Back to Journals » International Journal of Nephrology and Renovascular Disease » Volume 14
Prevalence of Anemia and Its Associated Factors Among Chronic Kidney Disease Patients Attending Selected Public Hospitals of Addis Ababa, Ethiopia: Institutional-Based Cross-Sectional Study
Authors Alemu B, Techane T , Dinegde NG , Tsige Y
Received 18 December 2020
Accepted for publication 20 February 2021
Published 5 March 2021 Volume 2021:14 Pages 67—75
DOI https://doi.org/10.2147/IJNRD.S296995
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Pravin Singhal
Birhie Alemu,1 Tesfaye Techane,1 Negalign Getahun Dinegde,2 Yosief Tsige2
1Saint Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia; 2School of Nursing and Midwifery, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia
Correspondence: Tesfaye Techane Email [email protected]
Background: Anemia is a global public health problem with high mortality and morbidity. It is becoming more prevalent in Ethiopia. Therefore, this study aimed to estimate the prevalence of anemia and its associated factors among chronic kidney disease patients at selected public hospitals of Addis Ababa, Ethiopia.
Methods: An institutional-based cross-sectional study was conducted on selected public hospitals in Addis Ababa for studies on anemia in CKD patients. Data was entered into EPI Info version 4.4.1 and exported to SPSS version 24 for analysis. Both bivariate and multivariate logistic regression analyses were used to identify independently associated factors of anemia in CKD patients. Multivariate analysis was used to control the possible effect of confounders.
Results: A total of 387 participants were included to estimate the prevalence of anemia among chronic kidney disease patients. The prevalence of anemia was 53.5% (95% CI). Being females were 2 times more likely to develop anemia as compared to their counterparts (AOR=2.04, 95%:1.18, 2.46) and hemodialysis history had two times higher odds for anemia (AOR=2.754, 95% CI: 1.218– 6.229, P=0.015) compared to patients without hemodialysis history.
Conclusion: The overall prevalence of anemia across all stages of CKD is 53.5%. From the stage of CKD, stage 5 CKD has a higher (90.9%) anemia prevalence compared to others, and females showed a higher frequency of anemia when compared with males. Therefore, situation-based interventions and country context-specific preventive strategies should be developed to reduce the prevalence of anemia in this patient group.
Keywords: chronic kidney disease, anemia, prevalence, Ethiopia
Background
Chronic kidney disease (CKD) is a progressive, irreversible deterioration of renal function in which the body’s ability to maintain metabolic, fluid electrolyte, and balance fail, which results in uremia or azotemia and can be classified into stages 1 to 5 based on the deterioration of glomerular filtration rate (GFR), with each increasing number indicating a more advanced stage of the disease.1
Anemia is a global public health problem of all developing and developed countries which affects not only human health but also both social and economic development and occurs at all age group of the population.2 Anemia is defined as a state in which the quality and/or quantity of circulating red blood cells is below normal or the established cut-off point defined by the World Health Organization.3 According to the World Health Organization (WHO) diagnostic criteria, Patients are classified as anemic when hemoglobin (Hgb) is < 12 g/dl for females and <13g/dl for males.4
Anemia with CKD is defined as a situation in which the concentration of hemoglobin in the blood is below the mean Hg level, corrected for age and sex and the main cause of anemia in CKD is the inadequate production of endogenous erythropoietin either due to functional or absolute iron deficiencies.5 The prevalence of anemia was high in non-dialysis chronic kidney disease patients from stage 1 to the last stage. Certain factors that are associated with the development of anemia in patients with CKD include; diabetic nephropathy (DN), chronic kidney disease (CKD) stages, body mass index (BMI), smoking, leukocyte count, and serum albumin.6 The occurrence of anemia was higher for patients with both end-stage renal disease and CKD. It has also a greater prevalence among older persons, persons with diabetes, cardiovascular disease, and hypertension than persons without these conditions.5
Anemia is the commonest complication of CKD which accounts significant burden of cardiovascular diseases. CKD decreases the quality of life of patients.7 It suggested that CKD had a significant association with anemia and is considered as a possible cause when the glomerular filtration rate (GFR) is <60mL/min/1.37m2 and, it is more likely to be the cause if the GFR is <30mL/min/1.73m2. The use of iron therapies and erythropoiesis-stimulating agents (ESAs) has allowed improvement in patients with anemia of CKD.8
A study done in Korea showed that, as CKD stages increase the anemia has also direct relation with the stages. Current smokers showed a lower risk of anemia compared to the previous smoker and lower body mass index (BMI) had a higher risk of anemia but there is no significant association between gender and age with anemia.6 Therefore, this cross-sectional study aimed to estimate the prevalence of anemia in patients with CKD and its associated factors at selected public hospitals in Addis Ababa Ethiopia.
Methods
Study Design
A hospital-based cross-sectional study was conducted from November 15 to May 15/2020.
Study Setting
The study was conducted in Addis Ababa. According to the data obtained from Addis Ababa City Administration Health Bureau, there are 11 public hospitals in Addis Ababa, which we are giving different services to the public. Three hospitals (St Paul Hospital, Tikur Anbessa Specialized Hospital, and Zewditu Memorial Hospital) were purposely selected to be the study areas among the public hospitals. These hospitals provide service to patients with the diverse socio-economic background. Besides, there is a high load of CKD patients in these three hospitals as compared to the other public hospitals in the city.
Eligibility Criteria
CKD patients (age >18 years old) who were attended Tikur Anbessa Specialized Hospital, St Paul Hospital, and Zewditu Memorial hospital outpatient department for follow up purpose at the time of data collection and who were willing to participate in the study were included.
Sample Size Determination and Sampling Procedure
The sample size was determined using a formula of estimating a single proportion for a cross-sectional study. Since the population size is less than 10, 000, the final sample size was estimated using the correction formula. The final sample size obtained including a 10% non-response rate was 387. Then, the number of participants in each selected hospital was determined using the population proportionate sampling (PPS). It is estimated using the formula: (E) = C*D, (C*BD in a health facility)/C, where, BD = proportion of CKD patients in the study in a given hospital, E = Final sample size obtained using correction formula (387), C is the total number of CKD patients in the selected hospitals.
Study Instrument
Interviewer structured questionnaire and checklist for chart review was used for data collection. The tool was adopted from a previous study at Gondar University Hospital, North West Ethiopia.9
Operational Definition
- CKD: CKD is defined as kidney damage or glomerular filtration rate (GFR) of less than 90 mL/min/1.73 m2 for three months or more, irrespective of cause or evidence of kidney damage.10
- Stage of CKD: is classified as stage 1, 2, 3, 4, and 5 based on GFR category (Stage 1 CKD: eGFR 90 or Greater, Stage 2 CKD: eGFR between 60 and 89, Stage 3 CKD: eGFR between 30 and 59, Stage 4 CKD: eGFR between 15 and 29, Stage 5 CKD: eGFR Less than 15).11
- Anemia: Anemia was defined as serum hemoglobin levels ≤12 g/dL in women and ≤ 13 g/dL in men age ≥18years old, as recommended by the National Anemia Action Council and the World Health Organization.3
- Cigarette smoking: participants who have current and previous cigarette smoking history at the start of study.12
- Alcohol consumption: Excessive Alcohol Consumption in this study is defined as consumption of ≥6 drinks and ≥4 drinks on a single occasion in men and women respectively.13
- BMI: is a person’s weight in kilograms (kg) divided by his or her height in meters squared. <18.5kg/m2 for underweight, 18.5-24.9 for normal-weight, and ≥ 25.0 kg/m2 for overweight.14
- GFR: can be estimated from calibrated serum creatinine and estimating equations, such as the Cockcroft-Gault formula (normalized for the body surface area [BSA]): (140 - Age [years]) x weight (kg) x (0.86, if female) x 1.73/72 x serum creatinine (mg/dl) x BSA (m2).15
Data Analysis
Data was entered into EPI Info version 4.4.1 and exported to SPSS version 24 for analysis. Descriptive statistics, such as median was used to compute continuous variables and counted with percentage for categorical variables. Both bivariate and multivariate logistic regression analyses were used to identify independently associated factors of anemia in CKD patients. The bivariate analysis was exported to multivariate analysis when P<0.2 to control the possible effect of confounders. Adjusted odds ratio (AOR) with 95% CI and P-value <0.05 were used to select variables associated with anemia in CKD patients.
Result
Socio-Demographic Characteristics of the Participants
In the present study, about 387 participants were included. Out of these more than half, 220 (56.8%) of the participants were females. The median age of the participants was 55, while the minimum and maximum ages of the patient were 19 and 85 years respectively. About one third (32.6%) of participants were in the age group of 46–60 years old category. Two hundred twelve (54.8%) and two hundred eighty-five (73.6%) participants were urban dwellers and married respectively. More than 27% of the respondents were self-employed and 88 (22.7%) participants were civil servants. Around 25% of the respondents had a certificate of college achievement (Table 1).
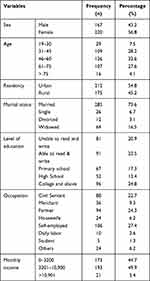 |
Table 1 Socio-Demographic Characteristics of Adult CKD Patients Attending Outpatient Department at Addis Ababa Public Hospital, Addis Ababa, Ethiopia, 2020 (N= 387) |
Clinical Characteristics of Respondents
One hundred fifty-four (39.8%) and two hundred twenty-two (57.4%) of respondents were hypertensive and diabetes mellitus patients respectively. Of all participants, more than half (54.0%) of the respondents had CKD of less than one-year duration. About two-thirds (66.1%) of the participants had normal weight with a BMI label of between 18.5–24.9 (Table 2).
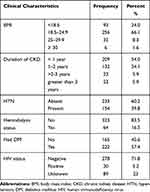 |
Table 2 Clinical Characteristics of Adult CKD Patients Attending Outpatient Department at Addis Ababa Public Hospitals, Addis Ababa, Ethiopia, 2020 (n=387) |
History of hemodialysis was present in 64 (16.5%) of the respondents. As few as 20 (5.2%) of the respondents were HIV positives confirmed by the laboratory (Table 2).
Nearly 70% of the participants were diagnosed having stage 3 and stage 4 CKD while only 2.8% of them were at an early stage (stage 1) of CKD. [Figure 1].
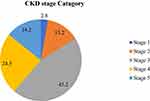 |
Figure 1 CKD stages of adult CKD patients attending outpatient department at Addis Ababa Public Hospital, Addis Ababa, Ethiopia, 2020 (n= 387). |
Behavioral Characteristics of Respondents
Three hundred thirty-two (85.8%) of the respondents were not smoking currently. But 72 (18.6%) of the respondents were smokers before and stopped smoking currently. Sixty-four (16.5%) of the participants still take alcohol (Table 3).
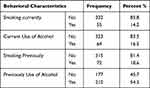 |
Table 3 Behavioral Characteristics of Adult CKD Patients Attending Outpatient Department at Addis Ababa Public Hospital, Addis Ababa, Ethiopia, 2020 (n=387) |
Prevalence of Anemia
Study participants’ charts were reviewed to assess the prevalence of anemia, and the clinical characteristics of the respondents. Accordingly, from a total of 387 CKD patients who were on follow-up at Zewditu Memorial, St. Paul, and Tikur Anbessa specialized hospitals, a total of 207 (53.5%) CKD patients were found anemic. Hence, the current study confirms an overall 53.5% prevalence of anemia in CKD patients.
The prevalence of anemia concerning age was 33.3 for those aged 46–60 years. Married CKD patients had a higher prevalence (76.3%) of anemia as compared to unmarried. The prevalence of anemia was 28% in respondents who attended at least college. (Table 4). Furthermore, the prevalence of anemia is increasing as the stage of CKD is increasing. Two (18.8%) among 11 stage one CKD patients, 14 (27.5) out of 52 stage 2 CKD patients, 72 (41.1) among 175 stage 3 CKD patients, 69 (72.6%) among 95 stage 4 CKD patients, and 50 (90.9%) out of 55 stage 5 CKD patients were anemic (Figure 1). Among the respondents who were hypertensive and diabetics, 68.8% and 59.5% were found anemic respectively (Figure 2).
 |
Table 4 Distribution of Anemia by Demographic, Clinical, and Behavioral Characteristics Among Adult CKD Patients Attending the Outpatient Department |
 |
Figure 2 Distribution of anemia in HTN and DM patient among adult CKD patients attending outpatient department at Addis Ababa Public Hospital, Addis Ababa, Ethiopia, 2020 (n=387). |
Higher anemia prevalence was noted in stage 3 and stage 4 CKD patients. More than 12% of the respondents who smoke currently and 13% of the respondents who used to smoke cigarettes have developed anemia. Nearly 50% of the respondents who had a history of alcohol consumption and 14.5% of the respondents who are currently taking an alcohol developed anemia (Table 4).
Factors Associated with Anemia Among CKD Patients
To identify factors associated with anemia among CKD patients, a logistic regression model was fitted. On binary logistic regression analysis, sex, CKD stages, previous history of alcoholic and smoking, presence of proteinuria, HTN, DM, BMI, hemodialysis, and duration of CKD were associated with anemia. Finally, after obtaining statistically significant variables at p<0.05 in binary logistic regression analysis, multiple logistic regression analysis was carried out to see the independent predictors of anemia among CKD patients. The multivariable logistic regression was carried out by taking anemia as a covariate in addition to those variables where the significant association was obtained in binary logistic regression. After adjusting potential cofounders, being female, presence of proteinuria, co-morbidity with HTN and DM, CKD stages, and hemodialysis remained statistically significant for the occurrence of anemia among CKD patients (Table 5).
 |
Table 5 Bivariate and Multivariate Analysis for the Occurrence of Anemia Among CKD Patients (N= 387) |
Discussion
This study aimed to synthesize evidence on the magnitude of anemia and its associated factors in patients with CKD at selected hospitals of Addis Ababa, Ethiopia. According to this study, the overall prevalence of anemia in patients with CKD was 53.5% which is higher than reported data by a study conducted in Korea (44.9%), Tanzania (33%), United States (15%), UK (6.76%), and Dessie, Ethiopia (39.5%).6,7,18,19 However, it is lower than the reports made by other researchers that showed, 77.5%, 67%, and 64.5% in Nigeria, Kenya and University of Gondor Ethiopia respectively.23,24 The low prevalence in our study is because of differences in the definition of anemia, study population, and survey period. The variation of the prevalence may be due to differences in the methodology, variation of quality of care and quality of reporting, policy, and strategic difference.
The prevalence of anemia was varied by the stages of CKD. This study demonstrated that 18.2%, 27.5%, 41.1%, 72.6% and 90.9% of stag1, 2, 3, 4 and stage 5 patients were anemic respectively. A comparable trend was also observed from the recent Korean cohort with a prevalence of 10% at stage 1 to 96.5% at stage 5.18 NHANES survey in the United States with a prevalence of anemia from 8.4% at stage 1 to 53.4% at stage 5.19 It is also similar to studies conducted in the University of Gonder Ethiopia study with a prevalence of 20% at stage 1 to 93.8% at stage 5 CKD.20 It indicates that an increasing prevalence of anemia was associated with the CKD stage.
This study has clinical implications in that the high magnitude of anemia in patients with CKD should guide healthcare professionals to minimize the risk of anemia by guiding the patient who could be detected in health checkups, give information about possible risk factors during routine patient care, and provide knowledge about the potential risk of anemia. Besides, identifying associated risk factors may help health care professionals treat anemia patients with CKD during their clinical care.
The current study indicated that patients with the presence of diabetes mellitus were 2 times more likely to develop anemia as compared to patients with non-diabetes (AOR = 2.156; 95% CI: 1.231, 3.776), and patient with HTN had 3 times more likely to develop anemia as compared to patients with non-hypertensive (AOR = 2.776; 95% CI: 1.597, 4.827). A similar study was conducted in Kenya.18 The result indicates that the prevalence of anemia among hypertensive and DM CKD respondents is higher than those who are not. This finding suggests that any CKD patient who presents with HTN and DM should be more closely monitored for anemia.
In this study, patients with hemodialysis history had two times higher odds for anemia (AOR=2.754, 95% CI: 1.218–6.229, P=0.015) compared to patients without hemodialysis history which is similar to the study in the university of Gonder Ethiopia.20 This is because hemodialysis requiring patients with advanced renal disease, in which the presence of anemia was prevalent.
The present study demonstrated that females were 2 times more likely to develop anemia as compared to their counterparts (AOR=2.04, 95% CI:1.18, 2.46). This finding is supported by previous studies conducted in the World Journal of Nephrology, Nepal, and Nigeria.17,21,22 This would suggest those female patients had lower HGB concentrations than male patients, which likely explains why females had a greater risk of developing anemia. However, sex was not significantly associated with anemia as reported in Korea study.6
The current study revealed that those patients who had urine protein were 2 times more likely to develop anemia as compared with patients who do not have proteinuria (AOR=2.320, 95% CI: 1.323, 4.067). This finding is in agreement with Korea.23 The evidence further supports that low serum albumin due to protein malnutrition and/or inflammation.
Conclusion and Recommendation
Prevalence of anemia was increased when stages of chronic kidney disease increased and worsened with a progressive decline in kidney function. HTN, DM, presence of proteinuria, CKD stages, and having hemodialysis history were independently associated with anemia in CKD patients. There is a need to improve multiple aspects of CKD management, including early diagnosis and treatment of anemia. Periodic screening and intervention for anemia in CKD patients should be practiced to prevent its complication.
Abbreviations
BMI, body mass index; BP, blood pressure; BUN, blood urea nitrogen; CKD, chronic kidney diseases; DM, diabetic mellitus; EGFR, estimated glomerular filtration rate; GFR, glomerular filtration rate; HCT, hematocrit; HGB, hemoglobin; HIV, huma immune virus; KDOQI, Kidney Disease Outcomes Quality Initiative; RBC, red blood cell; UK, United Kingdom; USA, The United States of America; WHO, World Health Organization.
Data Sharing Statement
The data analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
To confirm the Declaration of Helsinki (1964) and the Population Screening Act, Addis Ababa University Institutional Review Board approved the study. Participation was voluntary, and information was collected anonymously after obtaining written consent from each respondent. Confidentiality of data was ensured throughout the study.
Acknowledgments
Our gratitude goes to Addis Ababa University, College of Health Sciences, School of Nursing and Midwifery for their financial support. Our recognition also goes to data collectors, supervisors, nursing staff, and authorities of the three hospitals at the renal clinic who were facilitating the data collection. We would like to thank the study participants who were willing to take part in this study. Finally, we are glad to mention that the paper has been published as a thesis repository file in the Addis Ababa university college of health sciences school of the nursing library.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was conducted in collaboration with Addis Ababa University. Every step of the project was followed by Addis Ababa University, College of Health Sciences, School of Nursing and Midwifery. The university has no role in designing, analyzing, and writing the study.
Disclosure
The authors declare that they have no competing interests.
References
1. Chronic F, Disease K. Clinical practice guidelines.
2. Database WHOG. Worldwide prevalence of anemia. 2005
3. WHO. The Global Prevalence of Anemia in 2011. WHO. 1–48. 2011.
4. Walters D, Kakietek J, Eberwein JD, Shekar M. An investment framework for meeting the global nutrition target for anemia.. 2017;1–4.
5. Cases A, Egocheaga MI, Tranche S, et al. Special article Anemia of chronic kidney disease: Protocol of study, management, and referral to nephrology. 2018;8(1):8–12.
6. Kang E, Han M, Kim H, et al. Baseline general characteristics of the Korean chronic kidney disease: report from the Korean cohort study for Outcomes in Patients With Chronic Kidney Disease (KNOW-CKD). J Korean Med Sci. 2017;32(2):221–230. doi:10.3346/jkms.2017.32.2.221
7. Meremo AJ, Mwashambwa MY, Masalu MB, et al. Prevalence and predictors of anemia among patients presenting with kidney diseases at the University of Dodoma hospital in central Tanzania. Tanzan J Health Res. 2017;19(3):1–7.
8. Cases A, Egocheaga MI, Tranche S, et al. Anemia of chronic kidney disease: protocol of study, management, and referral to Nephrology. Nefrologia. 2018;38(1):8–12. doi:10.1016/j.nefro.2017.09.004
9. Keywords SA Prevalence of Anemia and Its Associated Factors among Chronic Kidney Disease patients at University of Gondar Hospital, North West Ethiopia: a Hospital-based Cross-Sectional Study.:1–17.
10. Journal O, Society I. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. 2013. 3(1).
11. Ckd DOF, Graded N. Chapter. 1: definition and classification of CKD. Kidney Int Suppl. 2013;3(1):19–62. doi:10.1038/kisup.2012.64
12. Who Global Report on Trends in the Prevalence of Tobacco Smoking 2000-2025.
13. Getachew T, Defar A, Teklie H, Gonfa G, Bekele A, Bekele A Magnitude and predictors of excessive alcohol use in Ethiopia: Findings from the 2015 national non-communicable diseases STEPS survey. 2015
14. Nishida C, Barba C, Cavalli-Sforza T, et al. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363(9403):157–163.
15. Shuwa K. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: improving Global Outcomes (KDIGO) J EROME R OSSERT, D ICK DE Z EEUW, T HOMAS H. H OSTETTER, N ORBERT L AMERIE, CONFERENCE. Kidney Int. 2005;67:2089–2100. doi:10.1111/j.1523-1755.2005.00365.x
16. Id TF, Adamu A, Tesfaye M, Gebreweld A. Prevalence of anemia in diabetic adult outpatients in Northeast Ethiopia.. 2019;1–12.
17. Ijoma C, Ulasi I, Ijoma U, Ifebunandu N High prevalence of anemia in predialysis patients in Enugu, Nigeria us eon m er al. 2010
18. Karimi PN, Nyamu D. Correlates and management of the anemia of chronic kidney disease in a Kenyan Tertiary Hospital CORRELATES AND MANAGEMENT OF ANAEMIA OF CHRONIC KIDNEY. 2018;2016(January).
19. Stauffer ME, Fan T. Prevalence of Anemia in Chronic Kidney Disease in the United States. 2014;9(1):2–5.
20. Adera H, Hailu W, Adane A, Tadesse A. Prevalence of anemia and its associated factors among chronic kidney disease patients at university of Gonder hospital, northwest Ethiopia: a hospital-based cross-sectional study. Int J Nephrol Renovasc Dis. 2019;12:219–228. doi:10.2147/IJNRD.S216010
21. Study CC. World J Nephrol. 2016;5(4):358–366.
22. Poudel B, Yadav BK, Jha B, Raut KB. Prevalence and association of anemia with CKD: a hospital-based cross-sectional study from Nepal Prevalence and association of anemia with CKD: a hospital-based cross-sectional study from Nepal. 2019.
23. Yi SW, Moon SJ, Yi JJ. Low-normal hemoglobin levels and anemia are associated with increased risk of end-stage renal disease in general populations: a prospective cohort study. PLoS One. 2019;14(4):1–12. doi:10.1371/journal.pone.0215920
24. Alemu B, Techane T, Negalign Getahun YT. College of Health Sciences School of Allied Health Sciences Department of Nursing and Midwifery College of Health Sciences School of Allied Health Sciences. Addis Ababa University, College of Health Sciences, School of Nursing. 2020.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
