Back to Journals » Clinical Ophthalmology » Volume 14
Prevalence of Subclinical Keratoconus Among Pediatric Egyptian Population with Astigmatism
Authors Sidky MK , Hassanein DH , Eissa SA , Salah YM , Lotfy NM
Received 10 January 2020
Accepted for publication 27 February 2020
Published 23 March 2020 Volume 2020:14 Pages 905—913
DOI https://doi.org/10.2147/OPTH.S245492
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Scott Fraser
Mohamed Karim Sidky, Dina Hosameldin Hassanein, Sherif A Eissa, Yehia M Salah, Nancy M Lotfy
Department of Ophthalmology, Faculty of Medicine, Cairo University, Cairo, Egypt
Correspondence: Mohamed Karim Sidky
Department of Ophthalmology, Faculty of Medicine, Cairo University, Al-Saray Street, El Manial, Cairo 11956, Egypt
Tel +201 222262629
Email [email protected]
Purpose: To report the prevalence of subclinical keratoconus (KC), and KC suspects, in pediatric Egyptian population with astigmatic error ≥ 2 diopters (D), with the use of CSO Sirius tomographer.
Methods: A cross-sectional study that included all the children aged 6– 18 years with an astigmatic error of ≥ 2 D who attended the pediatric ophthalmology clinic for routine checkups during the period between March 2017 and December 2017 was done. All subjects underwent comprehensive ophthalmic examination, and corneal imaging using CSO Sirius instrument, a pentacam with Scheimpflug technology.
Results: There were 547 subjects enrolled in our study. Mean age was 11.34 ± 3.03 (range 6– 18) years. There were 278 subjects (50.8%) between 6 and 12 years, and 269 subjects (49.2%) between 12 and 18 years. Males represented 48.4% and females represented 51.6% of patients. Among all the subjects, there were 26 (4.8%) KC patients, 24 (4.4%) KC suspects, and 497 (90.9%) were normal.
Conclusion: Prevalence of subclinical KC among pediatric age group with astigmatism is relatively high, hence the importance of corneal tomography screening of children with astigmatic error ≥ 2 D.
Keywords: pediatric keratoconus, subclinical keratoconus, astigmatism, Scheimpflug, Sirius, Egypt
Introduction
Keratoconus (KC) is a progressive asymmetric ectatic disorder that has biomechanical and structural effects on corneal collagen organization. It is characterized by progressive thinning of the cornea, which results in corneal protrusion, irregular astigmatism, and decreased visual function.1
Pediatric KC is more aggressive, with a higher rate of progression than adult KC; and that is mainly because of the dynamic nature of the young cornea.1
Epidemiology of pediatric KC was not extensively discussed in the literature,1 and to the best of our knowledge; this is the first study to report the prevalence of subclinical KC in pediatric age group with astigmatic error.
In Lebanon, El-Khoury et al reported an incidence of 0.53% in children 14 years or younger who were diagnosed in a tertiary referral eye center.2 Another study conducted in southwestern Nigeria reported 0.08% prevalence of blindness among school children due to KC.3 A recent study was conducted in Saudi Arabia reported 4.79% prevalence among the pediatric age group.4
Subclinical KC refers to a form of the disease in which the characteristic slit-lamp, keratometric, and retinoscopic findings are absent.5 The diagnosis of such cases is highly challenging, and the parameters of diagnosis should be adequately determined.6
Many methods with different corneal topographers have been used for the diagnosis of KC and subclinical KC. Pentacam is an advanced instrument that scans the anterior and posterior cornea with a rotating Scheimpflug camera. Reproducibility and repeatability of measurements of the corneal thickness and posterior elevation have been proved to be high.7
Studying the epidemiology of KC helps to apply adequate plans for the screening and early treatment of the condition. Early detection of cases is of utmost importance, as it allows us to advise the best treatment plan to prevent the progression of the disease.4
The aim of our study is to report the prevalence of subclinical KC, and KC suspects in pediatric Egyptian population with astigmatic error ≥2 D, with the use of CSO Sirius tomographer.
Methods
This is a cross-sectional study that was conducted in the pediatric ophthalmology clinic of (Eye Care center) in Cairo, Egypt.
The study adhered to the tenets of the declaration of Helsinki. The ethical approval process for clinical research for this study was achieved in agreement with the regulations of the Eye Care Center (approval number PRO170402).
The study included all the children aged (6–18) years with an astigmatic error of ≥2D, who attended the pediatric ophthalmology clinic for routine checkups. Children with an astigmatic error of less than 2 D, or giving a history of previous intraocular surgery, or showing keratometric, biomicroscopic or retinoscopic signs of unilateral or bilateral clinical KC were excluded from the study.
After obtaining informed consents from the parents or guardians, all included subjects were given a referral sheet including demographic data, medical and family history, and comprehensive eye examination.
Routine history recording of each patient included: age at the time of presentation, gender, symptoms (blurred and decreased vision, monocular diplopia, photophobia, headache, glare, ghosting of images and frequent changes in glasses prescription), history of ocular disease (allergy and vernal kerato-conjunctivitis VKC), systemic disease associated with Down syndrome (cardiac; mitral valve prolapse, atopic diseases), and family history of KC.
Clinical assessment of subjects included: measurements of Snellen visual acuity; monocular unaided, and optimal corrected distance visual acuity CDVA (with the best manifest correction), keratometry measurement, objective cycloplegic refraction measurement (streak retinoscopy) that may provide a sensitive assessment for detecting and confirming the diagnosis of early KC. It was used to determine the initial refractive error.
Slit-lamp biomicroscopy examination was performed to detect the presence or absence of biomicroscopic signs of KC, such as apical thinning, Fleischer’s ring, Vogt’s striae, and apical scars.
Imaging and Classification of Cases
All patients underwent bilateral corneal imaging by CSO Sirius instrument, a pentacam with Scheimpflug technology. The Sirius studies the tomography of the anterior ocular segment with a 3D corneal analysis. It merges the Scheimpflug technology with the placido disc topography for assessment of corneal elevations, and curvatures. All scans were taken by a single experienced ophthalmologist who was masked to the patients’ diagnoses. Subjects were instructed to blink completely just before the measurement, then to look at the fixation target with the opening of both eyes for proper alignment. The examiner then aligned the instrument and started to capture the scans of both eyes (25 images per second for each eye, with a resolution of 1024 x 768 pixels). The Scheimpflug camera acquires radius of curvature measurements in the flat and steep meridians on a 3 mm diameter field of the central cornea. The instrument was re-aligned before each measurement. Only scans with the comment (OK) in the quality specification window were taken. The image acquisition quality criteria of Scheimpflug images are coverage ≥ 90%, centration ≥ 90% and keratoscopy coverage ≥ 80%.
Two experienced cornea consultants were involved in studying each subject’s tomography scans. All patients with suspected or abnormal tomographic images detected by one or two consultants were invited for further clinical examinations, including slit-lamp biomicroscopy, fundoscopy, and corneal imaging by the CSO Sirius tomographer.
In absence of any clinical signs of KC; either keratometric, retinoscopic or biomicroscopic, the classification of cases into subclinical KC, KC suspect, and normal was based on: 1 – The computerized analysis obtained by the CSO Sirius tomographer, 2 – Studying the tomography maps of patients.
The KC screening software parameters obtained by the Sirius are: [symmetry index front (SIf), symmetry Index back (SIb), Baiocchi-Calossi-Versaci index (BCV) front, back and total, and 4.5 and 8 mm zone root-mean-square values per unit area (RMS/A) front and back].
Diagnosis of definite subclinical KC was established when there are two or more atypical tomography patterns (Inferior isolated steep cone/asymmetric bow tie with inferior steepness and I-S ratio over 1.4, or posterior elevation > 20 µm, or K Max > 49 D, Km > 47.2 D, in addition to classification (KC) by Sirius software analysis, in absence of any clinical signs of KC; either keratometric, retinoscopic or biomicroscopic).
However, we considered subclinical KC a suspect when only one tomographic criterion was found, in addition to classification (KC suspect) by Sirius software analysis, and in absence of any clinical signs of KC; either keratometric, retinoscopic or biomicroscopic.
In case of disagreements between consultants regarding classification, the decision was made according to the Sirius analysis results.
Statistical Analysis
Statistical analysis was performed using SPSS 21 software. All normally distributed continuous data were presented as means and Standard Deviations. The Chi-square test was used to determine whether gender or age is significantly associated with KC and KC suspects.
We used the following formula for a CI for a population proportion:
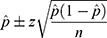
where 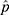 is the sample proportion,
is the sample proportion,  is the sample size, and
is the sample size, and  is the appropriate value from the standard normal distribution for our desired confidence level.
is the appropriate value from the standard normal distribution for our desired confidence level.
Multivariate logistic regression analysis was performed to explore the association between KC and KC suspect and the independent variables (age and gender). Adjusted odds ratios (OR) as a measure of association and 95% confidence intervals (CI) were calculated. All tests were two-tailed and P values lower than 0.05 were considered statistically significant.
Results
There were 547 subjects enrolled in our study, they attended the pediatric ophthalmology clinic of (Eye Care center) in Cairo, Egypt during the period between March 2017 and December 2017.
Mean age was 11.34 ± 3.03 (range 6–18) years. Regarding age groups; there were 278 subjects (50.8%) between 6 and 12 years, and 269 subjects (49.2%) between 12 and 18 years. There were 265 (48.4%) males and 282 (51.6%) females.
Among all the subjects, there were 26 KC patients, 24 KC suspects, and 497 were normal. Classification and frequency of the disease are demonstrated in (Table 1). (Figures 1–3) show representative CSO Sirius scans for KC, KC suspects, and normal eyes, respectively.
 |
Table 1 Classification and Frequency of KC, KC Suspects, and Normal Subjects |
 |
Figure 1 CSO Sirius scan image of a case of KC. |
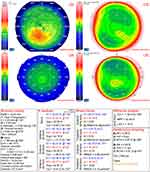 |
Figure 2 CSO Sirius scan image of a case of KC suspect. |
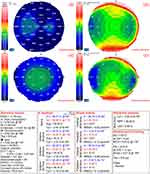 |
Figure 3 CSO Sirius scan image of a normal eye. |
The prevalence of disease laterality is shown in (Table 2).
 |
Table 2 Prevalence of Laterality of Cases |
The descriptive statistics for the tomographic numerical data are represented in (Table 3).
 |
Table 3 Mean Values of Sirius Parameters for Study Groups |
There was a significant difference between the percentages of KC with regard to gender (P = 0.03), and we found that the percentage of KC in males was significantly higher than in females (Table 4). However, there was no significant difference between the percentages of KC suspect with regard to gender.
 |
Table 4 The Prevalence of KC with Regard to Gender |
Moreover, we found a significant difference in the prevalence of KC with regard to age groups (P< 0.001), as it was significantly higher in the age group (12–18) years than (6–12) years (Table 5).
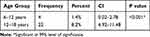 |
Table 5 The Prevalence of KC with Regard to Age Groups |
The prevalence of KC suspects was also significantly higher in the older age group (12–18) years. (P = 0.03).
Discussion
KC in pediatric age group was not extensively discussed in the literature, and few reports are available about the epidemiology of the disease.1
In our study, we report 4.8%, and 4.4% prevalence of subclinical KC and KC suspects, respectively, among pediatric Egyptian population with astigmatic error ≥2 D. This percentage is comparable to the results of a recent study that was conducted in Saudi Arabia; the study reported 4.79% prevalence of KC among pediatric Saudi children. However, that study relied mainly on the tomographic imaging without clinical examination of patients, also it did not differentiate between KC cases and KC suspects.4
Another study was conducted in Egypt to investigate corneal abnormalities among children with high myopia for early detection of KC. They reported a 9.2% prevalence of KC among the studied population. A value, which is higher than our results. However, this study had different inclusion criteria. First: the study group age was less than 14 years old. Second: they included high myopic cases with more than 6D either with or without astigmatism.8
Our results are much higher than the results of a study conducted in Lebanon which reported an incidence of 0.53% in children 14 years or younger who were diagnosed in a tertiary referral eye center.2 Another study conducted on a group of Lebanese medical students with a mean age of 23.6 revealed a 3.3 prevalence rate.9 In Iran, Hashemi et al reported a 2.5% prevalence of KC among students of Mashhad University with mean age 26.1.10 Awwad et al reported that: the prevalence of KC is 17.5–19.1% in pediatric first-degree relatives aged 6–18 years of patients with KC.11
KC induces progressive myopia and irregular astigmatism. In literature, few studies reported the association between prevalence of KC and refractive errors. A study conducted by Serdarogullari et al reported an overall percentage of 14.1% (6.3% cases of KC, and 7.8% subclinical KC) among the general population with astigmatic error ≥2 D. which is the same astigmatic error value (≥2 D) we used in our study to compare the prevalence of the disease in children. Serdarogullari et al study concluded that higher prevalence rates of KC will be associated with increased astigmatic errors.12 That prevalence rate was a higher estimate as compared to our study and other reported percentages in literature. Though this study may not actually represent the prevalence of KC among the general population with astigmatism, because of the small sample size (64 patients).12
Prevalence of KC in literature has shown a wide range of variations, and that could be attributed to genetic, geographical, ethnic, diagnostic and environmental factors.2 In 1986 Kennedy et al reported that the prevalence of KC in the population of Minnesota was 0.05%.13 This study was conducted over a 48-year duration, and it is the most frequently cited reference in this subject.4
Prevalence of KC is higher in the Middle East than in other areas, as proved by literature.9,10,14-17 This might be due to the high incidence of chronic allergies and spring catarrh.17
In our study, VKH was reported in 35% of cases with subclinical KC, which is comparable to the value obtained by Omar & I.A.N who reported a 37.5% prevalence of VKC in KC eyes in Egyptian children with myopia.8 Prevalence of eye allergies between Egyptian children is about 20.05%, and VKH represents about 3.95%. The association between KC and allergic conjunctivitis is controversial; however, most studies have reported a firm association between allergy with eye rubbing and keratoconus.17
In our study, we found that the percentage of subclinical KC in males 6.8% was significantly higher than in females 2.8%. Some studies in the literature have reported a higher prevalence of KC in males.2,18-21 Others have reported a higher prevalence in females,22 and some studies did not find any differences between males and females.5,13,23
The upper age limit for pediatric age groups in our study was 18 years old; (in accordance with the Egyptian law), and that is also the age limit for children officially accepted in the governmental hospitals in Egypt.
Also, we divided cases into two age groups in order to highlight the higher prevalence rate of the disease among older age groups, as we report a higher percentage (8.2%) of subclinical KC among the older age group (12–18) years as compared to (1.4%) in the younger age group. A result was consistent with Awwad et al study that reported an increased prevalence of KC among increasing age groups of pediatric first-degree relatives of KC patients.11
Pediatric KC is much more aggressive with higher rates of progression than in adults. This is attributed to the structural differences in the collagen cross-linking between the young and adult cornea.1 Early diagnosis of the disease with proper selection of the treatment plan is crucial, it can prevent deterioration of the disease that can cause severe ocular morbidity.1,11
This study is limited because of its cross-sectional design; a cohort study would provide a better understanding of the nature of the disease. Using a clinical sample, instead of a population-based sample for prevalence estimates is another limitation of our study. Accordingly, our results may not reflect the actual prevalence of subclinical KC among Egyptian children with astigmatic error ≥2 D. However, the high prevalence rate that we report should ring a bell. Further research with adequate population-based sample is required to corroborate the results of this study.
Differentiation between KC suspects and normal eyes with tomographic criteria is difficult and challenging, there are no universally accepted diagnostic criteria for KC suspects, and that represents a shortcoming for all KC screening methods.11,12
Scheimpflug-based tomographers such as Sirius have shown promise in early detection of KC cases and suspects. Since the Scheimpflug principle works with the highest depth of focus and the least image distortion, it delivers valuable information from the anterior corneal surface to the posterior lens surface.24
CSO Sirius specificity, sensitivity, and predictability have been discussed in some studies in the literature. Shetty et al reported that the Slb, BCV, and 8 mm RMS/A back screening indices have shown specificity equals to (100% each), and SIf, BCVf and 4.5 mm RMS/A back indices have shown sensitivity equals to (84.6%) in distinguishing KC from normal eyes. However, to differentiate subclinical KC from normal eyes, the highest specificity was seen for SIb parameter (100%).25
Another study was conducted by Safarzadeh M and N Nasiri to assess the anterior segment parameters of KC eyes at different stages of the disease by the Sirius tomographer, concluded that posterior corneal elevation, corneal thickness, RMS, BCVf and BCVb are the most important indices that need to be considered to diagnose different grades of keratoconus.25
Maresca et al study concluded that: Sirius showed precision and repeatability almost twice as much as ultrasound pachymetry.26
Wang Q et al conducted a study to comprehensively assess the precision and agreement of anterior corneal power measurements using eight different devices, which are Tomey RC-5000, Topcon KR-8000, IOLMaster, Allegro Topolyzer, Pentacam and Sirius. They concluded that anterior corneal curvature measurements obtained from the eight different devices present very good repeatability and reproducibility.27
A study conducted by Shetty et al concluded that although the screening indices from the Scheimpflug devices can effectively differentiate KC from normal, the cutoff values are still not adequate for differentiation between normal and sub-clinical cases.28 Hence, the importance of individual evaluation of the tomography maps of each patient.
Future studies may include the screening of children corneas using ultrahigh-resolution anterior segment optical coherence tomography (AS-OCT) that detects a smaller thickness of the epithelium and Bowman layer, with an epithelial donut pattern of eyes with KC, enabling for an earlier diagnosis.29
We report a high prevalence (4.8%) of subclinical KC in children with astigmatic error; therefore we recommend tomography screening for children with astigmatic error ≥2 D.
To the best of our knowledge, this is the first study to report the prevalence of subclinical KC in pediatric age group with astigmatism.
Conclusion
Pediatric KC is a serious disease. Early detection can prevent severe ocular morbidity. The development of Scheimpflug-based corneal imaging techniques enables the early diagnosis of the disease. Prevalence of subclinical KC among pediatric age group with astigmatism is relatively high, hence the importance of corneal tomography screening for children with astigmatic error ≥2 D.
Disclosure
The authors report no funding and no conflicts of interest in this work.
References
1. Mukhtar S, Ambati BK. Pediatric keratoconus: a review of the literature. Int Ophthalmol. 2018;38(5):2257–2266. doi:10.1007/s10792-017-0699-8
2. El-Khoury S, Abdelmassih Y, Hamade A, et al. Pediatric keratoconus in a tertiary referral center: incidence, presentation, risk factors, and treatment. J Refract Surg. 2016;32(8):534–541. doi:10.3928/1081597X-20160513-01
3. Ajaiyeoba AI, Isawumi MA, Adeoye AO, Oluleye TS. Prevalence and causes of blindness and visual impairment among school children in south-western Nigeria. Int Ophthalmol. 2005;26(4–5):121–125. doi:10.1007/s10792-005-4836-4
4. Torres Netto EA, Al-Otaibi WM, Hafezi NL, et al. Prevalence of keratoconus in paediatric patients in Riyadh, Saudi Arabia. Br J Ophthalmol. 2018;102(10):1436–1441. doi:10.1136/bjophthalmol-2017-311391
5. Li X, Rabinowitz YS, Rasheed K, Yang H. Longitudinal study of the normal eyes in unilateral keratoconus patients. Ophthalmology. 2004;111(3):440–446. doi:10.1016/j.ophtha.2003.06.020
6. de Sanctis U, Loiacono C, Richiardi L, Turco D, Mutani B, Grignolo FM. Sensitivity and specificity of posterior corneal elevation measured by Pentacam in discriminating keratoconus/subclinical keratoconus. Ophthalmology. 2008;115(9):1534–1539. doi:10.1016/j.ophtha.2008.02.020
7. de Sanctis U, Missolungi A, Mutani B, Richiardi L, Grignolo FM. Reproducibility and repeatability of central corneal thickness measurement in keratoconus using the rotating Scheimpflug camera and ultrasound pachymetry. Am J Ophthalmol. 2007;144(5):712–718. doi:10.1016/j.ajo.2007.07.021
8. Omar IAN. Keratoconus screening among myopic children. Clin Ophthalmol. 2019;13:1909–1912. doi:10.2147/OPTH.S225326
9. Waked N, Fayad AM, Fadlallah A, El Rami H. Dépistage du kératocône dans une population universitaire au Liban [Keratoconus screening in a Lebanese students’ population]. J Fr Ophthalmol. 2012;35(1):23–29. French. doi:10.1016/j.jfo.2011.03.016
10. Hashemi H, Khabazkhoob M, Yazdani N, et al. The prevalence of keratoconus in a young population in Mashhad, Iran. Ophthal Physiol Opt. 2014;34(5):519–527. doi:10.1111/opo.12147
11. Awwad ST, Yehia M, Mehanna CJ, et al. Tomographic and refractive characteristics of pediatric first-degree relatives of keratoconus patients. Am J Ophthalmol. 2019;207:71–76.
12. Serdarogullari H, Tetikoglu M, Karahan H, Altin F, Elcioglu M. Prevalence of keratoconus and subclinical keratoconus in subjects with astigmatism using pentacam derived parameters. J Ophthalmic Vis Res. 2013;8(3):213–219.
13. Kennedy RH, Bourne WM, Dyer JA. A 48-year clinical and epidemiologic study of keratoconus. Am J Ophthalmol. 1986;101(3):267–273. doi:10.1016/0002-9394(86)90817-2
14. Millodot M, Shneor E, Albou S, Atlani E, Gordon-Shaag A. Prevalence and associated factors of keratoconus in Jerusalem: a cross-sectional study. Ophthalmic Epidemiol. 2011;18(2):91–97. doi:10.3109/09286586.2011.560747
15. Hashemi H, Khabazkhoob M, Fotouhi A. Topographic keratoconus is not rare in an iranian population: the Tehran Eye Study. Ophthalmic Epidemiol. 2013;20(6):385–391. doi:10.3109/09286586.2013.848458
16. Shehadeh MM, Diakonis VF, Jalil SA, Younis R, Qadoumi J, Al-Labadi L. Prevalence of keratoconus among a palestinian tertiary student population. Open Ophthalmol J. 2015;9:172–176. doi:10.2174/1874364101509010172
17. Elbedewy HA, Wasfy TE, Soliman SS, et al. Prevalence and topographical characteristics of keratoconus in patients with refractive errors in the Egyptian delta. Int Ophthalmol. 2019;39(7):1459–1465. doi:10.1007/s10792-018-0965-4
18. Pearson AR, Soneji B, Sarvananthan N, Sandford-Smith JH. Does ethnic origin influence the incidence or severity of keratoconus? Eye. 2000;14(Pt 4):625–628. doi:10.1038/eye.2000.154
19. Owens H, Gamble G. A profile of keratoconus in New Zealand. Cornea. 2003;22(2):122–125. doi:10.1097/00003226-200303000-00008
20. Wagner H, Barr JT, Zadnik K. Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study: methods and findings to date. Cont Lens Anterior Eye. 2007;30(4):223–232. doi:10.1016/j.clae.2007.03.001
21. Godefrooij DA, de Wit GA, Uiterwaal CS, Imhof SM, Wisse RP. Age-specific incidence and prevalence of keratoconus: a nationwide registration study. Am J Ophthalmol. 2017;175:169–172. doi:10.1016/j.ajo.2016.12.015
22. Krachmer JH, Feder RS, Belin MW. Keratoconus and related noninflammatory corneal thinning disorders. Surv Ophthalmol. 1984;28(4):293–322. doi:10.1016/0039-6257(84)90094-8
23. Gokhale NS. Epidemiology of keratoconus. Indian J Ophthalmol. 2013;61(8):382–383. doi:10.4103/0301-4738.116054
24. Shetty R, Arora V, Jayadev C, et al. Repeatability and agreement of three Scheimpflug-based imaging systems for measuring anterior segment parameters in keratoconus. Invest Ophthalmol Vis Sci. 2014;55(8):5263–5268. doi:10.1167/iovs.14-15055
25. Safarzadeh M, Nasiri N. Anterior segment characteristics in normal and keratoconus eyes evaluated with a combined Scheimpflug/Placido corneal imaging device. J Curr Ophthalmol. 2016;28(3):106–111. doi:10.1016/j.joco.2016.06.003
26. Maresca N, Zeri F, Palumbo P, Calossi A. Agreement and reliability in measuring central corneal thickness with a rotating Scheimpflug-Placido system and ultrasound pachymetry. Cont Lens Anterior Eye. 2014;37(6):442–446. doi:10.1016/j.clae.2014.07.009
27. Wang Q, Savini G, Hoffer KJ, et al. A comprehensive assessment of the precision and agreement of anterior corneal power measurements obtained using 8 different devices. PLoS One. 2012;7(9):e45607. doi:10.1371/journal.pone.0045607
28. Shetty R, Rao H, Khamar P, et al. Keratoconus screening indices and their diagnostic ability to distinguish normal from ectatic corneas. Am J Ophthalmol. 2017;181:140–148. doi:10.1016/j.ajo.2017.06.031
29. Reinstein DZ, Archer TJ, Gobbe M. Corneal epithelial thickness profile in the diagnosis of keratoconus. J Refract Surg. 2009;25(7):604–610. doi:10.3928/1081597X-20090610-06
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
