Back to Journals » International Journal of Nanomedicine » Volume 14
Proliposomes for oral delivery of total biflavonoids extract from Selaginella doederleinii: formulation development, optimization, and in vitro–in vivo characterization
Authors Chen B , Wang X, Lin D , Xu D, Li S , Huang J, Weng S , Lin Z, Zheng Y , Yao H, Lin X
Received 6 May 2019
Accepted for publication 27 July 2019
Published 20 August 2019 Volume 2019:14 Pages 6691—6706
DOI https://doi.org/10.2147/IJN.S214686
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Bing Chen,*,1–3 Xuewen Wang,*,2,3 Dan Lin,2,3 Dafen Xu,2,3 Shaoguang Li,2,3 Jianyong Huang,4 Shaohuang Weng,2,3 Zhen Lin,2,3 Yanjie Zheng,2,3 Hong Yao,2,3 Xinhua Lin2,3
1Nano Medical Technology Research Institute, Fujian Medical University, Fuzhou, Fujian, People’s Republic of China; 2Higher Educational Key Laboratory for Nano Biomedical Technology of Fujian Province, Fujian Medical University, Fuzhou, Fujian, People’s Republic of China; 3Department of Pharmaceutical Analysis, School of Pharmacy, Fujian Medical University, Fuzhou, Fujian, People’s Republic of China; 4Department of Pharmaceutical, Fujian Medical University Union Hospital, Fuzhou, Fujian, People’s Republic of China
Correspondence: Xinhua Lin; Hong Yao
Department of Pharmaceutical Analysis, Fujian Medical University, No.1 Xueyuan Road, University Town, Fuzhou 350122, People’s Republic of China
Email [email protected]; [email protected]
*These authors contributed equally to this work
Purpose: Amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone are five major active ingredients in the total biflavonoids extract from Selaginella doederleinii (TBESD) with favorable anticancer properties. However, the natural-derived potent antitumor agent of TBESD is undesirable due to its poor solubility. The present study was to develop and optimize a proliposomal formulation of TBESD (P-TBESD) to improve its solubility, oral bioavailability and efficacy.
Materials and methods: P-TBESD containing a bile salt, a protective hydrophilic isomalto-oligosaccharides (IMOs) coating, were successfully prepared by thin film dispersion-sonication method. The physicochemical and pharmacokinetic properties of P-TBESD were characterized, and the antitumor effect was evaluated using the HT-29 xenograft-bearing mice models in rats.
Results: Compared with TBESD, the relative bioavailability of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone from P-TBESD were 669%, 523%, 761%, 955% and 191%, respectively. The results of pharmacodynamics demonstrated that both TBESD and P-TBESD groups afforded antitumor effect without systemic toxicity, and the antitumor effect of P-TBESD was significantly superior to that of raw TBESD, based on the tumor growth inhibition and histopathological examination.
Conclusion: Hence, IMOs-modified proliposomes have promising potential for TBESD solving the problem of its poor solubility and oral bioavailability, which can serve as a practical oral preparation for TBESD in the future cancer therapy.
Keywords: Selaginella doederleinii, proliposomes, sodium deoxycholate, oral bioavailability, antitumor
Introduction
Selaginella doederleinii Hieron, a medicinal herb widely distributed in southern China, has been used as a folk medicine for health promotion and treatment of various cancers since ancient times.1 S. doederleinii has been reported to have many medicinal properties, including anti-oxidation,2 anti-senescence,3 anti-inflammation,4 anti-hyperglycemic,5 anti-diabetic,6 anti-virus7 and anti-cancer activities.8,9 Phytochemical studies disclosed that the chemical components of S. doederleinii were mainly consisted of biflavonoids, including amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone, etc.10–12 Our current investigations have shown that the total biflavonoids extract of S. doederleinii (TBESD) could induce cancer cells apoptosis, significantly inhibit the tumor growth and enhance the antitumor immune response, and did not show apparent oral acute toxicity in vivo.13 Meanwhile, it has also been reported that the biflavonoid from S. doederleinii, delicaflavone, could trigger the autophagic death of lung cancer cells and exert powerful anti-lung cancer effect in vivo.14 All these suggested that TBESD possessed favorable anti-tumor potential and low toxicity, and deserved to be further developed as an anti-cancer candidate. However, these biflavonoids of TBESD with low aqueous solubility, low gastrointestinal permeability and high hepatic metabolism cause their poor oral absorption and bioavailability.15,16 The prominent anti-tumor effect of TBESD is therefore limited due to the above-mentioned factors.
Recently, some nanotechnology-based delivery systems have been developed for improving the oral bioavailability, such as liposomes,17 solid lipid nanoparticles (SLN),18 micelles,19 self-nanoemulsifying drug delivery system (SNEDDS)20 and porous absorbent particles.21 Among these drug delivery systems, liposomes are more preferable delivery system for herbal bioactives because of the ability to encapsulate both hydrophilic and hydrophobic components, absorption-enhancing capability, low toxicity, excellent biodegradability and biocompatibility.22–24 In addition, liposomes incorporated of phospholipid and cholesterol form a lipid bilayers vesicles, which can enhance the oral absorption by adhering to the mucosal membrane.25 However, despite liposomes enable an antitumor agent to improve solubility and bioavailability, inherent limitation such as aggregation, drug leakage, sedimentation, phospholipid susceptibility to oxidation and hydrolysis still exist.26,27 In order to overcome these issues, a proliposome formulation, dry and free-flowing granular powder, which can disperse to form liposomes suspension via addition of water, is adopted in the current study.28
The bioflavonoids are believed to be key active ingredients that are responsible for the pharmacological action of TBESD.12,15 It is mainly composed of five ingredients, including amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone with proportions of 10.38%, 3.75%, 4.44%, 5.35% and 3.52%, respectively. However, TBESD may contain many uncontrollable factors such as endotoxin and pyrogen except five major bioflavonoids. Among versatile administration route, oral delivery has been the most preferred route of drug administration for its safety, convenience and good patient compliance. It is worth noting that there are several challenges for oral delivery liposomes, including instability under the harsh conditions of the gastrointestinal tract (GIT) and poor permeability across cell membranes. Several studies have been reported that the insertion of bile salts into phospholipid bilayer could improve the stability of drugs in GI environment by offsets the destructive effects of outside bile salts.29 Besides, liposomes containing bile salts can promote the absorption of poorly soluble drugs.30 Polysaccharides are functional coating materials to prevent liposomes from the harsh GI environment and well-known cryoprotective agents to stabilize liposomes during freeze-drying processes.31 Isomalto-oligosaccharides (IMOs), a naturally derived mixture polysaccharides and widely applicable as food ingredients, possess synergistic antitumor effects and hydrolysis-resistant capability.32,33 Fairly few studies have been reported that IMOs-coating liposomes improve stability and the oral absorption of drugs in the GIT.34 Polysaccharide derivative of IMOs could be used as a protective agent of liposomes. IMOs can form the corona outer layer to protect the liposomes from hydrolytic degradation and counteract the aggregation and adhesion of liposomes. In the current study, we attempt to develop and evaluate the IMOs-modified proliposomal formulation of TBESD. The solid proliposome preparations not only afford higher chemical as well as physical stability during storage, but also remain improved solubility and bioavailability characteristics of the traditional liposome preparations.
In this study, a dual strategy of protective bile salt-containing and IMOs-coating was applied to improve the solubility and bioavailability of TBESD. Finally, the oral IMOs-modified TBESD proliposomes containing bile salt were successfully prepared and characterized. Furthermore, to evaluate the oral bioavailability and in vivo drug effect, the pharmacokinetic properties and antitumor effect on HT-29 xenograft-bearing mice models were examined.
Materials and methods
Material
The total biflavonoids extract of S. doederleinii (TBESD) was prepared according to our previously described procedure and its contents of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone in TBESD were 103.82 mg/g, 37.52 mg/g, 44.40 mg/g, 53.36 mg/g and 35.12 mg/g, respectively.8 Chrysin (purity ≧98%, internal standard, IS) was provided by Shanghai Winherb Medical Technology Co., Ltd. (Shanghai, China). Cholesterol, soy lecithin and sodium deoxycholate (NaDC) were purchased from Aladdin (Shanghai, China). Isomalto-oligosaccharides (IMOs) were acquired from VitaFiberTM (London, England). Acetonitrile and methanol of HPLC-grade were obtained from Merck (Darmstadt, Germany). Acetic acid of HPLC-grade was purchased from Aladdin (Shanghai, China). Dichloromethane and ethanol of analytical-grade were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). All other solvents used in this study were of HPLC-grade, and chemicals were of analytical grade. The reference standards of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone (purity ≧98%) were isolated from S. doederleinii and their structures were fully elucidated by UV, MS, 1H-NMR and 13C-NMR and confirmed by comparison with the literatures.10
Cell lines and culture
Human HT-29 colon cancer cells were obtained from the Cell Resource Center of Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (Shanghai, China) and maintained in a humidified incubator containing 5% CO2 at 37°C. HT-29 cells were cultured in RPMI 1640 medium (Hyclone, Utah, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, New York, USA) and 1% penicillin-streptomycin. Cells were grown in plastic tissue culture dishes and harvested using 0.25% trypsin-EDTA (Gibco, New York, USA) and rinsed with PBS (Hyclone, Utah, USA). 3-(4,5-dimethylthiazol-2yl)-2,5,diphenyltetrazolium bromide (MTT reagent) was from Sigma Aldrich (St Louis, MO, USA). Dimethylsulfoxide (DMSO) was purchased from Sinopharm Group Co. Ltd. (Shanghai, China). All experiments were performed on logarithmically grown cells.
Animals
Male Sprague-Dawley rats (n=12, 250±20 g) were supplied by the Laboratory Animal Center of Fujian Medical University. Six-week-old BALB/c male nude mice (n=30, 20±2 g) were purchased from National Rodent Laboratory Animal Resource (Shanghai, China) and maintained on the premises under standard animal house conditions. All the rats were housed with an ambient temperature of 25±2°C, relative humidity of 55±5%, under 12 hrs light/dark cycles with free access to water and food for one week before the experiment.
All animal experiments were approved by the Institutional Animal Care and Use Committee of Fujian Medical University. Animal welfare and experimental procedures were performed strictly in accordance with the Guide for the Animals Care and Ethics Committee of Fujian Medical University.
Preparation of P-TBESD
The technique of thin film dispersion-sonication followed by lyophilization was used to prepare TBESD proliposomes (P-TBESD).35,36 To briefly summarize the process, the prescribed amount of TBESD, cholesterol and soy lecithin were dissolved in a certain amount of methylene chloride and then evaporated at 35°C under vacuum to obtain a thin lipid film. A certain amount of sodium deoxycholate (NaDC) was dissolved in an appropriate amount of phosphate-buffered saline (PBS, pH 7.4) and heated 50±1°C to form an aqueous phase. The aqueous phase was added to hydrate for 30 mins at 50±1°C to obtain the liposome suspension. Subsequently, the suspension was ultrasonicated at 300 W, 5 mins in an ice-bath by an ultrasonic cell pulverizer (Ningbo Biological Technology Co, Ltd, Ningbo, China). Thereafter, the liposome suspension was lyophilized using IMOs as cryoprotectant (ratio of IMOs to lipid, 2:1, w/w) and reconstituted by deionized water before use.37 The proliposomes powder was kept at 4°C until used.
Optimization of P-TBESD formulation
Several elements were considered during the preparation process of TBESD liposome suspension. According to preliminary experiments, the main variables that affected the drug entrapment efficiency (EE) in the desired formulation were the concentration of soybean phospholipid (mg/mL), soybean phospholipid/drug ratio (w/w), soybean phospholipid/cholesterol ratio (w/w) and soybean phospholipid/sodium deoxycholate ratio (w/w) (labeled as A, B, C and D, respectively, in Table 1). In this study, a L9 (34) orthogonal test design was employed to further optimize the composition of P-TBESD. The orthogonal design scheme was composed of 9 experimental groups for examining the effects of four elements, each at three levels, which is very effective and economical.38 The P-TBESD powers were prepared by a freeze-drying method. A set of cryoprotectants was screened as a protective carrier during freeze-drying process. The TBESD liposomes suspension was lyophilized using a vacuum freeze dryer to obtain proliposome powders after adding various sugars (lactose, xylitol, mannitol and IMOs). The desirability function of EE of liposomes was at the maximum level, which was selected to optimize the P-TBESD formulation. In the current research, all the data were determined as the mean from triplicate samples.
 |
Table 1 The factors and levels of orthogonal test |
Characterization of P-TBESD
Morphology
The surface morphology and shape of the optimized P-TBESD was evaluated by transmission electron microscopy (TEM) (FEI, Oregon, USA). The liposomes were reconstituted by hydrating proliposome powders with 10 times of deionized water to obtain the appropriate liposomal concentration. The sample was dropped on a copper grid coated with carbon film with staining for 1 min and air-dried at room temperature. Thereafter, a drop of 0.2% phosphotungstic acid was added and stained. Finally, the air-dried samples were observed under TEM.
Particle size and zeta potential
The mean particle size, polydispersity index (PDI) and zeta potential of the reconstituted liposomes were measured by the dynamic light scattering (DLS) technique using NanoPlus zeta/nano particle analyzer (Micromeritics, Atlanta, USA). The samples were diluted to a suitable liposomal concentration with deionized water, and analysis was performed at a temperature of 25±1°C and a scattering angle of 90°. All tests were carried out in triplicate.
Determination of drug entrapment efficiency and drug loading
Since the contents of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone in S. doederleinii biflavonoids were relatively high (﹥1 μg/mL) in the formulation, an HPLC method was employed for their analysis. A Shimadzu LC-20AD HPLC system (Shimadzu Corporation, Kyoto, Japan) was used to determine biflavonoids. An Ultimate® XB-C18 (100×4.6 mm, 3.5 μm; Welch Materials, Inc., Ellicott, MD, USA) was used for the separation. The mobile phase composed of water (containing 0.05% acetic acid, A) and acetonitrile (B) was used at a flow rate of 1 mL/min while the column temperature was maintained at 30°C. The gradient elution program was set as follows: 0–4 mins, 30–40% B; 4–23 mins, 40–42% B; 23–25 mins, 42–46% B. The detection wavelength and the injection volume were set at 270 nm and 10 μL, respectively.
Entrapment efficiency (EE) is a key parameter to evaluate P-TBESD. The EE of the reconstituted liposomes, defined as the percentage of biflavonoids incorporated into the liposomal formulation, was determined by ultracentrifugation and selected to represent P-TBESD.39 A certain amount of P-TBESD was reconstituted in 1 mL of ultrapure water. The resultant suspension was vortexed for 2 mins and then centrifuged at 20,000 rpm for 30 mins at 4°C to separate unencapsulated biflavonoids from liposomes. The supernatant was mixed with a triple volume of methanol via sonication and centrifuged for 5 mins at 5000 rpm; the total amount of unencapsulated biflavonoids in the dilute solution was determined by high-performance liquid chromatography (HPLC) as Wf. Another resultant suspension was vortexed for 2 mins and then disrupted in triple the volume of methanol via sonication and centrifuged for 5 mins at 5000 rpm; the total amount of biflavonoids in the dilute solution was determined by HPLC as Wt. The weight of P-TBESD was documented by analytical balances (Sartorius, Gttingen, Germany) as Wp. The experiments were carried out in triplicate, and the EE and DL of total S. doederleinii biflavonoids were calculated using the following equations:
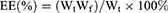
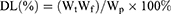
where Wf, Wt and Wp denote the amount of unencapsulated biflavonoids, the total amount of biflavonoids and the weight of P-TBESD, respectively.
Cytotoxicity assay
In vitro cytotoxicity of TBESD, P-TBESD and blank proliposomes were evaluated by MTT assay. Briefly, HT-29 cells were seeded in 96-well plates at a density of 4000 cells/well and permitted to incubate overnight. All the samples were added into each well at the designated concentration (1, 10, 25, 50, 75, 100 μg/mL) for equivalent TBESD. After 48 hrs, the cells were washed with PBS (pH 7.4) and 20 μL of MTT solution (5 mg/mL in PBS) was added to each well of the 96-well plates, and the plates were incubated for 4 hrs at 37°C. Before analysis, the medium in the plates were removed and replaced with 150 μL DMSO to dissolve the formazan crystals. The maximum absorbance was set at 490 nm, and the optical density (OD) of samples was obtained using a microplate reader (Multiskan Go, Thermo, Fisher Scientific, USA). The inhibitory rate was calculated as Inhibitory rate (%) = (1-ODsample/ODcontrol) ×100%, where ODsample and ODcontrol represent the OD of cells treated with the sample group and control group, respectively. All samples were repeated five times independently.
In vivo pharmacokinetic study
HPLC-ESI-MSn analyses
For quantitative analysis plasma concentration of biflavonoids (amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone), plasma sample was analyzed on HPLC-ESI-MSn as previously described using a Shimadzu LCMS-8040 triple quadruple mass spectrometer (Shimadzu, Kyoto, Japan) equipped with a Shimadzu LC-20AD HPLC system. Chromatographic separation was performed using a reversed-phase HPLC column (Ultimate® XB-C18, 100×2.1 mm, 3.5 μm; Welch Materials, Inc., Ellicott, MD, USA) protected by a guard column (Ultimate® XB-C18, 10×2.1 mm, 3.5 μm; Welch Materials, Inc., Ellicott, MD, USA). Column oven temperature was 30ºC. The mobile phase was composed of water (0.5% acetic acid, A) and acetonitrile (B) at a flow rate of 0.2 mL/min. The gradient elution program is described as follows: 0–1 min, 40–44% B; 1–14 mins, 44–49.5% B; 14–15 mins, 49.5–95% B; 15–17 mins, 95% B; and then 40% B for equilibration.
The mass spectrometer was equipped with an electrospray ionization (ESI) source and operated in the negative ion. Multiple reaction monitoring (MRM) transitions for amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin, chrysin (IS) and delicaflavone were m/z 537.08 → 375.00, m/z 537.08 → 309.10, m/z 539.10 → 387.15, m/z 541.11 → 237.09, m/z 253.24 → 143.00 and m/z 537.08 → 255.00, respectively. The collision energy of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin, chrysin (IS) and delicaflavone were 35 V, 40 V, 30 V, 45 V, 28 V and 52 V, respectively. System control and data analysis were carried out with LabSolutions LCMS Ver. 5.5 software.
Pharmacokinetic study
In this study, 12 SD rats were randomly partitioned 2 groups (n=6). Before drug administration, the rats were fasted with free access to water for 12 hrs. The formulations of the suspensions of TBESD and P-TBESD were orally administered at a single dose of 200 mg/kg to rats in groups A and B, respectively. Blood samples (200 μL) were collected into heparinized centrifuge tubes from the tail vein at 0, 3, 8, 15, 30, 45, 60, 90, 120, 240, 360, 480, 720 and 1440 mins post-dosing. After centrifugation at 3000 rpm for 10 mins, the supernatant of plasma samples was obtained and stored at −80°C until analysis.
Plasma samples were prepared by a protein precipitation method and plasma concentration of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone were determined by LC-ESI-MS/MS according to a previously described method.15
The pharmacokinetic parameters for TBESD and P-TBESD including maximum concentration (Cmax), time of the maximum concentrations (Tmax), area under the concentration-time curve (AUC), volume of distribution (Vd), elimination half-time (T1/2), clearance (CL) and mean residence time (MRT) were calculated by noncompartment model analysis using the DAS pharmacokinetic software Version 3.0 (Bontz Inc., Beijing, China).
In vivo antitumor efficacy study
A xenograft-bearing mice model of HT-29 tumor was established by our research group. Briefly, HT-29 colon cancer cells (1.0×107 cells/mice) were inoculated subcutaneously into the right front armpit area of BALB/c male nude mice. The tumor volume (V) was calculated as V = W2×L/2, where the width (W) and length (L) were measured by electronic caliper. The drug was administrated when the xenograft tumors had developed to an average size of 50–70 mm3.
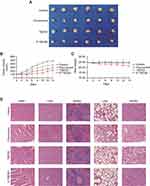
The in vivo antitumor test of TBESD and P-TBESD were evaluated according to previous experiments with slight modification.12,13,40 When the xenograft tumors had developed to an average size of 50–70 mm3, the mice were divided into control, positive, TBESD group (300 mg/kg), P-TBESD group (300 mg/kg total biflavonoids-equivalent for TBESD) (n=6) in a way that minimized weight and tumor size differences between the groups. Control or treatment groups were administered p. o. (0.2 mL/10 g) every day for 14 days. The positive group was administered i. v. 25 mg/kg Fluorouracil (0.05 mL/10 g) once every three days via tail-vein until sacrifice. The tumor size and body weight of mice were measured and recorded every 2 days. At day 14 after treatment, the mice were euthanized, and tumors were collected, weighted and photographed. Tissues (heart, liver, spleen, lung and kidney) were collected and fixed in 4% formaldehyde overnight for hematoxylin and eosin (H&E) staining.
Statistical analysis
All the data were presented as Mean ± SD. Statistical comparisons were performed by using Student’s t-test to determine statistical significance. In all cases, *P﹤0.05 was considered to be statistically significant and **P﹤0.01 was considered to be highly significant difference.
Results and discussion
Preparation of P-TBESD
TBESD proliposomes (P-TBESD) were prepared successfully by the film dispersion-sonication method, followed by lyophilization. The method of thin film dispersion was selected due to advantages of simplicity, high efficiency and material saving.41
In this study, stable proliposomes with high entrapment efficiency and drug loading are manufactured by mixing soybean phospholipid and cholesterol which are complex lipid materials. In addition, they have function that regulated the fluidity of the lipid bilayer and reduced the permeability of the liposomes membrane. In order to change the phase transition behavior of liposomes in the gastrointestinal tract, NaDC was used as a surfactant in the experiment.42 In the process of freeze-drying, sugars are well-known cryoprotective agents, which can improve the resistance of liposomes toward lyophilization stress and prevent agglomeration of proliposomes during freeze-drying cycles.43 A set of cryoprotectants was screened among different sugar, as shown in Table 2. IMOs were chosen as the cryoprotectants (ratio of IMOs to lipid, 2:1, w/w), because the values of mean diameter and zeta potentials for before and after lyophilization were not significantly different. Therefore, film dispersion-sonication followed by lyophilization is a simple, reliable and feasible method to prepare P-TBESD.
 |
Table 2 Effects of different cryoprotectants on the characteristics and EE of P-TBESD (mean ± SD, n=3) |
Optimization of P-TBESD
HPLC-DAD analyses
The main five biflavonoids in the TBESD exhibited favorable separation and lower background noise in the chromatographic method (Figure 1). The linearity for delicaflavone, 3’,3’’’-binaringenin, 2’’,3’’-dihydro-3’,3’’’-biapigenin and robustaflavone was obtained over a range of 0.5–100 μg/mL using a 1/X2 weighting. Linearity for amentoflavone was obtained over a range of 1–200 μg/mL also using a 1/X2 weighting. The calibration curves of the five biflavonoids showed good linearity over their concentration ranges with coefficient of determination r2≥0.999 (Table S1). The repeatabilities of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone were in the range of 1.26–2.91%. The precision of the five biflavonoids ranged from 0.72% to 2.51% (Table S2). The results proved that the method has good accuracy and precision.
Optimization of proliposomes formulation
Entrapment efficiency (EE) and drug loading (DL) are the key parameters for the assessment of the quality of proliposomes as a drug carrier.44 Several elements were influenced by EE of liposomes in the preparation process. In this study, the preparation conditions of liposomes that the concentration of soybean phospholipid (mg/mL), soybean phospholipid/drug ratio (w/w), soybean phospholipid/cholesterol ratio (w/w) and soybean phospholipid/sodium deoxycholate ratio (w/w) were optimized in terms of EE by the orthogonal experiment design of L9 (34). The orthogonal experiment design, which is a systematic, statistical method, is a rapid and reliable technique to optimize the preparation conditions of liposomes.45,46
In order to obtain stable and high-quality P-TBESD with high EE and proper DL, the preparation conditions of liposomes were performed with four elements and three values. The optimized compositions and the corresponding EEs of TBESD liposomes response values are listed in Table 3. The experiment results indicated that the ranking of the elements was A﹥D﹥C﹥B. Consequently, the maximum value of EE was obtained when the concentrations of soybean phospholipid (mg/mL), soybean phospholipid/drug ratio (w/w), soybean phospholipid/cholesterol ratio (w/w) and soybean phospholipid/sodium deoxycholate ratio (w/w) were 30 mg/mL, 10:1, 10:1 and 10:1 (A3B2C2D2), respectively.
 |
Table 3 Results of the orthogonal experiment (n=3) |
Characterization of P-TBESD
The particle analysis of the reconstituted liposomes for P-TBESD was evaluated by the dynamic light scattering system using NanoPlus zeta/nano particle analyzer. The mean particle size and PDI values of the formulation were 249.77±15.68 nm and 0.184±0.002, respectively (Figure 2B). The results show that the nanoparticle and distribution of P-TBESD are favorable. The zeta potential of the reconstituted liposomes was −21.89±1.51 mV, indicating a good stability (Figure 2C). It has been reported that nanoparticles with a relatively high surface charge could generate repulsion between the particles. In addition, nanoparticles with small particle size and negative surface charge exhibit preferential uptake by biological membrane, which could be beneficial for larger contact areas and electrostatic interaction.47,48
The morphology and particle size of the reconstituted liposomes for P-TBESD was confirmed by TEM. Figure 2A shows the structure of the reconstituted TBESD liposomes, suggesting that the liposomes were spherical in shape, well dispersed and with no aggregation. Light scattering measurements demonstrated a mean diameter of ~250 nm.
Since the biflavonoids of S. doederleinii are difficult to dissolve in water, free drug was separated by ultracentrifugation and determined by HPLC. In this study, the EE and DL of total biflavonoids of the optimal P-TBESD formulation were 91.39±0.88% and 7.59±0.08%, respectively. It is obvious that P-TBESD have a high encapsulation efficiency and proper drug loading for better delivery.
Cytotoxicity assay
The cytotoxicity of TBESD and P-TBESD was determined by MTT assay. HT-29 cells were treated with designated doses of the equivalent extract for 48 hrs. The results showed both TBESD and P-TBESD concentration-dependent cytotoxicity against HT-29 cells with mean IC50 values of 38.76 and 24.33 μg/mL, respectively (Figure S1). The IC50 of P-TBESD was 0.63-fold compared to free drugs. As can be seen from the aforementioned results, the optimal proliposomes increased the solubility of TBESD and improved cellular uptake of those ingredients with poor membrane permeability.
In vivo pharmacokinetic study
A comparative pharmacokinetic study was carried on TBESD and P-TBESD; the bioanalytical quantitation method by LC-ESI-MS/MS was sensitive and selective enough to allow the simultaneous quantification of 5 biflavonoids employed. Concentrations of 5 biflavonoids (amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone) were analyzed in rat plasma after oral administration. The plasma concentration-time curves of TBESD and P-TBESD are plotted in Figure 3. The main pharmacokinetic parameters of 5 biflavonoids are compiled in Table 4. The pharmacokinetic analysis was carried out by fitting the blood drug concentration-time using a noncompartmental model with DAS pharmacokinetic software Version 3.0 program.
 |
Table 4 Pharmacokinetic parameters of five active ingredients in rats following single oral administration of TBESD and proliposome formulations respectively (mean ± SD, n=6) |
Compared with the raw TBESD, the values Cmax, AUC0-∞ and MRT0-∞ for amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone in proliposomes significantly increased (Figure 3). The results show that the values of T1/2 of biflavonoids show no obvious difference between TBESD and P-TBESD. The values Cmax and AUC0-∞ of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone in proliposomes were 5.40/6.69, 1.96/5.23, 5.56/7.61, 7.18/9.55 and 2.59/1.91-fold higher than those of control, respectively, indicating that the absorption of five active ingredients in the form of P-TBESD substantially increased. The higher values of MRT0-∞ and lower values of CL of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone from P-TBESD groups relatively to TBESD groups suggested that proliposome formulation increased blood retention times and decreased the elimination rates of the five S. doederleinii components.49 The reasonable explanation is delayed release and reduced first pass metabolism by the transport mechanism from the proliposomes, and against degradation by the gastric fluid from sodium deoxycholate.30 However, the values of Vd were lower than those of the control (p﹤0.01), with for the exception of Vd for robustaflavone (p≤ 0.05). These results suggested that the proliposomes promoted the accumulation of the five active ingredients of S. doederleinii at the tumor site. Compared with TBESD, the relative bioavailability (RA) of amentoflavone, robustaflavone, 2’’,3’’-dihydro-3’,3’’’-biapigenin, 3’,3’’’-binaringenin and delicaflavone from P-TBESD were 669%, 523%, 761%, 955% and 191%, respectively. The elements will play impacts on the mechanisms of enhanced oral bioavailability from proliposomes: (1) the increased dissolution and absorption of proliposomes-loaded herbal bioactives;50 (2) the absorption of liposomal drugs can be enhanced by adhering to the epithelial cell membrane;18 (3) NaDC inserted into liposomal bilayers might have protection of drugs offsets in the destructive effects of outside bile salts;51 (4) the mechanism of endocytosis by epithelial cells led to reduced first pass effect; (5) TBESD-loaded liposomes coated with IMOs are able to withstand the damage caused by bile salts, gastric acid and enzymes in GIT, and improving the liposome stability.
In vivo antitumor efficacy study
To further validate whether P-TBESD formulations have improved oral bioavailability and enhanced the antitumor effect, HT-29 xenograft-bearing mice were treated with different TBESD formulations when tumors averaged 50–70 mm3 in size.
As shown in Figure 4B, the tumor volume of control group with the aggressive nature of the HT-29 tumor model increased rapidly. Compared with the control group, the tumor volumes of mice in all treatment groups were significantly decreased (P﹤0.01). Mice treated with P-TBESD displayed tumor suppression significantly better than TBESD (P﹤0.05), but Fluorouracil did much better than both TBESD formulations. The mice were euthanized in 24 hrs after the last lavage, and tumor tissues were collected and weighed (Figure 4A). The tumor growth inhibition (TGI) rate of TBESD, P-TBESD and Fluorouracil groups was 25.96%, 48.41% and 56.75%, respectively (Table 5).
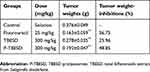 |
Table 5 The average weights and tumor weight-inhibitions of mice before and after treatment (mean ± SD, n=6) |
Meanwhile, as an indicator of drug-mediated toxicity, weight loss is used widely. In our experiments, body weights of mice were monitored every 2 days during treatment. As shown in Figure 4C, the body weights of mice for fluorouracil group were significantly lower than those of control groups (p﹤0.01) during the treatment period. There was no significant difference observed among TBESD, P-TBESD and control groups, possibly suggesting the slight systemic toxicity of TBESD and P-TBESD. Furthermore, during the treatment period of two weeks, no apparent signs of anorexia, dehydration, locomotor impairment or other symptoms associated with side effects were observed in the treatment groups except for fluorouracil group. After euthanizing the animals, the histopathological changes in vital organs were examined by H&E staining. As shown in Figure 4D, the morphology of the different tissue sections exhibited no appreciable abnormalities or noticeable organ except for the group of the fluorouracil. As marked by the red circle, small focal inflammation and lymphocytes; infiltration of liver and alveoli epithelial cells proliferation and alveoli collapse of lung were observed, suggesting that liver and lung have a slight degree of damage and necrosis. Furthermore, myocardial edema with vacuolar degeneration of heart and renal tubular epithelial cells edema of kidney were observed, indicating a comparatively serious systemic toxicity of fluorouracil. While slightly different between TBESD and P-TBESD treatment groups, there were not obvious pathological abnormalities for both groups.
All these results suggested that P-TBESD is more effective than TBESD, and P-TBESD and TBESD are safer than Fluorouracil in treating tumor, which efficiently enhanced the therapeutic effect without causing toxic and side effects, and improve the quality of life.
Conclusion
In this study, an oral dosage form of P-TBESD containing bile salts, which is used for insertion into lipid bilayer to improve stability in the gastrointestinal environment and promote absorption, were successfully prepared by thin film dispersion-sonication method and were characterized. The formulation composition was optimized using a L9 (34) orthogonal experiment design. P-TBESD exhibits a small size and a slightly negative surface charge, which involved preferential uptake by biological membrane. As compared to TBESD, P-TBESD significantly increased blood retention time in addition to prolonging intestinal absorption, which results in dramatically improved oral bioavailability of the total S. doederleinii biflavonoids. More importantly, P-TBESDdemonstrated strongly enhanced in vivo cytotoxic effect without systemic toxicity. These encouraging results demonstrate that proliposomes as a specific and efficient drug delivery system is a promising strategy to improve oral bioavailability, target efficiency and treat cancers.
Acknowledgments
The authors gratefully acknowledge the financial support of the National Natural Science Foundation of China (21775023), Joint Funds for the innovation of science and Technology of Fujian province (2017Y9123 and 2017Y9124), Social Development Guiding Programs of Fujian Province of China (2017Y0042), Natural Science Foundation of Fujian Province of China (2016J01767), Funds for scientific research start-up of high-level talents of Fujian Medical University (XRCZX2018020) and Sailing Funds of Fujian Medical University (2018QH1016). The authors also thank the Fujian Medical University Ethics Committee for their kind guidance in the animal experiments.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Editorial committee of Chinese Materia Medica. Zhong Hua Ben Cao. Shanghai: Shanghai Science and Technology Press; 1999:711–712.
2. Saroni Arwa P, Zeraik ML, Ximenes VF, Da Fonseca LM, Bolzani Vda S, Siqueira Silva DH. Redox-active biflavonoids from Garcinia brasiliensis as inhibitors of neutrophil oxidative burst and human erythrocyte membrane damage. J Ethnopharmacol. 2015;174:410–418. doi:10.1016/j.jep.2015.08.041
3. Park NH, Lee CW, Bae JH, Na YJ. Protective effects of amentoflavone on Lamin A-dependent UVB-induced nuclear aberration in normal human fibroblasts. Bioorg Med Chem Lett. 2011;21(21):6482–6484. doi:10.1016/j.bmcl.2011.08.067
4. Yu S, Yan H, Zhang L, et al. A review on the phytochemistry, pharmacology, and pharmacokinetics of amentoflavone, a naturally-occurring biflavonoid. Molecules. 2017;22(2):299–321. doi:10.3390/molecules22020299
5. Zheng XK, Li YJ, Zhang L, Feng WS, Zhang X. Antihyperglycemic activity of Selaginella tamariscina (Beauv.) Spring. J Ethnopharmacol. 2011;133(2):531–537. doi:10.1016/j.jep.2010.10.028
6. Zheng XK, Zhang L, Wang WW, Wu YY, Zhang QB, Feng WS. Anti-diabetic activity and potential mechanism of total flavonoids of Selaginella tamariscina (Beauv.) Spring in rats induced by high fat diet and low dose STZ. J Ethnopharmacol. 2011;137(1):662–668. doi:10.1016/j.jep.2011.06.018
7. Coulerie P, Nour M, Maciuk A, et al. Structure-activity relationship study of biflavonoids on the Dengue virus polymerase DENV-NS5 RdRp. Planta Med. 2013;79(14):1313–1318. doi:10.1055/s-0033-1350672
8. Li S, Zhao M, Li Y, et al. Preparative isolation of six anti-tumour biflavonoids from Selaginella doederleinii Hieron by high-speed counter-current chromatography. Phytochem Anal. 2014;25(2):127–133. doi:10.1002/pca.2478
9. Liu H, Peng H, Ji Z, et al. Reactive oxygen species-mediated mitochondrial dysfunction is involved in apoptosis in human nasopharyngeal carcinoma CNE cells induced by Selaginella doederleinii extract. J Ethnopharmacol. 2011;138(1):184–191. doi:10.1016/j.jep.2011.08.072
10. Li S, Yao H, Zhao M, Li Y, Huang L, Letters XLJA. Determination of seven biflavones of Selaginella doederleinii by high performance liquid chromatography. Anal Lett. 2013;46(18):2835–2845. doi:10.1080/00032719.2013.831426
11. Lin RC, Skaltsounis AL, Seguin E, Tillequin F, Koch M. Phenolic constituents of Selaginella doederleinii. Planta Med. 1994;60(2):168–170. doi:10.1055/s-2006-959443
12. Yao H, Chen B, Zhang Y, et al. Analysis of the total biflavonoids extract from Selaginella doederleinii by HPLC-QTOF-MS and its in vitro and in vivo anticancer effects. Molecules. 2017;22(2):325–342. doi:10.3390/molecules22020325
13. Sui Y, Li S, Shi P, et al. Ethyl acetate extract from Selaginella doederleinii Hieron inhibits the growth of human lung cancer cells A549 via caspase-dependent apoptosis pathway. J Ethnopharmacol. 2016;190:261–271. doi:10.1016/j.jep.2016.06.029
14. Sui Y, Yao H, Li S, et al. Delicaflavone induces autophagic cell death in lung cancer via Akt/mTOR/p70S6K signaling pathway. J Mol Med (Berl). 2017;95(3):311–322. doi:10.1007/s00109-016-1487-z
15. Chen B, Wang X, Zou Y, et al. Simultaneous quantification of five biflavonoids in rat plasma by LC-ESI-MS/MS and its application to a comparatively pharmacokinetic study of Selaginella doederleinii Hieron extract in rats. J Pharm Biomed Anal. 2018;149:80–88. doi:10.1016/j.jpba.2017.10.028
16. Yang S, Shi P, Huang X, et al. Pharmacokinetics, tissue distribution and protein binding studies of chrysocauloflavone I in rats. Planta Med. 2016;82(3):217–223. doi:10.1055/s-0035-1558159
17. Luo X, Liu M, Hu L, et al. Targeted delivery of pixantrone to neutrophils by poly(sialic acid)-p-octadecylamine conjugate modified liposomes with improved antitumor activity. Int J Pharm. 2018;547(1–2):315–329. doi:10.1016/j.ijpharm.2018.06.021
18. Hosny KM, Aljaeid BM. Sildenafil citrate as oral solid lipid nanoparticles: a novel formula with higher bioavailability and sustained action for treatment of erectile dysfunction. Expert Opin Drug Deliv. 2014;11(7):1015–1022. doi:10.1517/17425247.2014.912212
19. Yokoyama M, Okano T, Sakurai Y, Ekimoto H, Shibazaki C, Kataoka K. Toxicity and antitumor activity against solid tumors of micelle-forming polymeric anticancer drug and its extremely long circulation in blood. Cancer Res. 1991;51(12):3229–3236. doi:10.1016/b978-0-444-82027-3.50061-4
20. Tang J, Sun J, Cui F, Zhang T, Liu X, He Z. Self-emulsifying drug delivery systems for improving oral absorption of ginkgo biloba extracts. Drug Deliv. 2008;15(8):477–484. doi:10.1080/10717540802039089
21. Zhang Y, Che E, Zhang M, et al. Increasing the dissolution rate and oral bioavailability of the poorly water-soluble drug valsartan using novel hierarchical porous carbon monoliths. Int J Pharm. 2014;473(1–2):375–383. doi:10.1016/j.ijpharm.2014.07.024
22. Guan P, Lu Y, Qi J, et al. Enhanced oral bioavailability of cyclosporine A by liposomes containing a bile salt. Int J Nanomed. 2011;6:965–974. doi:10.2147/IJN.S19259
23. Torchilin VP. Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov. 2005;4(2):145–160. doi:10.1038/nrd1632
24. Alexander A, Ajazuddin PRJ, Saraf S, Saraf S. Recent expansion of pharmaceutical nanotechnologies and targeting strategies in the field of phytopharmaceuticals for the delivery of herbal extracts and bioactives. J Control Release. 2016;241:110–124. doi:10.1016/j.jconrel.2016.09.017
25. Jain S, Harde H, Indulkar A, Agrawal AK. Improved stability and immunological potential of tetanus toxoid containing surface engineered bilosomes following oral administration. Nanomedicine. 2014;10(2):431–440. doi:10.1016/j.nano.2013.08.012
26. Hiremath PS, Soppimath KS, Betageri GV. Proliposomes of exemestane for improved oral delivery: formulation and in vitro evaluation using PAMPA, Caco-2 and rat intestine. Int J Pharm. 2009;380(1–2):96–104. doi:10.1016/j.ijpharm.2009.07.008
27. Khan I, Yousaf S, Alhnan MA, Ahmed W, Elhissi A, Jackson MJ. Design characteristics of inhaler devices used for pulmonary delivery of medical aerosols. In: Ahmed W, Jackson MJ, editors. Surgical Tools and Medical Devices. Cham: Springer International Publishing; 2016:573–591.
28. Khan I, Yousaf S, Subramanian S, Albed Alhnan M, Ahmed W, Elhissi A. Proliposome tablets manufactured using a slurry-driven lipid-enriched powders: development, characterization and stability evaluation. Int J Pharm. 2018;538(1–2):250–262. doi:10.1016/j.ijpharm.2017.12.049
29. Zheng B, Teng L, Xing G, et al. Proliposomes containing a bile salt for oral delivery of Ginkgo biloba extract: formulation optimization, characterization, oral bioavailability and tissue distribution in rats. EurJ Pharm Sci. 2015;77:254–264. doi:10.1016/j.ejps.2015.06.007
30. Yang G, Zhao Y, Zhang Y, Dang B, Liu Y, Feng N. Enhanced oral bioavailability of silymarin using liposomes containing a bile salt: preparation by supercritical fluid technology and evaluation in vitro and in vivo. Int J Nanomed. 2015;10:6633–6644. doi:10.2147/IJN.S92665
31. Carafa M, Marianecci C, Annibaldi V, Di Stefano A, Sozio P, Santucci E. Novel O-palmitoylscleroglucan-coated liposomes as drug carriers: development, characterization and interaction with leuprolide. Int J Pharm. 2006;325(1–2):155–162. doi:10.1016/j.ijpharm.2006.06.040
32. Sorndech W, Nakorn KN, Tongta S, Blennow A. Isomalto-oligosaccharides: recent insights in production technology and their use for food and medical applications. Lwt-Food Sci Technol. 2018;95:135–142. doi:10.1016/j.lwt.2018.04.098
33. Singh DP, Singh J, Boparai RK, et al. Isomalto-oligosaccharides, a prebiotic, functionally augment green tea effects against high fat diet-induced metabolic alterations via preventing gut dysbacteriosis in mice. Pharmacol Res. 2017;123:103–113. doi:10.1016/j.phrs.2017.06.015
34. He H, Lu Y, Qi J, Zhu Q, Chen Z, Wu W. Adapting liposomes for oral drug delivery. Acta Pharmaceutica Sinica B. 2019;9(1):36–48. doi:10.1016/j.apsb.2018.06.005
35. Liu Y, Luo X, Xu X, Gao N, Liu X. Preparation, characterization and in vivo pharmacokinetic study of PVP-modified oleanolic acid liposomes. Int J Pharm. 2017;517(1–2):1–7. doi:10.1016/j.ijpharm.2016.11.056
36. Zhang L, Ren Y, Wang Y, He Y, Feng W, Song C. Pharmacokinetics, distribution and anti-tumor efficacy of liposomal mitoxantrone modified with a luteinizing hormone-releasing hormone receptor-specific peptide. Int J Nanomed. 2018;13:1097–1105. doi:10.2147/IJN.S150512
37. Khayata N, Abdelwahed W, Chehna MF, Charcosset C, Fessi H. Stability study and lyophilization of vitamin E-loaded nanocapsules prepared by membrane contactor. Int J Pharm. 2012;439(1–2):254–259. doi:10.1016/j.ijpharm.2012.09.032
38. Liu L, Zhang X, Zhang Y, et al. Optimal activation of carboxyl-superparamagnetic iron oxide nanoparticles bioconjugated with antibody using orthogonal array design. J Nanosci Nanotechnol. 2013;13(12):8137–8143. doi:10.1166/jnn.2013.7925
39. Li Y, Tang C, Zhang E, Yang L. Colistin-entrapped liposomes driven by the electrostatic interaction: mechanism of drug loading and in vivo characterization. Int J Pharm. 2016;515(1–2):20–29. doi:10.1016/j.ijpharm.2016.10.001
40. Katanasaka Y, Kodera Y, Yunokawa M, Kitamura Y, Tamura T, Koizumi F. Synergistic anti-tumor effects of a novel phosphatidyl inositol-3 kinase/mammalian target of rapamycin dual inhibitor BGT226 and gefitinib in non-small cell lung cancer cell lines. Cancer Lett. 2014;347(2):196–203. doi:10.1016/j.canlet.2014.02.025
41. Zhang H. Thin-film hydration followed by extrusion method for liposome preparation. In: Methods in Molecular Biology. Vol. 1522. New York: Humana Press; 2017. 17–22. doi:10.1007/978-1-4939-6591-5_2
42. Mann JF, Scales HE, Shakir E, et al. Oral delivery of tetanus toxoid using vesicles containing bile salts (bilosomes) induces significant systemic and mucosal immunity. Methods. 2006;38(2):90–95. doi:10.1016/j.ymeth.2005.11.002
43. Li B, Li S, Tan Y, et al. Lyophilization of cationic lipid-protamine-DNA (LPD) complexes. J Pharm Sci. 2000;89(3):355–364. doi:10.1002/(SICI)1520-6017(200003)89:3<355::AID-JPS7>3.0.CO;2-H
44. Gu Z, Wang Q, Shi Y, et al. Nanotechnology-mediated immunochemotherapy combined with docetaxel and PD-L1 antibody increase therapeutic effects and decrease systemic toxicity. J Control Release. 2018;286:369–380. doi:10.1016/j.jconrel.2018.08.011
45. Wang Q, Wei Q, Yang Q, et al. A novel formulation of [6]-gingerol: proliposomes with enhanced oral bioavailability and antitumor effect. Int J Pharm. 2018;535(1–2):308–315. doi:10.1016/j.ijpharm.2017.11.006
46. Gao D, Tang S, Tong Q. Oleanolic acid liposomes with polyethylene glycol modification: promising antitumor drug delivery. Int J Nanomed. 2012;7:3517–3526. doi:10.2147/IJN.S31725
47. Zheng B, Yang S, Fan C, et al. Oleic acid derivative of polyethylenimine-functionalized proliposomes for enhancing oral bioavailability of extract of Ginkgo biloba. Drug Deliv. 2016;23(4):1194–1203. doi:10.3109/10717544.2015.1101790
48. Venault A, Hsu KJ, Yeh LC, Chinnathambi A, Ho HT, Chang Y. Surface charge-bias impact of amine-contained pseudozwitterionic biointerfaces on the human blood compatibility. Colloids Surf B Biointerfaces. 2017;151:372–383. doi:10.1016/j.colsurfb.2016.12.040
49. Yadav J, Korzekwa K, Nagar S. Impact of lipid partitioning on the design, analysis, and interpretation of microsomal time-dependent inactivation. Drug Metab Dispos. 2019;47(7):732–742. doi:10.1124/dmd.118.085969
50. Nguyen TX, Huang L, Gauthier M, Yang G, Wang Q. Recent advances in liposome surface modification for oral drug delivery. Nanomedicine (Lond). 2016;11(9):1169–1185. doi:10.2217/nnm.16.9
51. Gagliardi A, Paolino D, Iannone M, Palma E, Fresta M, Cosco D. Sodium deoxycholate-decorated zein nanoparticles for a stable colloidal drug delivery system. Int J Nanomed. 2018;13:601–614. doi:10.2147/IJN.S156930
Supplementary materials
 |
Figure S1 Cytotoxicity of HT-29 cells after incubated with free TBESD and P-TBESD for 48 h. (n=5). |
 |
Table S1 The standard curve results of biflavonoids |
 |
Table S2 Precision and repeatabilities of biflavonoids from P-TBESD (n=6) |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.