Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
Prevalence and risk factors for COPD in greenhouse farmers: a large, cross-sectional survey of 5,880 farmers from northeast China
Authors Liu S, Ren Y, Wen D, Chen Y, Chen D, Li L, Zhang X, Zhang Y, Fu S, Li Z, Xia S , Wang D, Chen H, Zhao J, Wang X
Received 14 December 2014
Accepted for publication 29 June 2015
Published 1 October 2015 Volume 2015:10(1) Pages 2097—2108
DOI https://doi.org/10.2147/COPD.S79264
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Shuo Liu,1 Yangang Ren,1 Deliang Wen,1 Yu Chen,2 Donghong Chen,1 Liyun Li,3 Xuhua Zhang,1 Yibing Zhang,4 Shuang Fu,5 Zhenhua Li,3 Shuyue Xia,6 Dongliang Wang,7 Hong Chen,8 Jian Zhao,9 Xiaoge Wang1
1The Fourth Affiliated Hospital, 2The Second Affiliated Hospital, 3The First Affiliated Hospital, China Medical University, 4The Shenyang Military General Hospital, Shenyang, 5The General Hospital of Fushun Mining Bureau, Fushun, 6Fengtian Hospital, Shenyang Medical College, 7202nd Hospital of People’s Liberation Army, Shenyang, 8The First People’s Hospital of Kazuo, Chaoyang, 9205th Hospital of People’s Liberation Army, Jinzhou, Liaoning, People’s Republic of China
Background: COPD is one of the most common chronic diseases, and more and more farmers who were frequently exposed to greenhouse environments were diagnosed with COPD. However, little information is available on the prevalence of COPD among the greenhouse farmers. This study was conducted to assess the prevalence of COPD and investigate the potential risk factors for COPD among the Chinese greenhouse farmers.
Methods: Cross-sectional studies involving a sample of greenhouse farmers living in northeast China were performed via stratified-cluster-random sampling. All subjects were interviewed using a uniform questionnaire and underwent pulmonary function tests between 2006 and 2009, based on the diagnostic criteria of the Global Initiative for Chronic Obstructive Lung Disease. Multiple logistic regression analysis was conducted to examine the risk factors for COPD.
Results: Of the 5,880 greenhouse farmers from northeast China who were originally selected for this study, 5,420 questionnaires were completed. The overall prevalence of COPD in greenhouse farmers was 17.5%. The COPD prevalence was significantly higher in elderly subjects (≥50 years), current smokers, in those with lower body mass index (≤18.5 kg/m2) and less education, in those who were exposed to mushroom, flowers and poultry, and in those living in mountain and coastal region. Multiple logistic regression analysis revealed that age over 50 years old (odds ratio [OR]=298.69, 95% confidence interval [CI]=121.57–733.84), smoking (OR=2.18, 95% CI=1.84–2.59), planting mushroom and flowers (OR=1.46 and 1.53, 95% CI=1.13–1.87 and 1.24–1.95), and living in mountain and coastal region (OR=1.68 and 1.35, 95% CI=1.37–2.06 and 1.10–1.65) were associated with the development of COPD among greenhouse farmers.
Conclusion: In northeast China, COPD is highly prevalent among greenhouse farmers, and advanced age, smoking, planting mushroom, and flowers, as well as living in mountain and coastal regions, are potential risk factors for this disease.
Keywords: COPD, epidemiology, greenhouse farmers, prevalence, risk factor
Introduction
Currently, plastic film greenhouses are horticultural and agricultural facilities that conserve energy and provide opportunities to grow plants year round in a controlled environment. In the People’s Republic of China, the greenhouse cultivation is an important component of agricultural economy. Liaoning Province has the largest greenhouse area with 67,800 ha in the People’s Republic of China, and there are more and more greenhouse farmers due to the upward trend in the number of greenhouses. However, several studies have recently shown high prevalence of respiratory symptoms such as rhinitis, chronic cough, dyspnea, chronic bronchitis, and farmer’s lung disease among greenhouse farmers.1–4 The major risks of long-term exposure to internal greenhouse environments are enclosed spaces, high temperature, high humidity, and inhalation of antigens derived from protozoa, molds, insects, bacteria, and chemicals such as pesticides, fertilizers, and other organic materials.2,5
COPD, one of the most common respiratory diseases, is characterized by incompletely reversible lung airflow limitation.6 Worldwide, by 2010, there were approximately 329 million people affected by COPD, and the number of deaths from COPD was 2.9 million.7,8 The main risk factors for COPD globally are tobacco smoking, air pollution, genetics, as well as occupational exposure to workplace dusts, fumes, and chemicals.9 Recently, in our clinical practice, more and more greenhouse farmers were diagnosed with COPD. However, there is no information about the prevalence of COPD among the greenhouse farmers. Therefore, the objective of this present study was to estimate the prevalence and to analyze the potential risk factors for COPD in Chinese greenhouse farmers by a screening survey in the suburbs of Liaoning Province in North People’s Republic of China from August 2006 to June 2009.
Methods
Study subjects
Greenhouse farmers from the suburbs of Liaoning Province in North People’s Republic of China were chosen as subjects using stratified-cluster-random sampling. We divided our study population into three levels based on geographic differences in Liaoning Province for a questionnaire. The sample size was calculated as follows: N = K* (1-P)/P, N indicates sample size, K is constant decided by the allowable error of research projects, and P represents the expected rate for a certain disease. When the error of sample rate limits the 10% of the overall rate and α equals to 0.05, K is 400. According to the prevalence of COPD from previous survey of 300 greenhouse farmers in each level, overall prevalence was P=0.20. Therefore, the estimated sample size was 1,600 for each level (three levels), and the total sample size (N) was 4,800. Considering the possibility of nonresponsive subjects and subjects being lost to follow-up, the sample size was increased by 10% to 5,334.
Questionnaire
We developed a special questionnaire including 99 items based on the international BOLD study, the previous COPD studies in the People’s Republic of China, and our previous studies on Farmer’s Lung in greenhouse farmers.4,10,11 Questions about personal and family information, medical history, allergic history, and respiratory symptoms (cough, cough up phlegm, and shortness of breath) were asked. In addition, the natural conditions for greenhouses, such as height, area, inside temperature, relative humidity, sunlight duration, ventilation frequency, as well as the pH value, temperature, humidity of the soil were also included. Moreover, present or previous smoking habits, years of work experience, working hours, and the application of pesticides were also asked. All questionnaires were completed by several well-trained respiratory physicians via face-to-face interviews with the subjects.
Data collection
All recruited residents were identified through employee registries, were contacted by phone calls, and were invited to participate in the study at their convenience. They were interviewed individually by our trained interviewers using a standardized questionnaire. Each of the participants was made fully aware of the study’s purpose and provided the informed consent. A total of 5,420 greenhouse farmers were interviewed and underwent physical examination and pulmonary function tests (spirometer model: REF 910520 spirotel, Rome, Italy). The spirometry was performed according to joint American Thoracic Society and European Respiratory Society (ATS/ERS) guidelines.12 Additionally, all subjects also underwent postbronchodilator test 15 minutes after receiving 400 μg of inhaled salbutamol. Pulmonary function tests were undertaken at least three times, and the best result of the three was used. Both the definition of COPD and the classification of severity of COPD were based on the diagnostic criteria of the GOLD. We defined COPD as the ratio of forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) <70% after administration of an inhaled bronchodilator, FEV1 ≥80% as mild obstruction, FEV1 between 50% and 79% as moderate obstruction, FEV1 between 30% and 49% as severe obstruction and FEV1 <30% as very severe obstruction.13,14 Body mass index (BMI) was calculated from the equation as BMI = weight (kg)/height (m2), which was classified into four groups: >18.5, 18.5–23.9, 24.0–27.9, and 28.0 or more based on World Health Organization guidelines and the distribution of our data.15 The current study was approved by the regional ethics committee.
Exclusion criteria
Subjects suffering from chronic respiratory diseases contracted prior to working in greenhouses and subjects who refused to participate in the study were not included in the survey. Subjects diagnosed with any of the following conditions within 2 months of the study’s initiation were ineligible for spirometry testing: thoracic or abdominal surgery, eye surgery (or retinal detachment) tuberculosis, heart attack, or hospitalization due to cardiovascular disease. Subjects with the following conditions were required to skip the test until they became eligible: respiratory infection in the past 3 weeks, pulmonary medication use in the past 24 hours, and smoking or vigorous exercise within 1 hour.
Quality control
All interviewers and spirometry operators were trained and accredited before the survey. All spirometry data were recorded, and no deletions or revisions were made. Each completed questionnaire and spirometry report was verified by a field supervisor. The results were double checked by the principal investigator and were repeated in each greenhouse farmer with <95% repetitive rate. All questionnaire data were coded and independently entered into a standardized excel database by two individuals: computer programs proof read for outlying values and logic errors.
Statistical analysis
These variables included the following: sex, age (<40, 40–49, 50–59, and ≥60), BMI (<18.5, 18.5–23.9, 24.0–27.9, and ≥28), smoking status, region, greenhouse type, working years, daily work hours, plant area, childhood greenhouse exposure, education and allergy history, which were all treated as categorical variables. The chi-square test was used for comparisons of two sample rates, and the chi-square segmentation method for additional comparisons. ORs and 95% CI values for COPD relative to potential risk factors were calculated using multiple logistic regression models. The variables mentioned above except sex were all forced into the final multiple logistic regression model. All data were analyzed using the SPSS 17.0 statistical software package (SPSS, Inc., Chicago, IL, USA). A P-value ≤ 0.05 was statistically significant.
Results
Sample demographics
Of the 5,880 greenhouse farmers from the Liaoning Province suburbs of the People’s Republic of China who were originally sampled, 5,498 (93.5%) were interviewed. Among these 5,498 interviewees, 78 failed to complete spirometry, and 5,420 questionnaires were received for evaluation, a response rate of 92.2% (5,420/5,880) (Figure 1). Reasons for nonresponse included patient refusal, failure to make contact with patients, and failure to complete spirometry.
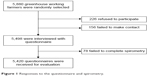  | Figure 1 Responses to the questionnaire and spirometry. |
The mean age of the study population was 44.76 (standard deviation, 10.66) and ranged from 22 to 74. The percentages of current smokers and nonsmokers in the study population were 35.1% and 64.9%, respectively. Smokers accounted for 51.7% of male subjects and 19.1% of female subjects. Subjects with <15 pack-years of smoking history accounted for 34.3% of study subjects. The smoking rates of the study population by sex, age, region, and greenhouse type are included in Table 1.
  | Table 1 Smoking status of responders by region, sex, age, and greenhouse type |
Prevalence of COPD in greenhouse farmers
The parameters of postbronchodilator pulmonary function, including mean FEV1 (%), FVC (%), and FEV1/FVC as well as the proportion of FEV1/FVC <0.70 and the proportion of FEV1/FVC >0.70 but FEV1 or FVC <80% by region, sex, age, and greenhouse type are presented in Table 2. According to the definition of COPD as postbronchodilator FEV1/FVC >0.70, the overall prevalence of COPD in greenhouse farmers from Liaoning Province was 17.5% (947/5,420); the prevalence of COPD in males was 18.1% (481/2,660); and the prevalence in females was 16.9% (466/2,760). The prevalence of COPD in greenhouse farmers older than 40 years was 25.5% (940/3,688). Most patients were elderly individuals and were older than 50 years. The current smokers were associated with a higher COPD prevalence (23.3%) compared to nonsmokers (14.3%).
The prevalence of COPD in greenhouse farmers exposed to plain areas was 15.6% (464/2,981), which was lower than that in both the mountains (20.2%, 246/1,220) and the coastal areas (19.4%, 237/1,219), a statistically significant difference (the values of χ2 were 13.04 and 9.35, respectively, all P<0.005). The prevalence of COPD in vegetable greenhouse farmers was 12.6% (273/2,168), which was lower than that observed in mushroom greenhouse farmers (24.3%, 263/1,084), poultry greenhouse farmers (20.8%, 169/813), and flower greenhouse farmers (17.9%, 242/1,355), a difference that was statistically significant (the values of χ2 were 77.35, 20.83, and 34.41, respectively, all P<0.0005). The prevalences of COPD organized by region and greenhouse type are included in Table 3.
Based on the GOLD criteria, the prevalences of stage I (mild), stage II (moderate), stage III (severe), and stage IV (very severe) COPD were 13.2% (715/5,420), 3.1% (169/5,420), 1.2% (63/5,420), and 0%, respectively.
Risk factors for COPD among greenhouse farmers
The stratification analysis results regarding potential risk factors for COPD among Chinese greenhouse farmers are shown in Table 4. On the basis of these chi-square analysis results, age, BMI, smoking status, demographics, greenhouse type, working years, daily work hours in greenhouse, plant area, heating materials in winter, childhood greenhouse exposure, childhood asthma symptoms, education and allergy history were forced into multiple logistic regression models. Some of them including smoking status, childhood greenhouse exposure, asthma history, education, and allergy history were categorized as shown in Table S1, and other variables were respectively set into dummy variables as listed in Table S2.
Through multiple logistic regression analysis as presented in Table 5, it was determined that aging, smoking (odds ratio [OR]=2.18, 95% CI=1.84–2.59), mushroom and flower greenhouses (compared with vegetable greenhouses, OR=1.46 and 1.53, 95% CI=1.13–1.87 and 1.24–1.95), mountain and coastal region (compared with plains, OR=1.68 and 1.35, 95% CI=1.37–2.06 and 1.10–1.65), and working in greenhouse for 3–5 years (OR=1.52, 95% CI=1.08–2.15) were the primary risk factors for COPD among greenhouse farmers. Additionally, BMI was negatively associated with COPD (BMI: 18.5–23.9 kg/m2, 24.0–27.9 kg/m2, and ≤28.0 kg/m2 compared to >18.5 kg/m2, OR=0.43, 0.38, and 0.20, 95% CI=0.25–0.75, 0.22–0.65, and 0.11–0.36).
  | Table 5 Risk factors for COPD among greenhouse farmers by multiple logistic regression analysis |
Discussion
In this population-based study, we demonstrated for the first time the prevalence of COPD among the greenhouse farmers and the potential risk factors for COPD. COPD is a significant public health problem, and experts predict that COPD will rise to the third leading cause of death by 2020 and that it will be the fifth most costly disease worldwide by that time.16,17 The global prevalence of COPD ranges between 0.23% and 18.3% based on previous studies.18 In this large-scale population and cross-sectional study in Liaoning Province in the People’s Republic of China, the overall prevalence of COPD among greenhouse farmers here was 17.5%. This result was much higher than the combined prevalence of COPD in 12 Asia-Pacific countries and regions (6.3%).19 It was also higher than the results determined by Nanshan Zhong’s large-scale spirometry-based population survey (8.2%) involving Chinese people older than 40 years,10 but it was lower than the results obtained by Salzburg of Austria (26.1%).20 According to the GOLD criteria, the prevalence of COPD has also been described in Finland (9.4% among adults 20–70 years)21 and Korea (17.2% among subjects older than 45 years).22 Although the results of those studies cannot be directly compared with ours, all of them have identified COPD as a global threat to human health.
As reported by the Obstructive Lung Disease in Northern Sweden Studies, the prevalences of COPD were 8.2%, 5.3%, 0.7%, and 0.1% for mild, moderate, severe, and very severe disease, respectively.23 As patients with stage IV (very severe) disease cannot work, there were no subjects with stage IV COPD in our study. Our study also demonstrated a higher prevalence of stage I COPD (13.2%) in greenhouse workers than in the Chinese general population (2.0%),10 which may be attributed to greenhouse work environments. Additionally, 1,200 farmers aged ≥40 years who live in the same regions with greenhouse farmers but have never worked in greenhouse were also invited to complete questionnaires and perform pre- and postbronchodilator spirometry. The results indicated that the prevalence of COPD among nongreenhouse farmers was 9.75% (117/1,200, data not shown), which was significantly lower than that of greenhouse farmers aged ≥40 years (25.5%, 940/3,681; χ2=132.3, P<0.0005). Taken together, it is of significant concern and explores the prevalence and risk factors for COPD among the greenhouse farmers.
As expected, the prevalence of COPD increased in conjunction with the ages of greenhouse workers in our study, which was consistent with the findings observed in the Chinese general population.10
Tobacco smoking is a well-documented risk factor that contributes substantially to COPD globally.24 As shown in our study, 22.8% (434/1,902) of greenhouse workers who smoked had COPD, which was much higher than nonsmokers (14.3%) as well as the general COPD prevalence (17.5%). There was no significant difference in COPD prevalence between male and female smokers (23.3% vs 21.65, χ2=0.657, P>0.05). Thus, these results indicate that smoking is an important risk factor for COPD in greenhouse farmers. Surprisingly, we did not find a strong dose–response relationship with pack-years of smoking. This could be partly explained that the sample size of >14 pack-years smokers (55 subjects) was insufficient to analyze its correlation with COPD. Therefore, large-scale population-based studies are necessary to be performed to further explore the relationship between pack-years of smoking and COPD.
In the present study, nonsmokers accounted for 48.3% of the male subjects and 80.9% of the female subjects. However, there was no significant difference between the overall COPD prevalence of female (16.9%) and that of male (18.1%) (χ2=0.948, P>0.05). It is rather remarkable that the prevalence of COPD in female nonsmokers was much greater than that in male nonsmokers (15.8% vs 12.5%, χ2=174.880, P<0.05) (Table S3). These findings suggest that risk factors other than tobacco smoking may be linked to COPD in female greenhouse workers. Recently, the role of biomass fuel smoke exposure in the development of COPD has attracted more attention. Biomass fuels, such as wood, crop residues, mildewed grasses, and organic animal dung, are commonly used for household cooking and heating in the villages of developing countries.25 Incomplete burning of biomass fuels can release a large amount of air pollutants into the environment, which include carbon monoxide, nitrous oxides, sulfur oxides, and suspended particulate matter.26 Generally, women are more likely to be exposed to high levels of indoor air pollution than men in developing countries. There is increasing evidence that exposure to biomass fuel smoke is an important risk factor for COPD globally, especially in a rural women population in developing countries.27–31 Thus, biomass fuel smoke probably leads to a high COPD prevalence for nonsmoking females.
Our studies also demonstrated that the greenhouse farmers who often used mildewed grasses (biomass fuel) for heating in winter had higher COPD prevalence (17.7%) than other materials users (electricity and coal), although there was no correlation in multiple logistic analysis models. Additionally, the results of the multiple logistic regression analysis indicated that mountainous and coastal environments were risk factors for COPD among greenhouse framers compared with plain areas (OR=1.68 and 1.35, 95% CI=1.37–1.87 and 1.24–1.95). Besides other possible explanations for these results such as altitude, high humidity and poor ventilation, heating with biomass fuels in winter, particularly in mountainous regions in which traffic problems and rich vegetation are commonplace, may be the primary cause. Therefore, the effect of biomass fuel smoke in the pathogenesis of COPD cannot be ignored.
As shown in the previous studies, the prevalence of COPD was significantly higher in subjects with lower BMI (>18.5 kg/m2).32–34 We also drew the same conclusion from the present study. This might be associated with the previous reports that undernutrition contributed to an increased susceptibility to infection or respiratory muscle wasting and subsequently abnormal pulmonary function.35,36 However, since weight loss commonly occurs among patients suffering from COPD, whether low BMI is a risk factor for COPD or a consequence of established disease is not clear. Thus, further investigation on the cause–effect relationship between BMI and COPD remains to be needed.
Our results revealed that the prevalence of COPD in mushroom greenhouse farmers (24.3%, 263/1,084) was higher than that in other types of greenhouses. Multiple logistic regression analysis also indicated that mushroom and flower were probably the risk factors for COPD among greenhouse farmers compared with vegetable (OR=1.46 and 1.53, 95% CI=1.13–1.87 and 1.24–1.95). These findings may be due to the existence of more allergens in mushroom and flower greenhouses. Surprisingly, we found that the subjects who worked in greenhouse for <5 years had less COPD compared with >3 years workers (OR=0.51, 95% CI=0.40–0.65). Why this should be the case and whether possible reasons such as enhancement of airway tolerability and subsequent decline of COPD susceptibility could explain this finding warrants further investigation.
Furthermore, it is surprising that there is no significant correlation between COPD and education background, asthma symptoms in childhood, allergic history, heating materials in winter as well as greenhouse area and daily time spending in greenhouse when assessed by multiple logistic regression analysis. This was probably due to multifactorial relationships among risk factors and the possibility of recall bias. Nevertheless, the results of logistic regression analysis imply that significant differences in daily working time and area of greenhouse do not constitute differences in working conditions, which may be beneficial for clinicians to implement preventive measures to reduce the prevalence of COPD in greenhouse farmers.
Despite these findings, the present study had several limitations. First, we did not further subdivide nonsmoker into “exsmoker” and “never smoker” in our survey. Second, in this study, a postbronchodilator fixed FEV1/FVC ratio of >0.7 was used as a criterion for COPD diagnosis, which can probably lead to overestimation of COPD prevalence in the elderly population reported by other studies.37,38 Recently, lower limit of normal criterion was instead of the fixed ratio criterion to minimize the potential misclassification.39,40 However, there is no current national predicted normative value of FEV1 in the People’s Republic of China. Alternatively, we adjusted the predicted normative value of FEV1 derived from the European Coal and Steel Community equations with conversion factors recommended by Zheng and Zhong to minimize the ethnic differences, which produced similar overall COPD prevalence and proportion of stage I disease with nonadjustment.41 Third, the numbers included in several variable categories such as subjects with experience of living in greenhouse during childhood, with asthma symptoms in childhood and with allergic history were so small that the contribution of these risk factors for COPD might be less significant.
Conclusion
In summary, this cross-sectional survey reveals that the prevalence of COPD is 17.5% among greenhouse farmers from Liaoning Province, which is much higher than the overall COPD prevalence in the People’s Republic of China (8.2%). There are numerous potential risk factors for the development of COPD among greenhouse farmers, including advanced age, tobacco smoking, planting mushroom and flowers, as well as living in mountain and coastal region. Our results highlight COPD as a major public health problem among Chinese greenhouse farmers and suggest that more attention should be devoted to the preventive measures and management of this disease. Further research will be needed to better understand the relationship between these determinants and the pathogenesis of COPD to reduce the burden of COPD in greenhouse farmers.
Summary
Chinese greenhouse cultivation is a special agricultural economy. Long-term exposure to internal greenhouse environments puts greenhouse farmers at risk for the inhalation of antigens; and therefore, the prevalence of respiratory diseases is higher in this population. This study showed the prevalence and risk factors for COPD in greenhouse farmers from northeast China.
Acknowledgments
The authors thank Professor Nanshan Zhong (Guangzhou Institute of Respiratory Disease, Guangzhou Medical College, People’s Republic of China), Jian Kang (The First Affiliated Hospital, China Medical University, People’s Republic of China), Quanying He (Beijing Chaoyang Hospital, Capital University of Medical Sciences, People’s Republic of China), and Yiming Zhao (The Third Hospital, Peking University, People’s Republic of China) for providing support and guidance. The authors thank the following investigators and local administrations for their technical assistance with this study: Lei Xu, Lingling Wang, Qun Wang, Yuan Ling, Lihua Wu, Xiaoling Yu, Fangzhi Li, Jing LI, Bo Zou, Lijian Wu, Yanqing Liu, Baoguo Jiang, Xiaoyu He, Mingjing Zhao, Lie Ma, Jiaying Sun, Guangdan Zhao, Hongguang Dong, Lijiao Zhang, Jie Zou, Hao Zhang, Xuewen Wang, Dan Ma, Fang Nie (all of whom are from The Fourth Affiliated Hospital, China Medical University, People’s Republic of China), Jun Tian (The Fourth People’s Hospital of Shenyang, People’s Republic of China), Yuhong Zhou (General Hospital of Liaoning, Anshan Iron and Steel Group, People’s Republic of China), and Wenliang Ren (People’s Hospital of Shenyang Metro sub-district, People’s Republic of China).
The study is supported by grants from the National Science and Technology Pillar Program of the People’s Republic of China during the Eleventh Five-year Plan Period (No 2007BAI24B04), the Medical Education Science Research Topic of China Medical University during the Twelfth Five-year Plan Period (No YDJK2012052), the Science Foundation of the Chinese Medical Association (Nos 07010320040 and 08020680146), and the Youth Foundation of Medical Education and Science Research of China Medical University (YDJK2012052).
Disclosure
The authors report no conflicts of interest in this work. None of the authors has a financial relationship with a commercial entity invested in the subject of this paper.
References
Monso E. Occupational asthma in greenhouse workers. Curr Opin Pulm Med. 2004;10(2):147–150. | ||
Zuskin E, Schachter EN, Mustajbegovic J. Respiratory function in greenhouse workers. Int Arch Occup Environ Health. 1993; 64(7):521–526. | ||
Nigatu AW, Bratveit M, Deressa W, Moen BE. Respiratory symptoms, fractional exhaled nitric oxide and endotoxin exposure among female flower farm workers in Ethiopia. J Occup Med Toxicol. 2015;10:8. | ||
Liu S, Chen D, Fu S, et al. Prevalence and risk factors for farmer’s lung in greenhouse farmers: an epidemiological study of 5,880 farmers from Northeast China. Cell Biochem Biophys. 2015;71(2):1051–1057. | ||
Illing HP. Is working in greenhouses healthy? Evidence concerning the toxic risks that might affect greenhouse workers. Occup Med. 1997;47(5):281–293. | ||
Decramer M, Janssens W, Miravitlles M. Chronic obstructive pulmonary disease. Lancet. 2012;379(9823):1341–1351. | ||
Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2163–2196. | ||
Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095–2128. | ||
Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013; 187(4):347–365. | ||
Zhong N, Wang C, Yao W, et al. Prevalence of chronic obstructive pulmonary disease in China: a large, population-based survey. Am J Respir Crit Care Med. 2007;176(8):753–760. | ||
Buist AS, Vollmer WM, Sullivan SD, et al. The burden of obstructive lung disease initiative (BOLD): rationale and design. COPD. 2005; 2(2):277–283. | ||
Miller MR, Hankinson J, Brusasco V; ATS/ERS Task Force. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. | ||
Dewar M, Curry RW Jr. Chronic obstructive pulmonary disease: diagnostic considerations. Am Fam Physician. 2006;73(4):669–676. | ||
Bakke PS, Rönmark E, Eagan T, et al; European Respiratory Society Task Force. Recommendations for epidemiological studies on COPD. Eur Respir J. 2011;38(6):1261–1277. | ||
World Health Organization. Global Database on Body Mass Index: BMI Classification. Geneva: WHO; 2009. | ||
Lopez AD, Murray CC. The global burden of disease, 1990–2020. Nat Med. 1998;4(11):1241–1243. | ||
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442. | ||
Halbert RJ, Isonaka S, George D, Iqbal A. Interpreting COPD prevalence estimates: what is the true burden of disease? Chest. 2003; 123(5):1684–1692. | ||
Regional CWG. COPD prevalence in 12 Asia-Pacific countries and regions: projections based on the COPD prevalence estimation model. Respirology. 2003;8(2):192–198. | ||
Schirnhofer L, Lamprecht B, Vollmer WM, et al. COPD prevalence in Salzburg, Austria: results from the burden of obstructive lung disease (BOLD) study. Chest. 2007;131(1):29–36. | ||
Jyrki-Tapani K, Sovijarvi A, Lundback B. Chronic obstructive pulmonary disease in Finland: prevalence and risk factors. COPD. 2005; 2(3):331–339. | ||
Kim DS, Kim YS, Jung KS, et al; Korean Academy of Tuberculosis and Respiratory Diseases. Prevalence of chronic obstructive pulmonary disease in Korea: a population-based spirometry survey. Am J Respir Crit Care Med. 2005;172(7):842–847. | ||
Lindberg A, Bjerg A, Ronmark E, Larsson LG, Lundback B. Prevalence and underdiagnosis of COPD by disease severity and the attributable fraction of smoking Report from the Obstructive Lung Disease in Northern Sweden Studies. Respir Med. 2006;100(2):264–272. | ||
Xu F, Yin X, Zhang M, Shen H, Lu L, Xu Y. Prevalence of physician-diagnosed COPD and its association with smoking among urban and rural residents in regional mainland China. Chest. 2005; 128(4):2818–2823. | ||
Fullerton DG, Bruce N, Gordon SB. Indoor air pollution from biomass fuel smoke is a major health concern in the developing world. Trans R Soc Trop Med Hyg. 2008;102(9):843–851. | ||
Kim KH, Jahan SA, Kabir E. A review of diseases associated with household air pollution due to the use of biomass fuels. J Hazard Mater. 2011;192(2):425–431. | ||
Mahesh PA, Jayaraj BS, Prabhakar AK, Chaya SK, Vijaysimha R. Identification of a threshold for biomass exposure index for chronic bronchitis in rural women of Mysore district, Karnataka, India. Indian J Med Res. 2013;137(1):87–94. | ||
Salvi SS, Barnes PJ. Chronic obstructive pulmonary disease in non-smokers. Lancet. 2009;374(9691):733–743. | ||
Liu S, Zhou Y, Wang X, et al. Biomass fuels are the probable risk factor for chronic obstructive pulmonary disease in rural South China. Thorax. 2007;62(10):889–897. | ||
Johnson P, Balakrishnan K, Ramaswamy P, et al. Prevalence of chronic obstructive pulmonary disease in rural women of Tamilnadu: implications for refining disease burden assessments attributable to household biomass combustion. Global Health Action. 2011;4:7226. | ||
Hu G, Zhou Y, Tian J, et al. Risk of COPD from exposure to biomass smoke: a metaanalysis. Chest. 2010;138(1):20–31. | ||
Daldoul H, Denguezli M, Jithoo A, et al. Prevalence of COPD and tobacco smoking in Tunisia – results from the BOLD study. Int J Environ Res Public Health. 2013;10(12):7257–7271. | ||
Lee SJ, Kim SW, Kong KA, Ryu YJ, Lee JH, Chang JH. Risk factors for chronic obstructive pulmonary disease among never-smokers in Korea. Int J Chron Obstruct Pulmon Dis. 2015;10:497–506. | ||
Harik-Khan RI, Fleg JL, Wise RA. Body mass index and the risk of COPD. Chest. 2002;121(2):370–376. | ||
Thurlbeck WM. Diaphragm and body weight in emphysema. Thorax. 1978;33(4):483–487. | ||
Chandra RK. Cell-mediated immunity in nutritional imbalance. Fed Proc. 1980;39(13):3088–3092. | ||
Ko FW, Woo J, Tam W, et al. Prevalence and risk factors of airflow obstruction in an elderly Chinese population. Eur Respir J. 2008; 32(6):1472–1478. | ||
Pothirat C, Chaiwong W, Phetsuk N, Liwsrisakun C. Misidentification of airflow obstruction: prevalence and clinical significance in an epidemiological study. Int J Chron Obstruct Pulmon Dis. 2015;10:535–540. | ||
Sorino C, D’Amato M, Steinhilber G, Patella V, Corsico AG. Spirometric criteria to diagnose airway obstruction in the elderly: fixed ratio vs lower limit of normal. Minerva Med. 2014;105(Suppl.3 to No.6):15–21. | ||
Hnizdo E, Glindmeyer HW, Petsonk EL, Enright P, Buist AS. Case definitions for chronic obstructive pulmonary disease. COPD. 2006; 3(2):95–100. | ||
Zheng J, Zhong N. Normative values of pulmonary function testing in Chinese adults. Chin Med J. 2002;115(1):50–54. |
Supplementary materials
  | Table S1 The categorization for variables |
  | Table S2 Categorical dummy variables coding |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




