Back to Journals » International Journal of Women's Health » Volume 14
Prevalence of Human Immunodeficiency Virus Among Pregnant Women
Authors Kurtay S, Hussein AI
Received 6 October 2022
Accepted for publication 17 December 2022
Published 22 December 2022 Volume 2022:14 Pages 1803—1806
DOI https://doi.org/10.2147/IJWH.S392442
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Elie Al-Chaer
Sabri Kurtay, Ahmed Issak Hussein
Mogadishu Somalia Turkish Hospital, Department of Gynecology and Obstetrics, Mogadishu, Somalia
Correspondence: Sabri Kurtay; Ahmed Issak Hussein, Department of Obstetrics and Gynecology, Somalia Mogadishu Turkey Recep Tayyip Erdoğan Training and Research Hospital, Mogadishu, Somalia, Tel +90 536 339 93 63 ; +252 61 559 74 79, Email [email protected]; [email protected]
Background: While the prevalence of human immunodeficiency virus shows a decreasing trend globally, it is reported that the frequency of the infection and the associated mortality has increased in Somalia. HIV infection also plays an important role in maternal mortality and morbidity.
Objective: It is aimed to determine the HIV prevalence in pregnant women by observing the general population.
Methods: It was a quantitative, retrospective study conducted to examine the records of anti-HIV antibody test results of all patients. The sample size was calculated as 664 with a 99% confidence interval (CI) and a 5% margin of error. Anti-HIV results of selected patients in the electronic database were obtained. Mean, standard deviation, minimum and maximum values were calculated for the age. Percentages were calculated for prevalence.
Results: A total of 7959 pregnant were tested and 17 pregnants were found to be positive (0.2%). The results of 199,936 patients were analyzed. A total of 699 patients were found to be positive (0.3%). Of the 699 positive cases, 293 (41.9) were female and 406 (58.1%) were male. HIV seroprevalence in men and women was 0.3% and 0.4%, respectively.
Conclusion: Within the scope of WHO’s global HIV elimination goal, Somalia needs effective national and international initiatives.
Keywords: HIV/AIDS, pregnancy, Somalia
Introduction
World Health Organisation (WHO) estimates that 19.3 million women were living with Human Immunodeficiency Virus (HIV) worldwide, of whom 660.000 were detected in 2020. The count of children living with HIV was 1.3 million in 2020.1 HIV can be transmitted from mother to baby during pregnancy, during childbirth, or through breastfeeding after birth. Nine percent of new HIV cases in 2017 were patients who acquired it by vertical transmission. Vertical transmission plays an important role in the WHO’s plan to eliminate HIV/AIDS by 2030. In this context, it is aimed that at least 95% of pregnant women should have at least one antenatal visit, the HIV test rate in pregnant women should be more than 95%, and the antiretroviral treatment rate in positive pregnancies should be more than 95%.2 There are national and international guidelines for the strategy of preventing mother-to-child transmission (PMTCT).3,4 Another ambitious but achievable target set by The Joint United Nations Programme on HIV/AIDS (UNAIDS) in 2015 is as follows; by 2020, 90% of all people living with HIV will know their HIV status, 90% of all people with diagnosed HIV infection will receive sustained antiretroviral therapy, 90% of all people receiving antiretroviral therapy will have viral suppression (90-90-90 target).5 According to UNAIDS 2020 data, it is estimated that there are more than 8.000 HIV-positive adults and children in Somalia, nearly half of whom are women older than 15 years of age, and the HIV prevalence in women aged 15–49 is less than %0.1.6
Globally, while the incidence of HIV is decreasing, HIV cases in the Middle East and North Africa region (MENA) increased by 7% between 2010 and 2020. In 2020, only 61% of people living with HIV were aware of their HIV status, 43% of people living with HIV were receiving treatment, and only 37% of people living with HIV had achieved viral suppression. Women between the ages of 15–49 constitute 34% of new HIV cases in this region. In Somalia, the annual number of new HIV infections in women over 15 years of age is less than 100 people, the number of AIDS-related deaths is less than 200 people, the number of people living with HIV is 3900, and the prevalence of HIV in people aged 15–49 is estimated to be less than 0.1%. The proportion of women living with HIV who know their status, women living with HIV who are on treatment, and women living with HIV who are virally suppressed was 62%, 44%, and 39%, respectively. According to these ratios, the approximate number required to reach the 90-90-90 target is 20,000, 30,000, and 30,000 respectively, in Somalia. Regarding the elimination of vertical transmission, the percentage of pregnant women living with HIV accessing antiretroviral drugs is 0% in 2010, compared to 33% in 2020. However, the target in services to prevent vertical transmission could not be reached.7
Due to the lack of standard health services, there is no systematic follow-up of pregnant women in Somalia. This leads to the deprivation of many protective services at the national level. If HIV positivity is detected during pregnancy, a diagnosis and treatment plan for the newborn can also be made. As far as is known, the number of studies on the frequency of HIV/AIDS in Somalia is insufficient in the literature. This study aims to determine the prevalence in pregnant women by examining the frequency of HIV in the population.
Methods
It was a quantitative, retrospective study conducted to examine the records of Anti-HIV antibody test results of all patients admitted to the Mogadishu Somalia Turkish Hospital (MSTH) between January 01, 2015, and September 30, 2021. Using the Raosoft ® sample size calculator (http://www.raosoft.com/samplesize.html), the sample size was calculated as 664 with a 99% confidence interval (CI) and a 5% margin of error. A total sampling technique was used for sample selection. Anti-HIV results of selected patients in the electronic database were obtained. Mean, standard deviation, minimum and maximum values were calculated for the age variable. Percentages were calculated for prevalence. SPSS 22.0® software was used for statistical analysis. The data used in the study were obtained from the hospital’s electronic archive.
Results
A total of 199,936 patients who were admitted to the hospital over 6 years were tested for HIV under appropriate indications. The distribution of patients by gender was Female/Male: 82,169/117,767 (41.1% / 58.9%). Among 199,936 patients, 699 patients were found to be positive (0.3%). Of the 699 positive cases, 293 (41.9%) were female and 406 (58.1%) were male. Among the people tested in the study were 7959 pregnant women, of whom 17 pregnant women were found positive. The rate of HIV positivity among pregnant women was 0.2% (Table 1). The rate of positive pregnant women (n=17) among HIV-positive women (n=293) was 5.8% (Table 2).
 |
Table 1 Characteristics of Tested Patients |
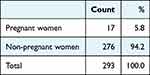 |
Table 2 Rate of Pregnant Women Among HIV-Positive Women |
Discussion
The UNAIDS data estimated the prevalence of HIV/AIDS in the general population, males and females to be <0.1% for each of these groups.6 HIV prevalence in pregnant women in 2007 and 2010 was 1.3% and 1.1% in Somaliland, and 0.7% and 0.3% in Puntland (respectively). However, data were unavailable for the South Central region, including the capital Mogadishu.8 In our study, new and updated information was added to the literature by revealing the prevalence of pregnant women in Mogadishu to be 0.2%.
HIV screening during pregnancy has an important role in strategies to reduce vertical transmission.9,10 However, there is no routine follow-up during pregnancy in Somalia; therefore, pregnant women cannot be screened for HIV. In this study, it is seen that 5.8% of HIV-positive women are pregnant women. This poses a risk of vertical transmission. Knowing the HIV prevalence in pregnant women is a guide for determining a strategy for reducing vertical transmission in Somalia.
Following the WHO recommendations, it was observed that the frequency of HIV in newborns decreased following the initiation of treatment for HIV-infected pregnant women in Thailand and South Africa.11,12 In these examples, success has been achieved by adhering to principles such as creating appropriate guidelines, effective planning, widespread education activities, cooperation with international and national organizations, and recording data. HIV elimination can be initiated in Somalia via similar organizations by national healthcare providers.
One of the effects of the civil war is the difficulty in diagnosis, treatment, and surveillance of infectious diseases in women, newborns, children, and adolescents.13 Considering the case detection and the low treatment rates in the detected cases, it is concluded that more precautions should be taken than in the current situation. Local governments, non-governmental organizations, and health service providers should be in effective coordination with international organizations for the prevention of HIV/AIDS in the country and the treatment of detected cases.
Limitations
Since it was a prevalence study, the subsequent clinical status of patients who were found to be HIV positive could not be evaluated.
Declaration of Patient Consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that her name and initials will not be published and due efforts will be made to conceal the identity, but anonymity cannot be guaranteed.
Disclosure
The authors declare no competing interests in this work.
References
1. World Health Organization. Global Progress Report on HIV, Viral Hepatitis and Sexually Transmitted Infections, 2021: Accountability for the Global Health Sector Strategies 2016–2021: Actions for Impact: Web Annex 2: Data Methods. World Health Organization; 2021.
2. World Health Organization. Global Guidance on Criteria and Processes for Validation: Elimination of Mother to Child Transmission of HIV, Syphilis and Hepatitis B Virus. Geneva: World Health Organization. Licence: CC BY-NC-SA 3.0 IGO; 2021.
3. Start free, stay free, AIDS free — final report on 2020 targets; 2021. Available from: https://www.unaids.org/en/resources/documents/2021/start-free-stay-free-aids-free-final-report-on-2020-targets.
4. Ministry of Health Ethiopia. National Guideline for Prevention of MTCT HIV, Syphilis and Hepatitis B Virus. Ministry of Health Ethiopia; 2021.
5. UNAIDS. 90-90-90. An ambitious treatment target to help end the AIDS epidemic; 2014. Available from: https://www.unaids.org/en/resources/documents/2017/90-90-90.
6. UNAIDS. Country factsheets Somalia; 2020. Available from: https://www.unaids.org/en/regionscountries/countries/somalia.
7. UNAIDS. UNAIDS Data. Geneva: Joint United Nations Programme on HIV/AIDS; 2021.
8. Bozicevic I, Riedner G, Calleja JM. HIV surveillance in MENA: recent developments and results. Sex Transm Infect. 2013;89(Suppl 3):iii11–iii16. doi:10.1136/sextrans-2012-050849
9. Marston M, Risher K, Mahy MI. HIV acquisition in pregnancy: implications for mother-to-child transmission at the population level in sub-Saharan Africa. J Int AIDS Soc. 2021;24(Suppl 5):e25783. doi:10.1002/jia2.25783
10. Gunn JK, Asaolu IO, Center KE, et al. Antenatal care and uptake of HIV testing among pregnant women in sub-Saharan Africa: a cross-sectional study. J Int AIDS Soc. 2016;19(1):20605. doi:10.7448/IAS.19.1.20605
11. Bailey H, Zash R, Rasi V, Thorne C. HIV treatment in pregnancy. Lancet HIV. 2018;5(8):e457–e467. doi:10.1016/S2352-3018(18)30059-6
12. Thisyakorn U. Elimination of mother-to-child transmission of HIV: lessons learned from success in Thailand. Paediatr Int Child Health. 2017;37(2):99–108. doi:10.1080/20469047.2017.1281873
13. Singh NS, Ataullahjan A, Ndiaye K, et al. Delivering health interventions to women, children, and adolescents in conflict settings: what have we learned from ten country case studies? Lancet. 2021;397(10273):533–542. doi:10.1016/S0140-6736(21)00132-X
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
