Back to Journals » OncoTargets and Therapy » Volume 13
Primary Resistance to Immune Checkpoint Blockade in an STK11/TP53/KRAS-Mutant Lung Adenocarcinoma with High PD-L1 Expression
Authors Kwack WG , Shin SY , Lee SH
Received 16 July 2020
Accepted for publication 24 August 2020
Published 7 September 2020 Volume 2020:13 Pages 8901—8905
DOI https://doi.org/10.2147/OTT.S272013
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nicola Silvestris
Won Gun Kwack,1 So Youn Shin,2 Seung Hyeun Lee1
1Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Kyung Hee University School of Medicine, Seoul, South Korea; 2Department of Radiology, Kyung Hee University School of Medicine, Seoul, South Korea
Correspondence: Seung Hyeun Lee
Division of Pulmonary, Allergy, and Critical Care Medicine, Department of Internal Medicine, Kyung Hee University School of Medicine, Kyungheedae-ro 23, Dongdaemun-gu, Seoul 02447, South Korea
Tel +82 2 958 8511
Fax +82 2 968 1848
Email [email protected]
Abstract: Several studies have shown that STK11 and TP53 mutations have different effects on the susceptibility to immune checkpoint blockade in KRAS-mutant non-small cell lung cancer (NSCLC). However, the impact of STK11/TP53 co-mutations on treatment outcomes in the same clinical setting has never been reported. We recently encountered a case of a 70-year-old man who was diagnosed with advanced lung adenocarcinoma with high-programmed death-ligand 1 (PD-L1) expression. He received pembrolizumab monotherapy as a frontline treatment; however, the tumor did not respond to this therapy and showed deleterious outcome. Next-generation sequencing revealed that the tumor harbored a rare STK11/TP53/KRAS triple mutation. Our case suggests that these compound mutations may constitute a distinct, aggressive subset that is resistant to immunotherapy even when the tumor strongly expresses PD-L1. In addition, this report highlights the importance of using molecular profiling to detect co-mutations that can be associated with primary resistance or disease progression to improve survival even in the immunotherapy setting.
Keywords: lung cancer, KRAS, STK11, TP53, immune checkpoint inhibitors, prognosis
Introduction
KRAS mutations are the most prevalent oncogenic driver of non-small cell lung cancer (NSCLC), accounting for 20–30% of the lung adenocarcinoma cases.1 They are associated with cigarette smoking and have been linked to poor prognosis without available targeted therapy.2 The emergence of immune checkpoint inhibitors (ICIs) in recent years has enriched treatment options for NSCLC; however, clinical data on their efficacy in KRAS-mutant tumors has been inconsistent across different clinical settings.3,4 A recent study demonstrated that the treatment outcomes with ICIs were similar between KRAS-mutant and KRAS–wild type NSCLC and the response was better in programmed death-ligand 1 (PD-L1)-positive patients in both populations.5 Interestingly, evidence suggests that concurrent genomic alterations can affect the response and prognosis of KRAS-mutant NSCLC.6–9 Concurrent mutations in STK11 have been associated with reduced survival in patients with KRAS-mutant tumors after chemotherapy.6–8 La Fleur et al evaluated the mutational status of 82 genes using targeted sequencing in 352 patients with NSCLC and demonstrated that STK11 and TP53 mutations were linked to poor patient survival in KRAS-mutant subtypes.9 These studies suggest that it is not KRAS mutations themselves, but co-mutations in other genes that determine the aggressive behavior of tumors and treatment outcomes in patients with the driving mutation.
Previous studies have shown that STK11 and TP53 mutations seldom overlap in treatment-naïve NSCLC patients,7,10 and the impact of these compound mutations on the efficacy of immunotherapy for KRAS-mutant NSCLC remains to be elucidated. Here, we report a case of lung adenocarcinoma harboring an STK11/TP53/KRAS triple mutation which showed poor response to first-line pembrolizumab monotherapy even though the tumor strongly expressed PD-L1.
Case Presentation
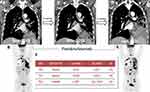
A 70-year-old male presented at our institution in September 2019 with a complaint of a persistent cough that had lasted for three months. He never smoked and had been taking statin and anti-hypertensive drugs for several years. His vital signs were stable and physical examination revealed no significant findings. Chest computed tomographic (CT) scan and positron emission tomography (PET) revealed a 4.8 cm x 4.3 cm-sized malignant lung mass in the left lower lobe (LLL) invading the corresponding bronchus, and multiple mediastinal lymph nodes and bone metastases suggesting that the cancer was at stage IV (Figure 1A and B). Bronchoscopy showed irregular mucosal changes and a protruding mass at the orifice of the LLL, and histopathology revealed invasive mucinous lung adenocarcinoma positive for thyroid transcription factor 1 (TTF-1). Although routine mutation profiling ruled out several genetic alterations, including epidermal growth factor receptor mutations and anaplastic lymphoma kinase (ALK) or ROS proto-oncogene 1 (ROS1) fusions, the PD-L1 tumor proportion score (TPS), evaluated using the 22C3 pharmDx assay, was 100%. We initiated first-line pembrolizumab monotherapy (200 mg intravenously administered every three weeks) as a first-line treatment, expecting a favorable response. The first response evaluation conducted in November 2019 showed slight increase in the size of primary tumor mass and the mediastinal lymph nodes (Figure 1C). Taking the possibility of pseudoprogression into account, we continued with an additional two cycles of treatment, which was well tolerated by the patients without the occurrence of immune-related adverse events. However, the patient’s cough was difficult to control despite the use of antitussives, inhaled bronchodilators, and systemic corticosteroids. Follow-up of patients with chest CT and PET scans conducted in January 2020 showed a marked enlargement of both the primary lung tumor and the mediastinal lymph nodes as well as several newly developed bone metastases (Figure 1D and E). The carcinoembryonic antigen level also elevated to 57.2 ng/mL from the baseline level of 21.9 ng/mL. We immediately discontinued the frontline treatment and switched to cytotoxic chemotherapy (pemetrexed and carboplatin). At the same time, we performed next-generation sequencing (NGS) to isolate genetic alterations that might be related to the poor response to pembrolizumab in our patient. NGS revealed three concurrent mutations in the KRAS (p.G12V), TP53 (p.E286K), and STK11 (p.Y36fs*) genes (Figure 1F). Soon after the first cycle of chemotherapy, the patient developed pneumonia. Serum C-reactive protein and procalcitonin levels elevated to 12.7 mg/dL (normal: < 0.5 mg/dL) and 1.27 ng/mL (normal: < 0.046 ng/mL), respectively. Additionally, chest CT scan showed definite bronchial obstruction at LLL while no ground-glass opacities or airspace consolidations were evident in the right lung. This indicates an association of the pneumonia with bronchial obstruction rather than immunotherapy and subsequent chemotherapy. Despite intensive care with broad-spectrum antibiotics and mechanical ventilation, the pneumonia quickly progressed to sepsis from which the patient succumbed in February 2020.
Discussion
Cancer immunotherapy with ICIs has shown promising outcomes in numerous clinical trials and has provided one of the most important breakthroughs in the management of solid tumors, including lung cancers.11 Antibodies against PD-1 and PD-L1 have become the standard of care for second-line treatment for all comers with NSCLC, and first-line treatment for the patients with high PD-L1 expression. PD-L1 expression has been consistently shown to correlate with an increased efficacy of anti-PD-1/PD-L1 treatments.12–14 The US Food and Drug Administration approved the use of anti-PD-L1 clones, 22C3 and 28–8, as a companion diagnostic method for pembrolizumab and a complementary diagnostic method for nivolumab, respectively, in 2015. However, PD-L1 expression alone is not sufficient to predict a patient’s response to ICIs.15 Tumor mutational burden (TMB) and concurrent genetic alterations have emerged as novel predictive biomarkers for immunotherapy with immune checkpoint blockade.16
STK 11 (also known as liver kinase B1, LKB1) is a tumor suppressor gene encoding a serine/threonine kinase 11 that directly phosphorylates AMP-activated protein kinase (AMPK), regulating lipid, cholesterol, and glucose metabolism in a variety of tissues.8 SKT11-AMPK signaling negatively regulates the mammalian target of rapamycin pathway which is commonly dysregulated in human malignancies.17,18 Somatic mutations in SKT11, which primarily induces a loss-of-function, are present in approximately 30% of the NSCLC patients with an increased prevalence seen in smokers and patients harboring KRAS mutations.19,20 STK11 mutations alone do not appear to predict response to chemotherapy; however, the occurrence of STK11/KRAS co-mutations have been associated with reduced survival in NSCLC patients in a variety of treatment settings.6–8 TP53 is a DNA binding transcription factor that regulates DNA repair, metabolism, and apoptosis.21 TP53 mutations alone have been associated with poor response and survival in NSCLC patients treated with chemotherapy or surgical resection,22,23 while their prognostic significance in KRAS-mutant tumors remains inconclusive.6,7,9
Current evidence suggests that mutations in STK11 and TP53 exert opposite effects on clinical outcomes of immunotherapy.4,7 STK11 mutations induce T-cell exhaustion and secretion of immunosuppressive cytokines, resulting in uninflamed tumor microenvironment (TME) with weak PD-L1 expression, and have been linked to adverse clinical outcomes in NSCLC patients treated with immune checkpoint blockade therapy.4,7,24 In contrast, tumors harboring TP53 mutations are characterized by frequent somatic mutations, an inflamed TME, and a favorable response to PD-1/PD-L1 inhibition.4,7 STK11/TP53/KRAS triple mutations are exceptionally rare; only seven in 1385 NSCLC patients harbored this mutation.10 Thus, the clinical significance of these compound mutations in immunotherapy has never been discussed. Our case demonstrated de novo resistance to frontline pembrolizumab monotherapy despite strong PD-L1 expression. To the best of our knowledge, this is the first case describing an adverse clinical outcome after immune checkpoint blockade for NSCLC harboring KRAS/STK11/TP53 co-mutations. A recent study by Bange et al demonstrated that STK11/TP53/KRAS triple mutation in advanced NSCLC conferred a better prognosis after first-line therapy compared with STK11/KRAS co-mutations or SKT11 alone.10 Among the sixty-two enrolled patients in that study, fifty-one (82%) were treated with platinum-doublet chemotherapy, while only five patients (8%) received immunotherapy as a first-line treatment.10 Therefore, the effect of STK11/TP53/KRAS co-mutations on survival was evaluated mainly in patients treated with chemotherapy. In addition, only seven patients with STK11/TP53/KRAS co-mutations were enrolled in that study. Those previous results and ours suggest that the survival impact of the compound mutations can be different depending on treatment settings. The mechanism underlying the primary resistance of STK11/TP53/KRAS-mutant lung adenocarcinoma to immune checkpoint blockade in our case remains unclear; however, it may be partially explained by the strong proliferative drive and profound immune escape mediated by the mutations in each of these genes as described in previous studies.6,7 Interestingly, previous studies have demonstrated that TP53 has four potential binding sites in the STK11 promoter region, suggesting that it may act as a regulator of STK11.25,26 Thus, it may be possible that mutations in TP53 modulate STK11 signaling, which may accentuate the uninflamed phenotype of TME. This hypothesis should be validated by future studies.
In summary, we described a case of rare STK11/TP53/KRAS-mutant lung adenocarcinoma which demonstrated resistance to anti-PD-1 therapy. Although further evidence should be accumulated, our report suggests that this compound mutation may be a strong predictive biomarker for adverse treatment outcomes in ICIs, and may constitute a distinct aggressive subtype requiring unique therapeutic strategies other than immunotherapy, even when the tumor strongly expresses PD-L1. In addition, our case emphasizes the importance of comprehensive molecular profiling in routine clinical practice, even in this treatment setting, and supports the development of clinical trials to facilitate “personalized immunotherapy” for genetically defined subsets of NSCLC.
Consent for Publication
Written informed consent was obtained from the patient’s wife for the publication of both the case report and its accompanying images.
Data Sharing Statement
All data generated or analyzed during this study are available in this manuscript.
Ethics Statement
The publication of this case report was approved by the Institutional Review Board at Kyung Hee University Hospital (KHUH 2020-06-010).
Acknowledgments
The authors thank the patient and his family for their permission to publish this case report along with the accompanying images.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Lee SH. Chemotherapy for lung cancer in the era of personalized medicine. Tuberc Respir Dis (Seoul). 2019;82(3):179–189. doi:10.4046/trd.2018.0068
2. Cancer Genome Atlas Research N. Comprehensive molecular profiling of lung adenocarcinoma. Nature. 2014;511(7511):543–550.
3. Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373(17):1627–1639. doi:10.1056/NEJMoa1507643
4. Skoulidis F, Goldberg ME, Greenawalt DM, et al. STK11/LKB1 mutations and PD-1 inhibitor resistance in KRAS-mutant lung adenocarcinoma. Cancer Discov. 2018;8(7):822–835. doi:10.1158/2159-8290.CD-18-0099
5. Jeanson A, Tomasini P, Souquet-Bressand M, et al. Efficacy of immune checkpoint inhibitors in KRAS-mutant Non-Small Cell Lung Cancer (NSCLC). J Thorac Oncol. 2019;14(6):1095–1101. doi:10.1016/j.jtho.2019.01.011
6. Arbour KC, Jordan E, Kim HR, et al. Effects of co-occurring genomic alterations on outcomes in patients with KRAS-mutant non-small cell lung cancer. Clin Cancer Res. 2018;24(2):334–340. doi:10.1158/1078-0432.CCR-17-1841
7. Skoulidis F, Byers LA, Diao L, et al. Co-occurring genomic alterations define major subsets of KRAS-mutant lung adenocarcinoma with distinct biology, immune profiles, and therapeutic vulnerabilities. Cancer Discov. 2015;5(8):860–877. doi:10.1158/2159-8290.CD-14-1236
8. Schabath MB, Welsh EA, Fulp WJ, et al. Differential association of STK11 and TP53 with KRAS mutation-associated gene expression, proliferation and immune surveillance in lung adenocarcinoma. Oncogene. 2016;35(24):3209–3216. doi:10.1038/onc.2015.375
9. La Fleur L, Falk-Sorqvist E, Smeds P, et al. Mutation patterns in a population-based non-small cell lung cancer cohort and prognostic impact of concomitant mutations in KRAS and TP53 or STK11. Lung Cancer. 2019;130:50–58. doi:10.1016/j.lungcan.2019.01.003
10. Bange E, Marmarelis ME, Hwang WT, et al. Impact of KRAS and TP53 co-mutations on outcomes after first-line systemic therapy among patients with STK11-mutated advanced non-small-cell lung cancer. JCO Precis Oncol. 2019;3:1–11.
11. Dall’Olio FG, Maggio I, Massucci M, Mollica V, Fragomeno B, Ardizzoni A. ECOG performance status >/=2 as a prognostic factor in patients with advanced non small cell lung cancer treated with immune checkpoint inhibitors-A systematic review and meta-analysis of real world data. Lung Cancer. 2020;145:95–104.
12. Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–1550. doi:10.1016/S0140-6736(15)01281-7
13. Reck M, Rodriguez-Abreu D, Robinson AG, et al. Updated analysis of KEYNOTE-024: pembrolizumab versus platinum-based chemotherapy for advanced non-small-cell lung cancer with PD-L1 tumor proportion score of 50% or greater. J Clin Oncol. 2019;37(7):537–546. doi:10.1200/JCO.18.00149
14. Rizvi NA, Mazieres J, Planchard D, et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a Phase 2, single-arm trial. Lancet Oncol. 2015;16(3):257–265. doi:10.1016/S1470-2045(15)70054-9
15. Kim T, Cha YJ, Chang YS. Correlation of PD-L1 expression tested by 22C3 and SP263 in non-small cell lung cancer and its prognostic effect on EGFR mutation-positive lung adenocarcinoma. Tuberc Respir Dis (Seoul). 2020;83(1):51–60. doi:10.4046/trd.2019.0026
16. Kim HC, Choi CM. Current status of immunotherapy for lung cancer and future perspectives. Tuberc Respir Dis (Seoul). 2020;83(1):14–19. doi:10.4046/trd.2019.0039
17. Wullschleger S, Loewith R, Hall MN. TOR signaling in growth and metabolism. Cell. 2006;124(3):471–484. doi:10.1016/j.cell.2006.01.016
18. Shackelford DB, Shaw RJ. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer. 2009;9(8):563–575. doi:10.1038/nrc2676
19. Matsumoto S, Iwakawa R, Takahashi K, et al. Prevalence and specificity of LKB1 genetic alterations in lung cancers. Oncogene. 2007;26(40):5911–5918. doi:10.1038/sj.onc.1210418
20. Facchinetti F, Bluthgen MV, Tergemina-Clain G, et al. LKB1/STK11 mutations in non-small cell lung cancer patients: descriptive analysis and prognostic value. Lung Cancer. 2017;112:62–68. doi:10.1016/j.lungcan.2017.08.002
21. Olivier M, Hollstein M, Hainaut P. TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb Perspect Biol. 2010;2(1):a001008. doi:10.1101/cshperspect.a001008
22. Mitsudomi T, Hamajima N, Ogawa M, Takahashi T. Prognostic significance of p53 alterations in patients with non-small cell lung cancer: a meta-analysis. Clin Cancer Res. 2000;6(10):4055–4063.
23. Kandioler D, Stamatis G, Eberhardt W, et al. Growing clinical evidence for the interaction of the p53 genotype and response to induction chemotherapy in advanced non-small cell lung cancer. J Thorac Cardiovasc Surg. 2008;135(5):1036–1041. doi:10.1016/j.jtcvs.2007.10.072
24. Koyama S, Akbay EA, Li YY, et al. STK11/LKB1 deficiency promotes neutrophil recruitment and proinflammatory cytokine production to suppress T-cell activity in the lung tumor microenvironment. Cancer Res. 2016;76(5):999–1008. doi:10.1158/0008-5472.CAN-15-1439
25. Rizvi NA, Hellmann MD, Snyder A, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015;348(6230):124–128. doi:10.1126/science.aaa1348
26. Van Allen EM, Miao D, Schilling B, et al. Genomic correlates of response to CTLA-4 blockade in metastatic melanoma. Science. 2015;350(6257):207–211. doi:10.1126/science.aad0095
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.