Back to Journals » Journal of Inflammation Research » Volume 15
Pulsed-Ultrasound Irradiation Induces the Production of Itaconate and Attenuates Inflammatory Responses in Macrophages
Authors Yamaguchi A, Maeshige N , Ma X, Uemura M , Noguchi H, Matsuda M, Nishimura Y, Hasunuma T , Kondo H, Fujino H
Received 9 February 2022
Accepted for publication 7 April 2022
Published 13 April 2022 Volume 2022:15 Pages 2387—2395
DOI https://doi.org/10.2147/JIR.S361609
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Atomu Yamaguchi,1 Noriaki Maeshige,1 Xiaoqi Ma,1 Mikiko Uemura,1 Hikari Noguchi,1 Mami Matsuda,2 Yuya Nishimura,2 Tomohisa Hasunuma,2,3 Hiroyo Kondo,4 Hidemi Fujino1
1Department of Rehabilitation Science, Kobe University Graduate School of Health Sciences, Kobe, Japan; 2Graduate School of Science, Technology and Innovation, Kobe University, Kobe, Japan; 3Engineering Biology Research Center, Kobe University, Kobe, Japan; 4Department of Food Science and Nutrition, Nagoya Women’s University, Nagoya, Japan
Correspondence: Noriaki Maeshige, Department of Rehabilitation Science, Kobe University Graduate School of Health Sciences, 7-10-2 Tomogaoka, Kobe, 654-0142, Hyogo, Japan, Tel +81 78 796 4582, Email [email protected]
Background: Itaconate is a key metabolite in the innate immune system and exerts strong anti-inflammatory effects in macrophages. For the production of itaconate in macrophages, immune-responsive gene 1 (IRG1) is an imperative enzyme, and activating the IRG1-itaconate pathway is reported to alleviate inflammatory diseases by upregulating nuclear factor-erythroid 2-related factor 2 (NRF2). However, there are very few reports on strategies to increase itaconate production. Ultrasound therapy is a widely used intervention for anti-inflammatory and soft-tissue regeneration purposes. Here we show the effect of ultrasound irradiation on the production of itaconate in macrophages.
Methods: Murine bone marrow-derived macrophages (BMDMs) were exposed to pulsed ultrasound (3.0 W/cm2) for 5 minutes. Three hours after irradiation, the intracellular levels of metabolites and mRNA expression levels of Irg1 and Nrf2 were measured using CE/MS and qPCR, respectively. To evaluate macrophage inflammation status, 3 h after irradiation, the cells were stimulated with 100 ng/mL lipopolysaccharide (LPS) for 1.5 h and the mRNA expression levels of pro-inflammatory factors (Il-1β, Il-6, and Tnf-α) were measured. Student’s t-test, one-way ANOVA and Tukey’s multiple comparison test were used for statistical processing, and the significance level was set to less than 5%.
Results: Ultrasound irradiation significantly increased the intracellular itaconate level and the expression levels of Irg1 and Nrf2 in BMDMs. Upregulation of Il-1β, Il-6, and Tnf-α by LPS was significantly suppressed in BMDMs treated with ultrasound. Ultrasound irradiation did not affect cell viability and apoptosis.
Conclusion: Ultrasound irradiation induces the production of itaconate by upregulating Irg1 expression and attenuates inflammatory responses in macrophages via Nrf2. These results suggest that ultrasound is a potentially useful method to increase itaconate production in macrophages.
Keywords: itaconate, IRG1, NRF2, pulsed-ultrasound, macrophage, inflammation
Introduction
Itaconate is a derivate of the tricarboxylic acid (TCA) cycle and known to have anti-oxidative, anti-tumor, and anti-microbial effects.1 Recently, itaconate has been shown to serve as a critical anti-inflammatory metabolite, suggesting a novel biological effect of itaconate beyond its antimicrobial function.2 In particular, its anti-inflammatory effect on macrophages has been attracting attention.
Macrophages play a pivotal role in the innate immunity and represent the forefront of defense against invading bacterial pathogens.3 Upon detection of lipopolysaccharide (LPS), macrophages are activated in pro-inflammatory direction characterized by overexpression of interleukin-1β (IL-1β), IL-6, and tumor necrosis factor-α (TNF-α).4 In several inflammatory diseases, it is very important to suppress excessive inflammatory responses of macrophages, and itaconate is a potential target for this purpose. In fact, a previous study reported that itaconate significantly reduced the production of pro-inflammatory mediators in LPS-stimulated macrophages and alleviated sepsis in a vivo model.5
Itaconate exerts strong anti-inflammatory effects in macrophages via upregulation of nuclear factor-erythroid 2-related factor 2 (NRF2).2 NRF2 activation in myeloid cells is reported to alleviate inflammation by inhibiting the expression of inflammatory factors such as IL-1β and IL-6.6
Immune-responsive gene 1 (IRG1) has been identified as a gene that codes for immune-responsive gene 1 protein/cis-aconitic acid decarboxylase, an enzyme catalyzing the production of itaconate by decarboxylating cis-aconitate.7 The levels of IRG1 and itaconate in macrophages are known to be increased by infection with several infectious diseases.8 However, there are yet few reports on strategies to increase the production of itaconate in macrophages, and a non-invasive method to increase itaconate production has not been reported.
Ultrasound (US) has seen development not only as a diagnostic imaging modality but as a therapeutic modality in which energy is deposited in tissues to induce various biological effects.9 It is mainly used in the treatment of soft tissue injuries, for the acceleration of wound healing, the resolution of edema, and softening of scar tissue. Ultrasound therapy is advantageous over other treatments for its non-invasive nature and its ability to selectively target organs. Although Zhang et al reported that low-intensity US irradiation have an anti-inflammatory effect on macrophages by suppressing the toll-like receptor 4-nuclear factor κB signaling pathway, they concluded the detailed mechanism is still unclear and requires further studies.10 Furthermore, the physiological mechanisms by which US exerts its effects are diverse and the itaconate-mediated effect of US irradiation on macrophages has not been clarified.
Here we show that US irradiation enhances the production of itaconate by upregulating Irg1 expression in macrophages, exerting an anti-inflammatory effect on LPS-induced inflammation.
Materials and Methods
Cell Culture
To generate bone marrow-derived macrophages (BMDMs), the bone marrow cells from femurs and tibias from 7-week-old male C57BL/6NCrSlc mice were harvested and cultured in Petri dishes with RPMI 1640 supplemented with 10% fetal bovine serum (FBS), 1% Penicillin/Streptomycin, 1% L-Glutamine, and 25% L929 cell supernatant for eight days.11 Differentiated macrophages were harvested and plated in 35 mm tissue culture dishes at a density of 7.5×105/dish with macrophage culture media (RPMI 1640 supplemented with 10% FBS, 1% Penicillin /Streptomycin, 1% L-Glutamine, and 10% L929 cell supernatant).
This study was approved by the Institutional Animal Care and Use Committee and was performed according to the Kobe University Animal Experimentation Regulations.
US Irradiation
US irradiation was conducted by positioning the probe of a medical US device (SZ-100, MINATO Medical Science Co., LTD, Japan) under the bottom of the culture dish. Coupling gel was applied between the probe and the dish, and a sterilized silicone was suspended in the culture media 2 mm above the cell monolayer (Figure 1).12,13 Parameters of US were as follows: intensity of 3.0 W/cm2 in SATP, duration of 5 min, duty cycle of 20%, acoustic frequency of 1 MHz, pulse repetition frequency of 100 Hz, and effective radiation area of 8.0 cm2. The beam nonuniformity ratio (BNR), which is the ratio of the maximum intensity to the average intensity of the US, was 2.4, indicating the safety of the probe. These US parameters refer to the settings clinically used to treat patients. After US irradiation, the temperature of the culture media was measured to monitor it was below 37 degrees Celsius. Control samples were handled identically to US-treated samples to minimize discrepancies in temperature between the control group and the US-treated group.
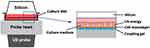 |
Figure 1 In vitro ultrasound (US) irradiation system. US waves are sent out from the probe placed under the culture dish, and surplus energy is absorbed by the silicon. |
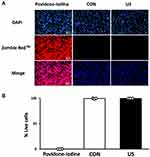
Cell Viability Analysis by Zombie RedTM Immunofluorescence Staining
BMDM viability was assessed 3 h after US irradiation or treatment with 1% povidone-iodine (positive control) using Zombie RedTM. This reagent is an amine-reactive fluorescent dye that is non-permeant to live cells but permeant to cells with a compromised plasma membrane. BMDMs were washed twice with PBS, stained with Zombie Red™ (1:1000) (red) for 15 min, and fixed with 4% paraformaldehyde (PFA) for 30 min. Nuclei were counterstained with DAPI (blue) for 5 min. Immunofluorescent staining patterns were observed with a BX50 fluorescence microscope at ×200 magnification (Olympus, Tokyo, Japan) and recorded with a digital camera (EOS Kiss X4, Canon, Tokyo, Japan), and the numbers of total cells (blue) and dead cells (red) were counted. Then the percentage of live cells to total cells was calculated. Triplicate cell cultures and 10 random fields of each dish were analyzed for each condition tested.
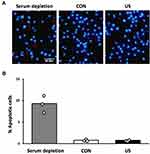
Morphological Detection of Apoptosis
The occurrence of apoptosis in BMDMs was evaluated using DAPI staining:14 24 h after US irradiation, the cells were fixed with 4% PFA for 30 min and stained with DAPI for 5 min. As positive control, BMDMs were cultured in serum-depleted medium for 24 h to induce apoptosis.15 Immunofluorescent staining patterns were observed with a BX50 fluorescence microscope at ×200 magnification (Olympus, Tokyo, Japan) and recorded with a digital camera (EOS Kiss X4, Canon, Tokyo, Japan). Apoptotic cells were identified by chromatin condensation and fragmentation. Triplicate cell cultures were analyzed. Nuclei from 10 random fields of each dish were examined.
Metabolite Analysis
At 3 h after US irradiation, BMDMs were washed with PBS and lysed using 80:20 methanol: water containing 50 μM (+)-10-camphorsulfonic acid, 400 μM L-methionine sulfone, and 400 μM piperazine-1,4-bis(2-ethanesulfonic acid) (PIPES) as internal standards for mass analysis. After incubation at −80°C for 15 min, the cells were scraped and centrifuged at 14,000 g for 5 min at 4°C. The supernatant was collected and filtered using a Millipore 3 kDa cut-off membrane to remove solubilized proteins. After evaporation of the aqueous-layer extracts under vacuum using a FreeZone 2.5 Plus freeze dry system (Labconco, Kansas City, MO), the dried metabolites were dissolved in Milli-Q water. The concentrations of intracellular metabolites were analyzed using a CE/MS system (CE, Agilent G7100; MS, Agilent G6224AA LC/MSD TOF; Agilent Technologies, Palo Alto, CA) controlled by MassHunter Workstation Data Acquisition software (Agilent Technologies), as described previously.16 The relative abundance of each metabolite to the control group was calculated. Triplicate cell cultures were analyzed for each condition tested.
mRNA Analysis
mRNA was isolated using TRIzol RNA Isolation protocol and used to make cDNA using iScriptTM cDNA Synthesis Kit (Bio-Rad). The StepOne™ Real-Time PCR System was used to analyze the samples under the following conditions: 95 °C (3 min), 40 cycles of 95 °C (10 sec), and 60 °C (30 sec). The reaction mixture consisted of 8 μL cDNA, 1.5 μL 10× buffer, 0.3 μL 10 mM dNTPs, 1.5 μL 5 μM primers for each gene used in the study (F+R), 3.58 μL H2O, 0.075 μL Go Taq DNA polymerase, and 0.045 μL 2× SYBR green (Invitrogen). Target genes were Irg1, Nrf2, and pro-inflammatory factors Il-1β, Tnf-α, and Il-6. For the analysis of pro-inflammatory genes, the cells were stimulated with 100 ng/mL LPS for 1.5 h. Relative expression values for target genes were calculated by normalization to the expression of hypoxanthine phosphoribosyltransferase (Hprt). Data were analyzed using the delta/delta CT method.17 Obtained results are expressed as relative values with the control group or LPS-unstimulated cells. Sequences for qRT-PCR primers are shown in Supplementary Material 1. Quadruplicate cell cultures and technical duplicates for each sample were analyzed.
Statistical Analysis
All values are expressed as mean and individual data points. Statistical analysis was conducted using Statistical 4 (OMS, Tokyo, Japan). For two-group comparison, Student’s t-test was used and for multiple comparisons, ANOVA (Tukey’s multiple comparison test as post-hoc) was used. Power analysis using G Power software18 was conducted to determine sample size for each experiment to provide a power of at least 0.8 at a significance level of 0.05 (α = 0.05, β = 0.2). The results of statistical power analysis are presented in Supplementary Material 2.
Results
US Irradiation Has No Cytotoxicity on BMDMs
To examine the cytotoxicity of US irradiation to BMDMs, the cell viability of BMDMs 3 h after US irradiation was assessed by Zombie RedTM immunofluorescence staining. As shown in Figure 2, US irradiation at 3.0 W/cm2 did not cause cell damage while the cells treated with 1% povidone-iodine showed a significant decrease in viability.
US Does Not Affect BMDM Apoptosis
To assess the effect of US on BMDM apoptosis, nuclear morphological changes were observed using DAPI staining 24 h after US irradiation. As shown in Figure 3, US irradiation at 3.0 W/cm2 did not induce apoptosis in BMDMs while serum depletion for 24 h significantly induced apoptosis.
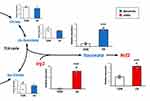
US Irradiation Induces the Production of Itaconate by Upregulating Irg1 Expression
To evaluate the effect of US irradiation on the production of itaconate in BMDMs, intracellular levels of the metabolites were quantified 3 h after US irradiation. As the factors related to the itaconate pathway, the mRNA expression levels of Irg1 and Nrf2 were measured. While US irradiation did not increase intracellular levels of citrate, cis-aconitate, and isocitrate, itaconate was significantly upregulated 3 h after US irradiation. Furthermore, US irradiation significantly increased the mRNA expression of Irg1 and Nrf2 (Figure 4).
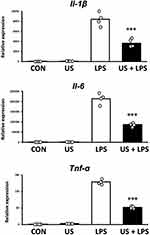
US Irradiation Attenuates LPS-Induced Inflammatory Responses in BMDMs
Three hours after US irradiation, BMDMs were treated with 100 ng/mL LPS for 1.5 h as an inflammation model. To assess the effect of US irradiation on macrophage inflammation, mRNA expression levels of pro-inflammatory Il-1β, Il-6, and Tnf-α were measured. As shown in Figure 5, LPS significantly upregulated the expression levels of Il-1β, Il-6, and Tnf-α in BMDMs and US irradiation significantly prevented the upregulation of those factors.
Discussion
In this study, we found that US irradiation increased the level of itaconate and the expression levels of Irg1 and Nrf2 in macrophages, and prevented LPS-induced inflammatory response. Also, US irradiation did not induce apoptosis and a reduction in cell viability.
Itaconate has been reported to alleviate various kinds of diseases such as sepsis,5 abdominal aortic aneurysm,19 pulmonary fibrosis,20 and COVID-1921 by suppressing inflammatory responses in macrophages. Furthermore, it is reported that itaconate inhibits the inflammatory responses of macrophages in a concentration-dependent manner.22 Therefore, increasing the production of itaconate in macrophages is of great significance in the treatment of various diseases.
Overexpression of IL-1β, IL-6, and TNF-α contributes to the development and progression of a range of diseases. Suppression of these pro-inflammatory factors has been reported to alleviate various inflammatory diseases and trials examining blockade of these factors have also been conducted.23 Hence, the regulation of pro-inflammatory factors by US irradiation suggests the possibility of a new therapeutic strategy for inflammatory organs using US.
In the present study, US irradiation specifically increased the expression level of Irg1 and the level of itaconate without upregulating the levels of citrate, cis-aconitate, and isocitrate in macrophages. As IRG1 is known to catalyze the production of itaconate from cis-aconitate,7 it is assumed that US increased the production of itaconate in macrophages via upregulation of Irg1. Stimulation with LPS or TNF-α has been reported to activate the IRG1-itaconate pathway in macrophages,24 but this method strongly induces inflammation and is not clinically applicable. Meanwhile, in this study, US irradiation activated the IRG1-itaconate pathway without causing inflammatory responses in macrophages. This point is a great advantage for the purpose of suppressing macrophage inflammation by targeting itaconate.
A previous study has reported that the activation of NRF2 is required for the anti-inflammatory action of itaconate.2 NRF2 has been found to directly bind to the promoters of inflammatory factors such as IL-1β and IL-6 and inhibit the expression of these factors by blocking the activation of RNA polymerase II.25 Therefore, since the Nrf2 expression level was upregulated in this study, it is assumed that the activation of Nrf2 following the US-induced increase in itaconate led to the suppression of the inflammatory response of macrophages. The regulatory system of NRF2 has been recognized as an attractive drug target.26 In human clinical trials, Tecfidera (dimethyl fumarate), an NRF2 inducer, was approved for the treatment of multiple sclerosis based on its anti-inflammatory function.27 Thus, activating NRF2 non-invasively by US has great significance.
While this study showed that US irradiation increased itaconate production via increasing the expression of Irg1 in macrophages, the mechanism by which US increased Irg1 expression is not clarified yet. A previous study elucidated that chelation of extracellular calcium inhibited Irg1 increase induced by LPS.28 Furthermore, Meijering et al reported that US promotes calcium influx into cells.29 Thus, US-induced increase in calcium influx into the cells is likely to be involved in the activation of the Irg1-itaconate pathway by US, but further investigation using methods such as RNA-sequencing is required to elucidate more detailed mechanisms. In addition, a verification using human-derived cells is expected to make this method feasible for clinical application.
Conclusion
We demonstrated that US irradiation increases the production of itaconate via elevation of Irg1 expression and exerts anti-inflammatory effects by upregulating Nrf2 expression in macrophages. These findings suggest that US has a potential capacity to be used as a method for suppression of macrophage inflammation targeting itaconate.
Acknowledgments
This study was supported by JSPS KAKENHI [grant number 21H03852] and the grant of Hyogo Science and Technology Association 2021. We are grateful to Dr. P Kent Langston (Department of Immunology, Harvard Medical School and Evergrande Center for Immunologic Diseases, Harvard Medical School, Boston, MA, USA) for providing insightful advice.
Disclosure
The authors declare no competing interests in this work.
References
1. Sano M, Tanaka T, Ohara H, et al. Itaconic acid derivatives: structure, function, biosynthesis, and perspectives. Appl Microbiol Biotechnol. 2020;104(21):9041–9051. doi:10.1007/s00253-020-10908-1
2. Yu XH, Zhang DW, Zheng XL, et al. Itaconate: an emerging determinant of inflammation in activated macrophages. Immunol Cell Biol. 2019;97(2):134–141. doi:10.1111/imcb.12218
3. Weiss G, Schaible UE. Macrophage defense mechanisms against intracellular bacteria. Immunol Rev. 2015;264(1):182–203. doi:10.1111/imr.12266
4. Orecchioni M, Ghosheh Y, Pramod AB, et al. Corrigendum: macrophage polarization: different gene signatures in M1(LPS+) vs. classically and M2(LPS-) vs. alternatively activated macrophages. Front Immunol. 2020;11:234. doi:10.3389/fimmu.2020.00234
5. Zhang S, Jiao Y, Li C, et al. Dimethyl itaconate alleviates the inflammatory responses of macrophages in sepsis. Inflammation. 2021;44(2):549–557. doi:10.1007/s10753-020-01352-4
6. Kong X, Thimmulappa R, Craciun F, et al. Enhancing Nrf2 pathway by disruption of Keap1 in myeloid leukocytes protects against sepsis. Am J Respir Crit Care Med. 2011;184(8):928–938. doi:10.1164/rccm.201102-0271OC
7. Tallam A, Perumal TM, Antony PM, et al. Gene regulatory network inference of immunoresponsive gene 1 (IRG1) identifies interferon regulatory factor 1 (IRF1) as its transcriptional regulator in mammalian macrophages. PLoS One. 2016;11(2):e0149050. doi:10.1371/journal.pone.0149050
8. Michelucci A, Cordes T, Ghelfi J, et al. Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production. Proc Natl Acad Sci USA. 2013;110(19):7820–7825. doi:10.1073/pnas.1218599110
9. Miller DL, Smith NB, Bailey MR, et al. Bioeffects Committee of the American Institute of Ultrasound in Medicine. Overview of therapeutic ultrasound applications and safety considerations. J Ultrasound Med. 2012;31(4):623–634. doi:10.7863/jum.2012.31.4.623
10. Zhang X, Hu B, Sun J, et al. Inhibitory effect of low-intensity pulsed ultrasound on the expression of lipopolysaccharide-induced inflammatory factors in u937 cells. J Ultrasound Med. 2017;36(12):2419–2429. doi:10.1002/jum.14239
11. Langston PK, Nambu A, Jung J, et al. Glycerol phosphate shuttle enzyme GPD2 regulates macrophage inflammatory responses. Nat Immunol. 2019;20(9):1186–1195. doi:10.1038/s41590-019-0453-7
12. Maeshige N, Langston PK, Yuan ZM, et al. High-intensity ultrasound irradiation promotes the release of extracellular vesicles from C2C12 myotubes. Ultrasonics. 2021;110:106243. doi:10.1016/j.ultras.2020.106243
13. Ma X, Yamaguchi A, Maeshige N, et al. Enhancement of astaxanthin incorporation by pulsed high-intensity ultrasound in LPS-stimulated macrophages. J Med Ultrason. 2022. doi:10.1007/s10396-022-01189-4
14. Torii K, Maeshige N, Aoyama-Ishikawa M, et al. Combination therapy with butyrate and docosahexaenoic acid for keloid fibrogenesis: an in vitro study. An Bras Dermatol. 2017;92(2):184–190. doi:10.1590/abd1806-4841.20176198
15. Xu JX, Morii E, Liu Y, et al. High tolerance to apoptotic stimuli induced by serum depletion and ceramide in side-population cells: high expression of CD55 as a novel character for side-population. Exp Cell Res. 2007;313(9):1877–1885. doi:10.1016/j.yexcr.2007.03.006
16. Kato H, Izumi Y, Hasunuma T, et al. Widely targeted metabolic profiling analysis of yeast central metabolites. J Biosci Bioeng. 2012;113(5):665–673. doi:10.1016/j.jbiosc.2011.12.013
17. Rao X, Huang X, Zhou Z, et al. An improvement of the 2ˆ(-delta delta CT) method for quantitative real-time polymerase chain reaction data analysis. Biostat Bioinforma Biomath. 2013;3(3):71–85.
18. Kang H. Sample size determination and power analysis using the G*Power software. J Educ Eval Health Prof. 2021;18:17. doi:10.3352/jeehp.2021.18.17
19. Song H, Xu T, Feng X, et al. Itaconate prevents abdominal aortic aneurysm formation through inhibiting inflammation via activation of Nrf2. EBioMedicine. 2020;57:102832. doi:10.1016/j.ebiom.2020.102832
20. Ogger PP, Albers GJ, Hewitt RJ, et al. Itaconate controls the severity of pulmonary fibrosis. Sci Immunol. 2020;5(52):eabc1884. doi:10.1126/sciimmunol.abc1884
21. Olagnier D, Farahani E, Thyrsted J, et al. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat Commun. 2020;11(1):4938. doi:10.1038/s41467-020-18764-3
22. Lampropoulou V, Sergushichev A, Bambouskova M, et al. Itaconate links inhibition of succinate dehydrogenase with macrophage metabolic remodeling and regulation of inflammation. Cell Metab. 2016;24(1):158–166. doi:10.1016/j.cmet.2016.06.004
23. Möller B, Villiger PM. Inhibition of IL-1, IL-6, and TNF-alpha in immune-mediated inflammatory diseases. Springer Semin Immunopathol. 2006;27(4):391–408. doi:10.1007/s00281-006-0012-9
24. Strelko CL, Lu W, Dufort FJ, et al. Itaconic acid is a mammalian metabolite induced during macrophage activation. J Am Chem Soc. 2011;133(41):16386–16389. doi:10.1021/ja2070889
25. Kobayashi EH, Suzuki T, Funayama R, et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat Commun. 2016;7:11624. doi:10.1038/ncomms11624
26. Suzuki T, Motohashi H, Yamamoto M. Toward clinical application of the Keap1-Nrf2 pathway. Trends Pharmacol Sci. 2013;34(6):340–346. doi:10.1016/j.tips.2013.04.005
27. Gold R, Kappos L, Arnold DL, et al. Placebo-controlled Phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med. 2012;367(12):1098–1107. doi:10.1056/NEJMoa1114287
28. Lee CG, Jenkins NA, Gilbert DJ, et al. Cloning and analysis of gene regulation of a novel LPS-inducible cDNA. Immunogenetics. 1995;41(5):263–270. doi:10.1007/BF00172150
29. Meijering BD, Juffermans LJ, van Wamel A, et al. Ultrasound and microbubble-targeted delivery of macromolecules is regulated by induction of endocytosis and pore formation. Circ Res. 2009;104(5):679–687. doi:10.1161/CIRCRESAHA.108.183806
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.