tibia of hindlimb-suspended mature fisher 344 x Brown Norway male rats. Appl Physiol Nutr Metab. 2012;37:1179–1188. doi:10.1139/h2012-099
10. Shakibaei M
11. Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. Aaps J. 2013;15(1):195–218. doi:10.1208/s12248-012-9432-8
12. Gupta SC, Patchva S, Koh W, Aggarwal BB. Discovery of curcumin,a component of golden spice, and its miraculous biological activities. Clin Exp Pharmacol Physiol. 2012;39(3):283–299. doi:10.1111/j.1440-1681.2011.05648.x
13. Bharti AC, Takada Y, Aggarwal BB. Curcumin (diferuloylmethane) inhibits receptor activator of NF-Kappa B ligand-induced NF-Kappa B activation in osteoclast precursors and suppresses osteoclastogenesis. J Immunol. 2004;172:5940–5947.
14. Hie M, Yamazaki M, Tsukamoto I. Curcumin suppresses increased bone resorption by inhibiting osteoclastogenesis in rats with streptozotocin- induce diabetes. Eur J Pharmacol. 2009;621(1–3):1–9. doi:10.1016/j.ejphar.2009.08.025
15. Rohanizadeh R, Deng Y, Verron E. Therapeutic actions of curcumin in bone disorders. Bonekey Rep. 2016;5. doi:10.1038/bonekey.2016.20
16. Folwarczna J, Zych M, Treciak HI. Effects of curcumin on the skeletal system in rats. Pharmacol Rep. 2010;62:900–909. doi:10.1016/S1734-1140(10)70350-9
17. Riva A, Togni S, Giacomelli L, et al. Effects of a curcumin-based supplementation in asymptomatic subjects with low bone density: a preliminary 24-week supplement study. Eur Rev Med Pharmacol Sci. 2017;21:1684–1689.
18. Fröjdö S
19. Sharma S, Anjaneyulu M, Kulkarni SK, Chopra K. Resveratrol, a polyphenolic phytoalexin, attenuates diabetic nephropathy in rats. Pharmacology. 2006;76(2):69–75. doi:10.1159/000089720
20. Kim DH, Jung YJ, Lee JE, et al. SIRT1 activation by resveratrol ameliorates cisplatin-induced renal injury through deacetylation of p53. Am J Physiol Renal Physiol. 2011;301(2):F427–F435. doi:10.1152/ajprenal.00258.2010
21. Soares TJ, Volpini RA, Francescato HDC, Costa RS, da Silva CGA, Coimbra TM. Effects of resveratrol on glicerol-induced acute renal failure in rat kidney. Life Sci. 2007;81(8):647–656. doi:10.1016/j.lfs.2007.06.032
22. Chander V, Chopra K. Protective effect of resveratrol,a polyphenolic phytoalexin on gentamicin-induced acute renal failure in rat kidney. Ren Fail. 2006;28(2):161–169. doi:10.1080/08860220500531112
23. Morales AI, Buitrago JM, Santiago JM, Fernández-Tagarro M, López-Novoa JM, Pérez- Barriocanal F. Protective effect of trans-resveratrol on gentamicin-induced nephrotoxcicity. Antioxid Redox Signal. 2002;4(6):893–898. doi:10.1089/152308602762197434
24. Silan C, Uzun O, Comunoglu NU, Gokcen S, Bedirhan S, Ceniz M. Gentamicin-induced nephrotoxicity in rats ameliorated and healing effects of resveratrol. Biol Pharm Bull. 2007;30(1):79–83. doi:10.1248/bpb.30.79
25. Chander V, Tirkey N, Chopra K. Resveratrol, a polyphenolic phytoalexin protects against cyclosporine-induced nephrotoxicity through nitric oxide dependent mechanism. Toxicology. 2005;210(1):55–64. doi:10.1016/j.tox.2005.01.011
26. Sun LJ, Sun YN, Chen SJ, Liu S, Jiang GR. Resveratrol attenuates skeletal muscle atrophy induced by chronic kidney disease via MuRF1 signaling pathway. Biochem Biophys Res Commun. 2017;487(1):83–89. doi:10.1016/j.bbrc.2017.04.022
27. Holthoff JH, Woodling KA, Doerge DR, Burns ST, Hinson JA, Mayeux PR. Resveratrol, a dietary polyphenolic phytoalexin, is a functional scavenger of peroxynitrite. Biochem Pharmacol. 2010;80(8):1260–1265. doi:10.1016/j.bcp.2010.06.027
28. Wang DT, Yin Y, Yang YJ, et al. Resveratrol prevents TNF-α-induced muscle atrophy via regulation of Akt/mTOR/FoxO1 signaling in C2C12 myotubes. Int Immunopharmacol. 2014;19(2):206–213. doi:10.1016/j.intimp.2014.02.002
29. Xiaonan H, Wang H, Mitch WE. Muscle wasting from kidney failure a model for catabolic conditions. Int J Biochem Cell Biol. 2013;45(10):2230–2238. doi:10.1016/j.biocel.2013.06.027
30. Tisdale MJ. The ubiquitin-proteasome pathway as a therapeutic target for muscle wasting. J Support Oncol. 2005;3(3):209–217.
31. Kimmel PL, Phillips TM, Simmens SJ, et al. Immunologic function and survival in hemodialysis patients. Kidney Int. 1998;54(1):236–244. doi:10.1046/j.1523-1755.1998.00981.x
32. Fanzani A, Conraads VM, Penna F, Martinet W. Molecular and cellular mechanisms of skeletal muscle atrophy: an update. J Cachexia Sarcopenia Muscle. 2012;3(3):163–179. doi:10.1007/s13539-012-0074-6
33. Resmi H. The combination of bortezomib and resveratrol may prevent muscle wasting in diabetes. Med Hypotheses. 2011;76(2):291–292. doi:10.1016/j.mehy.2010.10.026
34. Attaix D, Ventadour S, Taillandier D, Combaret L. The ubiquitin-proteasome pathway: limitations and opportunities. Support Oncol. 2005;3(3):221–222.
35. Carrero JJ, Chmielewski M, Axelsson J, et al. Muscle atrophy, inflammation and clinical outcome in incident and prevalent dialysis patients. Clin Nutr. 2008;27:557–564. doi:10.1016/j.clnu.2008.04.007
36. Ornstrup MJ
37. Zhao L, Wang Y, Wang Z, Xu Z, Zhang Q, Yin M. Effects of dietary resveratrol on excess-iron-induced bone loss via antioxidative character. J Nutr Biochem. 2015;26(11):1174–1182. doi:10.1016/j.jnutbio.2015.05.009
38. Zaky A, Bassiouny A, Farghaly M, Bassma M. A combination of resveratrol and curcumin is effective against aluminum chloride- induced neuroinflammation in rats. J Alzheimers Dis. 2017;60(1):S221–S235. doi:10.3233/JAD-161115
39. Tarantino G, Vinciguerra M, Ragosta A, et al. Do transferrin levels predict haemodialysis adequacy in patients with end-stage renal disease? Nutrients. 2019;11:1123. doi:10.3390/nu11051123
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
