Back to Journals » Journal of Asthma and Allergy » Volume 16
Quick Treatment of Very Severe Refractory Hand and Foot Eczema with Dupilumab: A Case Report and Literature Review
Authors Li Y , Xiao J, Sun Y , Fang H, Qiao J
Received 30 November 2022
Accepted for publication 21 December 2022
Published 5 January 2023 Volume 2023:16 Pages 1—8
DOI https://doi.org/10.2147/JAA.S399340
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Luis Garcia-Marcos
Yali Li,1 Jialing Xiao,2 Yi Sun,3 Hong Fang,1 Jianjun Qiao1
1Department of Dermatology, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, People’s Republic of China; 2Department of Stomatology, Zhejiang Hospital, Hangzhou, People’s Republic of China; 3Department of Dermatology, Jingzhou Central Hospital, The Second Clinical Medical College, Yangtze University, Jingzhou, People’s Republic of China
Correspondence: Jianjun Qiao, Department of Dermatology, The First Affiliated Hospital, School of Medicine, Zhejiang University, 79# Qingchun Road, Shangcheng District, Hangzhou, 310003, People’s Republic of China, Tel +86-571-87235857, Email [email protected]
Abstract: Previously case reports showed dupilumab may benefit for hand eczema treatment, but relatively comprehensive assessments are lacking. A 45-year-old male with multiple severe vesicles, bullae and pustule on the palmar aspects of both hand and foot diagnosed dyshidrotic eczema by pathology was treated with dupilumab at an initial dose of 600 mg subcutaneously, followed by 300 mg every 2 weeks. The physician’s assessment of the patient revealed an excellent response to the treatment with dupilumab; the lesions and symptoms achieved dramatic improvement on the third day, and at 6 weeks, the hands and feet became completely normal without relapse in the past 1.5 years of discontinuation. Systematic literature searches were performed, and 6 case reports, 5 case series, 2 prospective observational studies and 1 retrospective review with a total of 150 patients were identified to describing the evaluation of efficacy and safety of dupilumab treatment for hand and foot eczema. Dupilumab appears to be safe and well tolerated with clinical benefit in recalcitrant hand and foot eczema. Larger randomized controlled trials using validated outcome measures and detailed hand eczema type and population classification are needed before dupilumab can be applied in clinical settings.
Keywords: dupilumab, hand eczema, dyshidrotic eczema, treatment, quick
Introduction
Hand eczema is a socially significant disease because of its high prevalence, morbidity and the associated loss of working time due to sick leave.1 Clinically, this disease can manifest only on the hand or foot or both on the hand and foot at the same time, and its manifestations vary from vesiculous and erosive to hyperkeratotic and desquamative with a wide spectrum of severity and chronicity.2 Because of the wide diversity of etiological and clinical factors, standard treatment approaches are frequently difficult.3 According to the current knowledge of the clinical phenotype of atopic dermatitis (AD) in adults, 65% AD patients present hand dermatitis, and, therefore, recently, new therapeutics, such as biologics and topical Janus kinase inhibitors or PDE4 inhibitors, for the treatment of AD have shown that hand eczema patients may benefit from them.4
Dupilumab is the first FDA (Food and Drug Administration)-approved human monoclonal antibody for the treatment of moderate-to-severe atopic dermatitis. However, new possible indications that dupilumab was used in non-AD dermatologic conditions are increasingly being explored. Skin diseases, such as prurigo nodularis, nummular eczema, allergic contact dermatitis, chronic hand eczema, spontaneous chronic urticaria, bullous pemphigoid, and alopecia areata.5,6 Additionally, nonskin diseases, such as asthma, chronic rhinosinusitis with nasal polyposis, eosinophilic gastrointestinal disorders and food allergies, are new research fields.7,8 Here, we describe a patient with very severe and refractory hand and foot eczema who was quickly treated with dupilumab. Furthermore, we systematically reviewed the literature to identify studies of hand eczema treated with dupilumab.
Case Report
A man as a gym teacher in his 45s suddenly suffered very severe hand and foot eczema, multiple severe vesicles, bullae and pustule on the palmar aspects of both hand and foot recurred for 1.5 years, and he experienced no episodes of spontaneous improvement but progressively worsened. He and his family had no previous or concurrent atopy diseases and no allergies. His quality of life was severely affected by this disease, and he had to stop the teacher work.
From 2018 to 2020, he was treated with different medical treatments in other clinics, including high-potency topical corticosteroids (halometasone 0.05%, clobetasol propionate 0.05%), potassium permanganate baths, bifonazole cream 1%, and oral itraconazole beyond three months due to suspected mycoses but no improvement. After that, the biopsy of the palm showed parakeratosis, epidermal spongiosis and intraepidermal vesicles, and neutrophils, lymphocytes and a few eosinophils were seen in the blister as well as a mixed-cell perivascular dermal infiltrate, consistent with dyshidrotic eczema. Patch testing did not reveal any contact allergens. Laboratory test results showed eosinophils 1.15 × 10e9/L and serum total IgE >200 IU/mL but no positive allergen-specific IgE. Subsequently, he was treated intermittently with a multitude of systemic drugs, including oral compound glycyrrhizin, tripterygium glycoside, and corticosteroid therapy, intermittently. All of those systemic therapies just improved the lesions briefly, but on the contrary, lesions were exacerbated with every recurrence. The first time he came to our clinic, the lesions on the palm and plantar showed not only yellowish vesicles, blisters, bullae, and even bloody bulla, swelling and scabs accompanied by intense itching and pain that made it incapacitated for the hands and feet (Figure 1A and B). His dyshidrotic eczema was very severe according to photographic guide. The patient explicitly refused to accept the previous local and systemic medications, so we just gave cyclosporine as the therapy in order to control the symptoms as soon as possible. After one week of oral cyclosporine, 50 mg twice a day, he could not tolerate the itching and painful symptoms and required a new treatment regimen. Then, 600 mg dupilumab subcutaneously initiated with a loading dose followed by 300 mg once every 2 weeks was added to the treatment. Just three days later, the lesions and symptoms achieved dramatic improvement; one week later, the blisters dried up, swelling subsided, and symptoms were completely cleared. Three weeks after the first dose, the patient almost healed, with only a small amount of blood scab remaining, so he was able to resume his work, and cyclosporine was discontinued. At week 6, the hands and feet were completely normal without relapse (Figure 1C and (D), and after 16 weeks of treatment, there was no recurrence or adverse events at 1.5 years of discontinuation. This study was approved by the ethics committee of Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University (No. 2022-1024). Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
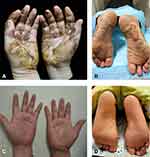 |
Figure 1 Clinical improvement of patients after 6 weeks of dupilumab treatment. (A and B) at baseline; (C and D) after 6 weeks of treatment. |
Discussion
This case highlights the speed and effectiveness of dupilumab in a patient with hand and foot dyshidrotic eczema that was extremely refractory to therapy and severely reduced the patient’s quality of life. A literature search of PubMed, Web of Science, Embase, and ClinicalTrials.gov was performed using the following search terms: “dupilumab”, “dyshidrotic eczema”, “hand eczema”, “hand dermatitis”, “foot eczema”, “foot dermatitis”, “hand and foot dermatitis”, “treatment”, and “anti-IL- 4”, “anti-IL- 13”. Cross-references were retrieved, but these did not identify additional studies, and studies published until October 2022 were included. Six case reports, 5 case series, 2 prospective observational studies and 1 retrospective review with a total of 150 patients were identified and summarized in Table 1. Three trials exploring dupilumab treatments in patients with hand and foot eczema are presently being recruiting or active but are not recruiting (ClinicalTrials.gov, 19-Oct-22) and no trial with results available. The numbers of male and female patients were 77 vs 73.
 |  |  |
Table 1 Published Patient Cases with Hand Eczema Treated with Dupilumab |
Patients were treated with the standard approved dosing of dupilumab, 600 mg subcutaneously at day 0, then 300 mg every two weeks, and one patient used 300 mg every 3 weeks due to the high cost.14 The duration of hand eczema ranged from 6 months to 68 years. Patients and their family history of atopy, patients’ skin lesion localizations did not receive much attention. Almost all treatment options were exhausted when dupilumab treatment was started; the prior treatments included emollients, topical corticosteroids, calcineurin inhibitors, antimicrobials, ultraviolet-A, narrowband ultraviolet-B, ultraviolet-B, psoralen ultraviolet-B, psoralen ultraviolet-A, and systemic drugs such as histamine antagonists, corticosteroids, methotrexate, cyclosporine, antimicrobials, alitretinoin, acitretin, azathioprine, mycophenolic acid, mycophenolate mofetil, tacrolimus, apremilast and psoriasis-related biologic medications. Every patient was treated with at least one or more systemic drugs. Efficacy evaluation for dupilumab showed that two patients achieved 1 week of improvement and 6 weeks of clearance after initial treatment.15,16 The skin lesions were clear as fast as 4 weeks.12 Most patients achieved complete skin clearance at 16 weeks. The two prospective observational studies including the same patients from the BioDay registry showed no difference in response between hand eczema subtypes at 16 weeks and 52 weeks.20,21
Regarding dupilumab-associated adverse events, 1 case report showed injection site pain and ongoing pruritic conjunctivitis;10 1 case series reported 2 facial redness and 1 ocular surface disease;19 2 cases mentioned no adverse events associated with dupilumab;13,14 the prospective observational study showed 27.8% mild conjunctivitis, 3 severe conjunctivitis with limbitis, 1 multiple verrucae filiforme and blood eosinophilia;21 and the other studies did not report related adverse reactions. That means dupilumab is safe in patients with hand eczema, consistent with dupilumabin special populations.23
Literature reports often focus on improving time and clearing time; 4 weeks and 16 weeks were the most reported, respectively. Case reports do not focus on the duration and efficacy of long-term treatment. One of the limitations of the research on hand eczema reported thus far is that patients in the prospective observational study and the retrospective review with AD, for hand eczema patients without AD, need more evidence. Additionally, the clinical manifestations and the affected area of hand and foot eczema are highly variable and involve pruritus and pain, and the subjective and objective evaluation tools are not unified. Existing reports have not quantified pruritus or pain in patients and have mainly focused on improvement in skin lesions.
Conclusion
Dupilumab appears to be a safe and effective alternative for the treatment of patients with refractory hand and foot eczema, which has a significant negative impact on patients’ activity level and quality of life. Larger studies with a homogenous design, including refinement of population, disease categories and standardized measures, are needed.
Acknowledgments
The Zhejiang Provincial Natural Science Foundation [LY20 H110001 to Y.L.L.] funded this work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Antonov D, Schliemann S, Elsner P. Hand dermatitis: a review of clinical features, prevention and treatment. Am J Clin Dermatol. 2015;16(4):257–270. doi:10.1007/s40257-015-0130-z
2. Elsner P, Agner T. Hand eczema: treatment. J Eur Acad Dermatol Venereol. 2020;34(Suppl. 1):13–21. doi:10.1111/jdv.16062
3. Diepgen TL, Andersen KE, Brandao FM, et al. Hand eczema classification: a cross-sectional, multicentre study of the aetiology and morphology of hand eczema. Br J Dermatol. 2009;160(2):353–358. doi:10.1111/j.1365-2133.2008.08907.x
4. Patruno C, Potestio L, Napolitano M. Clinical phenotypes of adult atopic dermatitis and related therapies. Curr Opin Allergy Clin Immunol. 2022;22(4):242–249. doi:10.1097/ACI.0000000000000837
5. Hendricks AJ, Yosipovitch G, Shi VY. Dupilumab use in dermatologic conditions beyond atopic dermatitis – a systematic review. J Dermatolog Treat. 2021;32(1):19–28. doi:10.1080/09546634.2019.1689227
6. Tancredi V, Licata G, Calabrese G, et al. Is itching in atopic dermatitis still an unmet need? A case series of 556 patients. J Eur Acad Dermatol Venereol. 2022;36(8):e650–e652. doi:10.1111/jdv.18115
7. Muñoz-Bellido FJ, Moreno E, Dávila I. Dupilumab: a review of present indications and uses out of indication. J Investig Allergol Clin Immunol. 2021;32(2):1–36. doi:10.18176/jiaci.0682
8. Napolitano M, Maffei M, Patruno C, et al. Dupilumab effectiveness for the treatment of patients with concomitant atopic dermatitis and chronic rhinosinusitis with nasal polyposis. Dermatol Ther. 2021;34(6):e15120. doi:10.1111/dth.15120
9. Oosterhaven JAF, Romeijn GLE, Schuttelaar MLA. Dupilumab treatment of very severe refractory atopic hand eczema. JAMA Dermatol. 2018;154(8):969–970. doi:10.1001/jamadermatol.2018.2027
10. Nanda S, Nagrani N, MacQuhae F, Nichols A. A case of complete resolution of severe plantar dyshidrotic eczema with dupilumab. J Drugs Dermatol. 2019;18(2):211–212.
11. Halling AS, Zachariae C, Thyssen JP. Severe treatment-resistant acute and recurrent vesicular chronic hand eczema successfully treated with dupilumab. Contact Dermatitis. 2020;83(1):37–38. doi:10.1111/cod.13501
12. Zhu GA, Honari G, Ko JM, Chiou AS, Chen JK. Dupilumab for occupational irritant hand dermatitis in a nonatopic individual: a case report. JAAD Case Rep. 2020;6(4):296–298. doi:10.1016/j.jdcr.2020.02.010
13. Weins AB, Biedermann T, Eyerich K, Moeckel S, Schnopp C. Successful treatment of recalcitrant dyshidrotic eczema with dupilumab in a child. J Dtsch Dermatol Ges. 2019;17(11):1165–1167. doi:10.1111/ddg.13929
14. Gan H, Gao YD. Occupational chronic hand dermatitis in hospital environment successfully treated with dupilumab: a case report. Iran J Allergy Asthma Immunol. 2022;21(4):484–487. doi:10.18502/ijaai.v21i4.10296
15. Matthew J, Zirwas MD. Dupilumab for hand eczema. J Am Acad Dermatol. 2018;79(1):167–169. doi:10.1016/j.jaad.2018.02.073
16. Gall RA, Peters JD, Brinker AJ. Two cases of recalcitrant dyshidrotic eczema treated with dupilumab. J Drugs Dermatol. 2021;20(5):558–559. doi:10.36849/JDD.5273
17. Weston GK, Hooper J, Strober BE. Dupilumab in the treatment of dyshidrosis: a report of two cases. J Drugs Dermatol. 2018;17(3):355–356.
18. Loman L, Diercks GFH, Schuttelaar MLA. Three cases of non-atopic hyperkeratotic hand eczema treated with dupilumab. Contact Dermatitis. 2021;84(2):124–127. doi:10.1111/cod.13693
19. Waldman RA, DeWane ME, Sloan B, Grant-Kels JM, Lu J. Dupilumab for the treatment of dyshidrotic eczema in 15 consecutive patients. J Am Acad Dermatol. 2020;82(5):1251–1252. doi:10.1016/j.jaad.2019.12.053
20. Oosterhaven JAF, Voorberg AN, Romeijn GLE, de Bruin-Weller MS, Schuttelaar MLA. Effect of dupilumab on hand eczema in patients with atopic dermatitis: an observational study. J Dermatol. 2019;46(8):680–685. doi:10.1111/1346-8138.14982
21. Voorberg AN, Romeijn GLE, de Bruin-Weller MS, Schuttelaar MLA. The long-term effect of dupilumab on chronic hand eczema in patients with moderate to severe atopic dermatitis-52 week results from the Dutch BioDay registry. Contact Dermatitis. 2022;87:185–191. doi:10.1111/cod.14104
22. Lee N, Chipalkatti N, Zancanaro P, Kachuk C, Dumont N, Rosmarin D. A retrospective review of dupilumab for hand dermatitis. Dermatology. 2019;235(3):187–188. doi:10.1159/000496481
23. Patruno C, Potestio L, Scalvenzi M, et al. Dupilumab for the treatment of adult atopic dermatitis in special populations. J Dermatolog Treat. 2022;33(7):3028–3033. doi:10.1080/09546634.2022.2102121
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
